Abstract
Contemporary conceptualizations on infant cognitive development focus on predictive processes; the basic idea is that the brain continuously creates predictions about what is expected and that the divergence between predicted and actual perceived data yields a prediction error. This prediction error updates the model from which the predictions are generated and therefore is a basic mechanism for learning and adaptation to the dynamics of the ever-changing environment. In this article, we review the types of available empirical evidence supporting the idea that predictive processes can be found in infancy, especially emphasizing the contribution of electrophysiology as a potential method for testing the similarity of the brain mechanisms for processing prediction errors in infants to those of adults. In infants, as with older children, adolescents, and adults, predictions involve synchronization bursts of middle-central theta reflecting brain activity in the anterior cingulate cortex. We discuss how early in development such brain mechanisms develop and open questions that still remain to be empirically investigated.
Contemporary conceptualizations of infant cognitive development focus on predictive processes as a basic and powerful underlying mechanism for infant learning (see comprehensive recent review in Köster et al., 2020), and some researchers have claimed that such predictions are based on Bayesian principles (Perfors et al., 2011). This view follows a modern philosophical and neuro-computational conceptualization of the brain as a predicting machine (Clark, 2016) that provides a unifying framework for all psychological phenomena. The basic idea is that the brain continuously creates predictions about what is expected and that the divergence between predicted and actual perceived data yields a prediction error. This prediction error updates the model from which the predictions are generated and therefore is a basic mechanism for learning and adaptation to the dynamics of the ever-changing environment. According to Friston (2010), the brain’s hierarchical organization and the existence of forward and backward connections fit perfectly with the idea that predictions can be formed at every level of the hierarchy. Predictions are feedforwarded to lower levels in the hierarchy, and prediction errors can be fed backward to higher levels in the hierarchy and improve the current predictive model on which the prior predictions were based. Accordingly, the primary brain-functioning principle is to minimize free energy, which is the information-theoretic equivalent of overall prediction error (Friston, 2010). The implementation of these concepts as a computational framework for cognitive development brought a modern twist to Piaget’s constructivist ideas of adaptation and accommodation (Johnson & Munakata, 2005), giving rise to what some researchers termed “rational constructivism” (Gopnik & Wellman, 2012). This new perspective postulates that children infer causal structure from statistical probabilistic information and Bayesian learning through their own actions on the world and through observations of the actions of others.
Recent research has provided highly interesting and significant empirical evidence supporting the claims that predictive processes can be found in infancy. In this article, we review the types of available evidence in this respect, especially emphasizing the contribution of electrophysiology as a method for testing how similar the brain mechanisms of processing prediction errors in infants are to those of adults and how early in development such brain mechanisms develop. These issues are fundamental because prediction errors have been shown to be relevant for enhancing learning, for example, by increasing the infant’s exploration of the “surprising” object and influencing their learning rate (Sim & Xu, 2017; Stahl & Feigenson, 2017). Prediction errors could be the basic mechanism for adapting the brain at early stages of development because top-down predictions are fed forward from areas such as the prefrontal cortex and may have a direct role in shaping the functional-response properties of lower perceptual areas such as the posterior visual cortex (Johnson, 2020). The relevant literature is currently quite scattered, although recently, some of its aspects have been separately reviewed. We point out and refer to such previous reviews of specific topics. Still, a more comprehensive organization of the extant literature is still missing, especially regarding the neural evidence for infant prediction errors, which we review in the present article. The accumulating empirical evidence is compelling, but there are still many open questions that remain to be answered; at the end of the article, we delineate some of the main ones.
What Infants’ Eyes Tell Researchers: Behavioral Evidence for Infant Predictions
Violation-of-expectation (VoE) paradigms are powerful and widely used tools for studying infants’ basic understanding about the physical and social world (see review in Köster et al., 2020). Infants orient toward events that violate their expectations regarding physical or social rules (whether these “priors” were based on “core knowledge” that the infant possessed or were acquired during the initial phase of the experiment). The basic VoE finding is that infants look longer at “impossible” events, that is, a display that violates a “rule.” Beginning in the 1980s with pioneer studies about object permanence (e.g., Baillargeon et al., 1985), this basic effect has been used for more than 40 years to learn about the basic expectations that infants have about the rules governing physical objects, actions, number, space, social agents (Spelke & Kinzler, 2007), and much more (see review of this literature in Stahl & Feigenson, 2017). Although, infant looking times in VoE paradigms are sensitive not only to novelty but also to familiarity, complexity, and informativeness and should not be considered exclusively as an index of prediction error (Zhang & Emberson, 2020). A series of recent studies have further modeled the patterns of infant looking times in VoE contexts beyond a basic dichotomy between “possible” and “impossible” events using a combination of empirical research and computational models (e.g., Kidd et al., 2012; Piantadosi et al., 2014; Teglas et al., 2007, 2011). These studies provided compelling behavioral evidence for prediction processing in infants. These articles were recently reviewed by Sim and Xu (2019), who concluded that looking times can be reconceptualized as reflecting “sophisticated statistical inference, captured by Bayesian probabilistic models.” In other words, longer looking times reflect prediction errors when the infant is faced with information that is incongruent with the “current” mental model, meaning that infants’ looking times are well predicted by the probability of observed events.
