Abstract
Background:
Polycystic ovary syndrome (PCOS) is the most common endocrine disorder in reproductive-aged women, linked to metabolic, hormonal, and psychological issues. Management typically involves lifestyle changes, including increased physical activity and reduced sedentary behaviour.
Objectives:
To compare the health profiles and behaviours of women with and without PCOS.
Design:
This study analysed data from the UK Biobank, which is a prospective cohort study.
Methods:
Women with PCOS in the UK Biobank were identified, while age- and body mass index (BMI)-matched controls were randomly selected. Data on factors associated with PCOS severity and self-reported lifestyle behaviours were analysed. Group differences were tested for significance, and participants were categorised by health behaviours to assess morbidity risk.
Results:
The study included 319 women with PCOS (mean age: 43.9 years) and 638 in each control group. Significant differences (p < 0.05) were observed in anthropometric (e.g. body weight, BMI, waist and hip circumference, and body fat), cardio-metabolic (e.g. blood pressure, triglycerides, and glycated haemoglobin), and androgenic (e.g. sex hormone-binding globulin) indices. Differences were most pronounced between PCOS and age-matched controls but remained when BMI was also considered. Women with PCOS engaged in less vigorous physical activity and had higher screen time and sedentary behaviours. Those with the lowest physical activity and highest sedentary time had the worst health profiles and highest morbidity risk, regardless of group.
Conclusion:
Women with PCOS exhibit poorer health despite only slight lifestyle differences. Across all participants, lower physical activity and higher sedentary behaviour were linked to increased health risks. Further research is needed to clarify causal relationships between lifestyle factors and PCOS.
Background
Polycystic ovary syndrome (PCOS) is a common endocrine disorder, affecting 10–15% of reproductive-aged women,1,2 although there is variability in prevalence depending upon the diagnostic criteria applied and/or geographical region. 3 PCOS is characterised by hyperandrogenism and ovulatory disruption and typically manifests in a range of symptoms such as acne, hirsutism, and infertility. 4 Women with PCOS are also at an increased risk of cardio-metabolic disorders, including all metabolic syndrome (MetS) components, that is, obesity (particularly central obesity with visceral and ectopic adipose tissue accumulation), insulin resistance, type 2 diabetes, dyslipidaemia, and hypertension.5 –8 The latest evidence-based guidelines 9 suggest that PCOS management should focus on lifestyle changes, incorporating increased physical activity, aiming to alleviate symptoms and lower the associated risk of type 2 diabetes and cardiovascular disease (CVD).
Increasing physical activity is a well-established approach for improving CVD risk factors in the general population. 10 There are also reported benefits (i.e. improved insulin sensitivity, lipid profile, and cardiorespiratory fitness, as well as decreased central adiposity and body mass index (BMI)) in individuals with PCOS following participation in exercise interventions,11,12 although the quality of evidence is low. What is even less clear in PCOS is how physical activity accumulated through activities of daily living and time spent in sedentary behaviours influence metabolic health and general well-being. Previous research indicates that women with PCOS are not less physically active,13 –17 and engage in similar durations of sedentary behaviours13,17,18 compared to women without PCOS, yet they consistently report lower health-related quality of life.13,19 –21 In any given population, people engage in varying degrees of physical activity and sedentary behaviours, but the lowest mortality and morbidity risks are generally noted in those who are the most active and engage in the lowest amount of sedentary behaviour. 22 If a diagnosis of PCOS significantly increases the long-term CVD risk, then those who are not physically active and spend more time sitting are likely further increasing their risk of disease.
As such, better understanding of the characteristics of individuals with PCOS, including risk factors that are modifiable (e.g. physical activity and sedentary behaviours), may lead to more effective and targeted interventions against these risk factors. Furthermore, this may allow earlier identification of those at higher risk of co-morbidity and thus enable early interventions which reduce risk, decrease morbidity and mortality, improve quality of life, and lower subsequent health care costs. 23 Therefore, we utilised data from the UK Biobank to better understand the health profile of women with PCOS and compare it to that of two additional cohorts (one age-matched, and another both age- and BMI-matched) of women without PCOS. For these three UK Biobank groups, we also analysed data on self-reported physical activity and sedentary behaviours to compare their respective health profiles and classify their health-risk behaviours.
