Abstract
It is becoming widely recognized that dissemination of research results to participants is an important action for the conclusion of a research study. Most research institutions have standardized consent documents or templates that they require their researchers to use. Consent forms are an ideal place to indicate that results of research will be provided to participants, and the practice of inserting statements to this effect is becoming more conventional. In order to determine the acceptance of this practice across Canada we conducted an assessment of 121 institutional consent document templates from 65 institutions (hospitals and universities) looking for language that endorsed results dissemination to participants. About half (51%) of the documents we examined had language included which stated that results should be made available. In an era where research participation in hospital settings and universities is becoming ubiquitous there should be a reciprocal expectation that results should be provided. The success of research should be measured in part by its accessibility and dissemination to all stakeholders.
Keywords
Introduction: Background
In the modern world of research, the participant is enjoying a newfound position of interest as researchers and research regulatory bodies recognize the benefits of engaging participants in more research processes. The purpose behind this push seems legitimately well intentioned, and both researchers and participants laud the efforts to make research more accessible and inclusive. One aspect of making research more participant-centred is the dissemination of research results to participants. There is a growing literature on this topic, including discussions of various considerations that should be made (Miller, 2008; Thorogood et al., 2014; MacNeil & Fernandez 2006b), and recommendations for various methods of participant feedback (Bell et al., 2004; Dixon-Woods et al., 2011; Haines et al., 2011; Partridge et al., 2005; Sood et al., 2009). However, much research is still conducted as if researchers are largely absolved from providing the participant with any feedback (Di Blasi et al., 2002; Rigby and Fernandez, 2005), even though both participants and researchers generally believe that provision of results is appropriate (Cox et al., 2011; Dixon Woods et al., 2006; Ferris and Sass-Kortsak, 2011; Partridge et al., 2003). Research institutions such as universities and hospitals which are equipped with institutional ethics review boards (IRBs) can help standardize expectations for research behaviours. Promotion of participant engagement through the provision of results can be part of the informed consent process, which falls under IRB purview. If institutions do not require researchers to discuss the provision of results in consent forms or in the consent process, this constitutes a missed opportunity to promote a just and fundamental aspect of participant engagement.
The Declaration of Helsinki (2013), Section 26, states, ‘All medical research subjects should be given the option of being informed about the general outcome and results of the study.’ Further, Section 36 indicates, ‘Researchers have a duty to make publicly available the results of their research on human subjects and are accountable for the completeness and accuracy of their reports.’ The Tri Council Policy Statement II (TCPS 2), which most researchers in Canada adhere to as a research guidance document, indicates in section 3.2f) that researchers should describe in the consent process ‘the measures to be undertaken for dissemination of research results …’ on the premise that participants should have a reasonable expectation that results will be made public in some way. Chapter 4 of the same document also notes that ‘… researchers should ensure that participating individuals, groups and communities are informed of how to access the results of the research’, and goes on to recommend formats for results to be provided. These influential documents are a baseline for what participants should expect following their participation in a research project, and they provide succinct guidance to researchers on expectations for distributing results.
Flaskerud and Anderson (1999) discuss community-based participatory research (CBPR) which centres on community participation in interpretation, analysis and dissemination of results to both participants and the communities where research is conducted. Likewise, Shippee et al. (2015) and Domecq et al. (2014) discuss what they term patient and service user engagement (PSUE) in biomedical and health services research, where dissemination to participants and lay persons is an integral component to research. Most research studies are not designed with the level of participant-focused engagement described in these articles; however, the concept of the participant as being elevated from simply an object of interest in research studies is emphasized in their work.
Longitudinal research projects rely on continued interest on the part of participants over years or decades. McCarty et al. (2011) looked at a Personalized Medicine Research Project (PMRP) where more than 20,000 participants contributed data to a biobank. No individual data were given to participants but they did receive a study newsletter two or three times a year. The project was designed to last decades, and therefore regular communication with participants was necessary to maintain awareness of the study and trust in the investigators. Feedback from the participants to researchers regarding content of the newsletters requested explanations of how research results might change healthcare and for context of the usefulness of the research nationally and globally. People who provide information or physical samples to a biobank which is designed for data to be anonymous may not be able to expect results from trials that their data might specifically have been used for. However, such large depositories could easily establish a website that could provide information on studies that are currently ongoing and summaries of completed studies that have utilized the data. The Ontario Health Study (2017), for example, has provided a ‘research’ tab on their website that is open to the public, which provides direct links to published articles, both scientific and media release, for some of the results for studies which have used information from their data.
