Abstract
Background:
Menopause has been linked to an array of symptoms, often with adverse effects on quality of life, work and relationships. Despite evidence of economic and social impacts, and a growing population of menopausal individuals, there are significant gaps in knowledge regarding menopause. Gastrointestinal (GI) symptoms in peri- and postmenopause are areas of uncertainty that warrant further investigation.
Objectives:
Following JBI guidance, this scoping review aimed to systematically map research on GI symptoms in ‘natural’ peri- and postmenopause, exploring the volume and conduct of research, and variables investigated that could influence symptom experience.
Eligibility criteria:
Studies assessing GI symptoms (constipation, diarrhoea, vomiting, nausea, abdominal pain, heartburn, faecal incontinence and bloating) in ‘natural’ menopause from all publication dates were included. Studies of medical/surgical menopause, with participants on hormone therapies, or not published in English were excluded.
Sources of evidence:
Results of comprehensive searches of databases (MEDLINE, Embase, PsycINFO, CINAHL, AMED, Scopus, Web of Science, CENTRAL, ProQuest Dissertations and Theses), trial registries, citation and web-searches were screened against eligibility criteria.
Charting methods:
Data were charted in a standardised template, including study designs, populations, countries, GI symptoms studied, methods used for their assessment, and variables investigated. Analyses included frequency counts and percentages, with findings presented in visual and tabular formats.
Results:
Overall, 122 studies were included, published between 1981 and 2024. Studies were predominantly quantitative in design, with constipation the most frequently investigated symptom (n = 58), in contrast to vomiting (n = 4). Results highlighted limited studies from South American and African regions, and insufficient reporting of the criteria used to determine menopausal stages and assess GI symptoms.
Conclusions:
While many studies were identified, findings indicate evidence gaps and methodological considerations pertinent to researchers and funders, regarding study designs, assessment and reporting of menopausal stages and GI symptoms, and the variables investigated. Recommendations are made for future research.
Introduction
In the UK, 13 million people are estimated to be peri- or postmenopausal, 1 with 80%–90% of women thought to be negatively impacted by menopausal symptoms. 2 ‘Natural’ menopause is reached when an individual has not had a menstrual cycle in the prior 12 months, due to a decline in ovarian function associated with ageing, rather than from surgical or medical intervention. 3 Menopause commonly occurs between 44 and 54 years of age; 4 however, it is proposed to be a ‘process’ rather than a single event. 5 The Stages of Reproductive Ageing (STRAW+10) outlines the stages of menopause, with the transition from reproductive life to one year following the final menstrual period (FMP) defined as the ‘perimenopause’. 6 The period leading up to the FMP is further categorised into the early and late menopausal transition (MT). Each stage is characterised by variability in menstrual cycle length and regularity, and significant fluctuations in levels of sex hormones including oestrogen, progesterone, luteinising hormone, and follicle-stimulating hormone. 7 This ‘reproductive hormonal milieu’ and the gradual decline in sex hormones following the FMP have been associated with an array of symptoms, 8 including hot flushes, night sweats, sleep disturbances, joint aches, vaginal dryness, brain fog, depression and anxiety. 7 Despite a growing global population of menopausal individuals, 3 and evidence of economic and social impacts associated with persistent menopausal symptoms,1,8,9 researchers argue that there are significant gaps in our understanding of menopause and the factors that influence the experience of symptoms.10,11
Gastrointestinal (GI) symptoms in peri- and postmenopause are one such area of uncertainty. GI symptoms as outlined by the National Institutes of Health (NIH) Patient Reported GI Symptom Scale 12 include diarrhoea, constipation, nausea, vomiting, abdominal pain, bloating, heartburn and faecal incontinence. These symptoms are highly prevalent in the general population, placing a significant burden on health systems.13,14 Recent news coverage and sources of patient information have suggested an association between menopause and heightened risk of GI symptoms.15,16 However, GI symptoms are not acknowledged as key menopausal symptoms in international and UK guidelines.17,18 This may be due to the omission of GI symptoms from menopausal symptom assessment scales commonly used in research,19–23 or the limited availability of high-quality evidence from human studies to underpin recommendations.
Exploratory searches were conducted using MEDLINE (Ovid), JBI Evidence Synthesis, Open Science Framework (OSF) and Epistemonikos (February 2024) to gauge available evidence on GI symptoms in the peri- and postmenopause. Searches identified primary studies, but few evidence syntheses, with one scoping review exploring swallowing difficulties and symptoms of the oral cavity only, 24 and one systematic review 25 examining the relationship between menopausal stage and irritable bowel syndrome (IBS), a condition with symptoms including abdominal pain and dysfunctional bowel habits. 26 While the systematic review by Adeyemo et al. 25 found limited evidence for an association, searches were restricted to the PubMed database, therefore potentially missing eligible studies, and focused on GI symptoms linked to IBS only. Furthermore, new research has been published since the searches by Adeyemo et al. 25 were completed in 2010. These new studies highlight conflicting findings regarding the relationship between menopausal stage and the frequency or severity of GI symptoms, including those common to IBS. For example, higher rates of constipation 27 and faecal incontinence 28 have been reported in postmenopausal compared with premenopausal individuals, while in contrast, other studies found no significant differences in the prevalence or severity of constipation,29–31 faecal incontinence, 32 bloating 33 or abdominal pain 34 across menopausal stages. Indeed, some studies have found that premenopausal women were more likely to be affected by GI symptoms, such as chronic constipation 35 and abdominal pain 30 , than women in later stages. These contradictory findings might be explained by the heterogeneity of definitions and methods used in these studies and indicate the need for an up-to-date review of research in this area.