Infant predictive processing in VoE tasks has also been studied using pupil size, gaze direction, and latency. A recent review of this literature can be found in Zhang and Emerson (2020). A relevant and compelling study using pupillometry as an index of prediction errors was conducted by Zhang et al. (2019). This study compared adults’ and 6-month-old infants’ pupil-dilatation changes when previously learned associations between sounds and pictures were violated. Both infants and adults showed larger comparative changes in pupil size for trials that elicited prediction errors, that is, trials in which the visual stimulus that should have followed the paired auditory cue was omitted, compared with trials that confirmed the participants’ predictions that were established in the initial “implicit learning” phase of the audiovisual associations. As for studies of infants’ predictions using anticipatory gaze direction, one clear example is the study by Romberg and Saffran (2013) with 12-month-old infants in which they measured anticipatory eye movements to targets that appeared in one of two possible locations and found that the infants adjusted their predictions to reflect changes in the target location on the previous trial and that such flexibility was more pronounced in infants with more variable prior experience regarding target locations. In other words, infants’ anticipatory behavior reflected their prior experience. Moreover, the individual differences in such flexibility and adaptability of anticipatory looking behavior at an early age seemed to be related to and to predict individual differences in language acquisition. This was later shown in a longitudinal study by Reuter et al. (2018; i.e., in vocabulary size), suggesting a causal role in learning, which is in line with error-based models of learning (see also Mani & Huettig, 2012). In this context, note that prediction processes have not solely been studied in infants regarding predictions of location or perceptual features of objects, but in fact, evidence supporting the ability of infants to extract statistical information and generate predictions, accordingly, has become dominant in the modern literature on language development (Golinkoff et al., 2013) and the developmental literature on perception and interpretation of the actions of others (Daum et al., 2012; Gredebäck & Daum, 2015; Gredebäck et al., 2018; Kayhan, Heil, et al., 2019, and more). Overall, as summarized by Köster et al. (2020), the PP [predictive processing] perspective offers a plausible explanation for infants’ VoE responses across social and physical knowledge domains. VoE responses indicate infants’ processing of prediction errors, which require them to refine prior predictions (i.e., update their predictive models) or, put more simply, learning. (p. 566)
The Predicting Brain
The brain structures related to prediction errors have been empirically studied using electrophysiological methods in human adults (Cavanagh & Frank, 2014) and primates (Phillips et al., 2014; Womelsdorf et al., 2010). Here we briefly review the adult mechanisms in order to compare them with what is present in infancy. For a more complete review of this extensive adult literature, see Alexander and Brown (2019).