Participants surveyed by Beskow and Dean (2008) raise the idea of researchers offering information about the research as fair exchange for the participants providing their data in the first place, especially in the case of biobanks, where there is rarely a direct benefit to participation. The concept of reciprocity is also discussed by Lévesque (2011) as important for upholding the ethical principle of justice in relation to provision of feedback to participants. If participants are going to provide their time, biologic samples, test outcomes etc. for research purposes, the knowledge gained from the research should be translated back to them.
Participants and researchers seem to be both largely agreeable to the concept of providing research results, and therefore the formal statement of such provision should be consistent in the devices which engage participants in studies in the first place – informed consent documents. It is not clear how frequently these documents communicate that research results will be made available to the participant. Hospitals and universities tend to be the institutions which support the bulk of publicly funded research and therefore would be most likely to have publicly available templates/documents for consent form construction. The objective of this article is to examine how well Canadian institutions support the provision of study results to participants in their informed consent templates and what they are suggesting to be appropriate statements about the receipt of research results.
Methods
Consent form templates, guidelines and checklists for Canadian universities and teaching hospitals were collected through an online search using the website http://www.researchinfosource.com/ (last accessed 20 August 2017) as a main source, as it had updated lists of the most recognized research institutions in Canada. It was felt that these institutions would be most likely to have publicly available ethics websites with documents for viewing. The top 40 research universities and the top 20 research hospitals listed for 2015 and 2016 on this site were searched online. Some institutions redirected to other institutions or entities for research consent forms; St. Josephs’ Health Care and Hamilton Health Sciences, for example, both redirect to the Hamilton Integrated Ethics Board. Other institutions referred to the Tri-Council Policy Statement for consent development (Laurentian, Lakehead and University of New Brunswick). The team felt it was important that there be representation from all provinces; therefore, as some provinces did not have an institution represented on these lists, searches were made specifically for institutions in the following provinces: Newfoundland, Manitoba, Yukon and the northern territories. Nunavut does not have a University, although Arctic College in Nunavut has partnerships with Dalhousie and with Carleton Universities. NorthWest Territories does not have a university but does have Aurora College, which serves the entire territory so the team considered it appropriate to review the documents for this institution as representing this territory. North West Territories did not have a hospital with a webpage that provided consent form templates. Some large agencies that are known to review research (e.g. Cancer Care Ontario) were also included in the search as it was decided that such organizations do have authority in certain contexts to review consent documents, and they did offer templates to be used.
Documents were obtained by searching the website of each institution/agency. By using the keywords ‘research ethics’ in a search engine such as Google, with the university or hospital name, institutional ethics webpages with consent documents available for download usually came up immediately. Otherwise, a search term of ‘research ethics’ was entered in the search field of the main institutional website, and consent documents were found through this link. Documents were reviewed for language that indicated that participants could, should or must have results provided to them. Since we did not expect any specific standard text to exist across institutions, it was left up to the reviewers to interpret whether the documents had language that was adequate to represent the idea of providing results to participants. For example, Nova Scotia Health Authority states, ‘Briefly describe the participant’s access to the study results when the study is completed.’ L’Hôpital de Montréal pour enfants indicates, ‘You may ask to receive a copy of the results of this research project; these will only be available after the entire project has been completed.’ Data were entered into an Excel spreadsheet using columns of ‘yes’ or ‘no’ to indicate the existence of appropriate text. Most of the documents stated in their title what type of document they were, and this was recorded as well as the actual statements related to provision of results.
