Abstract
Although most researchers are familiar with the application of the Common Rule in research, fewer are aware of specific requirements and restrictions for conducting human subjects research when employees of the US Department of Defense (DoD) will be participants. Because of the additional regulations concerning DoD employees as participants, federal regulations and research policies require researchers to submit their human subjects research proposals through a DoD review process to ensure compliance with DoD research policies, regardless of a non-DoD IRB’s approval. These research policies recognize the vulnerability of military subjects within a hierarchical organization to potential coercion and abuse during research studies. This article describes some of the unique requirements to conduct human subjects research within the DoD (in which the participants are military or civilian). The article concludes with steps researchers can follow to facilitate the DoD review process for research, and timely research within the DoD.
Keywords
Introduction
Most researchers are familiar with the application of the Common Rule, the role of the Institutional Review Board (IRB) process, and the requirements of Title 45 Part 46 US Code of Federal Regulations (45 CFR 46) when conducting human subjects research (Department of Health and Human Services, 2001). Many researchers are already frustrated by the IRB review process (Pech et al., 2007); discovering that the US Department of Defense (DoD) has additional requirements after a non-DoD IRB approval often necessitates another round of revision and IRB review. This article discusses how civilian researchers can better prepare themselves and their IRBs to obtain approval to conduct social science human subjects research within the DoD.
The Common Rule expresses the ethical standards for research recommended within the Belmont Report (Lynn and Nelson, 2005). Many IRBs within education and research institutions apply 45 CFR 46 to the composition and processes of their IRBs as a means to oversee all research activities whether funded by public funds or not (Riordan and Riordan, 2009). Title 32 Part 219 of the US Code of Federal Regulations (32 CFR 219) duplicates 45 CFR 46 subpart A and references 45 CFR 46 subparts B through D for the conduct of human subjects research within the DoD (Department of Defense, 2001). However, members of the Department of Defense – especially those in uniform – can be at increased risk of mistreatment by researchers because of the hierarchical design of the military and the risks inherent in their occupations. Department of Defense Instruction 3216.02 (DoDI 3216.02), Protection of Human Subjects and Adherence to the Ethical Standards in DoD-Supported Research, further regulates the application of 32 CFR 219 within the DoD with unique requirements for human subjects research protection. It is these unique requirements that can cause consternation with researchers.
DoD IRB reviews
DoDI 3216.02 requires all researchers conducting non-exempt human subjects research within DoD organizations to be covered by a federal assurance (Department of Defense, 2011). Many researchers conduct studies reviewed by an IRB from an institution like a university that holds a federal assurance from the Department of Health and Human Services (DHHS), which meets this requirement. Independent researchers conducting non-exempt human subjects research with DoD employees or within DoD organizations must arrange for coverage under a DoD or DHHS assurance prior to requesting approval to conduct their research (DoD, 2011). When an individual or private organization is not covered under a federal assurance to conduct human subjects research, the researchers must use an Individual Investigator Agreement to affiliate themselves with an organization that does have a federal assurance (DoD, 2011).
DoD organizations can accept another IRB’s approval of a research proposal so long as the DoD organization and the non-DoD IRB complete an Institutional Agreement for IRB Review and the research proposal receives an administrative review by a DoD determination official (DoD, 2011). This agreement requires the covering IRB to comply with both DHHS and DoD policies for the conduct of human subjects research (DoD, 2011); the DoD administrative review verifies this compliance. Researchers might find that a research proposal approved by their own IRB fails to meet the DoD requirements and will therefore be disapproved by the DoD organization. One method to avoid DoD disapproval is to work with a researcher within the DoD and rely on a DoD IRB for approval. Otherwise, as with other multi-institutional research arrangements, it is best that researchers incorporate the DoD-specific requirements within their proposals prior to submission to their IRB so that all requirements from the approving organizations are met within the first IRB’s review. This can avoid additional IRB reviews and research delays. Concurrent IRB reviews are permitted (DoD, 2011) and are therefore encouraged. See Figure 1 for an example of the review steps.
