Abstract
Improving the informed consent process is a common theme in literature regarding biomedical human subjects research. Standards for appropriate language and required information have undergone scrutiny and evolved over time. One response to the call for improvement is the provision and use of informed consent templates (ICTs) to ensure that documents have a standardized format and quality of content. Little is known, however, about the prevalence of such ICTs or their effectiveness. This article discusses the rationale for creating and using templates, describes the prevalence of and commonalities between templates, and identifies the need for an evaluation of their effectiveness in terms of their ethical and practical implications. The websites of 144 Association of American Medical Colleges (AAMC)-accredited institutions in the United States, 21 institutes in the National Institutes of Health (NIH), and the World Health Organization (WHO) were searched for the presence of ICTs. A total of 105 (72.9%) medical schools, 3 (14.2%) NIH institutes, and the WHO had at least one ICT publicly available on their websites. The templates varied in format, length, style, language, and sections included. The prevalence of ICTs, variability in their content, and lack of published research regarding their effectiveness suggest that ICTs are available, but more research into their effectiveness and standardization of their development are needed.
Keywords
Introduction
Since the Nuremberg Code of 1947, the process of informed consent has been an important ethical and legal standard by which research involving human subjects must abide. This process involves communication between researchers, subjects, and institutional review boards (IRBs) in order to explain the study, discuss expectations and requirements of participation, present the risks and benefits, and protect the autonomy of individual participants. The history of the development of research protections, from the Nuremberg Code to the Belmont Report and beyond, has highlighted the imperative for the informed consent process to be continually evaluated and updated to meet the standards of contemporary times. Currently the US Department of Health & Human Services Office for Human Research Protections sets the legal standards and guidelines for the informed consent process, particularly in Title 45 sections 46.116 and 46.117 of the Code of Federal Regulations (US DHSS, 2010). To this end, higher education medical institutions throughout the United States have developed and used informed consent templates (ICTs). Because we found no previous surveys of the availability of informed consent templates, the purposes of this article are to discuss the rationale for creating and using templates, describe the prevalence of and commonalities between templates, and identify the need for an evaluation of their effectiveness in terms of their ethical and practical implications.
Methods
Internet websites from three sources were searched to assess the prevalence of ICTs currently used for biomedical research with adult participants in the United States: US medical schools accredited by the Association of American Medical Colleges (AAMC), institutes within the National Institutes of Health (NIH), and the World Health Organization (WHO).
Inclusion criteria: Operational definition of an informed consent template
Any document referring to informed consent was included as an ICT if it was explicitly labeled as such or if it was a pre-formatted document that could be directly adapted or edited, resulting in an informed consent form ready for IRB submission. Excluded were documents that provided instructions, guidelines, or requirements for the consent process, but still required a researcher to build a consent form in a separate document. Only ICTs that referred to written consent for adults participating in medical/biomedical/clinical research were included.
Identifying informed consent templates at AAMC institutions
The initial list of AAMC accredited institutions was obtained from the medical school directory on the AAMC website at: https://members.aamc.org/eweb/DynamicPage.aspx?site=AAMC&webcode=AAMCOrgSearchResult&orgtype=Medical%20School. All US schools, including Puerto Rico, were included. Each medical school’s website was searched between March and July 2015 for any document that met our inclusion criteria. If no ICTs were found, a Google search was conducted using the school’s name and terms such as “informed consent template” and “biomedical research consent template.” If both the manual search and Google search failed to produce a location for ICTs, then the search concluded at the page most likely to house ICTs when compared to similar manual searches. The actual search terms used and/or links followed to locate the ICTs from the medical school’s homepage are given in Appendix 1 (online).
Identifying informed consent templates from the World Health Organization and NIH institutes
A search of the homepage of WHO, http://www.who.int/en/, was conducted in July 2015 using the term “informed consent templates,” and appropriate links were followed. Similar searches were conducted on the websites of each NIH Institute. Names of affiliated institutes were found by selecting the “Institutes at NIH” tab on the NIH’s homepage, which led to their “Institutes, Centers, and Offices” page at http://www.nih.gov/icd/. See Appendix 2 (online) for search terms used and/or links followed to locate the ICTs.
Data extraction and limitations
Data extracted included the homepage/origin link, the name and location of the institution, availability of ICTs, and, if available, a description of the online pathway to the ICTs, the final website link, the effective date or date of latest update, and comments. When no ICTs were found, the information described what steps were taken to conclude the search.
There are several limitations to this study. The survey was limited to institutions within the United States, we excluded ICTs offered in submission portals requiring a login, and the focus of the survey was limited to an assessment of the prevalence of ICT, not on the comprehensiveness, types or quality of the content of the forms.
