Abstract
Chronic pelvic pain is one of the most prevalent syndromes in family medicine, gynecology, and urology. Chronic pelvic pain patients are often misdiagnosed and have unnecessary prolonged suffering. Both misdiagnosis and delay in treatment negatively impact the health and well-being of chronic pelvic pain patients while additionally creating an economic burden on healthcare systems. This review aims to combine current treatment guidelines for managing chronic pelvic pain into a comprehensive algorithm for healthcare providers, with a focus on early diagnosis and treatment. The algorithm encompasses pathophysiology, assessment, organ system dysfunction management, overlapping pain syndromes, integrative management, pharmacological management, non-operative multidisciplinary care, and surgical interventions for chronic pelvic pain. A review of medical literature using the keyword “Chronic Pelvic Pain” between January 1, 1988 and December 31, 2023, was conducted using the PubMed database. Four hundred three articles from the PubMed database were eligible for detailed review. Each publication was evaluated utilizing a grading system adapted from the framework established by the Oxford Centre for Evidence-Based Medicine. For guideline development, a panel of experts specializing in pelvic pain determined how physicians can best optimize the diagnosis and treatment of chronic pelvic pain by integrating current treatment guidelines, best practice recommendations, and available data. The recommendations advocate for a multimodal approach to chronic pelvic pain treatment, emphasizing the importance of early disease recognition, coordinated care, and treatment protocols similar to those commonly employed for well-defined medical conditions such as asthma and diabetes.1–5 This article introduces a comprehensive treatment algorithm designed to address the existing gaps in the management of patients suffering from chronic pelvic pain, as well as decrease the economic burden on the healthcare system that chronic pelvic pain currently places. The algorithm promotes a comprehensive multimodal approach to prevent the prolonged suffering that chronic pelvic pain patients currently must endure.
Keywords
Introduction
Chronic pelvic pain (CPP), one of the most prevalent syndromes in family medicine, gynecology, and urology, is defined as continual, non-cyclic pain lasting at least 6 months.6,7
CPP is estimated to affect up to 24% of women of reproductive age and ~14% of women at least once in their lifetime.8,9 The prevalence of CPP in men ranges between 10% and 16%.10 –12 Patients with CPP often report a lower quality of life, sexual function, mental well-being, and productivity. 13 In addition, 87% of female patients with CPP are diagnosed with endometriosis. 14 Both endometriosis and CPP are referred to as a “silent” disease process, which cannot be detected through standard exams such as blood work, cultures, or imaging.15 –18 Other common comorbidities include adenomyosis, fibroids, post-partum pelvic pain, interstitial cystitis (IC), bladder pain syndrome (BPS), irritable bowel syndrome (IBS), anxiety, and depression. 10
Despite ongoing research, there is currently no scientifically validated marker or clear etiology for CPP.6,7,10 CPP is understood to stem from the interplay between gynecological, urological, gastrointestinal, neurological, psychological, and neuro-musculoskeletal system dysfunctions.19 –21 The multi-factorial nature of CPP, coupled with the absence of definitive markers, often leads to delays in both diagnosis and treatment. As a result, patients frequently need to see multiple healthcare providers, which can result in “normal” workups that fail to validate their pain or define the source of pain generation.22 –24 Misdiagnosis is common among patients with CPP, particularly due to the lack of clarity regarding the underlying primary pain generators and the absence of an interdisciplinary treatment plan to address these issues.22 –24
This publication aims to create a comprehensive treatment algorithm for healthcare providers to treat and manage CPP. The algorithm should encompass pathophysiology, assessment, organ system dysfunction management, integrative management, pharmacological management, non-operative care, and surgical interventions while considering overlapping pain syndromes.
Methods
A panel of nine experts specializing in pelvic pain was convened by the Pelvic Rehabilitation Medicine Clinical Research Foundation. This panel assessed the key question: “How can physicians optimize the diagnosis and treatment of Chronic Pelvic Pain?” A review of medical literature between January 1, 1988 and December 31, 2023, was conducted using PubMed to identify articles addressing the pathophysiology, diagnosis, and management of CPP across various organ systems. A search using the keyword “Chronic Pelvic Pain” resulted in 468 articles. Of these, 50 were excluded because they did not pertain specifically to the disease under review. Subsequently, a search was performed combining the keyword “Chronic Pelvic Pain” and a keyword for each organ system assessed by this guideline for its predisposing factors and/or comorbidities that contribute or are associated with CPP: Gynecological, Urologic, Gastrointestinal, Colorectal, Musculoskeletal, Neurologic, Vascular, and Psychological. The searches yielded a total of 112 articles between 1988 and 2021. Of these, 15 were excluded and a total of 403 articles were reviewed. For each remaining article, the quality of evidence was assessed using a grading system modified by the Oxford Centre for Evidence-Based Medicine. 25 Articles were eligible for inclusion if they addressed adult populations with CPP, presented clinical outcomes, treatment approaches or diagnostic algorithms and were published in English. Articles were given a score from 0 to 5 for relevance to the research question, study design and level of evidence, methodological quality, data quality and reporting, clinical and scientific impact, publication quality and credibility, and recency and applicability, yielding a total score between 0 and 25. Of the articles evaluated, 131 were highly graded, reflecting high-quality evidence, relevance, and data transparency, and were incorporated into this review. Due to heterogeneity across study designs and outcomes, a formal meta-analysis was not performed. Instead, data were synthesized descriptively, with comparative narrative analysis across major themes (e.g. physical therapy, neuromodulation, psychological interventions, pharmacologic management).