Methods
To ensure transparency and completeness, this study was reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology statement (Supplemental Table 1). 24
The UK Biobank is a large open access national health resource in the UK, aiming to improve preventative measures, diagnostic processes, and treatment of a wide range of diseases. 25 Health-related data are collected for UK Biobank participants who also provide blood, urine, and saliva samples, allowing for data to be collected on a range of biomarkers, many of which are associated with CVD (e.g. blood levels of lipids, glucose, and C-reactive protein (CRP)) and/or PCOS (e.g. testosterone, oestradiol, and sex hormone binding globulin (SHBG)). To obtain the required data, an application was made to the UK Biobank (Application Reference Number 52771), based on which the objectives and methods of the current study were reviewed and approved. The UK Biobank is a prospective cohort study which has approval from the Northwest Multi-centre Research Ethics Committee as a Research Tissue Bank (RTB). This approval means that researchers do not require a separate ethical clearance and can operate under the RTB approval. When participants first joined the study, they all provided explicit consent for UK Biobank to access all their medical and health-related records, and for the long-term storage and use of any samples. The main data collection for the UK Biobank occurred between 2006 and 2010, with subsequent data releases regularly being made available; notably, all participant biomarker data were made available in 2019.
To be included as a case in the present study, participants had to be female and have a diagnosis of PCOS reported in their data. Control women (with no reported PCOS diagnosis) were then matched according to age and separately again for age plus BMI. No other parameters were specified for eligibility. The included outcomes of this study were selected according to diagnostic features of MetS (i.e. waist circumference, blood pressure (BP), blood glucose, and lipid profile), 26 as well as the physical (i.e. body weight, BMI, cardiorespiratory fitness, and inflammatory and androgenic biomarkers) 1 and psychosocial (i.e. anxiety, depression, mood, self-esteem, and quality of life) characteristics that are often associated with PCOS. 27 Outcomes related to self-reported physical activity and sedentary behaviour were also included.
Data handling
Following study approval, data were made available through the UK Biobank Application Management System. The final dataset included all female participants from the UK Biobank (~250,000 participants), from which we identified all women with PCOS using the International Classification of Disease (ICD) codes. When selecting case-control participants from datasets such as the UK Biobank, the case group should comprise all (or a high proportion of) participants diagnosed with the outcome disease (i.e. PCOS), whereas the referent or control group(s) should be a random sample that are free from this disease at the specified sampling point. 28 However, it is important that within each group, no participant is sampled more than once. The two comparator (non-PCOS) groups selected in the present study were age-matched and age- plus BMI-matched, respectively, to the PCOS group. These were randomly selected from the non-PCOS matched sample on a 2:1 ratio. In addition to the above data, the ICD codes were also used to capture non-PCOS morbidity incidence across all three study groups.
Data analysis
Statistical analysis was completed in Jamovi (The Jamovi project: v1.2.16 (computer software), Sydney, Australia).Descriptive statistics were presented as median and interquartile range (IQR) for each outcome according to individual cohort. Assumptions of normality were checked, and analysis of skewness and kurtosis values were conducted. None of the included outcomes met the assumption of normality across all three groups; therefore, the following non-parametric analysis was adopted in the analyses: a Kruskal–Wallis (one-way analysis of variance (ANOVA) on ranks) to compare outcomes in the three independent groups (PCOS versus age-matched versus age- plus BMI-matched). Levels of significance were set at p < 0.05, and to gauge the magnitude of influence, the epsilon squared (ε2) effect size, a suitable effect size estimator for use in ANOVA, 29 was also reported for each comparison. Whilst an ε2 of zero would indicate no effect, there is no formal way to determine the magnitude of an effect greater than zero. In this instance, recommendations from Rea and Parker 30 were used to report the strength of the observed effect. That is: <0.01 as negligible; 0.01 to <0.04 as weak; 0.04 to <0.16 as moderate; 0.16 to <0.36 as relatively strong; 0.36 to <0.64 as strong; and ⩾0.64 as very strong.
