Abstract
Limited research has been done among pregnant people participating in investigational drug trials. To enhance the ethical understanding of pregnant people’s perspectives on research participation, we sought to describe motives and risk perceptions of participants in a phase 1 trial of ledipasvir/sofosbuvir (LDV/SOF) treatment for chronic Hepatitis C virus (HCV) during pregnancy. Pregnant people with chronic HCV infection enrolled in an open-label, phase 1 study of LDV/SOF participated in semi-structured, in-depth interviews to explore their reasons for participation and experiences within the study. Pregnant people took 12 weeks of LDV/SOF and were interviewed at enrollment and at the end of study. We recorded the interviews, transcribed them verbatim, coded them using NVivo software, and performed inductive thematic analysis. Nine women completed the study yielding 18 interview transcripts. We identified two themes regarding motives and one regarding risk perception. Motives—(1) Women conceptualized study participation as part of the caregiving role they associate with motherhood; participating was viewed as an act of caregiving for their infants, their families, themselves, and other pregnant women with chronic HCV. (2) Women also noted that they faced multiple barriers to treatment prior to pregnancy that created a desire to receive therapy through trial participation. Risk perception—(3) Women acknowledged personal and fetal risk associated with participation. Acceptance of risk was influenced by women’s concepts of motherhood, preexisting knowledge of HCV and medical research, family members, intimate partners, or by the study design. Women enrolled in a phase 1 trial for chronic HCV therapy during pregnancy acknowledged risks of participation and were motivated by hopes for fetal and personal benefit and by lack of prenatal access to treatment. Ethical inclusion of pregnant people in research should acknowledge structural factors that contribute to vulnerability and data deficiencies for treatment in pregnancy.
Keywords
Introduction
Responsible inclusion of pregnant participants is now recommended for drug trials, especially where the infant can benefit (Fairlie et al., 2019; World Health Organization and Council for International Organizations of Medical Sciences, 2017). Exclusion of pregnant participants under the claim of vulnerability has created a lack of knowledge on safety or dosing of drugs, while other drugs are not available in pregnancy (Little and Wickremsinhe, 2017; Lyerly et al., 2008; Omer and Beigi, 2016). Pharmaceutical research among pregnant people is rare but these efforts to include pregnant people in drug trials can mitigate the paucity of data (Caritis and Venkataramanan, 2021; Roset Bahmanyar et al., 2021). These trials should accommodate the ethical complexities associated with research during pregnancy as well as the complexities associated with coexisting medical conditions or cultural beliefs that may affect research participation (Committee on Ethics, 2015; Hunt et al., 2017; Macklin, 2010; van der Graaf et al., 2018). Trial participants may have comorbid conditions that provide eligibility for an investigational drug, and that warrant additional ethical considerations for research in pregnancy. With a better understanding of the motives for trial participation and risk perceptions among pregnant people with comorbid conditions, researchers may be able to design trials that better support pregnant participants and potentially have higher uptake among pregnant people (Ballantyne et al., 2017).
National rates of HCV infection among childbearing people are rising due to the opioid epidemic (Jhaveri et al., 2021; Koneru et al., 2016; Zibbell et al., 2018). This is concerning for pregnant people and infants given the 4%–8% risk of perinatal transmission of chronic HCV (Benova et al., 2014; Hughes et al., 2017). Oral direct-acting antiviral (DAA) drugs such as ledipasvir/sofosbuvir (LDV/SOF) treat chronic hepatitis C virus (HCV) with fewer adverse effects and high cure rates compared to prior therapies (Afdhal et al., 2014; Tapper et al., 2017). However, chronic HCV diagnosed during pregnancy rarely results in postpartum treatment due to barriers such as loss of pregnancy-related Medicaid eligibility, decreased healthcare engagement and competing demands associated with parenting (Chou et al., 2020; Krans et al., 2016; Schillie et al., 2020). A lack of drug treatment options during pregnancy and these multiple barriers to healthcare utilization and access after delivery are significant barriers to chronic HCV treatment (Jhaveri et al., 2021).
To address these barriers, a phase 1 open-label investigational drug trial recently evaluated pharmacokinetic and safety data for ledipasvir/sofosbuvir (LDV/SOF) among pregnant women with chronic hepatitis C virus (HCV) (Chappell et al., 2020). Given the unique barriers experienced by people living with chronic HCV, pregnant people with co-existing chronic HCV may require unique research protections and ethical considerations. These considerations may be elucidated through understanding the perspectives of pregnant women living with chronic HCV participating in investigational research. Surveys and interviews among non-pregnant women with chronic HCV have shown that women may be interested in DAA treatment during pregnancy but did not assess perspectives on research in pregnancy (Kushner et al., 2018; Lally et al., 2008; Mitchell, 2017; Rich et al., 2016). To understand the motives and risk perceptions of pregnant people with chronic HCV participating in an investigational drug trial, we conducted a series of semi-structured in-depth interviews within the phase 1 open-label trial of LDV/SOF in pregnancy. We sought to understand participants’ views on research in pregnancy and use of investigational drug therapy during pregnancy, especially in light of their concurrent diagnosis of chronic HCV.
