Abstract
The length of participant information sheets (ISs) for research and difficulties in their comprehension have been a cause of increasing concern. We aimed to examine the information sheets in research proposals submitted to an Australian HREC in one year, comparing the results with national recommendations and published data. Information sheets in all 86 research submissions were analysed using available software. The work of Flesch was used for Reading Ease or Readability and that of Flesch and Kincaid for the level of education required for comprehension, the Reading Grade Level. The mean length of 86 information sheets was 3110 words; many had more than 5000 words. Using the Flesch scale of 0 to 100, with 0 meaning most difficult and 100 very easy to read, the mean readability level was 47. The mean length of education needed to easily grasp the information was 11.6 years, equivalent to senior secondary school. Information sheets in research projects submitted to an HREC were often too long to be read in a reasonable time and too difficult to be easily understood. Recommended standards for information sheets were infrequently met.
Introduction
Respect for human beings is the underpinning criterion of ethical review of human research and the voluntary, informed consent of the participant is the cornerstone of an ethically acceptable research proposal. The review and application of methods of obtaining voluntary consent and ensuring that potential participants receive adequate and intelligible information that allows them the choice of joining the research is a principal task of ethics review bodies such as human research ethics committees (HRECs). We have noted the criticism of information sheets in research projects (Loverde et al., 1989; Grossman et al., 1994; Sharp, 2004; Dingwall, 2005; Beardsley et al., 2007; Terblanche and Burgess, 2010; Gordon et al., 2012; Taylor and Bramley, 2012) and comments on increasing length of information sheets (Burman et al., 2003; Fortun et al., 2008; Kass et al., 2011). Terblanche and Burgess (2010: 158) called for participant information and consent forms (PICFs) to be short and written in comprehensive language, facilitating a greater level of understanding in potential research subjects. The objective of this paper is to promote these aims and the validity of Informed Consent by assessing the length and qualities of readability of ISs in research proposals submitted to the principal HREC in the Australian Capital Territory (ACT), the structure and function of which were described by Biggs and Marchesi (2013).
Ethical review of health and medical research in Australia is managed by 230 accredited HRECs. The accrediting body, the National Health and Medical Research Council (NHMRC), has provided guidance and regulations in The National Statement (2007). In 2012, NHMRC provided standard IS templates for various types of research (NHMRC, 2012) and that for interventional research, such as an adult drug study, has 20 numbered sections; proposed sentences are provided as are references to The National Statement.
The IS is to be at a reading grade level of 8 or below, that is, the information provided in a document should be easily comprehended by a person who has received only eight years of schooling. National authorities in other countries offer similar guidance to ethics review bodies. In Britain, the National Health Service (NHS) National Patient Safety Agency (NHS, 2013) includes emphasis that the information sheet is only one part of seeking consent of subjects and recommends discussion as the most effective way to ensure consent is truly informed. While we acknowledge and support the use of various methods in obtaining informed consent, the IS is internationally recognized as the primary tool and is thus the focus of our study.
We report on human research projects containing ISs approved by the ACT HREC in a year. The committee comprises eight members as required by The National Statement and an additional seven providing expertise in nursing, midwifery, intensive care and indigenous health. It reviews all research projects conducted in or by ACT Health. In this study we assessed the number of words, the readability and the reading grade level. Amendment to the IS is the most common reason for delayed approval by the committee. Accordingly, assessment of ISs was performed retrospectively, after approval was concluded. Low-risk projects, defined in The National Statement (p. 16) as those in which the only foreseeable risk is one of discomfort, were reviewed by a sub-committee and were not included in our assessment.
Methods
Assessment of length was done by word count; readability was measured using the formula devised by Flesch (1948) to assess syllables in words, length of sentences and use of passive tone. Ability to understand a document was measured in terms of the years of education needed and the reading grade level. This was assessed using the method of Kincaid et al. (1975), first used by the US Navy to ensure technical documents were understandable by new trainees. Use of these formulae in medical research has been described by many authors (Tarnowski et al., 1990; Grossman et al., 1994; Sharp, 2004; Beardsley et al., 2007; Terblanche and Burgess, 2010; Williamson and Martin, 2010; Kass et al., 2011; Gordon et al., 2012).