Results
Of the 144 medical schools that met the inclusion criteria, 105 (72.9%) had at least one template for informed consent in biomedical human subjects research publicly available. Ten (6.9%) schools provided information that ICTs were available but required a login to access them, and for 29 schools (20.1%) we were unable to locate an ICT (Figure 1). Websites that included ICTs for biomedical research often provided templates for verbal consent documentation, assent documentation, and/or ICTs for social/behavioral research. Schools that used an interactive online portal for human subjects research submissions such as IRBNet, eIRB, or WIRB varied in whether or not they had information about ICTs. Some had ICTs that were accessible outside the online portal, some described that they had ICTs available within the portal, and others did not mention what resources were available within the portal. It was unclear whether these sites provided an interactive format for consent building rather than a template, or if the interactive format was preferred by researchers. Several schools listed links for ICTs which led directly to the portal log-in site or were no longer active.
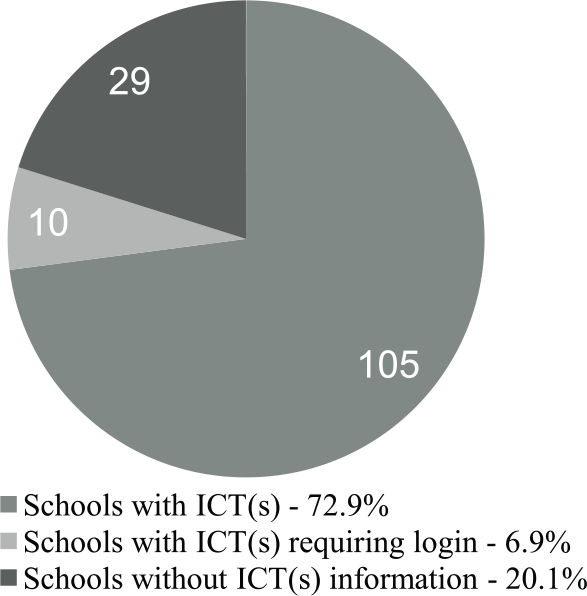
Prevalence of publicly available ICT(s) at AAMC institutions.
The templates varied in location, format, length, style, language, and sections included (Appendix 1, online). Common included sections referred to study title, introduction, study purpose, study procedures, potential risks of participation, potential benefits of participation, alternatives to study participation, voluntariness, withdrawal, confidentiality, conflict of interests, contact information for questions or concerns, and a consent statement involving signatures. Some institutions provided additional guidance and language documents, whereas others did not. There was also wide variation in the attention paid to readability. Some specifically described a standard of 6th or 8th grade reading level, some encouraged the use of lay terminology, others provided guiding terminology to choose from, and some reminded researchers to use language that would explain medical terminology. The offices that housed the links to ICTs or similar research-related templates at AAMC institutions varied in their names, including but not limited to: Institutional Review Board, Office of Research Integrity, Office for the Responsible Conduct of Research, Office of Human Subjects Research Protections Programs, and Office of Research Services. When updated/effective dates were available for either the medical school’s ICTs or the webpage hosting the ICTs, they ranged from 2008 to 2015.
The WHO provided several ICTs for research involving human subjects. At least one ICT from three NIH institutes for biomedical human subjects research were publicly available. One institute provided a template for documenting verbal consent, one provided a template for developing a research protocol, and one provided a guideline/template for developing a multi-site manual of operating procedures (Figure 2). Institutes that provided ICTs for biomedical research often provided protocol templates, other consent/assent documentation tools, and/or forms for data safety and monitoring. ICTs varied in page length and primarily used a delete-and-rewrite method. When updated/effective dates were available for either the WHO/NIH ICTs or the webpage hosting the ICTs, they ranged from 2013 to 2015. Appendix 2 (online) summarizes components of ICTs from WHO and NIH.
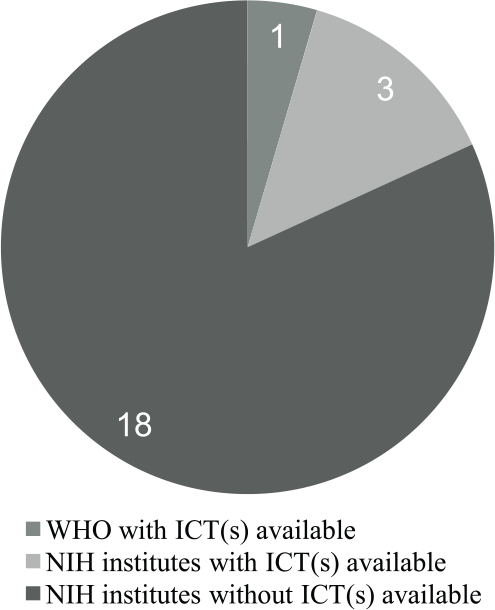
Prevalence of publicly available ICT(s) at WHO and NIH institutes.