Evidence suggests there may be ethnic and geographic variability in menopausal symptom prevalence and severity, 36 and that an individual’s experience of menopause can be influenced by many hormonal, psychological and sociocultural factors (see Figure 1). Sex hormones such as oestrogen and progesterone, are thought to play an important role in GI health.37–39 Fluctuations in these sex hormones associated with the MT could lead to GI symptoms, through altered perceptions of pain, 40 changes in gut motility, 41 intestinal mucosa barrier functions, 42 and immune and inflammatory processes. 37 The composition of the gut microbiome may be impacted by hormonal changes, while also contributing to sex hormone metabolism.43,44 Such changes within the gut could result in the onset of new GI symptoms45,46 or exacerbations in individuals diagnosed with IBS or inflammatory bowel diseases. 47 However, Yang et al. 48 suggest there is insufficient evidence to determine whether microbial changes triggered by hormonal fluctuations contribute to the experience of GI symptoms in the menopause.

Possible correlates and risk factors associated with increased risk of GI symptoms in peri- and postmenopause.
Psychological and social variables may also contribute to GI and menopausal symptoms. The timing of the MT may co-occur with multiple stressors, such as additional caretaking responsibilities for elderly parents or children, bereavement, alongside frustrations with work roles and changing personal identities. 53 Such sociocultural factors could contribute to increased susceptibility to psychological distress, 49 or negatively impact sleep quality and dietary patterns, which in turn could initiate or intensify existing GI symptoms.46,48
A lack of funding for women’s health research has resulted in knowledge gaps related to menopause.10,11,54 Menopause is growing in visibility in the UK media 55 and the recent Women’s Health Strategy for England 11 established menopause as a key priority for future research investment. In consequence, this is an opportune time to identify evidence gaps and key areas for further study. With no recent or ongoing evidence syntheses exploring GI symptoms in peri- and postmenopause, and sufficient studies for a scoping review, these are areas of uncertainty that warrant further investigation. In consequence, this scoping review aimed to systematically search for, select and map research on GI symptoms in ‘natural’ peri- and postmenopause to identify gaps in the evidence to inform future research. The review had the following three objectives:
To gauge the volume of evidence from published and grey literature examining patient-reported GI symptoms (nausea, vomiting, bloating, constipation, diarrhoea, heartburn, abdominal pain and faecal incontinence) in ‘natural’ peri- and postmenopause.
To consider how research on GI symptoms in ‘natural’ peri- and postmenopause has been conducted (including study designs; measures of GI symptoms; timeframes of symptom recall; menopausal stages studied, and the criteria used to determine stages).
To identify the key variables that have been measured in research on GI symptoms in ‘natural’ peri- and postmenopause (for e.g. correlates of GI symptoms: sex hormone levels, body mass index, perceived stress, ethnicity).
Methods
A scoping review methodology was selected to address the research objectives as it is deemed appropriate for broad questions with the goal of exploring the volume, type and characteristics of available evidence. 56
Methods for this review aligned with JBI guidance for the conduct of scoping reviews57,58 using systematic and reproducible methods to search for, select and map relevant studies, thus minimizing bias. Review reporting was guided by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses Scoping Review (PRISMA-ScR) extension 59 for transparency, with the completed checklist available in Supplemental Appendix 1.
As this is a review of existing literature, ethical approval was not necessary.
Protocol
A protocol was registered on OSF (https://osf.io/4w5xc/). Deviations from methods outlined in this protocol are reported with justifications in Supplemental Appendix 2.
Eligibility criteria
Eligibility criteria were pre-specified, defining the scope of this review, supporting development of search strategies, and facilitating decision-making regarding the relevance of records identified through comprehensive searches. 57 The JBI Participant, Concept, Context framework 57 was used to categorise eligibility criteria, as outlined in Table 1.
Eligibility criteria for the identification and selection of studies.

Population
This review focuses on GI symptoms in ‘natural’ menopause, which is characterised by fluctuations in sex hormones during perimenopause, followed by a gradual decline postmenopause. 6 Studies focusing on medically induced or surgical menopause were excluded, as these may cause an abrupt fall in oestrogen and progesterone, 36 and could contribute to more severe menopausal symptoms. 60 As hormone replacement therapies (HRT) may make it difficult to discern menopausal stage 61 and could influence the experience of GI symptoms, 62 studies recruiting individuals on HRT were excluded. Furthermore, as the menopausal stage of individuals with polycystic ovary syndrome, or those who have undergone hysterectomy, cannot be determined using menstrual cycle criteria, 6 studies focusing on these populations were excluded.
Variable nomenclature has been used to classify menopausal stages, 61 with the term ‘pre-menopausal’ inconsistently applied, sometimes being used to describe both the entire reproductive period, and also the 1–2 years prior to the cessation of menstruation. For clarity, studies of ‘pre-menopausal women’ only (aged <40 years with regular menstrual cycles) were excluded.