Methods
An open-label, phase 1 pharmacokinetic study of LDV/SOF during pregnancy (ClinicalTrials.gov Identifier: NCT02683005) included qualitative interviews. This qualitative analysis focuses on motivations for trial participation and assessment of risks involved in study participation. Pregnant people enrolled from October 1, 2016 to September 30, 2018 within UPMC Magee-Womens Hospital in Pittsburgh, Pennsylvania. This hospital is a tertiary care center with approximately 10,000 deliveries each year. The study protocol and recruitment materials received approval from the University of Pittsburgh Institutional Review Board. Food and Drug Administration Investigational New Drug Application (#129502) from the University of Pittsburgh allowed the experimental use of LDV/SOF in pregnancy. Prenatal or substance abuse clinicians referred women to the study, and we also used print advertisements. Participants provided written informed consent prior to screening for enrollment. Inclusion criteria were chronic HCV infection, age between 18 and 39 years old and gestational age between 23 and 24 weeks at the time of enrollment. After enrollment, participants completed a demographic questionnaire and all were invited to complete qualitative interviews alongside the investigational drug trial. Participants who declined investigational therapy were not invited for qualitative interviews.
Within the open label trial, participants took ledipasvir 90mg-sofosbuvir 400mg (LDV/SOF) for 12 weeks during pregnancy. Three pharmacokinetic visits occurred at 25–26, 29–30, and 33–34 weeks gestation. We called participants weekly to review drug adherence and discuss participant concerns. We obtained delivery information for women who delivered at Magee-Womens Hospital. Compensation for participation included $50 at screening, enrollment, each qualitative interview, and the postpartum visit, $250 for each intensive pharmacokinetic visit, $150 during delivery hospitalization and $50 for each infant follow up visits. Cab fare or bus fare for transportation was provided as needed. The full study protocol including inclusion and exclusion criteria can be found in the phase 1 trial publication regarding pharmacokinetic/safety data and virologic response (Chappell et al., 2020).
The phase 1 trial included qualitative interviews in order to understand motivations for participation in an investigational drug trial during pregnancy and preferences for research in pregnancy in general. We interviewed participants at study enrollment to understand their reasons for participation and assessment of risk. We interviewed participants again at the end of the study in order to understand how the experience of study participation may have influenced their original motivations for participation or altered their experiences of risk. Cure for chronic HCV is established 12 weeks after completing therapy, but participants may not have been able to recall their perceptions or feelings as accurately after such a delay. Thus, we performed end-of-study interviews during the final weeks of investigational therapy but before participants could know if they had been cured.
Study team members with clinical and research experience among pregnancy and people with chronic HCV created the semi-structured interview guides. All interviews were conducted in-person by experienced trained qualitative interviewers, and participants had not met the interviewers before their first interview. Interviewers were skilled in asking open-ended questions that allowed participants to direct the course of the interview. In all enrollment interviews, participants were asked: “How important do you think it is to research study medications in pregnancy?,” “What concerns are your concerns about using a medication that has not been used during pregnancy before this study?” In all end-of-study interviews, women were asked “Why was it important to you to receive treatment during pregnancy?” and “What questions or concerns do you have now that treatment is done?” Answers to these questions are the focus of this analysis.
Two researchers read the interview transcripts and developed a qualitative codebook by independently coding all the interviews and comparing codes. To create codes we used an editing approach to identify segments of transcript text that comprehensively included all relevant topics and themes and described interview content (Crabtree and Miller, 1999). The two researchers resolved the few differences in coding after interactive discussion and a final version of the codebook was agreed upon by both coders. A supervising experienced qualitative researcher was available to address any lasting disagreements in coding, but there were no ongoing discrepancies. Example codes from the codebook are: “Goals for Treatment in Pregnancy,” and “Perspectives on Research in Pregnancy.” Care was taken during coding to identify thematic saturation, when no new ideas are identified among the transcripts, signaling strength of the sample size to understand a population of interest (Crabtree and Miller, 1999). We used an inductive approach to identify themes from the coded interviews, and then reviewed these themes with the entire qualitative study team (Braun and Clarke, 2006). Final themes review participants’ motivations for joining the trial and assessment of risks of trial participation.