Results
In total, 121 documents were reviewed by two reviewers (DC and MK); 79 documents from University websites (Table 1) and 42 documents from healthcare institutions/agencies (Table 2). Of the documents reviewed, 93 were defined as templates/samples, 14 were checklists and 14 were guidance documents. There were 7 university and 5 health institution documents which were in conflict for the two reviewers (DC and MK), therefore they were provided to the 3rd author (TF) for resolution.
Summary of university documents reviewed.
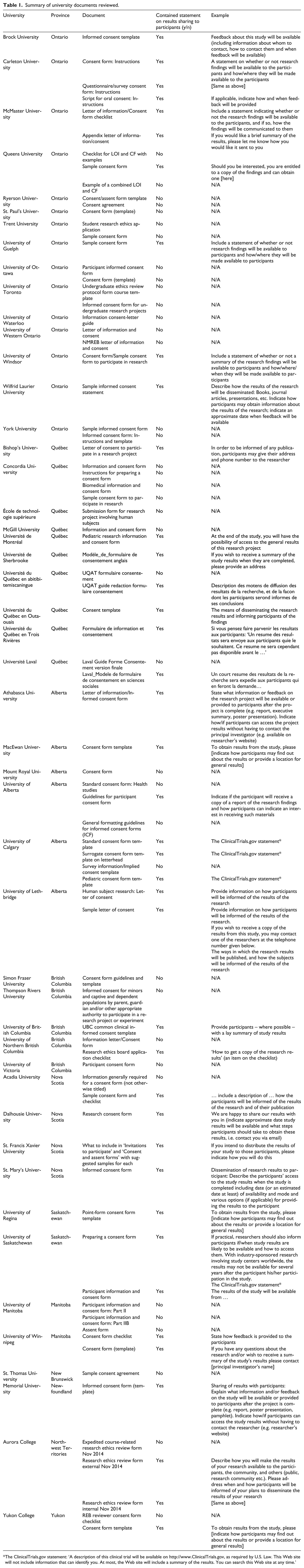
The ClinicalTrials.gov statement: ‘A description of this clinical trial will be available on http://www.ClinicalTrials.gov, as required by U.S. Law. This Web site will not include information that can identify you. At most, the Web site will include a summary of the results. You can search this Web site at any time.’
Summary of healthcare institution/agency documents reviewed.
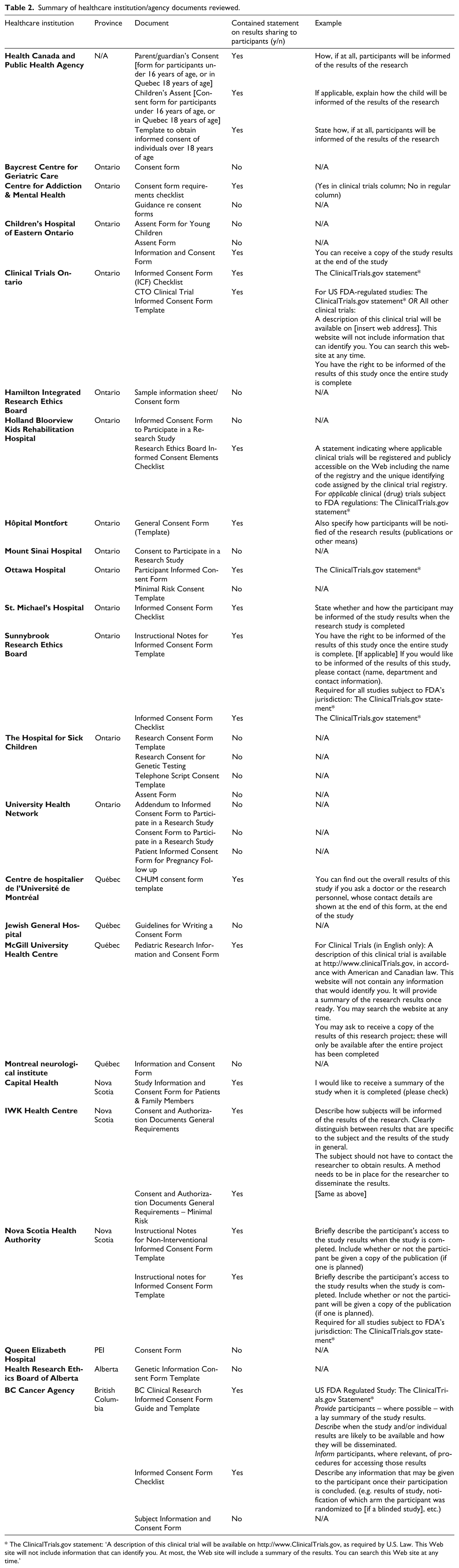
The ClinicalTrials.gov statement: ‘A description of this clinical trial will be available on http://www.ClinicalTrials.gov, as required by U.S. Law. This Web site will not include information that can identify you. At most, the Web site will include a summary of the results. You can search this Web site at any time.’