Concept
This scoping review focused on GI symptoms based on eight key categories described in the NIH Patient Reported GI Symptom Scale. 12 To avoid duplication with a recent scoping review, 24 studies on swallowing difficulties and oral symptoms were excluded. Patient-reported GI symptoms were the focus of this review, so studies reporting only laboratory or electrophysiological outcomes (such as GI motility or structural changes to the gut in menopause) were excluded.
Context
Studies from any country or region were included to facilitate identification of all variables (including ethnicity) studied that could impact on the experience of GI symptoms in peri- and postmenopause.
Language
Non-English publications were ineligible for inclusion due to translation costs, and the potential for language bias was noted. 63 Language limits were not applied to searches, but instead, non-English publications were excluded during screening, as recommended by Pieper and Puljak. 64
Publication dates
Studies from database inception to the search date were included, as this review aims to determine the volume of available evidence.
Types of evidence
To address objectives regarding the volume and conduct of available research, published and ongoing quantitative and qualitative primary research studies, and evidence syntheses were included. The following evidence sources were excluded: animal studies, laboratory studies (that did not assess patient-reported GI symptoms), editorials, case reports, literature reviews (that did not use systematic methods for the identification and selection of studies), and conference abstracts due to limited details of study methods and measures.
Sources of evidence
As recommended by JBI guidance, 57 searches were performed following a three-step process as outlined below:
Initial scoping searches
Text analysis of relevant studies from exploratory searches supported the identification of controlled vocabulary (e.g. Medical Subject Headings (MeSH)) and synonyms for the population (i.e. perimenopause) and the concept (i.e. named GI symptoms). An Information Specialist with expertise in evidence synthesis (NS) developed all search strategies. The Ovid MEDLINE strategy is presented in Box 1, with all other strategies reported in Supplemental Appendix 3.
Search strategy for Ovid MEDLINE.

Full database searches
The following bibliographic databases were searched on 4 March 2024 without date or publication type restrictions: MEDLINE, Embase, APA PsycINFO (Ovid); CINAHL, AMED (EBSCO); Cochrane Database of Systematic Reviews, Cochrane Central Register of Controlled Trials (Wiley); Web of Science Core Collection (Clarivate Analytics); Scopus (Elsevier) and ProQuest Dissertations and Theses Global. Search strategies were tailored for each database using appropriate syntax and controlled vocabulary.
Grey literature and supplementary searches
Searches of professional associations and key websites (e.g. British Menopause Society), Google/Google Scholar and clinical trials registries (ClinicalTrials.gov and the World Health Organization International Clinical Trial Registry Platform) were completed from April to June 2024 to identify grey literature. 65 Simplified search strategies were used for Google/Google Scholar, with the first ten pages of each search screened by one reviewer. Full details are provided in Supplemental Appendix 3. Forwards and backwards citation-searching of included studies from database searches was completed using CitationChaser in May 2024, 66 and bibliographies of key review articles were checked.
Selection of sources of evidence
All records from searches were imported into EndNote X9.3.3 (Clarivate Analytics) and duplicates removed using manual checks and EndNote functionality.
Pilot screening
Pre-specified eligibility criteria (Table 1) informed the selection of studies. Three reviewers (NS, CP, KT) independently reviewed titles/abstracts of a random sample of 100 records in Rayyan 67 to pilot eligibility criteria. Disagreements were discussed, and screening documentation was refined to ensure clarity.
Title/abstract and full-text screening
Screening by two independent reviewers is recommended to reduce the risk of bias or human error; 68 however, due to time constraints, one reviewer screened 100% of records (NS), with a second reviewer (KT) screening 50% at both stages of screening. Firstly, titles/abstracts were assessed against eligibility criteria, followed by the full text of selected articles. Articles not available through University Library services were excluded, though citations are listed in Supplemental Appendix 4. Disagreements at both screening stages were resolved through consensus.
Data charting methods
A Microsoft Excel template was developed to chart data items relevant to review objectives. The following data items were charted: citation; study design; populations studied; GI symptom(s) investigated, as well as the methods (e.g. Menopause-Specific Quality of Life (MENQOL) checklist) 69 and recall timeframe used in their assessment; definitions of menopausal stage (e.g. STRAW + 106); countries; and variables studied (i.e. predictors, correlates or interventions). Additional items (e.g. new variables) were added to the template as the reviewer became familiar with the included literature. As this review is exploratory and does not aim to synthesise findings, study results were not extracted. This is consistent with guidance for the conduct and appropriate use of scoping review methods. 57
The template was piloted by one reviewer (NS) on ten studies covering a range of evidence types. Independent data charting by two reviewers is recommended to minimise bias and human error; 58 however, resource limitations required that one reviewer charted all data.
Quality assessment
Quality assessment of included studies is not a mandatory step in scoping reviews 70 and was not completed for this review.
Data analysis and synthesis
Descriptive analyses focused on frequency counts of charted data, with percentages calculated using Excel (Microsoft; Version 2402) and presented in tabular, graphical or visual formats aligning with review objectives, as indicated in Table 2.
Analyses by review objective.

Results
Database and supplementary searches retrieved 13,111 and 3529 records, respectively. After duplicates were removed, 9569 records were assessed for eligibility from titles/abstracts. A total of 311 articles were retrieved for full-text screening, with 122 studies identified for inclusion. Figure 2 (PRISMA flow diagram) indicates the process of study selection. Details of excluded studies with reasons are available in Supplemental Appendix 4.