Results
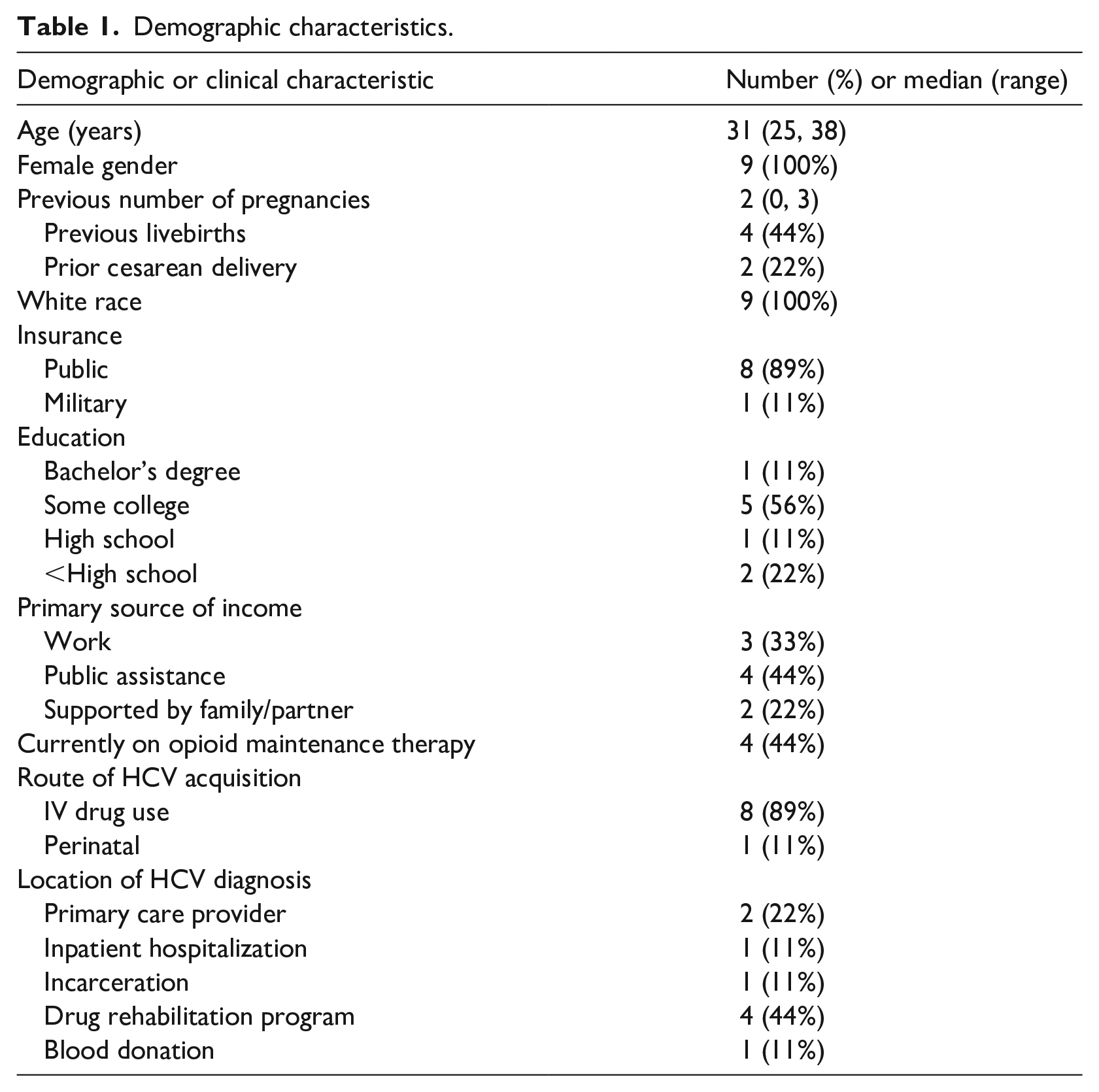
All nine participants of the phase 1 trial completed the enrollment and end-of-study interviews (Figure 1), resulting in 18 transcripts for analysis. Table 1 shows demographic characteristics of the sample population. All participants identified as women.
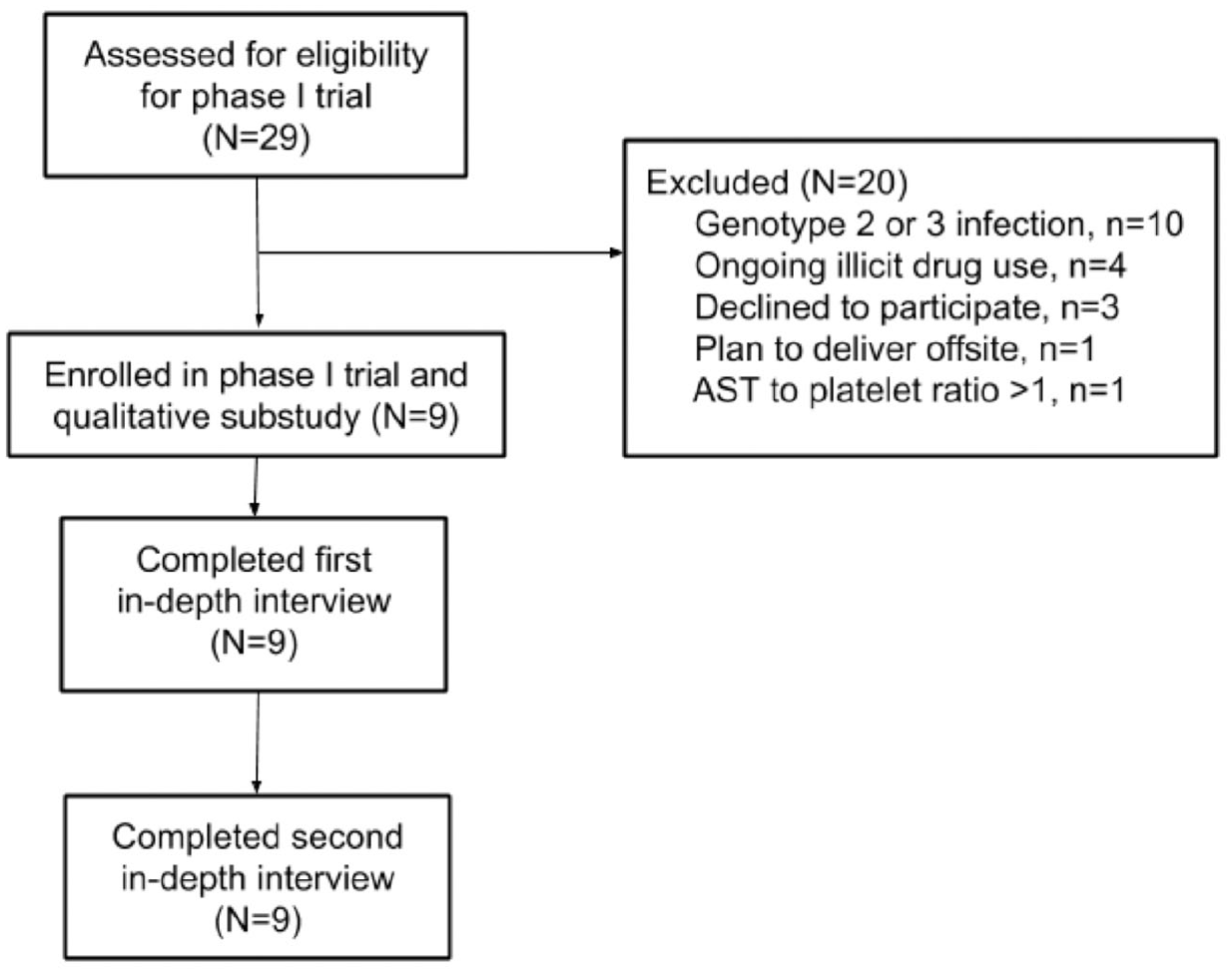
Flow diagram of participant inclusion and participation. For full inclusion/exclusion criteria and phase 1 open-label trial methodology, please see Chappell et al. (2020).
Demographic characteristics.
This qualitative analysis of the 18 transcripts focused on the components of the interview where participants discussed reasons for participating in research during pregnancy and assessment of personal and fetal risks. We analyzed all 18 transcripts and noted thematic saturation after we coded the first 10 transcripts. We used inductive analysis and identified three themes for discussion. We identified the following two themes regarding motives for participation, (1) Women conceptualized study participation as part of the caregiving role they associate with motherhood; participating was viewed as an act of caregiving for their infants, their families, themselves, and other pregnant women with chronic HCV. (2) Women also noted that they faced multiple barriers to treatment prior to pregnancy that created a desire to receive therapy through trial participation. We identified a third theme regarding risk perception, (3) Women acknowledged personal and fetal risk associated with participation. Acceptance of risk was influenced by women’s concepts of motherhood, preexisting knowledge of HCV and medical research, family members, intimate partners, or by the study design.
Theme 1. Women conceptualized study participation as part of the caregiving role they associate with motherhood; participating was viewed as an act of caregiving for their infants, their families, themselves, and other pregnant women with chronic HCV.