Articles were organized by topic: pathophysiology, diagnosis, or treatment. Pathophysiology articles were separated by nervous system dysfunction, organ system dysfunction (which were further divided into seven subcategories), and overlapping pain syndromes.
Treatment articles were sorted into medical, surgical, or interventional categories. By integrating available data from current treatment guidelines, best practice recommendations, randomized controlled trials, systematic reviews, and published case reports, consensus was reached by the panel on a comprehensive treatment algorithm for physicians.
Physical diagnostic assessment
As illustrated in Figure 1, when a patient presents with symptoms associated with CPP, the first course of action is a physical diagnostic assessment, which includes a complete medical history and a physical examination. A complete medical history encompasses a review of previous treatments, a detailed chronology of symptoms, past medical diagnoses, and surgical history. Diagnostic assessments for CPP also often include patient-reported outcomes (Figure 2).

Article selection process.
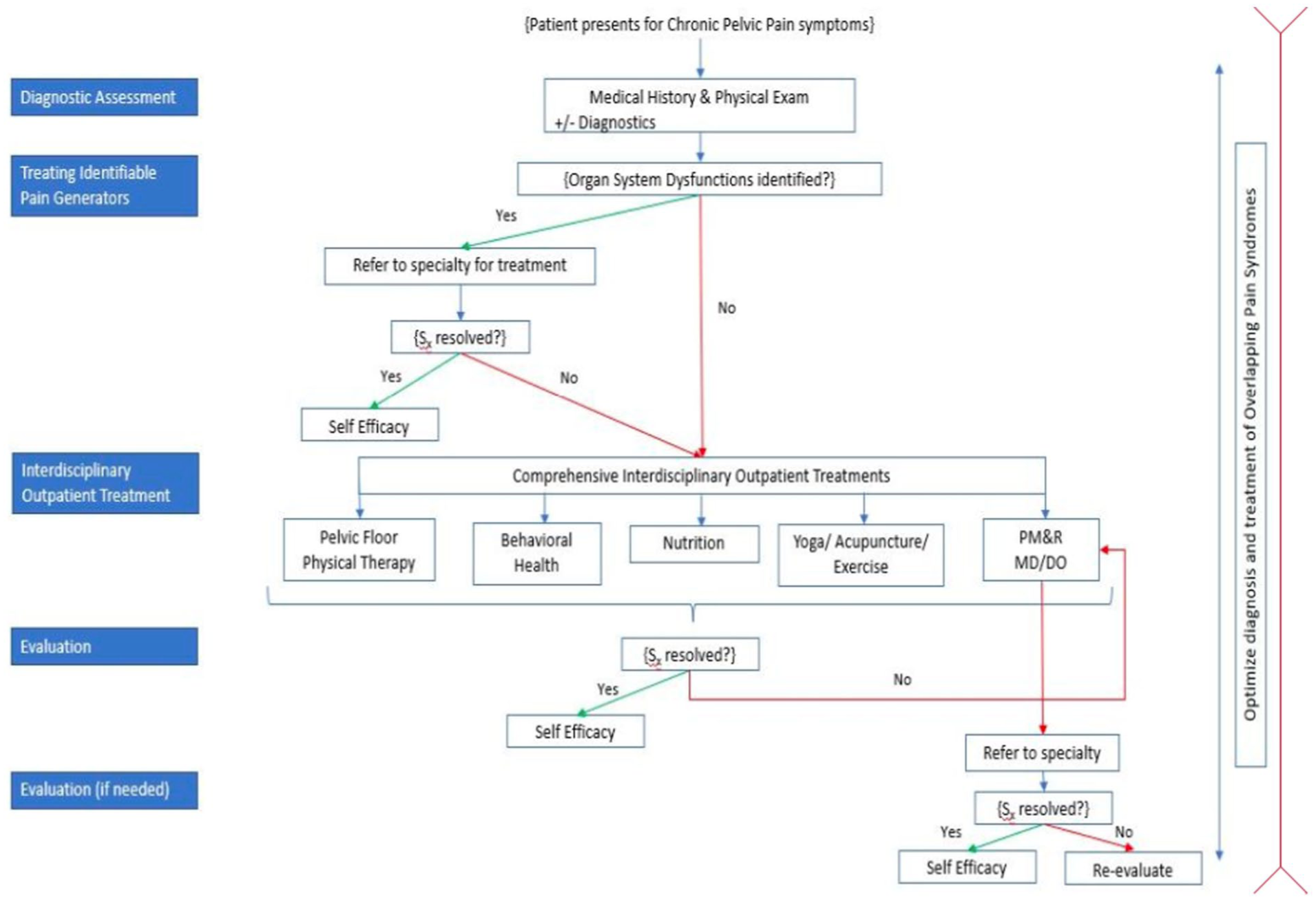
Physical diagnostic assessment and evaluation methods for patients with chronic pelvic pain.