PRISMA flow diagram: study identification and selection.
Characteristics of included studies
Results are reported in alignment with review objectives, considering:
The volume of evidence on GI symptoms in ‘natural’ peri- and postmenopause.
The conduct of research.
The variables investigated.
Study characteristics for 122 included studies outlining brief citation details, populations, GI symptom(s) assessed, study designs and variables investigated are provided in Supplemental Appendix 5.
Volume of evidence
Figure 3 shows the timeline of included studies, with 34% (n = 42) published within the last 5 years.
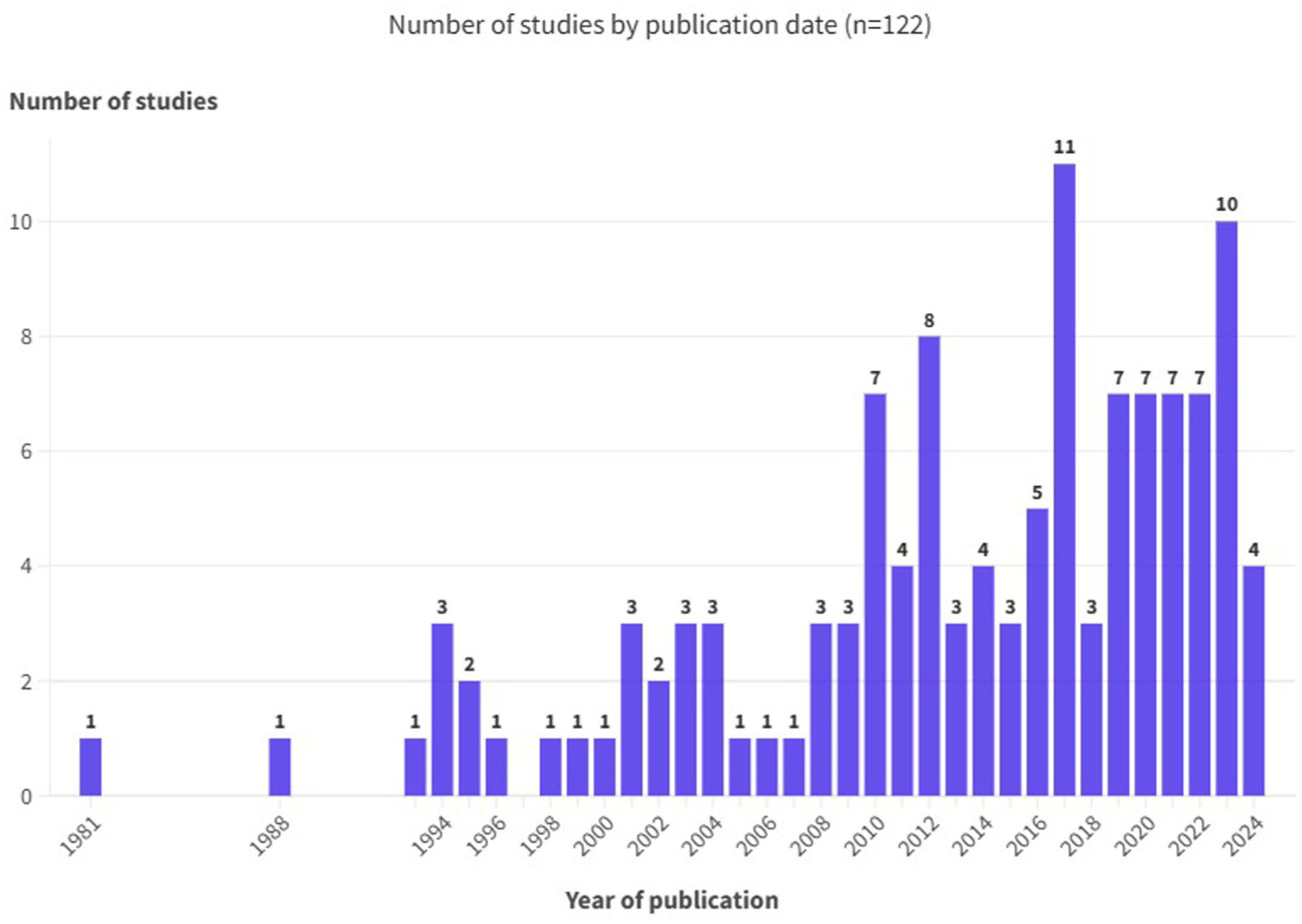
Number of included studies by publication date.
Table 3 provides summary characteristics for included studies relating to the volume of evidence. This shows that most studies (n = 116; 95.1%) were published as journal articles, with one dissertation, 72 two preprint publications,73,74 and three clinical trial registrations75–77 identified as ‘grey literature’. Studies were conducted in 35 countries represented by six geographic regions, 71 ranging from only 0.8% (n = 1) in South America, compared with 44.3% (n = 54) carried out in Asia. From 122 included studies, only 18% (n = 22) stated a primary objective that related to the study of GI symptoms in ‘natural’ peri- or postmenopause, while 82% (n = 100) either investigated a GI symptom as one of a range of menopausal symptoms or assessed menopause as one potential risk factor for a GI symptom. Constipation was the most studied GI symptom (n = 58; 47.5%), while only 4 (3.3%) studies investigated vomiting in ‘natural’ peri- or postmenopause.