We examined the information sheets included in 86 research proposals submitted to the ACT HREC in the year 2012–13. These included four types of research: pharmaceutical company-sponsored drug trials, usually international; national collaborative studies, Australian or Australian and New Zealand research by multiple researchers; investigator-led projects, usually in one hospital, university department or research institution; and student-led research. The classification is used in NHMRC reports. The number of words, the Flesch readability score and the Flesch-Kincaid reading grade level were measured in each by JB. We used the software program ‘Readability-Score.com’ 2011–14. Word length and readability scores were separately measured for the four types of research. All included the title of the project but not the consent pages. Readability scores have been graded from 0–29, very confusing; 30–49, difficult; 50–59, fairly difficult; 60–69, standard; 70–79, fairly easy; 80–89, easy; 90–100, very easy; documents scoring between 60 and 70 are readable by an average literate reader (Grossman et al., 1994: 2212; Terblanche and Burgess, 2010: 158; Garger, 2012: 2).
Results
1. Length of information sheets
The 86 ISs ranged in length from one to 35 pages and contained from 346 to 8955 words, with a mean of 3110 words. Twenty-three had more than 5000 words; five had more than 8000 words (Figure 1).
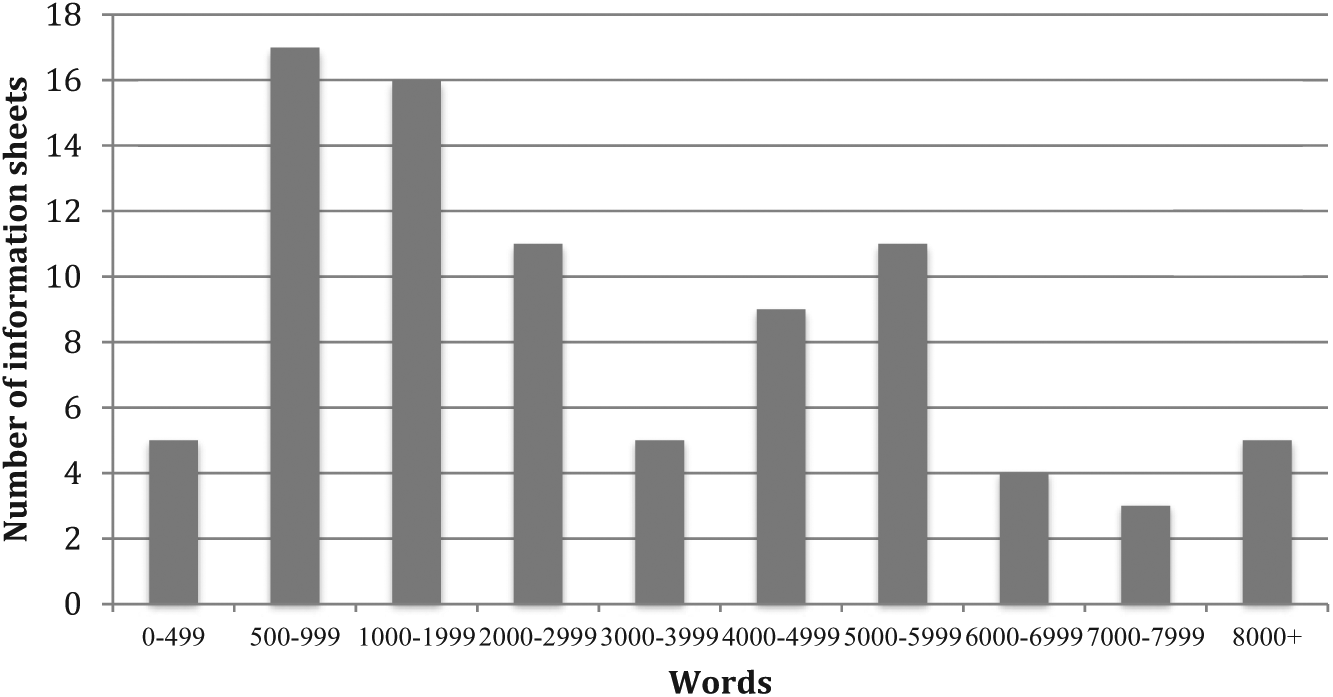
Length in words of information sheets submitted to ACT HREC. Prepared by John SG Biggs.