Patient-reported outcomes can provide insight into a patient’s pain experience and demonstrate improvements after proposed treatments. Typically, validated questionnaires for CPP patient-reported outcomes include the Visual Analog Scale (VAS), 26 Impact of Female CPP, 27 Female Sexual Function Index, 28 Chronic Prostatitis Symptom Index (CPSI), 29 Hospital Anxiety and Depression Scale, 30 and the International Index of Erectile Function. 31
Due to the multifaceted nature of CPP, it is essential to review diagnostic assessments from the perspective of the typical specialties that may be involved in caring for CPP patients, including gynecologists, urologists, colorectal surgeons, gastroenterologists, non-operative pelvic floor neuromuscular providers, and pelvic floor physical therapists. Therefore, this algorithm will detail best practices for each diagnostic assessment that may be involved in the care of CPP patients.
A diagnostic assessment with a gynecological provider should begin by evaluating the external vulvar area for any signs of swelling, discharge, growths, lesions, rashes, erythema, bruising, or tenderness.31,32 An internal exam follows to assess the cervix for color, discharge, blood, and lesions.31,32 The positioning of the external and internal os should also be checked, and samples of tissue may be taken as necessary. 32 Vaginal walls and both adnexa must be evaluated for masses and tenderness. 32 A rectovaginal exam can be done to assess the rectovaginal septum and posterior cul-de-sac to note any tenderness, polyps, hemorrhoids, and growths. 31 It is also essential to identify uterine fibroids, endometriosis, cervical polyps, and dysfunction of the pelvic wall and floor muscles.32,33 Laparoscopy is often necessary since abnormalities may not be detected during a physical examination and is considered the gold standard for visualizing endometriosis lesion phenotypes as peritoneal, cystic, or deep. 34 Imaging methods such as transvaginal ultrasound, transrectal ultrasound (TRUS), or MRI can also be employed to assess deep infiltrating endometriosis. 35 Pelvic and transvaginal ultrasounds can also be used to examine for signs of adhesions, such as adnexal abscesses, peritoneal inclusion cysts, and hydrosalpinx. 30 Pelvic and transvaginal ultrasounds and ultrasound imaging can be used to diagnose adenomyosis by inspecting the myometrium for non-vascularized lacunae and observing the uterus.36,37 Patients with adenomyosis often have an enlarged, heterogeneous uterus with wall asymmetry and possible linear striation that can be either hyperechoic or hypoechoic. 37 MRI is also useful for visualizing adenomyosis, pelvic masses, tubo-ovarian abscesses, thick adhesions, abnormalities in ovarian veins, and pelvic varicosities.37,38 A transvaginal ultrasound is often recommended when a patient is experiencing adnexal or uterine tenderness when there is suspicion of a pelvic mass or to evaluate for chronic pelvic inflammatory disease. 39 A Doppler ultrasound examination may also be performed to diagnose pelvic venous insufficiency (PVI) by allowing assessment of venous blood flow. 36 The Q-tip test should be used to diagnose vulvodynia and differentiate between involvement of the ilioinguinal or genitofemoral nerve or the pudendal or posterior femoral cutaneous nerve.21,40,41
Multiple tests are used in a urological diagnostic assessment. A critical test known as urinalysis covers urinary tract screening in addition to diabetes, kidney disease, liver disease, urinary tract infections (UTIs), and urothelial cancer.42,43 During a workup, urinalysis and additional tests such as mid-stream urine (MSU) cultures and two-glass prostate tests must be conducted to determine the involvement of a bacterial infection.42 –47 MSU cultures should be used to confirm UTIs. 44 If a UTI is ruled out, conditions such as overactive bladder (OAB), BPS, or IC may become more likely diagnoses. 48 OAB is characterized by the presence of urinary urgency in the absence of UTI or neurologic conditions. 49 Urgency, frequency, and nocturia are symptoms of BPS, IC, and OAB.44 –51 However, these conditions can be differentiated by additional symptoms: patients with BPS or IC may experience dyspareunia, pain during bladder filling, and pelvic pain, while those with OAB often experience urge urinary incontinence.44 –51 When IC is suspected, a cystoscopy can be performed to evaluate the lining of the bladder. 51 In addition to these diagnostics for assessment of CPP and related disorders, the 2-glass or 3-glass prostate test should be used to diagnose chronic prostatitis (CP) and CPP syndrome (CPPS).45 –47 Furthermore, a TRUS can be used to rule out a prostate cancer diagnosis as a source of CPP symptoms. 50
Diagnostic assessments for CPP in the context of colorectal and gastrointestinal disorders focus on examining and excluding common disorders.48,49,51,52 A digital rectal exam, perineal examination, proctoscopy, urogenital exam, endoscopy, and colonoscopy can be done in addition to a diagnostic colorectal and gastrointestinal assessment to evaluate for inflammatory bowel disease (IBD), IBS, and microscopic colitis as a potential source for gastrointestinal symptoms. 53 An IBS diagnosis relies primarily on information given to the physician by the patient and the exclusion of other potential pain generators. 52 Like IBS, proctalgia fugax can also be diagnosed by the exclusion of other pelvic and anorectal abnormalities. 54 During a diagnostic assessment for CPP, anal fissures, straining, and IBD can easily be excluded due to their perineal presentation. 52 Another potential avenue for CPP providers to explore is small intestinal bacterial overgrowth (SIBO). 55 SIBO can be identified through a hydrogen breath test, glucose or lactulose breath testing, or a culture of small bowel aspirates.55,56 A clinical evaluation should also include defecography, dynamic MRI, anal manometry, and an endo-anal ultrasound to determine the presence of obstructed defecation syndrome (ODS). 57