Discussion
The Organisation for Economic Co-operation and Development (OECD), a forum of 30 countries which address a number of global challenges, has published Guidelines on Human Biobanks and Genetic Research Databases (www.oecd.org/sti/biotechnology/hbgrd) which provide extensive and clear guidelines for obtaining informed consent and suggest specific information that should be included in consent forms. To our knowledge, however, this is the first survey of the prevalence and content of ICTs available for biomedical human subjects research within the United States. Although providing templates is a first step, it is important that the application and effectiveness of these documents be evaluated and updated as needed. Harvie et al. (2012) examined variability in the review processes of IRBs involved in multi-center minimal-risk studies and found that differing IRB requirements for standard formatting and language contributed to the need for revision in 86% of consent documents prior to IRB submission and 71% needing revision after submission. They suggested that standardized protocols may reduce variability, improve expediency, and still maintain high levels of research protection for participants.
In a cross-sectional survey of intensive care unit administrators and research coordinators to identify barriers to research involving critically ill patients during pandemics, Burns et al. (2013) found that respondents expressed a need for pre-approved template protocols and consent forms as well as a rapid approval process, and that such resources would be a positive influence in their decision to conduct such research. These findings suggest that a lack of guidance and resources for researchers may be a barrier to the conduct of research. Kelleher et al. (2013) evaluated the impact of a standard application form by assessing the review process 6 months prior to the adoption of the new form and 6 months after its adoption. They suggested that an ICT may decrease the need for post-submission revisions.
In order to meet ethical and legal standards, information regarding the research needs to be communicated in a way that is understandable by subjects. To this end, Nolan et al. (1999) suggested that providing a consent template can improve understandability and ensure that all pertinent information is included. Similarly, Reiman (2013) suggested that ‘standardized risk statement’ templates may also accomplish this, while also helping IRB members and clinical study coordinators who may not have detailed technical knowledge regarding some components of the research protocol.
Need for evaluation
Denzen et al. (2012) assessed whether ICTs developed by the National Cancer Institute and the National Heart Blood and Lung Institute increased patient comprehension of information. They found that the reading level of the consent documents was consistently higher than the reading level of the average person in the United States. This demonstrates the importance of not only developing, but also evaluating and revising ICTs.
ICTs should be evaluated within the research community for which they are created. This survey was limited to institutions within the United States and clearly should be expanded to other nations with sensitivity to local research cultures. Indeed, the need for improvement of consent documents in terms of content, comprehensibility, and understandability has been recognized in other countries such as India and Canada (Brehaut et al., 2012; Padhy et al., 2011). Survey and interview methods can be used to determine the current landscape of knowledge within the community, what difficulties have arisen, and what methods have worked well. After they are approved, ICTs should be periodically evaluated and updated to comply with advancements in research practices and address suggestions or concerns provided by the researcher and participant users. Follow-up evaluation of the ICTs should include examining the format and accessibility of the ICT, impact of the ICT on the submission and review process, and its effectiveness in terms of research participants’ understanding of the research. Evaluative measures will be most useful, in terms of practicality and ethics, when they take into consideration participant, researcher, and IRB perspectives.
Some ethical and practical implications
An ethical dilemma associated with informed consent that has yet to be resolved is therapeutic misconception. In the early 1980s, Appelbaum (Lidz and Appelbaum, 2002) discussed how a research subject’s inability to distinguish between the aims of research and the aims of treatment creates a serious barrier to obtaining truly informed consent, and Lidz (Lidz and Appelbaum, 2002) confirmed that this continued to be a problem decades later. A potential solution to therapeutic misconception may start with evaluating how ICTs differentiate and explain therapy/treatment vis-à-vis clinical research. The ICT provided by the National Human Genome Research Institute consistently refers to the research subject as a patient, despite clearly stating that they are consenting to participate in a ‘research study’ (http://www.genome.gov/27528182). It is possible that this choice of language may contribute to therapeutic misconception. Joffe et al. (2001) reported that 70 per cent of survey respondents in a cancer clinical trial did not understand the unproven nature of the research treatment and only 46 per cent of providers understood that a primary goal of the trial was to benefit future patients. They also found that use of the National Cancer Institute ICT was associated with increased knowledge.
ICTs can also be used as a pedagogical tool so that young researchers are better prepared to submit or participate in human research protocols. Templates can be introduced in the classroom as dynamic documents collectively agreed upon by researchers. This may facilitate the updating of the ICT and serve as a model for students of the level of professionalism and collaboration that will be expected of them. Moreover, becoming familiar with tools for informed consent, as well as the theory behind informed consent as a process, may help students become more confident in conducting research and save time when they create a protocol for IRB submission. As these students then become researchers and IRB members themselves, they will be able to make contributions to the field of clinical research that actually employs the theories surrounding the consent process and continues to push our expectations for best practices.
Conclusion
This survey adds to the currently published research evaluating the use of ICTs in human subjects research. The wide variation in page length, formatting, and components of the forms, however, suggests that there is a lack of agreement or knowledge within the research community about how to design ICTs to best meet the needs of researchers, IRBs, and potential subjects. Further research is needed to evaluate outcomes associated with use of more standardized ICTs.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