Summary of study characteristics relating to the volume of available evidence (n = 122 included studies).

Conduct of studies
Table 4 presents summary data on the conduct of studies on GI symptoms in ‘natural’ peri- and postmenopause.
Summary of study characteristics relating to the conduct of research.

Study designs
Study designs were predominantly quantitative (n = 118; 96.7%), with only two (1.6%) qualitative,163,176 and one mixed-methods study (0.8%) included. 162 One systematic review 25 met eligibility criteria, and 73% of included studies were cross-sectional (n = 89), with other studies of longitudinal design (n = 14; 11.5%), randomised controlled trials (n = 5, 4%), non-randomised studies (n = 3; 2.5%), case-control (n = 3; 2.5%), and one retrospective study (0.8%).
Populations
Most included studies recruited individuals described as postmenopausal (n = 107; 87.7%), with 57 studies (46.7%) including those in perimenopause. Further analysis indicated that just over a third of included studies (n = 42; 34.4%) focused on individuals of one menopausal stage only (perimenopausal only: n = 9; postmenopausal: n = 33). Criteria or definitions used to determine menopausal stage were not stated in 36% of studies (n = 44), with age ranges used to identify individuals for inclusion in a further nine studies (n = 7.4%). STRAW/STRAW + 106, 184 were cited as the criteria used for categorising menopausal stages in 13 studies (10.7%), while 56 (45.9%) provided alternative definitions (e.g. studies cited an adapted version of the WHO classification, 185 or classified individuals as perimenopausal if they had experienced menstrual bleeding in the prior 12 months, but not in the last 3 months).
Measures of GI symptoms
Studies used a range of measures to assess GI symptoms, with 66 studies (54.1%) citing a validated menopausal or GI symptom screening tool (e.g. MENQOL, 186 Wexner Incontinence Scale 187 ). The remaining studies developed new assessment tools (n = 47; 38.5%) or did not describe the method used to assess GI symptoms (n = 9; 7.4%). The timeframe for recall of GI symptoms was not reported in 84 studies (68.8%), with other studies using varied recall periods ranging from less than or equal to a week (n = 6; 4.9%) to 3–12 months (n = 13; 10.7%).
Variables investigated
Included studies investigated a range of variables in association with GI symptoms in peri- and postmenopause (see Figure 4). These were categorised into: demographics, lifestyle factors, biomarkers, clinical markers and comorbidities, interventions, and menopause-related variables.
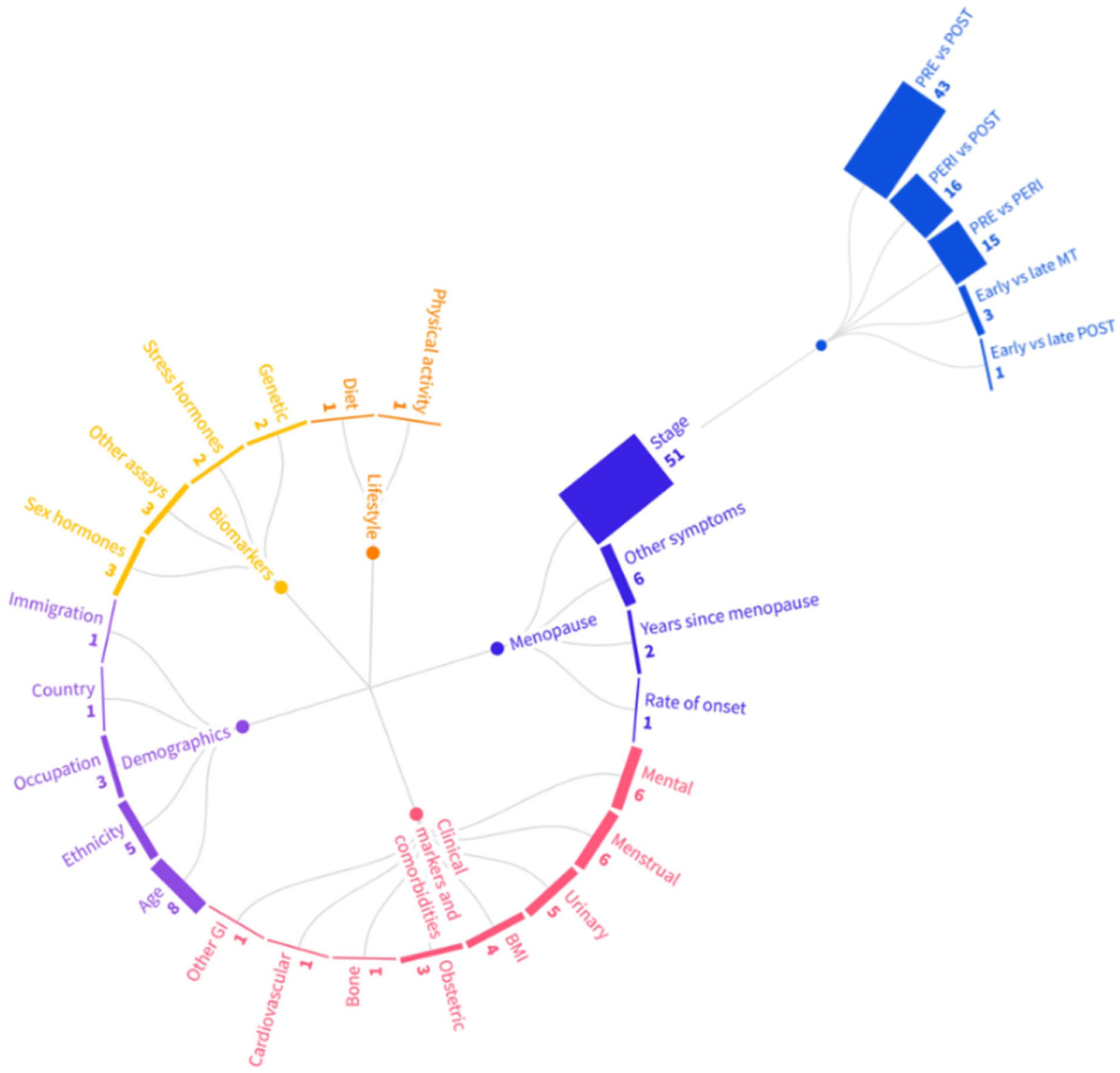
Number of studies by variables investigated in research on GI symptoms in peri- and postmenopause, including comparisons by menopausal stage.