Neuromuscular diagnostic assessments for CPP patients are often completed by non-operative pelvic floor neuromuscular providers.58 –60 These specialists focus on the pelvic neuromusculoskeletal system, abdominal system, and visceral examination to uncover neuromuscular CPP etiologies. They include physical medicine and rehabilitation specialists, family medicine doctors, and nurse practitioners.58 –60 When evaluating for CPP, specialists should conduct an internal pelvic floor examination consisting of palpating the levator ani and obturator internus muscles,17,61 a complete lumbosacral examination, a bilateral hip examination, 21 and an abdominal exam. 62 Specialist evaluation by a pelvic floor physical therapist should include an examination of the patient’s posture, gait, and range of motion 17 ; stability and alignment of the spine while sitting, standing, and during other functional positions and movements; breathing techniques and abdominal wall; and movement patterns as well as mobility and tonicity of the spinal, abdominal, and pelvic areas.63,64 To fully assess pelvic floor muscles in CPP patients, physical therapists should first conduct a neuromuscular assessment to examine coordination and relaxation of muscle activity using various palpation techniques.17,63 Next, the provider should examine the buttocks and ab muscles; and, finally, use a gloved finger to palpate the pubococcygeus, iliococcygeus, coccygeus, and obturator internus internally both vaginally and rectally with gentle turning motions while observing any pain reaction for each muscle, and assessing the tone, coordination, and tautness in the muscles and connective tissues. 58
A multimodal interdisciplinary treatment approach for CPP
After a diagnostic treatment is performed, it is essential for each specialist to treat each identifiable pain generator. Due to the interdisciplinary nature of CPP, as shown above, it is essential to utilize this multimodal approach in order to address all symptoms and improve the quality of life for patients.
Various gynecological conditions associated with CPP and diagnosed during assessment for CPP, such as endometriosis, adenomyosis, vulvodynia, adhesions, fibroids, polycystic ovary syndrome (PCOS), vaginismus, and persistent genital arousal disorder (PGAD), require tailored treatment approaches.7,13,32,40,41,65 –68 For endometriosis, management may involve NSAIDs, 65 hormonal therapy, 69 and laparoscopic surgery or resection, 70 alongside lifestyle strategies like meditation. Patients diagnosed with adenomyosis often necessitate interventions, including uterine artery embolization (UAE) or cytoreductive surgery.71,72 Vulvodynia treatments may include anti-inflammatories, 73 neuromodulators like gabapentin, cognitive behavioral therapy (CBT), 74 pelvic floor physical therapy (PFPT), 75 or last-resort surgeries to remove hypersensitive tissue. 76 Adhesions might require surgical lysis 77 or medication like antidepressants or gabapentinoids. 78 Fibroids can be addressed through myomectomy,79,80 UAE, magnetic resonance-guided focused ultrasound surgery, or vaginal occlusion of uterine arteries.80,81 PCOS management focuses on lifestyle modifications to decrease BMI. 66 Vaginismus treatment typically involves PFPT, 67 medication, 82 and CBT. 83 PGAD may require a multidisciplinary approach involving medication, PFPT, and sex therapy. 68
Urological conditions such as IC/BPS and CP/BPS also require specialized treatment strategies. For IC/BPS, interventions may be physical and pharmacological. Typical interventions include myofascial physical therapy, 84 global therapeutic massage, complementary and alternative medical treatments, 85 botulinum toxin-A (BTX-A) injections,86 –88 and sacral neuromodulation. 60 When engaging in myofascial physical therapy, patients were referred for a full history and evaluation by a pelvic floor physical therapist, to assess posture, neuromuscular imbalances, musculoskeletal comorbidities, and pelvic floor tone and strength. In CP/BPS management, α-adrenergic antagonist therapy like tetracycline hydrochloride may be prescribed. 89 Physical and pharmacological treatments are often combined for OAB treatment. OAB patients often undergo PFPT with myofascial release, 90 in addition to anti-muscarinic and β3-adrenoceptor medications 49 or specific medications such as solifenacin with mirabegron91,92 intravaginal estradiol cream with tolterodine 93 to address symptoms effectively.
For gastrointestinal and colorectal conditions like IBS, a multifaceted approach is also often employed. Management typically includes dietary adjustments such as a low FODMAP diet to alleviate symptoms. 94 For diarrhea-predominant IBS, anti-diarrheal medications like loperamide may be recommended. 95 Pain associated with IBS can be addressed using tricyclic antidepressants (TCAs), selective serotonin reuptake inhibitors (SSRIs), or specific agents like tricyclic serotonin reuptake inhibitors or serotonin-norepinephrine reuptake inhibitors (SNRIs), 96 alongside probiotics 97 for symptom relief. For constipation-predominant IBS, bulking agents such as psyllium, calcium polycarbophil, or bran can help regulate bowel movements, and rifaximin may be prescribed to manage symptoms effectively. 98 Treatment for SIBO often involves antibiotics such as rifaximin and tegaserod. 99 Herbal medicines and probiotics may also be considered as adjunctive therapies.56,100 In cases of Clostridium difficile infection, fecal microbiota transplant is a well-established treatment option. 101 Proctalgia fugax, characterized by fleeting rectal pain, can be managed conservatively with measures like heating pads or rubber rings alongside medications such as SNRIs, TCAs, gabapentin, or cyclobenzaprine, 102 and potentially BTX-A injections. 103 For ODS, interventions may include a high-fiber diet, hydration, and laxatives, 104 as well as biofeedback, enemas, suppositories, 105 and BTX-A injections. 106 Surgical options like transvaginal, transabdominal, or transanal procedures may be considered for refractory cases. 107 These treatments aim to alleviate symptoms and improve the quality of life for individuals with these gastrointestinal and colorectal conditions.