Most women described how their belief in the role of the mother and caregiver prompted their participation in the study. They described that pregnancy uniquely altered their perception of chronic HCV and highlighted their desire for cure. One woman explained that becoming pregnant changed the way she thought about her diagnosis of HCV, stating “it’s not just affecting me, it’s affecting my child.” Another woman described her beliefs about motherhood by stating, “I’m supposed to be a caretaker. . .I’m supposed to help people when they’re sick. . .and care for them, not give them things to make them sick. . .No it’s not what a mother does.” Grounded in this sense of responsibility and belief of the mother as a caretaker, women went on to discuss the desire to participate in the study to prevent fetal transmission, to improve personal health, and to create better options for all pregnant women with chronic HCV.
Most women noted that preventing vertical transmission was the primary motivator for participating in research. Women described fetal benefit in the context of avoiding negative health consequences for a baby, but also described their perceptions of motherhood and guilt related to transmission as incentives for participation. In relation to motherhood, one participant explained her participation in the study by stating she desired to avoid vertical transmission and that, “When you’re a mother your children come first period. No matter what.” Another woman described that her participation was motivated by a desire to assuage feelings of guilt given that her HCV acquisition was due to drug abuse. She stated that she was, “Doing it for him. . .And for myself, but mainly him. ‘Cause it’s not fair to him, the decisions I’ve made prior.” Interestingly, the mother of a participant who had acquired HCV through vertical transmission was also described as feeling relieved that her daughter was being treated. The participant noted, “I think [my mother] has some level of guilt about giving it to me. . .she’ll say, ‘I’m sorry that I gave that to you.’ So I think she was relieved as a mom herself, knowing that her child will not have to deal with something.” The concept of a caregiver extended to family members. Women noted that trial participation and ultimate cure would reduce their fears about transmission to family and household members for example if they experience inadvertent cuts or bleeding, or if their young toddlers accidentally bit them.