Menopause-related variables were the most studied category (n = 60; 49.2%). This category included associations of GI symptoms with other menopausal symptoms (such as hot flushes; n = 6; 4.9%),74,92,105,107,144,155 years since the FMP (n = 2; 1.6%)88,122 and rate of onset of menopause (n = 1; 0.8%). 137 Comparisons of GI symptom frequency or severity by menopausal stage were observed in less than half of the included studies (n = 51, 41.8%). Symptom experience was compared in premenopausal with postmenopausal individuals in 43 studies (35.2%), while only three studies31,34,182 (2.4%) made a comparison between perimenopausal individuals in the early and late MT.
Demographic variables were assessed in 18 studies (14.8%), with 8 studies (6.6%) considering the impact of age31,34,86,91,110,146,168,171 on the experience of GI symptoms in menopause. Ethnicity or race,115–117,119,120 type of occupation,86,111,158 immigration status 118 and country of residence 112 were also considered in the included studies.
With regard to studies investigating comorbidities and clinical markers (n = 27; 22.1%), mental health including perceived stress and anxiety,31,34,93,133,144,177 and menstrual symptoms (such as dysmenorrhoea and premenstrual syndrome)78,99,108,156,160,166 were both assessed in six studies, while other included studies considered associations with body mass index,79,91,130,150 obstetric history or complications during childbirth,125,157,163 urinary disorders,28,135,138,161,183 other GI symptoms, 117 cardiovascular disease,120,167 and bone health. 73
Few studies examined lifestyle factors, with only one study each examining the relationship between GI symptoms and dietary intakes (tryptophan) 91 and physical activity. 72
Included studies assessed biomarkers, including assays of sex hormones (n = 3; 2.4%),31,34,91 stress hormones (n = 2; 1.6%),31,34 genetic markers (n = 2; 1.6%)141,142 and other assays such as urinary tryptophan, melatonin metabolites and serum allopregnalone (n = 3, 2.4%).91,174,180
Discussion
This is the first scoping review to systematically map research on GI symptoms in ‘natural’ peri- and postmenopause. The most significant and most unexpected finding was the large volume (n = 122) of eligible studies identified, with a notable increase in publications in recent years. However, only 22 studies had a primary objective relating to GI symptoms in menopause. Furthermore, review findings highlight several methodological considerations and evidence gaps of interest to researchers and funders. These relate to the volume and conduct of research, and variables investigated, and are discussed further below. A summary of recommendations for future research is provided in Box 2.
Recommendations for future research.

Volume of evidence
This review mapped studies to at least one of eight GI symptoms, 12 with constipation being the most investigated symptom. While this review did not summarise study results, conflicting research findings27,29–31,35 combined with the large number of studies identified, suggest that constipation is a potential area for synthesis in a systematic review. In contrast, only four studies assessed vomiting in peri- or postmenopause. This may reflect the low prevalence of this symptom, or its omission from menopausal symptom scales commonly used in research.19–23,69,188–191 This is in contrast to symptoms such as constipation, diarrhoea, bloating, and abdominal pain, which feature in several instruments.69,188–191 Alternatively, symptom reporting may be influenced by an individual’s stereotype of ‘the menopausal woman’, with symptoms only reported that are considered to be linked to the menopause (e.g. hot flushes), rather than all those experienced (i.e. vomiting). 192 These menopausal stereotypes could vary across cultures, with Im et al. 119 finding that while nausea and vomiting was documented in 18.9% of a sample of midlife women, reports of symptom frequency and severity varied by ethnicity. Interestingly, a recent study 193 excluded from this review, found women placed high value on symptom relief from vomiting during menopause. However, it is not clear whether this study included individuals taking HRT, which could be a contributory factor in the aetiology of vomiting. 194 Further research in diverse populations (i.e. varied ethnicities; HRT users vs non-users) and in consultation with menopausal individuals, may be warranted to determine if vomiting (and other GI symptoms) are indeed related to ‘natural’ menopause.
