Abstract
Administration of Dienogest prior to hysteroscopic polypectomy is empirically performed, but the physiological effects of Dienogest on endometrial polyps are unclear. We aimed to investigate the effects of Dienogest on the proliferation and inflammation of endometrial polyps. We conducted a retrospective case study on 40 menstruating women who underwent hysteroscopic polypectomy at our hospital. We collected clinical data, and the polyps were divided by morphological appearance. The specimens obtained were immunostained for Ki67 as a marker of cellar proliferation and CD138 as a marker of plasmacytes, which are a hallmark of chronic endometritis. Dienogest significantly suppressed the proliferation status of EPs because Dienogest treatment prior to the operation significantly reduced the Ki67 index (41.25 ± 16.85 vs 7.18 ± 9.82, p < 0.01). We found that sessile-type polyps showed a significantly lower Ki67 index than the pedunculated type (12.28 ± 11.12 vs 2.09 ± 2.73, p = 0.026). The presence of CD138-positive cells was more pronounced in sessile-type polyps than in pedunculated polyps (p = 0.018). However, Dienogest treatment showed no apparent effect on inflammation status, as detected by CD138-positive cells. We revealed that Dienogest suppressed cellular proliferation, and morphological classification of endometrial polyps could be used to predict the responsiveness to Dienogest. However, Dienogest might not affect cellular inflammation.
Introduction
Endometrial polyp (EP) is defined as hyperplastic overgrowth of endometrial glands and stroma. This condition frequently causes atypical uterine bleeding in both menstruating and postmenopausal women. EP can also cause heavy menstrual bleeding and infertility. The treatment of EP is principally hysteroscopic removal of polyps because large polyps exceeding 15 mm will not be eliminated unless they are removed by surgery. 1
Dienogest (DNG) is a progestogen that is primarily used for the treatment of endometriosis and adenomyosis. 2 It binds to the progesterone receptor with high specificity, possesses endometrial anti-estrogenic effects, and it is hypothesized to inhibit the proliferation of endometrial epithelial cells. 3 DNG significantly decreased endogenous estrogen levels in endometriotic tissue, increased apoptosis of endometriotic cells, and reduced the production of pro-inflammatory cytokines from endometriotic stromal cells. 4 Moreover, some studies have indicated that administration of DNG prior to hysteroscopic polypectomy was effective in reducing the thickness of the endometrium, the severity of bleeding, and the operation time. 3 This treatment also enables us to schedule the operation day regardless of the menstrual cycle of patients. However, the physiological effects of DNG on EPs remain unclear because most of the in vitro studies using DNG use endometriotic stromal cells.
Our previous report indicated that EP could be morphologically divided into the sessile type and the pedunculated type, and sessile polyps exhibit pronounced regression after treatment with oral conceptive pills (OCP). 5 Our findings indicated that the shape of the polyps is helpful information for predicting treatment modalities, and these morphological differences might suggest the presence of different physiological features.
Here, we aimed to investigate the effects of DNG on the proliferation status and inflammation of EPs and assessed the effect of morphological classifications.
Materials and methods
We conducted a retrospective case study on 40 menstruating women under 50 years old. This study was approved by the ethics board of University of Tokyo Hospital and written informed consent was obtained from all patients in this study. The subjects underwent hysteroscopic resection of EPs following the hysteroscopy and were diagnosed with EP by histopathologic examination from March 2011 to December 2016 in our institution. Twenty of 40 received DNG treatment. DNG treatment (2 mg/day: 10–30 days: median 17.5 ± 5.63 days) was initiated on days 3–5 of the early follicular phase of menstruation. We surveyed the following parameters by the electronic patient records: age, menstrual cycle, pathological diagnosis, prescription of DNG prior to the operation, the presence of endometriosis or other complications, infertility, and pregnancy after polypectomy. Endometriosis was defined as the presence of endometrioma exceeding 2 cm or past operation history. Infertility and pregnancy after polypectomy were surveyed until April 2019.
We divided the patients into 4 groups of 10 patients each by the shape of the polyp (sessile type or pedunculated type) and DNG treatment prior to the operation. The shape of the polyp was determined by the images from hysteroscopy, and the polyps possessing pedicles were defined as the pedunculated type. All tissue samples of the EPs were obtained by hysteroscopic surgery. Hematoxylin–eosin staining was conducted for the diagnosis of EP. Immunohistochemistry was conducted for Ki67 to detect proliferating cells and CD138 to detect plasmacytes. Immunohistochemical staining was carried out using the following antibodies: Monoclonal Mouse Anti-Human Ki67 Antigen, clone MIB-1 (M7240; Dako, Carpinteria, CA, USA) and ready-to-use mouse anti-human syndecan-1 monoclonal IgG (B-A38; Nichirei Biosciences, Inc., Tokyo, Japan).