Women did discuss their own health as motives for participation, but often noted that improving their health status would allow them to better care for others. Women described hopes that the cure would give relief from immediate health issues such as fatigue, and this would let them have more time with their children. Participants also described the desire for participation to prevent future complications and anticipated a more engaged relationship with their children, “Maybe me being cured can help me have a longer relationship, a longer life with my kids. . .that could be a very good benefit.”
Many women also described care for other mothers as motivation for trial participation. Women discussed that research during pregnancy in general was important to help women make decisions regarding their own healthcare, especially because pregnancy is a unique motivator for decision making. When asked if she thought studying medications in pregnancy was important, one woman stated, “it’s pretty important because they shouldn’t just research medications on people that aren’t pregnant because I think the more we know about medications on pregnant women, the easier it’ll be for people to make decisions when the point of their life comes about when they’re about to have a child”
Other women felt that research in pregnancy was important because it would allow overall prevention of maternal to child transmission, stating, “. . .if you can prevent a mother passing any disease to the fetus, why not?” Still another woman noted that she was encouraged to participate to help other pregnant women, “I feel that. . .I helped get this treatment out there for other women.”
Theme 2. Women also noted that they faced multiple barriers to treatment prior to pregnancy that created a desire to receive therapy through trial participation.
Women discussed that preconception diagnosis and attempts to receive care for chronic HCV was stigmatized and challenging. After these prior experiences of stigma and barriers to care, women discussed that trial participation was an opportunity to get the care they felt they needed. Women described fearing that outside the study they would never be able to obtain treatment. One participant described taking study drug as a “once in a lifetime opportunity.” Women discussed preconception barriers such as diagnosis challenges, and lack of prenatal access to HCV therapy due to insurance barriers, cost of the medication, lack of cirrhosis, stigma from medical providers, and costs associated with the medication.
Women discussed that confirming diagnosis itself was difficult due to fractured communication. In Table 1, we show self-reported location of diagnosis from the enrollment questionnaire. Participants reported diagnosis happening at their primary care provider (PCP) office (n = 2, 22%), during inpatient hospitalization (n = 1, 11%), while incarcerated (n = 1, 11%), during a drug rehabilitation program (n = 4, 44%), or while attempting to donate blood (n = 1, 11%). However in the qualitative data, several participants report needing multiple steps to learn of their diagnosis. Women discussed first being tested at one location such as a health fair or at their PCP’s office and receiving no report of the results, and then learning of the diagnosis formally after being tested at a drug rehabilitation center. Another woman discussed seeking the test but never learning her results, instead finding she was positive years later after being tested by her PCP.
After learning of their diagnosis, women described feelings of disappointment and inadequate counseling regarding the diagnosis. In particular, one participant stated that they felt “devastated” after being diagnosed with chronic HCV, and like the diagnosis was a “death sentence.” Others explained that they were disappointed by their diagnosis of HCV because the diagnosis felt like a “consequence of the choices” made while using opiates, such as sharing needles. When prompted, participants also explained that they felt poorly about the diagnosis because they didn’t feel they had a lot of knowledge about HCV and did not know it was curable. Indeed, most participants described that they initially did not feel follow-up for chronic HCV was needed because healthcare providers did not convey a sense of urgency or gravity to the diagnosis. Study participants did persevere through disappointment and poor counseling to seek treatment before trial participation, however women described a continuation of barriers and stigma when they attempted to seek care.
Women described challenges to care such as insurance barriers, cost concerns, or being denied therapy based on their lack of cirrhosis. Women reported that after seeking care they found that therapy was not covered by their insurance. One woman highlighted the lack of insurance coverage and cost of the medication as a significant motivation for study participation, I would have paid the money. . . but the cost of the treatment. . .was a hundred thousand dollars. . .and I would have paid whatever portion that I could if the insurance would cover part of it. . . I was willing to make that financial investment in my own health and for the baby’s health but knowing that I didn’t have to pay for it at all is a very positive part of this experience
Others noted that following up with a specialist was not feasible often because their liver function had not deteriorated enough to be offered treatment. For example, one participant who had tried to seek treatment through a specialist explained, “. . .I had like just found out I got it and. . .my liver enzymes weren’t very high and I don’t know, he just wanted me to come back every six months. . . .and it was just not a good feeling. I thought I was gonna get it taken care of and I mean it was a lot of work, it was a lot of appointments, a lot of blood work. . . sonograms of my liver. . .it was a lot of time invested just to be told “Well, we’ll keep an eye on it.” And I mean he did say eventually yes, I would be treated, it just wasn’t gonna be as soon as I wanted.”
Another woman explained that when she was denied therapy due to lack of cirrhosis, her desire to avoid cirrhosis as a mother reinforced her desire to seek therapy, “because I was pregnant and knew I wanted to get it done and they said I wasn’t sick enough, I didn’t have enough liver damage. I knew [treatment] would not. . .be available to me for another 5 or so years, until I had liver damage and I’d be raising a kid. . . not being able to go out and do stuff with him, ‘cause I’m sick. So, I wanted to get it taken care of.”