Middle-frontal theta activity (4–8 Hz) has long been known to be elicited in situations requiring cognitive control (novelty, conflict, punishment, and error). This activity is reflected in a series of extensively studied event-related-potential (ERP) components of negative voltage polarity that can be measured at middle-frontal scalp areas. These include: the ERN (error-related negativity), which appears approximately 100 ms post incorrect responses (Hajcak et al., 2005); the FRN (feedback-related negativity), which appears at about the same time and at the same scalp localization as the ERN, and also has the same wave-form pattern as the ERN, but it appears following negative feedback (Miltner et al., 1997) and is thought to encode a quantitative reward prediction error (Walsh & Anderson, 2012); the N2 (second negative peak in the averaged ERP waveform), which appears around 200 ms from stimulus appearance and is mainly related to novelty or stimulus–response conflict (Cavanagh & Frank, 2014); and the N400, a negativity wave that peaks at about 400 ms following the appearance of semantically incongruent stimuli (Hanslmayr et al., 2008; Mesika et al., 2014). All of these ERP components seem to reflect bursts of phase-synchronization theta activity, leading to an increase of power in this frequency band, broadly signalizing deviations between expected and observed events (Cavanagh et al., 2012; Cavanagh & Frank, 2014; Luu et al., 2003). This theta activity seems to be elicited mainly “by” and “in” the anterior cingulate cortex (ACC), the main brain region considered to be strongly involved in monitoring and cognitive control (Botvinick et al., 2001, 2004; Bush et al., 2000; Cohen & Cavanagh, 2011; Gehring & Fencsik, 2001; Kerns, 2004; Mathalon et al., 2003; Rushworth et al., 2004). It then propagates to more distal locations, such as perceptual and motor brain areas (Cavanagh & Frank, 2014), becoming critical for learning (Cavanagh et al., 2010; Crivelli-Decker et al., 2018; Mas-Herrero & Marco-Pallarés, 2014). The functional correlation between cognitive-control processes and theta-band activity proceeding from the ACC has also been demonstrated in children (Adam et al., 2020; Meyer et al., 2019), adolescents (Buzzell et al., 2019), and nonhuman species using single-cell recordings (e.g., in macaques; e.g., Womelsdorf et al., 2010). Alexander and Brown (2019) suggested that the ACC has a broad role in computing a hierarchically structured surprise signal that is critical for learning models of the environment. According to the suggested model, prediction-error computations engage forwarding of error information up the hierarchy and prediction information (in the form of expected prediction error) downward. Such a model of the functioning of the neocortex is fully compatible with Friston’s (2010) ideas that were presented above at the beginning of this article.
Consistent with these ideas, the increase in theta power seems to be proportional to the degree of deviation from the expectations. In a series of studies (Tzur & Berger, 2007, 2009; Tzur et al., 2010), the brain reaction to VoE was explored using correct and incorrect arithmetic equations. The middle-frontal theta reaction to the erroneous solutions to the simple equations was sensitive to the error salience or magnitude; that is, stronger theta activity was found with larger or more deviant errors (e.g., 1 + 2 = 8) than with smaller or less deviant ones (e.g., 1 + 2 = 4). This brain activity was source-localized to the ACC (Tzur & Berger, 2009). Moreover, the time-frequency decomposition analyses indicated that theta activity was modulated by the magnitude of erroneous information in a nonlinear fashion in accord with Weber–Fechner’s logarithmic function or Stevens’s power functions (Tzur et al., 2010). In more modern “predicting brain” terms, the middle-frontal theta activity in the brain reflects the degree of the prediction error. Further studies suggested that the ACC signals should be more accurately defined as “unsigned” prediction errors reflecting the degree of “surprise” regardless of valence (Mas-Herrero & Marco-Pallarés, 2014).
Note that predictions can also occur at lower levels of the brain (e.g., locally at sensory areas). At sensory areas, the mismatch negativity (MMN) is the most studied ERP brain prediction error signaling the automatic detection of a deviation of perceptual features (e.g., in vision, unexpected spatial frequency, orientation, movement direction). A comprehensive review of the predictive-coding view of the MMN, especially in the visual domain, can be found in Stefanics et al. (2014), however, this type of predictions is outside the scope of the present review.
Neural Evidence for Prediction Errors in Infants
Following the findings in children, adolescents, and adults, tracing the development of prediction from infancy may serve to help understand the role of prediction in perception and action. This issue may be examined by tracing the origin and development of the neuronal activity found in the mature brain when the predictions of infants are violated. Using an adapted version of a classical VoE paradigm (Wynn, 1992), we (Berger et al., 2006) measured and analyzed the brain activity, as well as looking behavior, of a sample of 6- to 9-month-old infants presented with a videotaped series of “puppet” trials, including correct and incorrect solutions to simple arithmetic equations (see Fig. 1). At the behavioral level, Wynn’s (1992) looking-time VoE effect was replicated—the infants looked longer at incorrect solutions than at correct ones. At the brain-activity level, ERPs time-locked, at the same electrode locations in which such activity was found in adults, to the presentation of the solution also differed between conditions, with greater negative activity for the incorrect solution condition. The similarity between the infant and adult neural reactions to the perceived errors was striking in terms of both the topography and frequency of this activity. Although, there were also understandable differences in the magnitude of the effects (given the larger overall amplitude of the electroencephalogram [EEG] waves in infants) and their timing (which was slightly slower in the infants). These ERP findings were replicated and expanded on a different 7-month-old infant sample, including a time-frequency analysis that delineated the frequency of the effect, indicating that it was in the theta range (i.e., between 6 and 7 Hz; Berger et al., 2019). The 6- to 7-Hz band for middle-central response to perceived error was also found in a study with toddlers (Conejero et al., 2018). In this study, 16- to 18-month-olds observed the assembling of three-piece puzzles of pictures of an animal (e.g., a chicken) that were being formed either correctly or incorrectly (e.g., after the first two pieces built a body of a chicken, the third top part was of a head of an elephant). Observation of incorrect configurations produced increased potentials in midline channels and greater power of 6- to 7-Hz activity.