Nutritional modifications may also prevent worsening symptoms in CPP patients with BPS. Suggestions include monitoring the consumption of caffeine, soya, tomatoes, pepper, citric fruits, and food high in sodium content. 13 Patients with CP/CPPS have also shown worsening symptoms with grapefruit juice, spicy foods, alcohol, and caffeinated coffee. Patients showed improvement with alternative food, especially water and Prelief® acid reduction tablets. 85
In the realm of neuromuscular conditions, various treatment modalities are employed depending on the specific diagnosis. Underlying neuromuscular dysfunction exists in 87% of CPP patients. 59 Treating the underlying neuromuscular dysfunction in combination with a root cause analysis looking for potential underlying organ-specific causes is a must, as shown above. Patients should be referred to specialists to treat any underlying organ system dysfunction. General approaches often include analgesics like NSAIDs or acetaminophen, neuromodulators such as gabapentin or amitriptyline, and muscle relaxants for pain management. 17 Pelvic floor trigger points may be addressed through injections,17,108 biofeedback, or neuromodulators, excluding TCAs. 109 Nonrelaxing pelvic floor dysfunction may benefit from neuromodulators. 109 Labral tears or femoroacetabular impingement may undergo conservative treatments like rest, NSAIDs, or physical therapy,110,111 and in severe cases, surgical interventions like labral repair or debridement. 105 Hernias, both inguinal and femoral, typically require surgical repair.112 –114 Sacroiliac joint dysfunction may be managed conservatively through physical therapy, chiropractic care, medication, or nerve block injections.115 –117 Symphysis pubis dysfunction treatments often include physical therapy, analgesics, acupuncture, and pelvic support garments, with pelvic belts or girdles used for additional support.118,119
To treat neurologic and vascular conditions, tailored treatment strategies are essential for addressing specific diagnoses. CPP patients with pudendal neuralgia may be treated through a combination of nerve blocks, 120 PFPT, and medication. 121 More invasive options like perineural blocks, spinal cord modulation, or laparoscopic nerve surgery may be considered if conservative measures fail.121 –123 Treatments for ilioinguinal, iliohypogastric, and genitofemoral neuralgias include nerve rest, posture correction, exercise, or local anesthetic injections like lidocaine to targeted areas.122,124 In cases of refractory neurologic symptoms, procedures such as genitofemoral laparoscopy or open neurectomies may be warranted. For PVI, medical management with hormonal therapy like medroxyprogesterone acetate or goserelin,125,126 contraceptive measures such as Implanon, 127 and interventional options like transcatheter embolization or embolotherapy are commonly employed.38,128 In severe cases of CPP, surgery such as hysterectomy coupled with hormone therapy may be recommended,129,130 however, this is rarely used as modern endovascular treatments are more effective and less invasive. 38
Lastly, psychological conditions such as depression and anxiety often occur in conjunction with CPP. Women with CPP have demonstrated a significantly higher prevalence of anxiety and depression than those without pain, with significant impacts in physical, psychological, and social domains, demonstrating a lower quality of life. 8 To address this, a multifaceted approach of psychotherapy, lifestyle medication, and pharmacological management is often recommended. For CPP patients experiencing depression and anxiety, interventions may include lifestyle changes such as regular exercise, psychotherapy, medications such as SSRIs or benzodiazepines, and electrotherapy.131 –135 CBT may also prove beneficial. In a study of women with vulvodynia, those who participated in CBT demonstrated significantly greater improvement in pain severity during physician examination, sexual function, and treatment satisfaction than those who participated in supportive psychotherapy. 74 In women with dyspareunia, those who received group CBT showed significantly more reduction in both pain and pain catastrophizing, and significantly higher treatment satisfaction than women who just received a topical steroid. 78 Yoga, specifically as an exercise lifestyle modification, has been found to improve quality of life outcomes for CPP patients.136 –138 In a study of 60 female patients living with CPP, patients who participated in 8 weeks of yogic intervention showed a significant decrease in VAS pain scores and increase in psychological and social domain scores. 136
These approaches aim to address both the biological and psychological aspects of the CPP experience, helping individuals manage symptoms and improve their overall well-being.