2. Readability of information sheets
The 86 ISs showed a readability range of 28.6 to 67.5, with a mean level of 47.1, considered as difficult. Two ISs were in the very confusing range while the greatest number (49) was in the difficult range. Four returned a score in the standard range; none were considered fairly easy or easy to read.
3. Reading grade levels for ready understanding of information sheets
Reading grade levels, using the Flesch-Kincaid formula, are translated into US school grades, and Australian equivalents are used in this paper. The mean level of education required to readily understand the ISs in 86 research applications was 11.6 with a range of 7.5 to 14.9. Figure 2 shows a nearly normal distribution about the mean, with 16 ISs at grade 7 to 9 and 10 at grade 13 to 14, equivalent to university study. Only six research project ISs met the NHMRC standard of grade 8 or below.
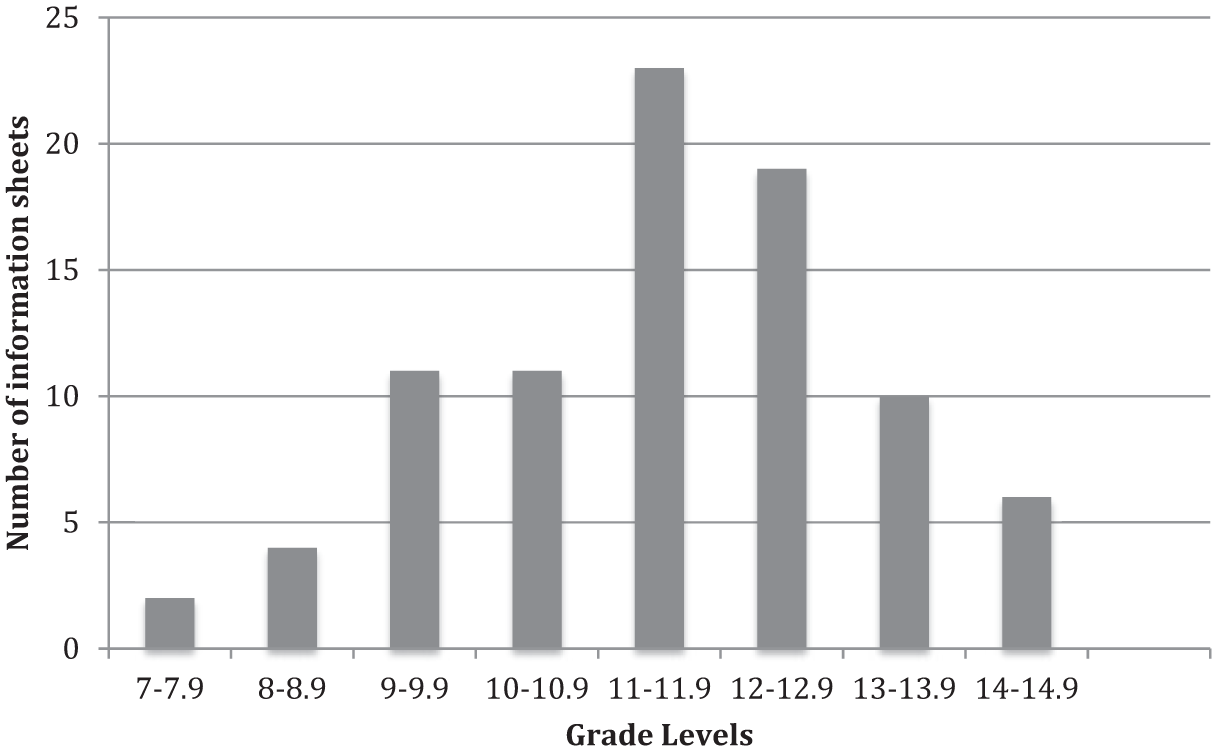
Reading grade levels of ACT information sheets. There were no values below 7.0 and none above 14.9. Prepared by John SG Biggs.