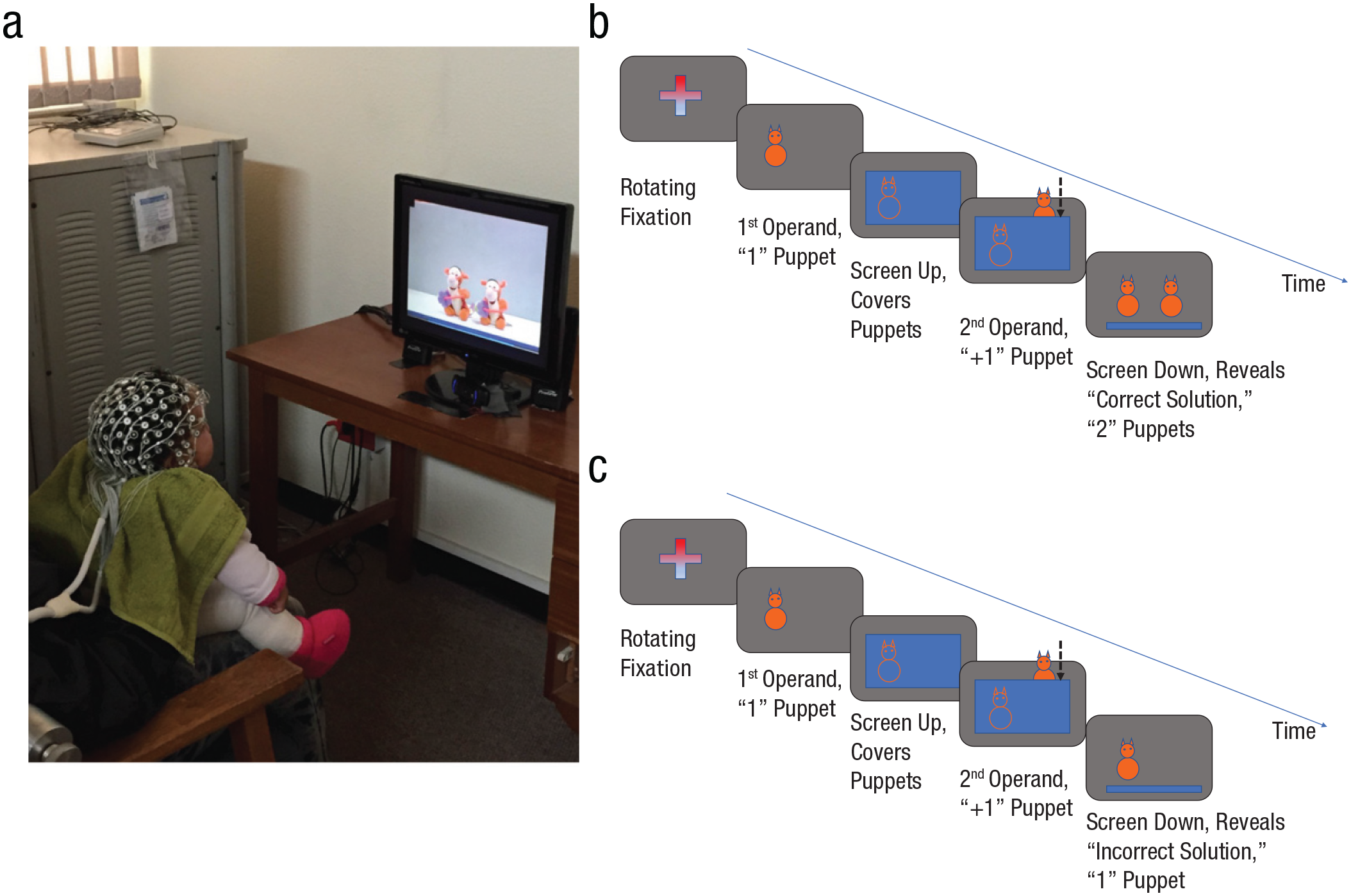
Experimental setup and design used in Berger et al. (2006, 2019). (a) One of the participants in the study wearing the geodesic net and watching the stimuli portrayed in a videotaped puppet version of the different mathematical equations (1 + 1 = 1; 1 + 1 = 2; 2 − 1 = 1; and 2 − 1 = 2), which were edited to keep an exact timing of the series of events in each type of trial. (b) A schematic representation of the events for the correct equation 1 + 1 = 2; one puppet is displayed, a screen is raised up, a hand enters the scene with another puppet, the screen comes down, the correct solution of two puppets is revealed. (c) A schematic representation of the events for the incorrect equation 1 + 1 = 1; one puppet is displayed, a screen is raised up, a hand enters the scene with another puppet, the screen comes down, the incorrect solution of one puppet is revealed.