Pelvic floor neuromuscular treatment for CPP with PRM protocol™
The PRM Protocol™ is a simple, safe office-based procedure to treat CPP in the setting of hypertonic pelvic floor musculature.139 –146 The PRM Protocol™ consists of a series of selective pelvic peripheral nerve block and trigger point injections to address the triad of myofascial pain, peripheral sensitization, and central sensitization, concomitantly promoting neuroplasticity.139 –146 The office-based procedure is external, ultrasound-guided, with a smaller than a traditional needle (27 G).139 –146 No anesthesia and downtime are required.139 –146 The PRM Protocol™ treats the cross-sensitization between adjacent pelvic nerves as well as using high-volume ultrasound-guided injections directed into spaces in the pelvis that are restricted and hypertonic to create space and release fascial restrictions, which contribute to neural ischemia and inflammation.139 –146 The goals of the neurogenic inflammation, decreasing muscle tension, and removing fascial restrictions to reverse PRM Protocol™ are to retrain the central and peripheral nervous system by decreasing local neuronal ischemia of pelvic peripheral nerves.139 –146 After this is achieved, the next step is to lengthen and strengthen pelvic, hip, and deep spine musculature with a neuromuscular reeducation program.139 –146 A summary of treatment options discussed can be found below (Table 1).
Treatment options in summary.
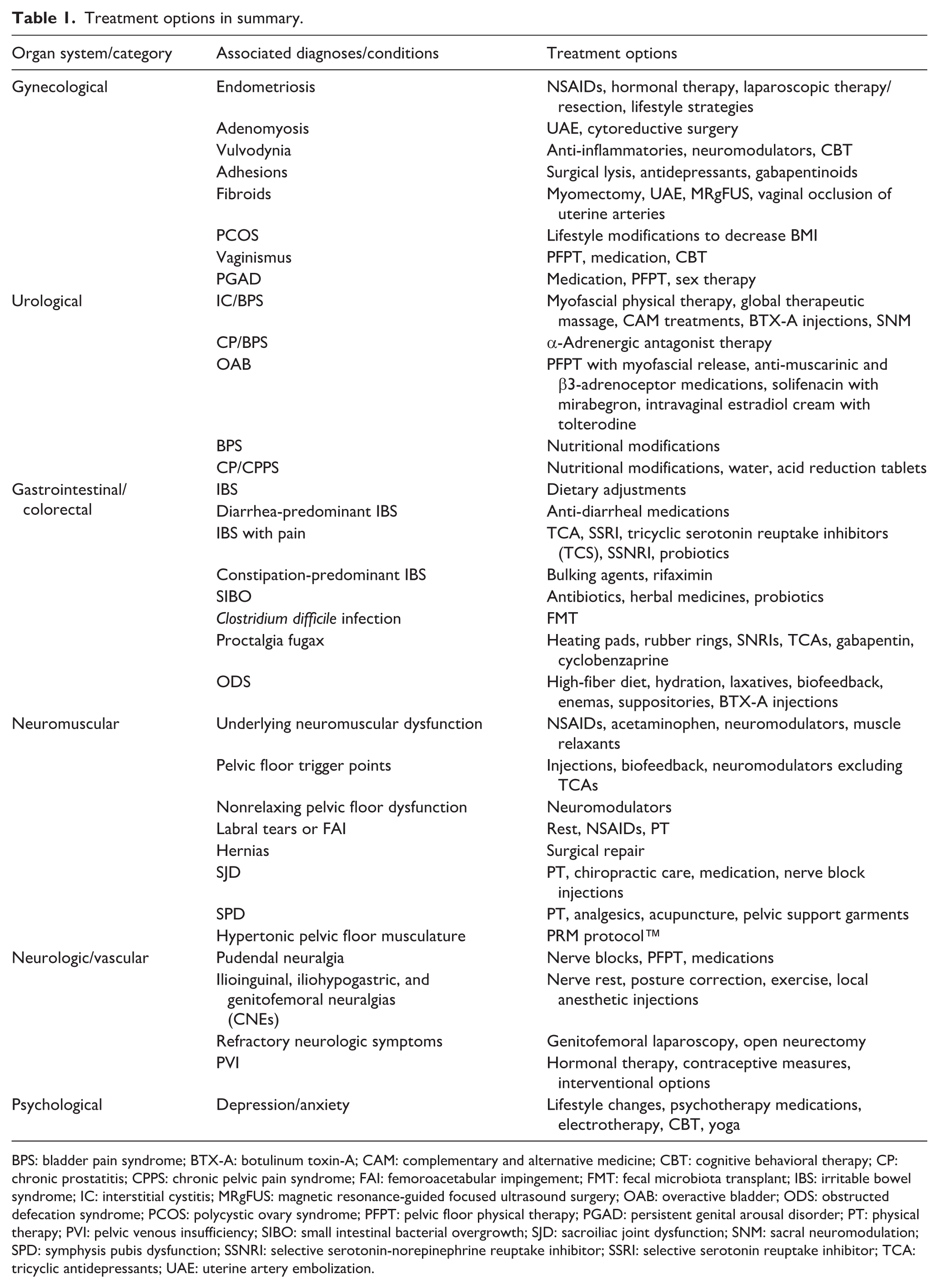
BPS: bladder pain syndrome; BTX-A: botulinum toxin-A; CAM: complementary and alternative medicine; CBT: cognitive behavioral therapy; CP: chronic prostatitis; CPPS: chronic pelvic pain syndrome; FAI: femoroacetabular impingement; FMT: fecal microbiota transplant; IBS: irritable bowel syndrome; IC: interstitial cystitis; MRgFUS: magnetic resonance-guided focused ultrasound surgery; OAB: overactive bladder; ODS: obstructed defecation syndrome; PCOS: polycystic ovary syndrome; PFPT: pelvic floor physical therapy; PGAD: persistent genital arousal disorder; PT: physical therapy; PVI: pelvic venous insufficiency; SIBO: small intestinal bacterial overgrowth; SJD: sacroiliac joint dysfunction; SNM: sacral neuromodulation; SPD: symphysis pubis dysfunction; SSNRI: selective serotonin-norepinephrine reuptake inhibitor; SSRI: selective serotonin reuptake inhibitor; TCA: tricyclic antidepressants; UAE: uterine artery embolization.