The number of Ki67-positive stained nuclei and total number of epithelial gland cells were counted, and the percentage of the positive nuclei (Ki67 index) in five random magnification fields (400×) was calculated.4,6,7 Because four cases in the non-DNG group were in the secretory phase or an unknown phase of the menstrual cycle, their Ki67 indexes were not calculated. The number of CD138-positive plasmacytes was counted in 10 random nonoverlapping magnification fields (400×), 8 and the presence of inflammation was defined when six or more plasmacytes were observed. Less than five cells was graded as negative (−), from 6 to 50 cells was positive (+), and more than 51 cells was strongly positive (++).
Statistical analysis was carried out using R-3.4.0. 9 Fisher’s exact test was used for comparison of binary date in unpaired samples. Wilcoxon rank sum test was used for comparing continuous data between two groups. A p value of less than 0.05 was considered to be statistically significant for all tests.
Results
Table 1 shows the clinical characteristics of the patients, and they were divided in the sessile and pedunculated polyp groups. There were no statistically significant differences in all parameters.
Clinical data of patients.

Results are given as mean ± SD or percentage and number.
DNG: Dienogest, NS: not significant.
Exact Wilcoxon rank sum test.
Fisher’s Exact Test.
Infertility was defined as 1 year regardless of unprotected intercourse.
Polyps without DNG treatment exhibited a relatively high Ki67 index and the type of polyps did not affect the Ki67 index (pedunculated: 37.54 ± 18.84 vs sessile: 44.13 ± 14.48, p = 0.697; Figure 1). DNG treatment significantly decreased the Ki67 index in both the pedunculated and sessile polyp groups, indicating the anti-proliferative effects of DNG. Furthermore, we found that sessile-type polyps exhibited a lower Ki67 index than the pedunculated type in the DNG-treated group. (12.28 ± 11.12 vs 2.09 ± 2.73, p = 0.026; Figure 1).
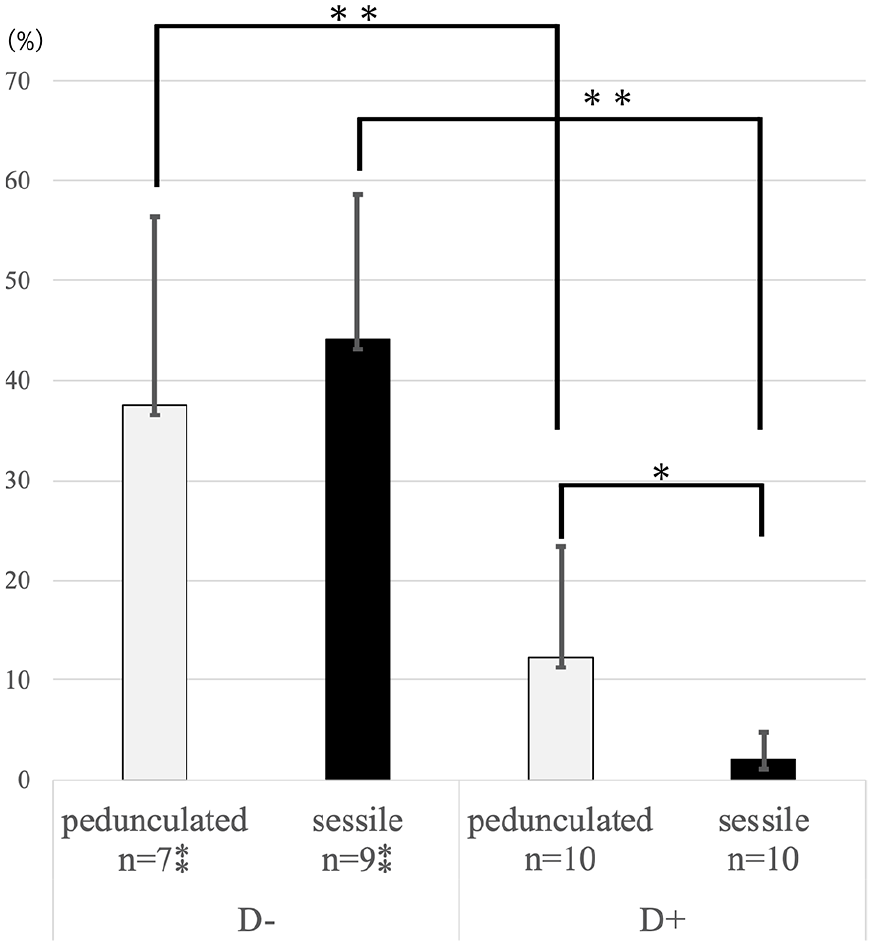
Analysis of the Ki67 index. The Ki67 index without DNG treatment was approximately 40% in both pedunculated and sessile polyps, and there was no significant difference between these groups. DNG treatment significantly decreased the Ki67 index in both groups, and we found that sessile-type polyps exhibited a lower Ki67 index than pedunculated-type polyps in the DNG-treated group (12.28 ± 11.12 vs 2.09 ± 2.73, p = 0.026). D− denotes the group without DNG treatment and D+ denotes the group with DNG treatment.