Increase of theta power at fronto-central electrodes and larger amplitudes have also been found for predictions and anticipations of actions in 9-month-olds when presented with simple action sequences completed in an unexpected way (e.g., the action of eating ended by placing food in the ear instead of the mouth; Reid et al., 2009; although their 7-month-olds did not show the effect). In this case, the ERP component for which the amplitudes were calculated was defined as an N400. Following this study, Kaduk et al. (2016) showed that individual differences in these effects in infants at 9 months of age were related to language productive proficiency at 18 months. We note several issues regarding the findings in these two studies. First, as is typical in infant ERP data when compared with adult data, the time window of the N400 that they found was later for infants relative to adults. Second, for the infants, the scalp localization of the N400 was more parietal, and the researchers suggested that this was because it was masked in frontal and central sites by the large negative component (Nc) that was induced by the stimuli (see also Monroy et al., 2019). The Nc is an ERP component of negative voltage largely studied in infants in response to novel/unfamiliar/unpredicted stimuli (Kayhan, Meyer, et al., 2019; Langeloh et al., 2020; Luyster et al., 2014; Monroy et al., 2019; Peykarjou et al., 2020), commonly but not exclusively in the context of face perception. At young ages, it is located in the middle-central area of the brain and is mostly evident at the Cz electrode scalp location, although some studies report increasing right-lateralization with age (Luyster et al., 2014).
A more recent interesting ERP study on VoE in agent actions was conducted by Langeloh et al. (2020). They assessed infants’ ERPs when presented with videos showing a model performing unusual and inefficient actions (e.g., turning on a lamp with the forehead as opposed to turning it on with the hand), adapting the well-known behavioral paradigm first introduced by Meltzoff (1988). They analyzed the amplitudes of the same two ERP components mentioned above, however, they measured both at the same middle-central location: the Nc component in an earlier time window (400–600 ms), which they considered to reflect the amount of attentional engagement (Reynolds & Richards, 2005), and the “N400-like” component in a slower window (736–868 ms), which they considered to reflect the reaction to the semantic violation. In this case, it was found that 12- to 14-month-olds’ but not 9-month-olds’ expectations were violated; in other words, the younger infants were “surprised” when observing an inefficient action, as indicated by increased Nc and N400-like amplitudes.
Recent studies by Köster and colleagues (2019, 2021) found that for 9-month-old infants, the increase in theta brain activity to stimuli that violated their expectations lasted a few seconds (in these studies, the usual immediate ERP amplitude effect at about 200 ms was not found). Köster et al. (2019, 2021) tested four different core knowledge domains in which looking-time VoE effects have been demonstrated—action, number, solidity, and cohesion. The researchers interpreted the infant theta middle-central response to erroneous information that violated the infants’ expectations as reflecting prediction errors. The fact that the same brain response to the unexpected stimulus was found in all of the four tested domains supports the idea of a broad general mechanism eliciting the prediction response. Moreover, the theta-rhythm effect was general over the brain, in particular over parietal areas. This finding may fit well with the early development and importance of the parietal orienting network as a control system during infancy (Posner et al., 2014).
Note that a relationship between frontal theta activity and learning has been demonstrated in infancy (Begus et al., 2015). In the study of Begus et al. (2015), the EEG activity was collected while 11-month-old infants explored new objects. The researchers found that theta power predicted the infants’ subsequent recognition of these objects in a preferential-looking test. For a comprehensive review about the involvement of theta oscillations in active learning in infancy, including sustained attention, self-controlled attention, and self-guided object exploration, see Begus and Bonawitz (2020).