Alongside these barriers to follow-up, stigma continued to pervade women’s experiences with chronic HCV care. Many participants revealed a history of disrespectful communication or breach of confidentiality from healthcare providers related to the diagnosis of HCV infection. One participant described her frustrations, “I’ve had some providers. . .whenever they find out that I have hepatitis. . . they’re complete jerks about it. Like they look at you like you’re the scum of the earth. . . .yeah their views on drugs are different. . .yeah we chose to do drugs and everything, but you don’t have to look at us like. . .you have some uncurable disease like the plague. . . .it’s not okay. . . . . .if somebody has a different kind of disease. . .you don’t really treat them any different. Just be nice about it. . . You’re in a field of dealing with people. Maybe you should be a people person.”
Another participant explained that she did not disclose her diagnosis to her mother, but that her mother was told while she was hospitalized which was not her choice. These acts of disrespect were communicated as barriers to care and frustrations with the healthcare system in general. With these background experiences of stigma and difficulties obtaining care as noted above, women often returned to the positive experience of the study and how glad they were to receive therapy through study participation.
Theme 3. Women’s acknowledged personal and fetal risk associated with participation. Acceptance of risk was influenced by a history of drug addiction, their concept of motherhood, preexisting knowledge of HCV and medical research, family members, intimate partners, or study design.
All women acknowledged a sense of taking on risk of personal or fetal harm due to involvement in research, but women also described that they were able to accept this risk and choose to participate in research. In describing their concerns for adverse fetal effects, most women described fear for anatomic, mental, or immunologic risks. Women described that they would feel a sense of relief after delivery, that seeing “ten fingers and ten toes,” and that “his ears are not on top of his head,” would all be reassuring. Some women described that their biggest concern about research participation was fear that this medication would not improve fetal health but may cause “another complication that potentially could be worse, like to me, a mental defect would be way worse than him being born normally but with hepatitis C.”
Despite this acknowledgment of risk, women chose to participate in the trial. Women often described accepting the risk of trial participation as an extension of their sense of motherhood, discussing the risk of fetal harm from perinatal acquisition of HCV as a greater threat than trial participation. For example, one participant described her risk acceptance in the following terms, “the way I live my life is, specially now, is the risk and reward factor. . . .does the risk outweigh the reward? Or does the award outweigh the risk? And right now the award is outweighing the risk. So that’s why I’m doing this.” Another participant explained that navigating competing risks is a part of motherhood and pregnancy, “Even if you take an antibiotic that is pregnancy approved. . .you can get a side effect from that. You can get a side effect from the nicotine patch. . .you could get a side effect from walking down the sidewalk. Who knows? Anything’s a risk and a gamble. . .you just hope for the best outcome.”
Preexisting knowledge about HCV and about medical research also factored into women’s assessment of risk for themselves or their infants. One woman discussed that she felt many people have chronic HCV and have babies and this reassured her about participation. She stated, “I would probably have some concern, but. . .they deliver babies with people with Hep C all the time. So, I’m not a worry wart. . . I know I’ll be ok no matter what.” Other women discussed their sense of fetal risk in context with their trust in medical research. For example, a participant stated, “I don’t think they would continue to do [the research] if there was a problem. . .that affected babies.” Other participants had taken part in research before, considered themselves “scientifically minded,” or had relatives that were in the medical field or had participated in research. These women stated that having these prior experiences with healthcare or research helped them to participate. One participant even described being able to discuss her research participation with her brother, “He did like a placebo for. . . a cancer medicine or something. . . and was so was super excited about it. He was like, “Oh” like it was something we could kind of share and like, “Oh, what did you do in your study?” “Oh, what did you do in yours?””
Women frequently described discussing research involvement with the father of the baby, often stating that the father had “a right to know.” One woman also discussed that the father was her “life partner. . . he gives me advice and he consoles me. . . so I need to talk to him about stuff that’s going on.” Participants discussed that their babies’ fathers were sometimes worried about adverse fetal effects from study medication, but that written materials from the study were helpful to address their fears. Other participants stated that the father was “excited,” and another participant stated that after getting involved in the study her partner then was able to get information on treatment for HCV and sought treatment himself. Some participants discussed that their intimate partners were happy that the participant would possibly have cure from HCV for her own benefit as well as reducing the chance of sexual or vertical transmission.
Women continued to assess risk during the study, and during end-of-study interviews noted that features of the study itself helped to assuage the sense of risk. This included learning that other women had completed the study with undetectable viral loads, no adverse fetal effects, and negative HCV testing for those children. Another woman explained that learning her viral load after each test was heartening, stating, “I got really good news that my levels have gone down and [the medicine is] really helping. . .it makes me feel a little bit better.”
Women reported that they would continue to assess the risk of their involvement until after study completion. During end-of-study interviews, while most women noted that if given the choice, they would participate in experimental treatment again, several women reported that they could not predict if they would choose to participate again. These women discussed that their feelings about involvement in the study would change if they were to experience adverse effects. When asked if she would choose to participate again, one woman responded, “depends on the outcome of the delivery, couldn’t answer that right now. ‘cause if I have the baby and there is something wrong with the baby, absolutely not, I wouldn’t. But if I had it and there was nothing wrong with it and everything went fine, then yeah I would do it.”