Figure 2 shows the immunohistochemical analysis of CD138-positive cells in the polyps. The number of patients diagnosed with inflammatory polyps was 32 in 40 (80%) in total. We found that a significant number of polyps had CD138-positive cells. Figure 2(a) shows the difference in CD138 positivity between the pedunculated- and sessile-type polyps without regard to DNG treatment. When Fisher’s exact test was conducted, there was a significant difference (p = 0.018), and sessile-type polyps tended to have strong inflammation. Moreover, considering the DNG treatment, there was no difference in each group (DNG group: p = 0.314, non-DNG group: p = 0.178; Figure 2(b)).
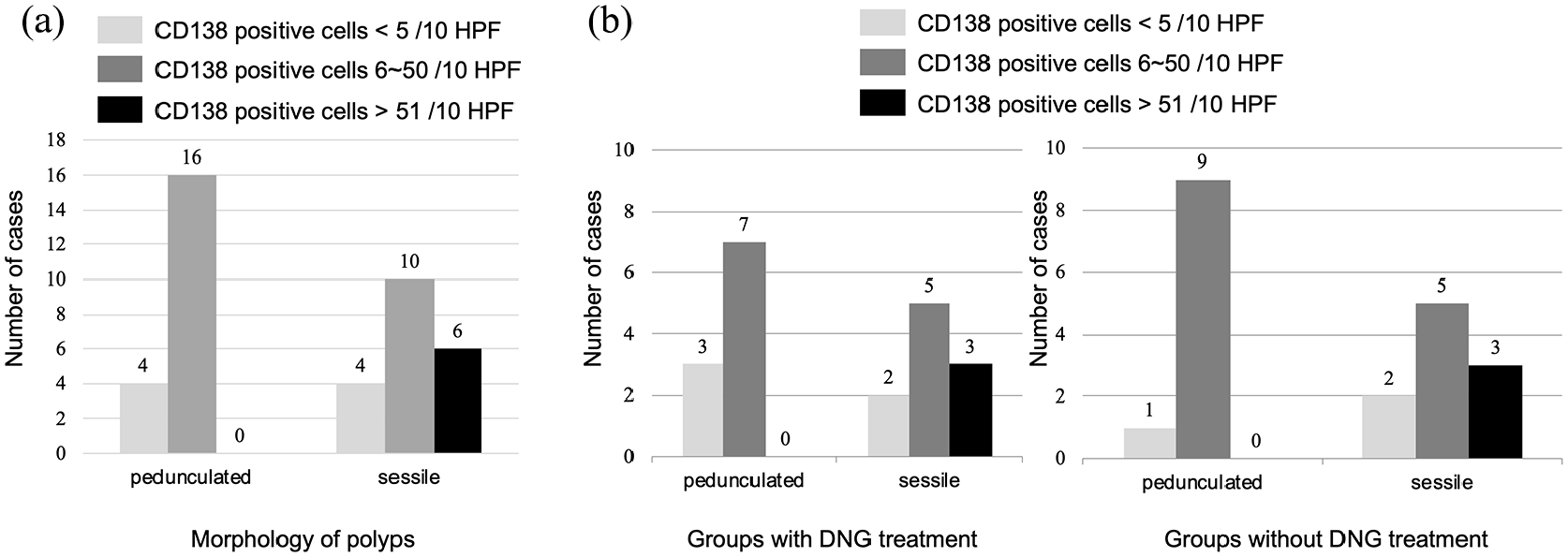
CD138 positivity and morphological classification. Results of the immunohistochemical analysis of CD138 are shown. CD138-positive cells in 10 HPF (400×) were confirmed; (−) denotes less than five cells were positive for CD138, (+) denotes 6–50 cells were positive for CD138, and (++) denotes more than 51 cells were positive for CD138. (a) Considering treatment of DNG, there was no significant difference between the sessile type and pedunculated type. (D+: p = 0.314, D−: p = 0.178). (b) However, when Fisher’s exact test was conducted between pedunculated-type and sessile-type polyps regardless of the DNG treatment, there was significant difference (p = 0.018). D− denotes the group without DNG treatment and D+ denotes the group with DNG treatment.