Discussion
CPP is a condition that significantly challenges both patients and healthcare providers. This study delves into key aspects of CPP, including its etiology, diagnosis, management, and the broader implications of this complex and often perplexing condition. CPP is frequently underdiagnosed or misdiagnosed, leading to prolonged suffering for patients. 24
The differential diagnosis in the root cause analysis for causes of CPP include gynecological, urological, gastrointestinal, musculoskeletal, neurological, vascular, rheumatological, and psychological factors.7,8,10,14,15,21,24 The complexity of CPP is further compounded by the fact that many patients may have a combination of these factors contributing to their pain.
Although CPP is a multifaceted condition with a diverse range of potential underlying causes, one constant is the presence of concomitant underlying pelvic floor neuromuscular dysfunction. In this discussion, we encourage a multimodal approach to diagnosing any underlying organ system dysfunction while simultaneously treating the concomitant neuromuscular dysfunction in CPP. Recognizing and addressing this complexity while understanding the importance of treating the underlying neuromuscular dysfunction that exists is crucial for effective diagnosis and management.
Diagnosing CPP can be a formidable task due to the wide array of potential causes and the intricate interplay of physical and psychological factors. Patients often undergo a battery of tests, including imaging, laparoscopy, and laboratory assessments, to identify the underlying cause of their pain. 147 Enhancing diagnostic accuracy and timeliness is an ongoing challenge in the field. Continued research is essential to improve our understanding of the causes and mechanisms of CPP and to refine treatment strategies. Furthermore, increasing awareness about CPP and the myofascial pain, peripheral sensitization, and central sensitization complex that occurs among healthcare providers is crucial to expedite diagnosis and intervention. Continued advances in imaging techniques, genetic research, and the development of targeted therapies hold promise for improved CPP patient outcomes.
This article presents a comprehensive treatment algorithm specifically developed to address the existing gaps in managing patients afflicted by CPP, including delayed diagnosis, delayed treatment, and a lack of a root cause analysis on potential underlying pathophysiology. The algorithm advocates early diagnosis and treatment to prevent the CPP patient from spiraling into the chronic pain cycle. A cornerstone of this is a multimodal treatment consisting of office-based procedures aimed to rehabilitate the dysfunctional pelvic nerves and muscles in combination with PFPT and behavioral therapy. Non-operative pelvic pain specialists coordinate the care of CPP patients and bring in the necessary specialists, such as gynecology, urology, gastroenterology, colorectal, psychology, and integrative care, in the coordinated care team, depending on the patient’s needs. A pelvic pain specialist is a non-operative provider specializing in treating pelvic pain by focusing on the nerves and muscles of the pelvis. They may belong to various medical specialties, including Physiatry (Physical Medicine and Rehabilitation), Primary Care Medicine (MD/DO), or Primary Care Nursing (NP). These specialists have expertise in diagnosing and managing pelvic pain conditions through non-surgical approaches, such as medication management, PFPT, nerve blocks, and other interventional techniques. They play a crucial role in providing comprehensive care to patients suffering from pelvic pain, helping to improve their quality of life and overall well-being.
The clinical outcome wheel, depicted in Figure 3, offers a comprehensive framework for assessing patient outcomes in the management of pelvic pain. It guides treatment decisions and optimizes patient care by utilizing self-assessment forms such as the VAS, CPSI, erectile dysfunction, Female Sexual Function Profile, and Promis-29, among others. This framework evaluates various dimensions, including symptom severity, pain management, functional impact, social impact, sexual well-being, opioid use, healthcare utilization, prior diagnostics, procedures, and surgeries.