One woman explained that her assessment of the study would depend on both fetal effects and on whether or not she would be cured, “I was hoping that it didn’t affect the baby in a harmful way. . . that was my main concern. . .my other concern was, if I do take this medicine, and follow through with this program like I’m supposed to, will I be cured? . . .. And am I gonna stay cured for the rest of my life, or is it ever gonna come back?”
Discussion
Our findings identified themes related to motivation and to risk perception. In regards to motivation, our analysis shows that pregnant women with chronic HCV are willing to participate in early phase drug trials and that they anchor their participation in their concept of motherhood and a desire to prevent transmission to their loved ones, including their infants. Prior qualitative studies among pregnant women at risk for HIV infection taking investigational pre-exposure prophylaxis (PrEP) have shown similar findings, with trial participation motivated by a desire for fetal benefit (Pintye et al., 2017, Sullivan et al., 2020). Interviews among women using investigational probiotics, novel vaccines, and anti-malarial drugs also show a willingness to enter research (Ballantyne et al., 2017; Lyerly et al., 2012; Osarfo et al., 2021). Guidelines promote the responsible inclusion of pregnant people in research, and the interest and engagement in research by pregnant people shows an alignment among pregnant people with the intent of these guidelines (van der Graaf et al., 2018; World Health Organization and Council for International Organizations of Medical Sciences, 2017).
The women in our study described desire for research participation inside the context of significant structural, financial, and interpersonal barriers to accessing care for chronic HCV. Prior work among pregnant H1N1 vaccine trial participants discussed similar perceptions of enhanced opportunity to receive a beneficial therapy and a greater sense of safety through trial participation compared to routine care (Lyerly et al., 2012). Yet our work is among a less affluent community, with 80% of participants using public insurance (Lyerly et al., 2012). Indeed, our population of pregnant women show features of a vulnerable population due to the structural conditions that coexist with drug misuse and chronic HCV infection (Committee on Ethics, 2015). Participants in the trial discussed financial insecurity, poor insurance access, experiences of stigma, and difficulty accessing HCV care when not pregnant. These barriers to HCV care can also be seen as structural factors that contribute to vulnerability among the participants.
Vulnerability does not arise from pregnancy itself, but vulnerability from broader factors can coexist with a pregnant state (Committee on Ethics, 2015). The Council on International Organizations of Medical Sciences (CIOMS) provides that vulnerability should be contextualized for the individual or the group involved in research and special protections be provided for both enrollment and ongoing research participation to safeguard their rights and well-being (World Health Organization and Council for International Organizations of Medical Sciences, 2017). In regards to enrollment, protecting autonomy by avoiding coercion is crucial when conducting research among vulnerable groups. Agrawal’s discussion of research at the end of life separates vulnerability from coercion, and astutely posits that “having poor options is consistent with making a free choice” (Agrawal, 2003). Pregnant people with chronic HCV who enrolled in this trial have decreased access to care for chronic HCV when pospartum, but this situation was not created by the researchers. Although participants may face vulnerability, their inclusion in research is not unethical. Robust informed consent can ensure autonomy during enrollment for trials involving pregnant trial participants that face poor options through structural vulnerability (Fisher, 2013).
CIOMS also states that there is a responsibility to safeguard rights and well-being during ongoing research participation. Hurst states that vulnerability can be considered as “an identifiably increased likelihood of incurring additional or greater wrong,” where special protections during research are “to be defined starting from the sorts of wrongs likely to occur” (Hurst, 2008). In our study, participants may be more vulnerable after trial participation ends where they continue to face lack of access to healthcare and increased stigma from healthcare providers. However, Hurst argues that it is not the responsibility of the institutional review board (IRB) or the researcher to correct all wrongs of humanity, and would not be reasonable for researchers to be obligated to attempt to fund efforts to combat structural inequity (Hurst, 2008). Counter to the promotion of research in pregnancy, it is likely financially prohibitive and too broad in scope to uniformly require researchers to ameliorate structural inequity. However, failure to acknowledge vulnerability created by structural factors such as financial insecurity, stigma, and lack of access to care outside of pregnancy, can compound the experience of marginalization and cause participants to experience trauma. Attending to the experiences of structural vulnerability during research by acknowledging their existence, enhancing access to post-trial care through connection to community resources, and promoting efforts through national organizations can minimize the ongoing issues that participants face when research ends (Grady, 2005; van der Graaf et al., 2018; Zong, 2008).
In regards to risk perception, women discussed acknowledging that risks of experimental drug therapy were unknown and there could be harm to the infant, and went on to describe that participation outweighed their perception of possible risks. While some women clearly described uncertain fetal risk due to investigational drug use, other women did demonstrate therapeutic misconception, believing that the trial would be conducted only if fetal benefit was certain. This is similar to prior research showing some pregnant women understanding the potential for risk, while other pregnant women believed their research participation had no risk (Ballantyne et al., 2017; Kenyon et al., 2006). Qualitative work among researchers and clinicians to elucidate perspectives on research in pregnancy revealed that professionals resist choosing either maternal or fetal safety as paramount and acknowledge that maternal and fetal risks are intertwined (Beima-Sofie et al., 2019). Professionals preferred trials with low fetal risk and high maternal benefit, but would be more amenable to trials with higher fetal risk if the maternal disease was severe. Similarly, women in our study discussed that their perception of the trial would be dramatically altered if they were to have infants with serious adverse events, especially given that the risks of chronic HCV were believed to be overall low by comparison to adverse fetal outcomes like mental deficits.