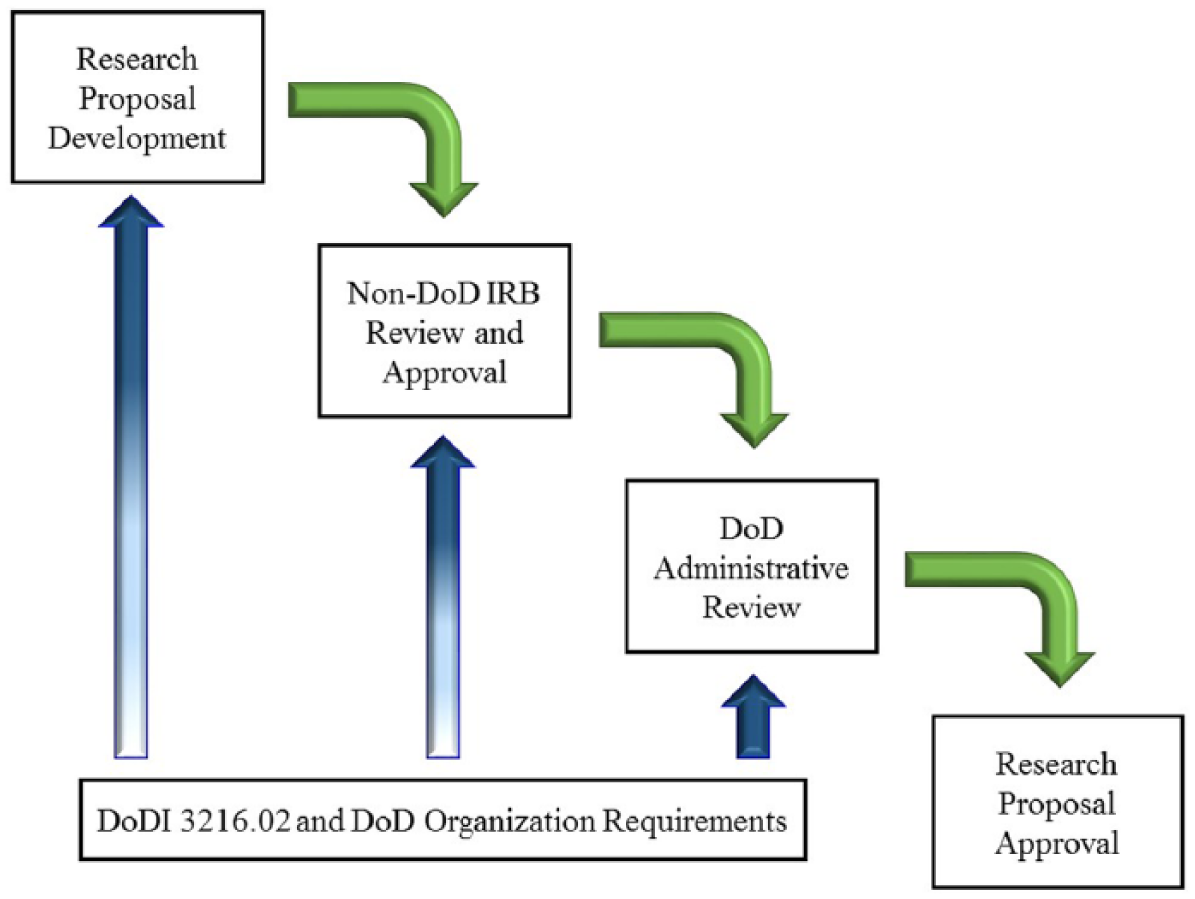
The DoD administrative review uses DoDI 3216.02 and local requirements to concur with an IRB human subjects determination and research approval, and these unique requirements should inform the development and non-DoD IRB review of a proposal to ensure that the DoD organization can approve the proposal
The DoD review of an external proposal is intended to ensure that all requirements of the DoDI 3216.02 are included in the study design and that the external IRB is aware of its responsibility to oversee the researchers’ conduct of the research (DoD, 2011). This review is not normally a second IRB review that would second-guess the external IRB’s determination or the researchers’ methodology. However, all IRBs have the responsibility to ensure the protection of the subjects of human research and therefore can exercise some authority over a researcher’s proposal when its research design is flawed or places subjects at unnecessary risk (Klitzman, 2013; Lynn and Nelson, 2005). DoDI 3216.02 requires that an IRB (whether DoD or external) reviewing a protocol consider either the scientific merit of a protocol (if the IRB is external) or have a formal scientific review as part of the IRB review (if the IRB is DoD). This portion of the administrative review ensures that the proposed study’s methodology is sound and will contribute to the body of knowledge (DoD, 2011). The DoD administrative review might result in required revisions. These IRB-directed revisions should be considered contributions to a better research proposal rather than impediments to IRB approval (Lynn and Nelson, 2005).