Of the total documents reviewed, 59 (49%) did not have any standard statements or wording that participants should be provided with study results. Universities and healthcare institutions were very similar in terms of whether or not their documents used language that required participants to receive results (Table 3), with 39 (49%) documents from universities and 20 (48%) documents from healthcare institutions not having any language discussing provision of results to participants. When provision of results was indicated, the most consistent language used was the ClinicalTrials.gov statement. In 16 (13%) of the total documents reviewed, institutions required researchers to use the following standard descriptor from the US Department of Health and Human Services, Food and Drug Administration (February 2012), regardless of whether or not the research would be performed in the United States or supported by a US funding agency:
Institution type and presence of statement in consent document.
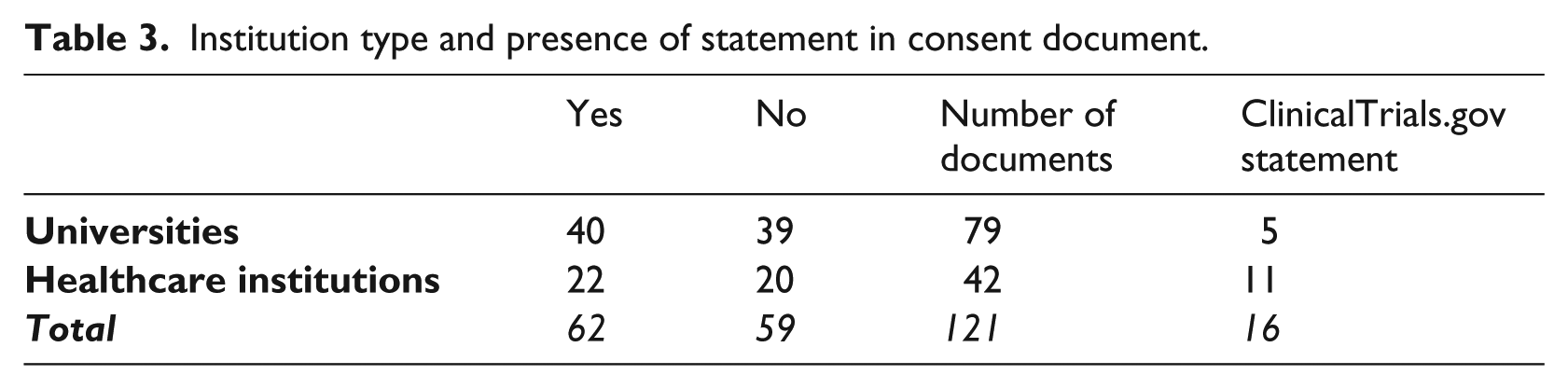
A description of this clinical trial will be available on http://www.ClinicalTrials.gov, as required by U.S. Law. This Web site will not include information that can identify you. At most, the Web site will include a summary of the results. You can search this Web site at any time.
The majority of these institutions were healthcare institutions, which may be more likely to conduct clinical research projects than universities.