4. Data for types of research projects
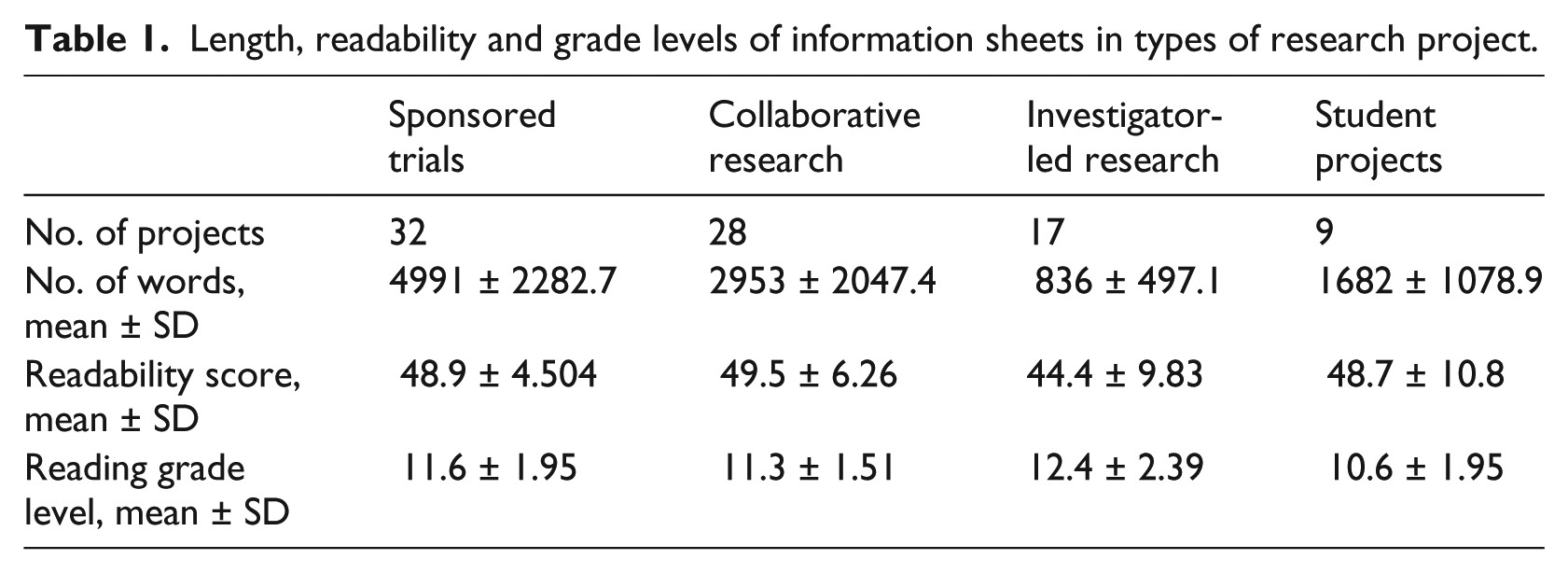
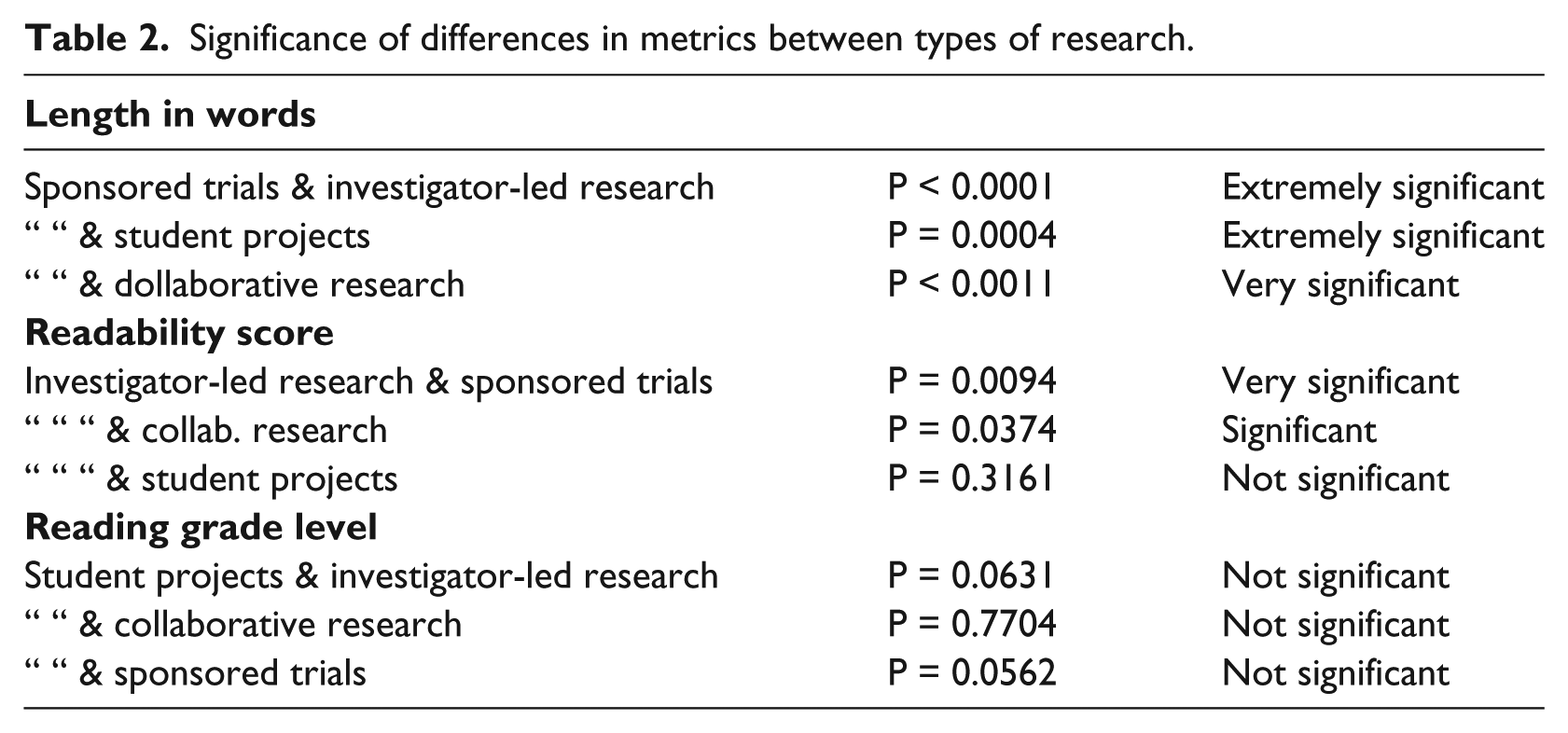
The data for types of projects, described above, showed differences that were notable in length of information sheets, but relatively small for other metrics (see Table 1). The mean length for sponsored trials was nearly double that for collaborative research and six times that of investigator-led research. Readability scores were similar for sponsored, collaborative and student research and slightly lower for investigator-led research. Reading grade level means were mostly similar, at about grade 11–12 or end of secondary school; the exception was student projects with a mean about one year lower. The differences between types of research are highly significant for length of information sheet, significant for readability scores and not significant for reading grade level.
Length, readability and grade levels of information sheets in types of research project.