Prior studies have demonstrated a placebo arm may also reduce willingness of pregnant people to enter a trial, but this phase 1 study did not include placebo and participants did not discuss that potential (Ballantyne et al., 2017). Our findings did show that participants discussed trial participation with their partners, but did not indicate that partner’s agreement to trial participation was necessary for their enrollment. Prior work among pregnant women living with HIV demonstrate heterogenous opinions where some women desired partner participation and consent in order to safeguard against fetal risks during trial participation while others desire that pregnant people should be able to decide alone (Raciti et al., 2021). When vertical transmission or exposure of the fetus to investigational drug therapy is possible as in the case trials among people with HIV and chronic HCV, researchers should consider the role of partners in consent. While partner consent may not be necessary for informed consent, researchers should consider that participants may value discussing the trial with their partner and should support the pregnant persons’ ability to do so.
Our study was limited by sample homogeneity, all participants were white and identified as female. This is likely due to the demographics of HCV infection in our region, with greater than 90% of pregnant women with chronic HCV identifying as white (Chappell et al., 2018). The other challenges are selection and subject bias. Regarding selection bias, our cohort may represent a small subset of highly motivated participants who may weigh their risks and benefits of trial participation differently than others. A recent survey among women with chronic HCV demonstrated that women may be interested in DAA therapy in pregnancy, but that cohort had a median age of 60 and did not include any women who were pregnant (Kushner et al., 2018). Thus, our work among pregnant women may be more representative of the desires of reproductive-aged women with chronic HCV. We did not attempt to conduct qualitative interviews among the three women who declined investigational drug therapy, and so cannot report the motivations or risk perceptions among women who decline investigational trial participation during pregnancy. For subject bias, when asked regarding concerns for fetal safety or motivation for participation, women may feel pressured to discuss their infant as their primary priority and so may be less likely to reveal personal motivations for study participation or stronger fears for fetal safety. Subject bias appears to have minimally impacted this group, given that most women did discuss their hopes for personal benefit and also named fears for fetal safety. Additionally, study compensation may have influenced women’s motivations for trial participation, although our participants did not discuss financial motivations during the interview. The amount of compensation was not considered coercive given the 13-hour duration of pharmacokinetic visits. As well, in the face of structural barriers such as poor access to prenatal care or extreme cost of DAA outside of the trial, too low a financial compensation may be coercive (Fisher, 2013). Fair compensation may be higher for those with higher burdens due to structural inequity and experiences of poverty (Persad et al., 2019). Lastly, this qualitative work was situated within a pharmacokinetic trial, and so a priori sample size was set by pharmacokinetic considerations. Qualitative methodology typically seeks thematic saturation to determine sample size, but this was not evaluated during interview collection. However, during analysis of the 18 transcripts, we did see thematic saturation after analyzing the first 10 transcripts and so we feel the findings have qualitative integrity.
Conclusion
Our study is the first to address motivations and risk assessment among pregnant women with chronic HCV participating in a trial for investigational LDV/SOF therapy. Our findings reveal participants’ nuanced risk assessments for their own health and fetal health and describe significant hope for cure and actualization of their ideal of motherhood by participating in the trial. These findings are within a United States cohort of pregnant women and can guide future research among pregnant women with chronic HCV and pregnant women in general. To continue to understand the experience of pregnant trial participants with chronic HCV, further work is needed to identify experiences among people of color, gender non-binary individuals, and among larger cohorts. Pregnant people deserve robust data to elucidate potential risks and benefits to therapy during pregnancy. By acknowledging that there are structural factors that might enhance research participation, that participants may wish to discuss the trial with their partners, continuing with enrollment after full informed consent, and considering options for post-trial access to care, researchers can promote appropriate research involving pregnant people who face structural vulnerabilities.
Footnotes
Author note
Yasaswi Kislovskiy is now affiliated to Department of OB/Gyn, Allegheny Health Network, USA.
Funding
All articles in Research Ethics are published as open access. There are no submission charges and no Article Processing Charges as these are fully funded by institutions through Knowledge Unlatched, resulting in no direct charge to authors. For more information about Knowledge Unlatched please see here: ![]()
This work was funded by the National Institutes of Health/Eunice Kennedy Shriver National Institute of Child Health and Human Development (1R21HD089456-01), the National Institutes of Health/Office of Research on Women’s Health (5K12HD43441-14), and Gilead Sciences (CO-US-337-2117). Gilead provided the study drug, performed pharmacokinetic assays in their commercial laboratory, and provided consultation on the phase 1 trial study design. The National Institutes of Health (NIH) had no role in the study design, collection, analysis, interpretation of data. The NIH and Gilead had no role in the writing of the report, or the decision to submit the report for publication. Catherine Chappell and Yasaswi Kislovskiy have served as consultants for Gilead Sciences. Elizabeth Krans and Catherine Chappell are receiving research funding from Merck.