Discussion
Overall, our findings indicate that the provision of research results to participants is not consistently endorsed by universities and research institutions that actively support research. Despite the endorsement of major stakeholders such as journals (who promote the registration of trials on recognized websites) and the continued updates of national and international guidance documents such as the TCPSII and the Declaration of Helsinki, it is unfortunate that consistency does not exist at the institutional level. Inconsistency in statements related to results sharing was also problematic in documents within institutions: some institutions provide recommendations in one documentation source but not in another. For example, Yukon College posted a Research Ethics Board Checklist which has no item listed for participants to be provided with information on the results of the study; however, the Sample Informed Consent document from the same institution does have a statement for researchers to include on how participants can find out the results, or directing them to a location for general results. It is important for researchers to be presented with consistent information regarding what should be in a consent form, and people who are responsible for reviewing consent documents for the institution should have the same understanding as the researcher. A previous study by MacNeil and Fernandez (2006a) looked at Canadian university research ethics boards policies on returning research results, and found that most of the REBs that responded to their survey (9/22 institutions) did not have a policy that governed return of results to participants. Though their sample was small, it indicated a lack of support at the time for returning research results. Our project reveals a continuing lack of support for return of research results from a much larger cohort of research institutions.
Given that consent forms represent the institutional support of the project, as they have been vetted by an REB directly affiliated with the institution, it is important that such documents reflect expectations of best practice for researchers. Consent forms are also provided to the participants so that they know what to expect from the research process and the research team, which makes it a logical place to state that results will be provided at a future point. In a survey by Cox et al. (2011), patient respondents indicated stronger agreement with the idea of a consent process around the offer of trial results for cancer research than researchers did. In this same study, researchers indicated that introducing the option of feedback during the consent process would help to facilitate the practice of offering feedback in general.
Feedback to research participants can take various forms, including assessment feedback, general results, incidental results and individual research results, and research projects can offer one or a combination of results to participants. Most projects offer incidental findings under the ethical principles of beneficence and justice, as such findings may have direct consequences for participants; most consent forms reviewed in this current project had standard language regarding provision of these kinds of results for interventional research. This is appropriate, as many clinicians juggle dual roles as both healthcare providers and researchers, and thus engage their patients in clinical trials to either supplement or replace standard care, especially in the field of oncology. However, there is a real conflict of interest for a clinician who is also a researcher who must ensure that the care of their patient is prioritized while ensuring that the integrity of a clinical trial is not compromised. It is inarguable that there is an ethical responsibility for the clinician to the study participant who is also their patient to give feedback should the patient’s care be impacted by participation in a research study. This is because professional codes of ethics put the patient at the focal point of clinical care, and it is the duty of a clinician to treat a patient with that person’s best interest in mind. However, it can also be argued that, although the clinical importance of certain specific individual information is imperative to the patient, the communication of general research results is also important because it respects the patient in the role of the study participant; the role of the clinician/researcher must accommodate the communication of both types of information. Most people come to health research through a clinical interaction; the patient sees the clinician because they are the person who will help them with their health problem, not because the clinician is a researcher. The first relationship is what the patient and clinician accept first; the research is a second relationship which may overlap the first, and this then has implications for duty and obligations, including disclosure of results of a study. The natural expansion of providing patients with appropriate clinical knowledge about their condition or issue and following up with them is to also provide research knowledge related to a patient’s participation in research, especially if the clinician is directly involved in the research. With the modern overlap of the clinician/researcher role, the clinician should acknowledge and be prepared to act on similar responsibilities of both roles.
Many participants prefer to have individual research results (Dorsey et al., 2008), which may or may not be reasonable from the perspective of the research design; studies do not necessarily have resources to produce individual results, or the project may not be designed to elucidate such results.