An important finding is the paucity of studies identified from South American and African countries, compared with Asian, North American and European regions. While this may be partly due to the exclusion of foreign language articles, an assessment of excluded publications indicates only a few non-English language studies were conducted in these regions. With evidence that there is ‘no universal menopausal syndrome’, 195 and cultural or ethnic differences in menopausal symptom reporting, 196 it is important that future research is conducted in varied locations to capture diverse experiences across cultures and geographies. This is consistent with findings of the recent Menopause Priority Setting Partnership 197 which highlighted the need for research to explore menopausal symptoms across countries, cultures and ethnic backgrounds.
Conduct of studies
This scoping review revealed that qualitative research was notably underrepresented in this area, with only two articles identified.163,176 This could be due to a focus in menopausal research on ‘known’ or clinically defined symptoms, rather than women’s actual experience of menopause. 198 Qualitative research is important for capturing insights into lived experiences at different stages of menopause, the impacts of symptoms (e.g. on quality of life) beyond those on ‘established’ symptom checklists as well as the acceptability of different interventions for symptom management.199–201 Furthermore, studies predominantly utilised quantitative cross-sectional designs (n = 89), as is consistent with findings of evidence syntheses assessing a diverse range of symptoms in menopausal research.202–205 While cross-sectional study designs provide a convenient methodological approach for investigating symptom prevalence, it is difficult to draw causal inferences due to the measurement of exposure (i.e. menopausal stage) and outcome (i.e. GI symptoms) at a single timepoint.206,207 Longitudinal studies are needed to assess the trajectory of symptoms across pre-, peri- and postmenopausal stages,208,209 to differentiate pre-existing and new onset symptoms210,211 and obtain accurate estimates of symptom durations. 212 However, this review identified only 14 longitudinal studies, and less than half of all included studies made a comparison of GI symptom experience across different menopausal stages. Only three studies compared GI symptoms in early versus late MT, despite evidence of differing menopausal symptomatology across these stages.6,213 These findings highlight a need for further longitudinal research to effectively answer questions about the relationship between menopause and GI symptoms, as well as qualitative studies to better understand menopausal lived experiences.
Consistency in use of menopausal terminology and application of validated criteria for the classification of menopausal stages are key to understanding the impact of the MT on symptom experience, and to support comparisons of results from different studies.61,214 However, the present review findings indicated that criteria for determining menopausal stage were not reported in over a third of studies, with only 13 studies citing STRAW or updated STRAW + 10 criteria.6,184 Failure to document criteria for menopausal staging has been reported in previous research. 61 An alternative approach using age ranges as a proxy for menopausal stage was applied in nine studies; however, this strategy has been criticised 40 due to the variability in age of MT and FMP. 215 To strengthen the reliability and validity of future research, and minimise the risk of misclassification of menopausal stages, studies should utilise and clearly report validated criteria such as STRAW + 106.
Similarly, review findings revealed that many studies did not use a validated tool to assess GI symptoms, with nine studies not reporting the measure used and 47 studies developing a study-specific tool. This issue has been noted in other reviews of menopausal symptoms more widely.216–218 While it may be considered necessary to adapt measures for cultural appropriateness, it has been argued that validated tools are required to standardise data collection, and to facilitate rigorous comparisons between studies. 219 Variation in the timeframe reported for symptom recall was also considerable: from not being described, to immediate capture on an app, or up to 12 months. This too is consistent with findings of evidence syntheses assessing other menopausal symptoms.216,217,220,221 Furthermore, 6-to 12-month recall periods could be too lengthy, limiting the accuracy of reporting of menopausal symptoms,192,222 whereas daily symptom diaries may be burdensome for study participants. 223 Future studies should use validated measures to assess GI symptoms, identify appropriate tools (e.g. digital diaries or mobile applications) and timeframes for their recall, and clearly report details of measures used, as is also recommended by the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guidance. 224 This will support the synthesis of findings from multiple studies that could inform policy and practice.
Variables investigated
The diversity of factors and variables studied suggests there is limited agreement on those thought to be most important in influencing menopausal symptom presentation, as is also described in a scoping review of correlates of palpitations during menopause. 225 Multiple factors may influence ovarian function, hormonal levels and GI symptom experience, including ageing, dietary patterns, smoking, body mass index, psychological and social variables, and it is argued these should all be considered in menopause research.117,213,222 The influence of ageing was only considered in eight included studies, despite evidence demonstrating the prevalence of GI symptoms such as constipation increases with age, 226 and psychological health was only considered in six studies. Furthermore, evidence gaps were identified, with no included studies assessing the impacts of smoking, sleep disturbances, or gut microbiome composition on GI symptoms in menopause. Interestingly, two ongoing studies75,77 are investigating probiotic interventions in menopausal individuals with constipation and diarrhoea, implying that researchers believe there could be a role for changes to gut microbial composition in managing GI and other menopausal symptoms. With menopause occurring at a time of significant biological, psychological and sociocultural change, it is important that future research considers potential confounding variables and correlates to understand their impacts on GI symptoms. This may support the development of targeted lifestyle, behavioural or hormonal interventions for effective GI symptom management in peri- and postmenopause.
A summary of recommendations and methodological considerations for future research on GI symptoms in peri- and postmenopause is outlined in Box 2.