In sum, there is consistent EEG/ERP evidence relating middle-frontal theta to prediction errors in infants; although some clear inconsistencies should be noticed between the studies, such as some differences in the reported frequency ranges of the effects reported in different studies. For example, Berger et al. (2019) reported theta activity at a frequency of 6 to 7 Hz, Köster et al. (2021) reported theta activity at 4 to 5 Hz, Reid et al. (2009) and Begus et al. (2015) reported theta activity at 3 to 5 Hz, and so on. Some of these variations can probably be explained by the differences in the paradigms that these studies used (videos vs. visually entrained oscillations vs. live object explorations; visual, audiovisual), domains (prediction of basic features such as color and location; number; action sequences; action efficacy), and more. To provide a more coherent picture of prediction processes in infancy, these differences should not be ignored, and they certainly call for further research. Orekhova et al. (2006) suggested that there might be a shift in the specific frequency boundaries of the theta wave involved in exploration behavior and cognitive control, from 3.6 to 5.6 Hz in infancy to 4 to 8 Hz from preschool and on, which definitely deserves further investigation. Still, it seems safe to claim at this point that there is substantial electrophysiological data in 6-month-old and older infants supporting the idea that middle-central theta activity proceeding from the anterior cingulate reflects prediction errors, similar to what is found in adults. This activity is supposed to be feedforwarded to sensory areas and affects perception and learning.
In the literature on adults, there is a distinction between the source of attention control and the site at which it operates (Petersen & Posner, 2012). We have argued that a likely source is the ACC, and among the sensory sites at which it operates is the posterior occipital cortex. Although there is no available direct evidence that ties together the whole idea in infants, there is a study by Kouider et al. (2015) with 12-month-old infants that looked at the neurophysiological effect of the prediction in the occipital perceptual areas. The first phase of the study was a learning phase in which they created cross-modal associations, pairing between sounds (sounds A, B) and visual stimuli (flowers, faces). In the test phase of the study, the infants heard the sound and then the picture was presented, but in 25% of the trials, the picture was of the wrong category, that is, it induced a prediction error. At about 1,000 ms after stimulus onset, the overall activity at posterior brain areas (i.e., the ERP amplitude at these electrode sites, reflecting the perceptual processing of the stimuli) was enhanced for the unexpected stimuli. This enhancement was interpreted by the researchers as reflecting the attention and processing created by the surprise. This effect was preceded, at an earlier time following the stimulus presentation, by an opposite enhanced processing of the expected stimuli, that is, the amplitude of the P1 component was enhanced for the expected stimuli. Such early enhanced processing of the expected stimuli, which was in the opposite direction of the late and longer-lasting processing of the unexpected surprising stimuli, was attributed by the researchers to the cross-modal paradigm-cuing manipulation used in this study.
Development of Neural Connectivity Supporting Prediction Errors
Resting-state MRI has revealed evidence for a midprefrontal node of activity at the ACC, present even in newborns (Gao et al., 2015, 2017). However, this area shows little connectivity to other brain areas until later in development (Gao et al., 2011). The slow development of longer brain connections may reflect on the ability of activity in the ACC to influence sensory and motor behavior. Specifically, a question arises regarding the ages at which the connections in the infant brain could be mature enough to support prediction-error information being feedforwarded to posterior visual brain areas. A recent study of visual perception in infants by Nakashima et al. (2021) relates to the youngest age at which the infant brain is developed enough to enable prediction-error information to be feedforwarded to sensory areas, altering perception and influencing learning. In this study, 3- to 9-month-old infants were presented masked visual objects. A target object was either presented simultaneously with a mask or after a delay. In adults, the delayed mask prevents perception of the target, but the simultaneous mask does not. Only the 7- to 8-month-old infants showed evidence of such pattern masking. The younger infants looked longer at face targets in both simultaneous and delayed conditions. The authors concluded that younger infants were able to distinguish the presence of a face even under a condition in which adults could not. The authors interpreted this superior performance by infants as being due to poor top-down stimulation by pathways from frontal areas. Only at 7 months old were these pathways sufficiently mature to produce object masking. This is consistent with the ages in studies of theta-error detection, reviewed above, at which infants detect errors in perceptual displays, suggesting that these same top-down pathways could convey error information to influence perception. However, Nakashima et al.’s study suggests that these brain pathways might not yet be developed in 3-month-old infants. It seems likely that the