There is some debate regarding what the actual provision of results to participants should look like. Miller et al. (2008) discuss the complexities of providing results in the context of different types of research (e.g. interventional, non-interventional) and the options for results to be communicated (e.g. aggregate results vs individual results). Partridge and Winer (2002) and Bell et al. (2004) also discuss the obligations of researchers to provide results in a form that participants can understand or that is relevant to them. As there is growing pressure to post research results on publicly accessible databases, the provision of general results regarding the primary outcomes of a research project should be more readily available to participants. Indeed, ClinicalTrials.gov is one of the favoured mechanisms for research dissemination noted in the consent forms we reviewed, and although we did accept this as meeting a basic requirement for provision of research results, it turns out that this website does not require lay summaries of results to be provided on project completion, and by the end of 2016 only about 10 percent of over 200,000 registered protocols had results posted at all (Zarin et al., 2016). Thus, providing a weblink to a public registry is clearly not the same as a statement committing to providing results directly to a participant. The evolution of such public databases to require lay person summaries is a logical next step which would mitigate not only the issue with lack of dissemination but also concerns that researchers might have regarding costs of dissemination as well, as these databases are provided and maintained by stakeholder organizations and not the researchers themselves.
Although ClinicalTrials.gov and several other clinical trials websites have been established to improve transparency in clinical research, it is not only clinical trials but also behavioural and non-interventional research that should provide results. The lack of provision of results is not just a medical research issue but also a general research issue (Fernandez et al., 2003; Ferris and Sass-Kortsak, 2011), and all consent forms should be written to ensure dissemination to participants.
Funding bodies such as the Government of Canada’s Canadian Institutes of Health Research (CIHR), the Natural Sciences and Engineering Research Council (NSERC), and the Social Sciences and Humanities Research Council (SSHRC) fund numerous studies across Canada annually. In 2015, these bodies updated their guidance documents to ensure that researchers would build provision of results into their project protocols, stating that research results should be ‘… as widely available and accessible as possible …’, http://www.science.gc.ca/default.asp?Lang=En&n=9990CB6B-1 (accessed 17 September 2017). Many researchers use funds for project results dissemination exclusively to attend conferences which are attended by their peers and not frequently attended by the public, thus limiting the dissemination of results. Other options that researchers utilize dissemination funds for include publication in open-access journals. However, Fernandez et al. (2003) argue that referring research participants to results published in literature is not enough to meet a requirement of providing results to participants.
Encouraging the planning of dissemination of results to participants and making the costs of dissemination part of the research design from the beginning could enhance ease of results provision to participants (Fernandez et al., 2004; Shalowitz and Miller, 2005). As a simple example, every scientific article is required to have an abstract, and many of these would likely be written in such a way as to be accessible to lay people. Expanding an abstract to be a summary-type document for research participants should not be overly burdensome, as the researcher is frequently required to produce a summary document for different contexts, including peers, funding agencies or stakeholders. Many people now have access to email and the internet, which also makes communication to large groups of people easier and more cost-effective than historical research cohorts would have experienced.
Conclusions
Research institutions have a responsibility to ensure that research participants are treated justly and respectfully, and this includes promoting the provision of research results in an appropriate manner. Many institutions fail to encourage this through the most accessible venue – the consent form. Other institutions, while stating that participants can access study results, refer participants to websites that frequently do not have results posted. This is a gap in participant engagement that should be remedied; as researcher expectations of the general populace to engage in research activities continue to rise, so too should participant expectations of researcher and research institution engagement in dissemination of results.
Limitations
There may well be additional documents related to consent that our search did not detect. In addition, we cannot account for any updates or other modifications to documents analysed after the review was completed.
Future directions
It is unclear whether other countries might be experiencing this same issue with provision of research feedback to participants; a future study might look at consent forms in other countries to determine how likely they are to provide statements for participant feedback. It is possible that ‘tokenism’ is also an issue whereby participants might be told that they will be provided an opportunity to receive information in the consent form, but the process for providing results for a particular study is not robust or may be so complex as to be inaccessible to the average research participant. Determining how many researchers actually adhere to the statement in their institution’s standard consent form to provide results would also be an appropriate next step for study.
Footnotes
Acknowledgements
The authors would like to acknowledge the invaluable assistance of both Kathleen Hoy for her help in sourcing the original consent documents, and Lauren Brandys for her help in sourcing consent documents, creating the institutional tables and helping to format the document.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
All articles in Research Ethics are published as open access. There are no submission charges and no Article Processing Charges as these are fully funded by institutions through Knowledge Unlatched, resulting in no direct charge to authors. For more information about Knowledge Unlatched please see here: ![]()