Discussion
The Declaration of Helsinki puts emphasis on potential participants understanding the information provided in research proposals (World Medical Association (2008: article 24). The length of information documents is an important factor. The finding in 86 research projects of a mean length of 3100 words and an average reading speed of 200–225 words per minute (Sharp, 2004: 573) means that a potential research participant will need about 15 minutes to read the information provided. A quarter of the projects had more than 5000 words and 6% more than 8000; reading would have occupied about 24 or 38 minutes, respectively. It must be asked whether participation will be encouraged by ISs of this length, and whether the information will be read. Sharp believed that people were unlikely to read documents of over 1000 words, noting that many people will need to read some parts over and over to understand them. He recommended that information sheets have a maximum length of 1250 words (2004: 574).
The mean length of ISs for sponsored trials in the ACT study was significantly longer than that of other types of research (see Table 2). While papers prepared for global use may seek to cover all eventualities, Sharp (2004) and Dingwall (2005) have surmised that their greater length has more to do with legal cover for the sponsor than protection of participants. The point may have been reached where the inclusion of all imaginable risks has become more important than gaining actual informed consent.
Significance of differences in metrics between types of research.
Our finding that information sheets in most of the 86 research projects were in the fairly difficult or difficult ranges of readability provides another indication that improvements are needed. Similar findings have been reported by Terblanche and Burgess (2010: 159), who described 84 information sheets with mean Flesch readability of 46.6, also in the difficult range; Grossman et al. (1994: 2214), whose 137 sheets had a mean readability of 52.6, in the fairly difficult range; and Sharp (2004: 272), whose 107 sheets had a mean readability of 45.68, in the difficult range. Of particular relevance is the study of Taylor and Bramley (2012), who studied Flesch-Kincaid readability of information sheets given to 40 patients receiving anaesthesia in Australia and New Zealand. The authors found a mean readability score of 11.9 in 40 studies and wrote that this exceeded the average ability to comprehend such documents in the two countries.
The problem of readability of information sheets is heightened by the prevalence of community illiteracy. Recent Australian work has described five skill levels of literacy, from 1, the lowest, to 5, the highest (Australian Bureau of Statistics, 2013). Level 3 is regarded as the lowest ‘compatible with working in the emerging knowledge-based economy’, and 44% of Australians at lower levels are deemed ‘functionally illiterate’. This has implications for researchers as they prepare information sheets for potential research participants. The data on reading grades is especially relevant in regard to NHMRC recommendations that information for research participants be presented at Grade 8 level or below, meaning that it will be understandable by persons with early secondary-school education (Kincaid et al., 1975). Since only 7% of ACT HREC projects, and similar proportions in reports of Beardsley et al. (2007) and Terblanche and Burgess (2010) met this expectation, there is a need for greater effort by researchers to simplify the content and language used in information sheets and greater rigor by ethics committees in reviewing the ethical acceptability of these documents.
The NHS recommendation that discussion is the best way to ensure consent is truly informed is supported by Olver et al. (1995), who assessed the impact of ISs on 100 patients, asking them at a scheduled treatment visit what was their main source of information before deciding to enter the research project. Twelve said the IS was their main source, 52 said it was a doctor and 26 a nurse. Clearly, there is more than the written word to aid a potential participant in knowing what is proposed, provide answers to their questions, and assist them in their decision to give informed consent.
Most consent forms we have seen contain the statement: ‘I have had the research or study explained to me and have been able to ask for more information’. It is contended that information on paper or on screen is evidence that the information was proffered, but it needs to be seen as an adjunct to detailed conversation with a member of the research team.
Conclusions
The length of information sheets provided to research participants is excessive, especially in sponsored studies, and the view that many find difficulty in reading and understanding them is supported by the literature. The reading grade level recommended for Australian research studies appears to be rarely attained, and this, together with the readability levels seen most commonly as difficult, suggests a pressing need for reform if the term informed consent is to have legal or moral weight. The recommended grade level of 8 or below should be a requirement for ethical approval, and consideration should be given to limiting the length of information, perhaps to the suggested 1250 words.
Footnotes
Conflict of Interest
There are no conflicting interests.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