Strengths and limitations
This review employed comprehensive search methods to identify existing and ongoing research, with a good-practice systematic approach to the selection and mapping of evidence. However, several limitations should be noted. GI symptoms are often not well described in article titles/abstracts, and in consequence, relevant studies may not have been retrieved by database searches. That said, the use of supplementary searches, including citation-searching may have helped mitigate this issue. 227 With regard to review methods, a second reviewer screened 50% of records, and data charting was completed by a single author, and the potential for bias and human error is noted. Quality assessment of included studies is not a mandatory step in scoping reviews, 70 and was not completed. While this may limit the review’s capacity to guide policy and practice, the review aim was exploratory, to identify gaps in the literature and inform future research.70,228 In addition, due to time limitations and the large number of included studies, research assessing the role of HRT as a cause of, or as an intervention for, GI symptoms was excluded. With evidence that HRT use may influence the presentation of GI symptoms in menopausal women,62,229–231 this is a key area that requires investigation in future reviews. As observed in other research on menopausal symptoms,220,232 surgical menopause, HRT or oral contraceptive use was often poorly reported, or separate data not provided, making it challenging to identify studies of ‘natural’ menopause. Furthermore, it was often difficult to discern whether symptoms were of GI origin, for example, chest pain was excluded but may have been associated with heartburn. Similarly, abdominal pain may have either a GI or gynaecological aetiology (i.e. menstrual cramping). In order to discern the origin of symptoms, patient-reported outcome measures could gather additional data, for example, about the location of pain using visual tools, 233 temporal patterns (i.e. after eating, exertion or associated with menstruation), and co-occurring symptoms. 234 Finally, as the results of studies were not extracted, it is not possible to draw conclusions about the prevalence of GI symptoms in ‘natural’ peri- and postmenopause. This is an important area for future study.
Conclusion
This review mapped the available evidence on GI symptoms in ‘natural’ peri- and postmenopause to inform future research. While many studies were identified, findings highlighted few longitudinal and qualitative studies, insufficient reporting of criteria used to determine menopausal stage and data collection methods used to assess GI symptoms, and no studies investigating key variables that could influence symptom prevalence. In consequence, this review has identified evidence gaps and methodological considerations with implications for researchers, funders and priority setting partnerships. 10 Findings may help facilitate the prioritisation of future research and the design of new primary studies to best answer questions about GI symptoms throughout menopause, disentangling the interacting factors that contribute to their lived experience. Evidence syntheses relating to GI symptoms in those taking HRT, and in surgical or medical-induced menopause, would be a valuable addition to the findings presented here.
Supplemental Material
sj-docx-1-whe-10.1177_17455057251387470 – Supplemental material for The volume and characteristics of research on gastrointestinal symptoms in ‘natural’ peri- and postmenopause: A scoping review
Supplemental material, sj-docx-1-whe-10.1177_17455057251387470 for The volume and characteristics of research on gastrointestinal symptoms in ‘natural’ peri- and postmenopause: A scoping review by Naomi Shaw, Rebecca Abbott and Clare Pettinger in Women's Health
Supplemental Material
sj-docx-2-whe-10.1177_17455057251387470 – Supplemental material for The volume and characteristics of research on gastrointestinal symptoms in ‘natural’ peri- and postmenopause: A scoping review
Supplemental material, sj-docx-2-whe-10.1177_17455057251387470 for The volume and characteristics of research on gastrointestinal symptoms in ‘natural’ peri- and postmenopause: A scoping review by Naomi Shaw, Rebecca Abbott and Clare Pettinger in Women's Health
Supplemental Material
sj-docx-3-whe-10.1177_17455057251387470 – Supplemental material for The volume and characteristics of research on gastrointestinal symptoms in ‘natural’ peri- and postmenopause: A scoping review
Supplemental material, sj-docx-3-whe-10.1177_17455057251387470 for The volume and characteristics of research on gastrointestinal symptoms in ‘natural’ peri- and postmenopause: A scoping review by Naomi Shaw, Rebecca Abbott and Clare Pettinger in Women's Health
Supplemental Material
sj-docx-4-whe-10.1177_17455057251387470 – Supplemental material for The volume and characteristics of research on gastrointestinal symptoms in ‘natural’ peri- and postmenopause: A scoping review
Supplemental material, sj-docx-4-whe-10.1177_17455057251387470 for The volume and characteristics of research on gastrointestinal symptoms in ‘natural’ peri- and postmenopause: A scoping review by Naomi Shaw, Rebecca Abbott and Clare Pettinger in Women's Health
Supplemental Material
sj-docx-5-whe-10.1177_17455057251387470 – Supplemental material for The volume and characteristics of research on gastrointestinal symptoms in ‘natural’ peri- and postmenopause: A scoping review
Supplemental material, sj-docx-5-whe-10.1177_17455057251387470 for The volume and characteristics of research on gastrointestinal symptoms in ‘natural’ peri- and postmenopause: A scoping review by Naomi Shaw, Rebecca Abbott and Clare Pettinger in Women's Health
Footnotes
Acknowledgements
The lead author would like to acknowledge Kate Taylor for their assistance with screening records from database and supplementary searches.
Ethical considerations
Not applicable. As this is a review of existing literature, ethical approval is not required.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data is available from the lead author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