DoDI 3216.02 requires all DoD organizations conducting or supporting research to have a published program that defines requirements and review processes within the organization. An Institutional Official supervises the DoD-issued assurance, the organization’s protections program, and some staff to conduct human subjects protection program administrative tasks (DoD, 2011). However, not all DoD organizations have an IRB or are covered by a DoD assurance to conduct non-exempt human subjects research. Where civilian institutions might delegate exempt research determination to the IRB (Lynn and Nelson, 2005), DoD organizations might have a human protections administrator or similar official not assigned to an IRB as an exempt determination official to review and approve exempt research. This official will forward non-exempt research to a supporting IRB for further review. This can be problematic for external researchers trying to identify who within a DoD organization must review a research proposal, because no two organizations might have the same review officials and processes. Establishing the identity of the point of contact within a DoD organization is therefore important to reducing the time required for proposal review.
The DoD organizational review specifically looks for how the researchers and the IRB approving the research incorporated the specific DoD requirements into the proposal. These requirements cover researcher qualifications, recruiting, risk assessment, the treatment of DoD employees as subjects, and the monitoring of research. Failure to include these specific requirements within a proposal can signal to a DoD IRB that the researchers and non-DoD IRB are not aware of policies for DoD research, and therefore might not be qualified to conduct the research.
Specific DoD requirements and restrictions
DoDI 3216.02 requires all DoD employees who conduct, supervise, review, or approve human subjects research to complete appropriate training and education to ensure that research is conducted ethically in accordance with DoD policies. When researchers external to the DoD conduct research within the DoD or with DoD employees, the DoD organization must review the training and education of all the researchers as well. Although many institutions accept training through the Collaborative Institutional Training Initiative program and research methods courses to certify researchers, DoD organizations might require additional training prior to approving research within the DoD. Each DoD organization establishes this education requirement and whether another certification program is comparable enough to not require additional education or training (DoD, 2011).
Although not a vulnerable population under the Common Rule, DoDI 3216.02 recognizes that military members are at a disadvantage when participating as subjects in research. Several DoD-specific requirements directly address the vulnerability of this population. When recruiting military subjects for a study and during the informed consent process, the researchers must ensure that supervisors (both military and civilian) are not present, so as not to create even the perception of supervisory influence on a military member’s decision to participate in the research (DoD, 2011). If supervisors are eligible to participate, then a separate recruitment presentation is necessary. Unit commanders must approve the participation of their subordinates when research activities occur during duty hours or might interfere with a service member’s performance even when the research activities are during non-duty hours. Military leaders should be made aware of any risk to the day-to-day activities of potential subjects prior to recruitment (DoD, 2011).
DoD civilians must also be recruited separately from their supervisors to avoid any perceived or actual influence on consent. Individual organization rules will determine the eligibility of DoD civilians to participate on- or off-duty. As with uniformed personnel as subjects, military commanders should approve the participation of civilian DoD employees when there is the potential that duty performance might be affected by an employee’s participation in the study. To ensure compliance with these recruiting procedures, the DoD organization’s IRB might appoint an ombudsman to observe the recruiting process (DoD, 2011).
Another potential vulnerability is the nature of the work performed by uniformed service members. When assessing the level of risk to subjects, the DoDI 3216.02 specifically requires IRBs to consider the definition of minimal risk – those activities ‘ordinarily encountered in daily life or during the performance of routine physical or physiological examinations or tests’ (DoD, 2011: 19) – to exclude the unique demands on military members in the performance of their duties. In other words, exposure to combat, intense physical training and conditions, or prolonged pain because of the service member’s occupation cannot be a justification for assessing research as minimal risk. IRBs must compare the proposed research activities to those experienced by a typical civilian in determining and mitigating risk levels (DoD, 2011).
By virtue of their status as federal employees, the DoDI 3216.02 and other federal laws limit compensation to DoD employees for their participation in a research study. Different rules apply based on duty status and whether the research is conducted by a DoD organization or by a non-DoD organization. For example, federal employees can be compensated up to $50 for blood draws whether on- or off-duty, but can only receive compensation for other research activities when those activities are during off-duty hours, are funded from a non-federal source, are comparable to local rates, and are approved by the DoD organization’s IRB (DoD, 2011). Researchers planning to compensate federal employees (whether uniformed or not) in any way should therefore be specific in the research protocol on the source of funding for compensation and when research activities will occur relative to the normal work hours for the DoD employees.