Discussion
In the present study, DNG showed robust anti-proliferative effects on EPs, especially on sessile polyps. Our previous report similarly showed that sessile-type polyps were more likely to respond to OCP treatment than pedunculated-type polyps. 5 The effect might be related to the progestin contained in OCP. DNG is a fourth-generation progestin and it exhibited anti-proliferative effects. Prior to hysteroscopic surgery, gonadotrophin-releasing hormone analogues (GnRHa), danazol, and DNG are commonly used for the purpose of thinning the endometrium, shortening the duration of surgery, and easing of surgery. 10 DNG has specific activities on progesterone receptor; thus, it is exempt from androgenic and estrogenic activities.3,11,12 The main clinical indication of the use of DNG is for reducing endometriosis, and several in vitro studies have indicated that DNG directly shows anti-proliferative activity on endometriotic lesions and suppresses the secretion of the cytokines in the endometriotic stromal cells. 13 Our results similarly demonstrated that EP responds DNG and the usefulness of DNG treatment prior to endometrial polypectomy is not only due to the effect of endometrial thinning but also to shrinking EPs; thus, hysteroscopic surgery might be easier and safer compared to that without pretreatment. This study first indicates that DNG shows strong anti-proliferative activity against EPs, which was evidenced by immunostaining of Ki67.
We also observed that sessile polyps show an indistinguishable appearance compared to micro-polyps observed in chronic endometritis; 14 thus, we further aimed to investigate the probable correlation between DNG treatment and the status of inflammation in polyps. CD138 positivity was high in all types of EPs in this study. We should note that strong positivity (++) of CD138 immunostaining appeared only in sessile-type polyps. To the best of our knowledge, only the study by Cicinelli 14 indicated the association between EPs and the presence of plasmacytes. According to their study, the CD138 positivity in patients with EPs was 76.7% (184 of 240), similar to our study. These facts suggest that women who possess sessile-type EPs should be assessed for chronic endometritis if the patients are infertile because the presence of plasmacytes is frequent in patient with EPs. Recent studies strongly suggest the usefulness of antibiotics therapy because it could improve their fertility.15–18 Although the efficacy of hysteroscopic inspection in all patients prior to in vitro fertilization (IVF)–embryo transfer (IVF-ET) treatment is supposed to be doubtful, we would like to suggest that the morphological classification of EP can be useful to detect the concomitant presence of chronic endometritis.
In case of patients with submucous myoma as well as EP, preoperative DNG administration can be a used for pretreatment prior to the hysteroscopic surgery. Management of myomas depends on the location, number and volume of them and hysteroscopic diagnosis and treatment is a gold standard for the treatment of G0 and G1 myomas.19,20 The diagnosis and treatment of EPs by hysteroscopy is also similar. However, the management of EPs by morphological classification has been poorly studied. Our study suggests that the difference of types can be an important clinical feature for the proper management of EPs. Furthermore, it is desirable that medicine alone can decrease clinical symptoms such as abnormal uterine bleeding by DNG administration. Incidentally, in case of patients who are concerned about gynecological examination or have difficulties in reaching the uterine cavity, hysteroscopy might be an unpleasant and painful procedure.21,22 Hysteroscopic polypectomy can be avoided, which will reduce patients’ anxiety and pain from the operation.
There are some limitations to our investigation. The first limitation is related to the study design, including the small sample size, retrospective study, and varied duration of DNG administration. It is desirable for a clinical study to include a large number of participants and to have a prospective design. Second, strict diagnosis criteria of chronic endometritis have not been established and there are no technical standards and conditions for CD138 immunostaining. 23 At present, the diagnostic method of chronic endometritis differs among institutions. It is necessary to standardize the diagnostic criteria and technique and CD138 immunostaining for a more accurate study.
Conclusion
DNG prescription prior to hysteroscopic surgery of EPs has inhibitory effects on cellular proliferation. We also found that patients with EPs might be significantly complicated by chronic endometritis. It is assumed that the immunohistological examination of CD138 could be important for infertile patients with EPs, especially those with sessile types. However, we should further investigate the physiological difference between micro polyps and sessile polyps because they are not regarded as the same ones so far.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grant-in-Aid for Scientific Research from the Ministry of Education, Science and Culture (grant no. 18k09248), the Japanese Foundation for Research and Promotion of Endoscopy Grant, the Japan Agency for Medical Research and Development (grant nos 17gk0110014h0002 and 18k0210018h0001), and Ministry of Health, Labor and Welfare.