influence of frontal areas of sensory systems may continue to increase well beyond infancy. In addition to the age limits, Nakashima et al.’s study suggests that prediction error is not the only basis for preference. When the researchers mixed faces and blanks, the older infants looked longer at faces, not because they predicted blanks and the faces deviated from prediction, but probably because faces are an especially preferred type of stimuli. Although at this point there is not much research in this direction, and therefore the conclusion should be considered with caution, Nakashima et al.’s findings raise the question of how early in infancy the connections between the different relevant areas of the brain develop, providing the infrastructure for predictive processes to occur. Feedforward predictions and prediction errors requiring connections between the ACC and sensory brain areas might be possible already at 6 to 7 months of age. Interesting supporting evidence in this respect has been found using the functional near-infrared spectroscopy technology. In a study by Emberson et al. (2015) with 6-month-old infants, the question of whether expectations about future sensory input can modulate brain activity in the infant visual cortex was tested using a cross-modal (audiovisual) omission paradigm. An expectation-based increase was found in the hemodynamic responses at the occipital cortex (when the auditory stimulus predicted the appearance of a visual one). Moreover, there was no such response in the visual cortex to auditory stimuli that were not predictive of a visual event. This pattern of results was interpreted as suggestive that “the young infant’s brain is already capable of some rudimentary form of expectation-based feedback.” Still, further development and maturation of the control networks is probably necessary before prediction errors can be reflected in the actual behavior of the child. For example, a purely behavioral study of the classical response slowing following an error in a Simple Simon game showed that it can be found in children 3 to 4 years old but not in younger toddlers (Jones et al., 2003).
Conclusions, Open Questions, and Future Research Directions
The extant empirical evidence reviewed above supports the idea that by at least 6 to 7 months of age, infants’ brain activity reflects surprise when predictions are violated in visual displays, which can be interpreted as prediction errors. Such neural responses involve theta-frequency activity, probably arising from the brain frontal midline including the anterior cingulate, which has been associated with learning in infants and adults and with increased brain connectivity in mouse studies (Piscopo et al., 2018; Weible et al., 2017).
This modern conceptualization is certainly intriguing and exciting. Yet there are many open questions arising from the presented ideas and claims that should guide future empirical investigations. It is still to be seen whether prediction errors in different domains and conditions all indeed involve one unified “control” mechanism. Moreover, it is yet to be seen whether the power of theta brain activity in infants is proportional to the discrepancy of the presented information from the predicted information and whether the perception of this discrepancy similarly follows the rules of perception of stimuli, meaning a logarithmic function, similarly to what was found by Tzur et al. (2010) in adults. At this point, there is only one study in infants (7-month-olds) that is somewhat related to this question. In the area of numerosity discrimination, both theta-band (4–6 Hz) and alpha-band (6–8 Hz) oscillatory power was found to differentiate between novel and familiar numerosities. However, at least in this case, only the power in alpha, but not in theta, was sensitive to the numerical ratio between the numerosities (Libertus et al., 2009). A further issue, as was discussed above in the section about the development of the neural connectivity supporting prediction errors, is the exact maturation timing of the brain pathways connecting the ACC and sensory areas that can support the flow of information required for the prediction processes. An intriguing opportunity that modern technology provides and that calls for serious research is the possibility to induce neuromodulation from scalp electrodes by enhancing middle-frontal theta activity, which might be able to strengthen the ACC signaling of prediction errors and therefore influence learning. Imagine, for example, using such technology for compensating the missing prediction error that the ACC should have signaled in individuals with neurodevelopmental psychopathologies involving deficits in this functioning, helping them to learn. Even theta stimulation too brief for white-matter change has been shown to produce behavioral change in attention (Reinhart & Nguyen, 2019) and memory (Roberts et al., 2018). A possible mechanism for such change is the enhanced synaptic plasticity that can be induced when rapid synaptic stimulation is synchronized to theta activity (Larson & Munkácsy, 2015). The intriguing possible modulating effects of theta induction, described in human adults and rodents, is still in very initial stages. At this point, in adults, theta electrical stimulation plus task training have enhanced behavioral performance and increased intrinsic theta (Voelker et al., 2020) but have failed to change structural white matter surrounding the ACC (Voelker et al., 2021) and have yet to be studied in infants and children.