Although the accepted vulnerable populations of 45 CFR 46 are also considered vulnerable populations within the DoD, the DoDI 3216.02 adds provisos to the DHHS regulations that researchers should understand. For example, the DoDI 3216.02 requires that the phrase ‘generalizable information’ replace the phrase ‘biomedical information’ when conducting research on pregnant women, fetuses, and neonates (DoD, 2011). Human subjects research on prisoners can only be approved by a full IRB regardless of risk determination, but can include some epidemiological research and research that would otherwise be exempt as two additional categories for research on prisoners (DoD, 2011). Finally, the DoDI 3216.02 establishes that all military personnel on active federal duty are considered adults rather than children for application of protections for research involving children. Researchers should nonetheless justify the inclusion of cadets and recruits under the age of 18 when proposing a study on military personnel otherwise considered legally as minors (DoD, 2011). Table 1 summarizes some of the unique requirements for research within the DoD.
DoD unique requirements on social human subjects research.

Source: DoDI 3216.02, Protection of Human Subjects and Adherence to Ethical Standards in DoD-Supported Research, Enclosure 3. Although not all-inclusive, this table provides the unique requirements commonly associated with social human subjects research for consideration during proposal development
Submitting your proposal
Conducting human subjects research within the DoD is similar to conducting multisite research involving more than one IRB. The additional requirements of the DoDI 3216.02 are akin to potential requirements by another IRB. With this in mind, researchers should follow a few simple steps to successfully navigate through the IRB review process.
Review the DoDI 3216.02 before you develop your proposal. Researchers are individually responsible for the conduct of ethical research (Cseko and Tremaine, 2013). Understanding the additional requirements for DoD-supported research should be a priority for researchers in developing viable research proposals and human subjects protections that will meet local and DoD IRB requirements. Relying on an IRB to suggest appropriate revisions can be time-consuming. Quality and detail of an IRB application are factors in how long an IRB will take to review a proposal and the number of revisions that will be required (Klitzman, 2013), so know what additional protections or requirements might be necessary for research within the DoD before submitting a research proposal or IRB application.
Contact the DoD organization’s human protections administrator or equivalent official early. This task can take some time because not all DoD organizations have an IRB or an official responsible for reviewing research proposals for exempt or IRB review determinations. Each department within the DoD has a Human Research Protections Office; you might have to start at the top and work down to the local DoD organization official. Once you have identified who will review your proposal, determine the specific requirements of the organization’s program. For example, the DoD organization might have an informed consent template that requires the addition of a DoD IRB contact as well as the researcher’s IRB contact information or a statement that indicates that the subject will not disclose classified information. An organization’s protection program might specify that a DoD researcher must be a member of the research team. Identify whether your institution’s researcher certification process is sufficient or if additional training will be required by the DoD organization. With the DoD official, determine which IRB will be responsible for approving the research proposal or if you will need to complete an Independent Investigator Agreement with the DoD organization’s IRB. The DoD official can also advise you whether an additional review and approval by another DoD institution such as the Army Research Institute is necessary when conducting surveys because of the requirements of Department of Defense Instruction 1100.13, DoD Surveys, and Department of Defense Instruction 8910.01, Information Collection and Reporting. Discuss your timeline with the DoD official and get input on your research design early in proposal development.
Explicitly state and cite DoDI 3216.02 requirements in the proposal and IRB application. Clearly indicating your knowledge of the DoD unique requirements within your proposal by articulating specific DoD requirements can assure the DoD reviewer that you are qualified to conduct research within DoD policy. Include required forms like the Institutional Agreement for IRB Review in your IRB application packet and clearly indicate that you are conducting research on DoD employees to alert your non-DoD IRB that it has additional responsibilities in reviewing your proposal. Consider including the DoD organizational point of contact in your IRB application in case the IRB reviewer has questions on DoD requirements. Including detailed information within the IRB application helps the IRB make a timely determination on your proposal (Pech et al., 2007); vague or omitted DoD requirements risk delays arising from additional IRB reviews prior to approval.
Conclusion
Whereas 32 CFR 219 duplicates almost all of the provisions of 45 CFR 46, the DoDI 3216.02 implements 32 CFR 219 and imposes additional requirements on the conduct of human subjects research within the DoD. Many of these unique requirements are based on the status of DoD employees as federal employees and the increased vulnerability of subordinates within hierarchical military organizations to superior–subordinate influence. Researchers planning to recruit DoD employees (civilian or military) should be knowledgeable on these DoD unique requirements prior to submitting an IRB application. As with a multi-site investigation, researchers should consult with a DoD IRB or determination official as well as the researchers’ IRB to ensure that requirements from all ethics review bodies are met within the research proposal. Meeting the requirements of both a civilian and a DoD IRB on the initial IRB application can go far to getting your IRB application, and research, approved in a timely manner.
Footnotes
Declaration of conflicting interests
The author declares there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