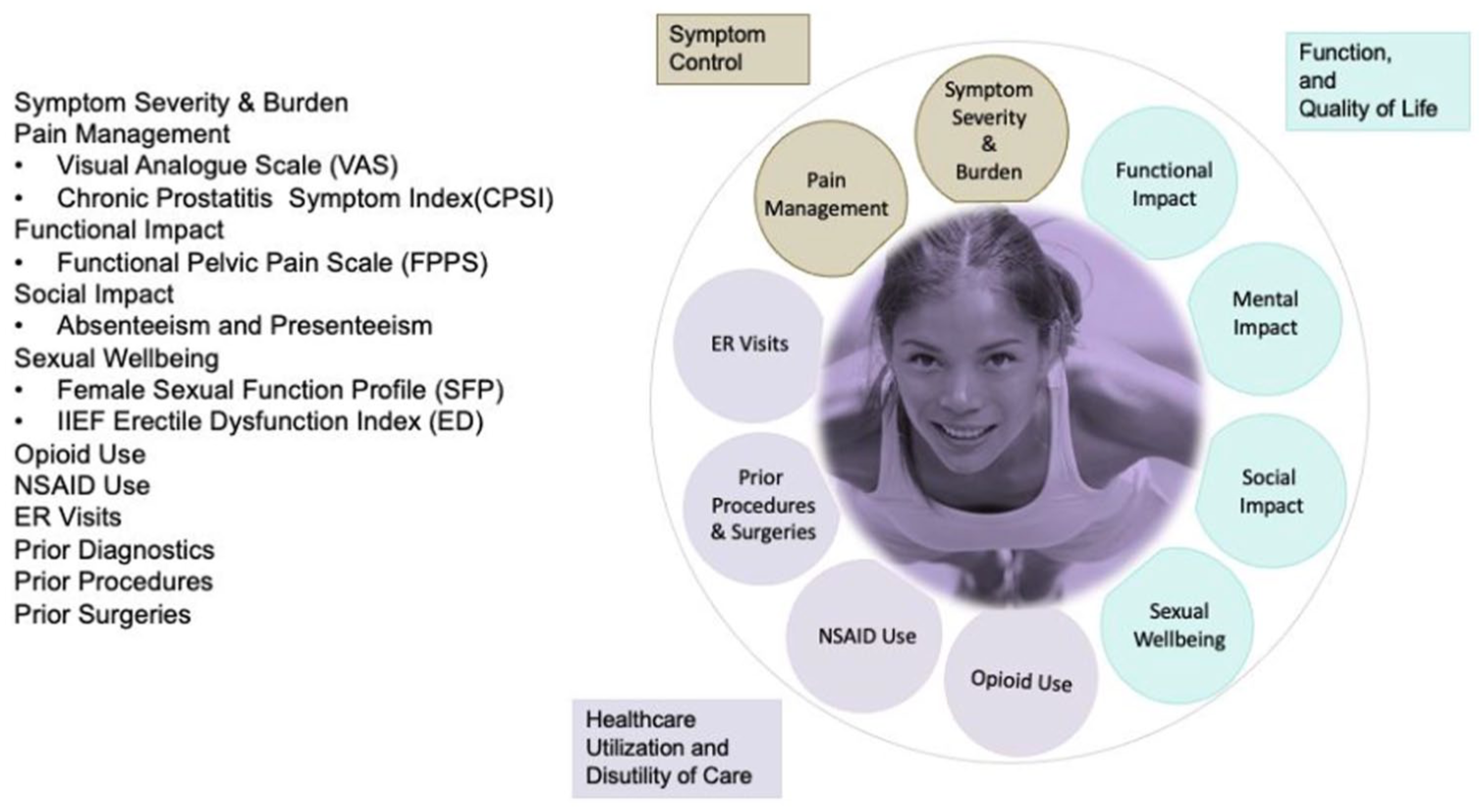
Clinical outcome wheel.
By incorporating these measures, healthcare providers can holistically evaluate patient progress and tailor treatments to improve outcomes and quality of life for individuals with pelvic pain.
Furthermore, the ultimate objective in managing CPP patients extends beyond mere pain reduction; it focuses on restoring functionality, thereby enabling patients to regain their quality of life. Outcome measures also include assessments of sexual function, urinary symptoms, psychological well-being, and overall health-related quality of life, allowing for a comprehensive evaluation of treatment effectiveness and patient satisfaction.
Limitations
Although this review offers a comprehensive overview of the pathophysiology, diagnosis, and management of CPP, there are some limitations to consider. First, the literature used for this review was solely obtained from the PubMed database, limiting the scope of the review as relevant studies published in other databases may have unintentionally been excluded. Furthermore, support for multidisciplinary treatment largely began around 2009. 148 Therefore, articles published prior to this point may reflect outdated or monomodal treatment suggestions. Lastly, although the treatment algorithm was created based on the findings from the literature, there is the potential for a degree of bias to be introduced by the expert panel as their clinical expertise and experiences may influence their recommendations.
Conclusion
This treatment algorithm aims to identify the gaps in the diagnosis and treatment of CPP patients, emphasizing early identification and treatment of pain generators and underlying pathology. The recommendations encourage providers, policymakers, and patients to take a proactive and multimodal approach to treating CPP to prevent the prolonged suffering that CPP patients currently must endure, as well as decrease the economic burden on America’s healthcare system that CPP currently places.
Footnotes
Acknowledgements
The study was performed at Pelvic Rehabilitation Medicine, Atlanta, GA; Chicago, IL; Dallas, TX; Houston, TX; Miami, FL; West Palm Beach, FL; New York City, NY; Great Neck, NY; Scarsdale, NY; Englewood, NJ; Florham Park, NJ; Troy, MI; and Bethesda, MD. Patient data was gathered from these clinics. All authors contributed to the design and implementation of the research, to the analysis of the Results, and to the writing of the article.
Ethical considerations
The protocol for this research project has been approved by a suitably constituted Ethics Committee of the institution, and it conforms to the provisions of the Declaration of Helsinki. Committee of The Feinstein Institutes for Medical Research, approval no. IRB #17-0761. Our study does not have a clinical trial number.
Consent for publication
Consent forms were waived due to the study design. Confidentiality was maintained when reporting all data, and no patient images were included.
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available on request. The data are not publicly available due to patient privacy.