For the analysis of differences between specific group pairings, the Dwass–Steel–Critchlow–Fligner (DSCF) post-hoc test was used where the ANOVA revealed a statistical effect. The DSCF test is a two-sided, non-parametric procedure that provides family-wise error (FWE) protection; FWE refers to the probability of making at least one type 1 error when completing a series of statistical tests. 31 The DSCF test procedure was used to calculate a Wilcoxon (W) rank sum test statistic on each pair of groups within each outcome family. 32 Where data are categorical (e.g. activity group based on International Physical Activity Questionnaire (IPAQ) data, health ratings, and health risk group membership), Chi-square (χ2) tests of frequencies were completed to determine whether statistical differences between expected and observed frequencies were present.
We also categorised participants according to their weekly physical activity level (metabolic equivalents of task (MET)-min/week from IPAQ responses) and their time spent (h/day) in sedentary behaviour. The four categories are: (1) high physical activity, low sedentary behaviour; (2) high physical activity, high sedentary behaviour; (3) low physical activity, low sedentary behaviour; and (4) low physical activity, high sedentary behaviour. It was considered that high physical activity and low sedentary behaviour are behaviours which carry a lower risk of morbidity/mortality (i.e. they have a health protective effect), whilst low physical activity and high sedentary time are higher risk behaviours.22,33 The groups were defined using quartiles from each respective outcome. Low physical activity included data from quartiles 1 and 2 of the MET-min/week outcome, while high physical activity from quartiles 3 and 4. Similarly, low sedentary behaviour used data from quartiles 1 and 2 of the summed sitting time variable, with high sedentary behaviour coming from quartiles 3 and 4. 34 Once identified, participants were allocated into a new categorical variable, whilst participants with missing data (i.e. missing MET-min/week or sitting time) were removed from subsequent analyses. Descriptive statistics (median and IQR) were reported, and a Kruskal–Wallis test was completed to assess variability between groups for each outcome. Where statistically significant effects were reported, pairwise comparisons (DSCF) were completed and results reported.
Finally, to calculate the probability of disease incidence between low-risk (i.e. High physical activity, low sedentary behaviour) and high-risk (i.e. Low physical activity, high sedentary behaviour) groups, logistic regression analyses were conducted. The diseases of interest were included in the model as the dependent variable in a dichotomous form (i.e. diagnosed or not diagnosed). The physical activity risk categories were selected as a categorical covariate in the model and the analysis run. The exponential of the coefficient (exp(β)) described the odds of disease incidence between the two groups. However, the odds ratio was transformed into a probability score (p = odds / (1 + odds)), which allowed reporting of the probability that a participant allocated in the high-risk group may have additional morbidities. 35
Results
In total, we identified 319 women with PCOS from the dataset, and we randomly assigned 638 women each to the age-matched and the age- plus BMI-matched groups. The mean age (±standard deviation (SD)) of the PCOS group was 43.9 ± 5.6 years, with the age-matched and age- plus BMI-matched groups being 44.05 ± 5.6 and 44.1 ± 5.6 years, respectively. The descriptive characteristics of the three groups are presented in Table 1. Tests of difference revealed statistical differences between groups for all included outcomes apart from age, stature, total cholesterol, low-density lipoprotein cholesterol (LDL-C), glucose, and oestradiol. Where statistical significance was identified, effect sizes ranged from ε2 = 0.005 (negligible) for sedentary time to ε2 = 0.097 (moderate) for waist circumference (Table 1).
Selected pertinent descriptive characteristics (median and IQR) of study participants and between group comparisons (Kruskal–Wallis), including outcomes relating to sedentary behaviour and physical activity.

IQR: interquartile range; BMI: body mass index; BP: blood pressure; cm: centimetres; CRP: C-reactive protein; HbA1c: glycated haemoglobin; HDL-C: high-density lipoprotein cholesterol; h/day: hours per day; IGF-1: insulin-like growth factor-1; kg: kilograms; kg/m2: kilograms per metre square; LDL-C: low-density lipoprotein cholesterol; MET: metabolic equivalent of task; min/week: minutes per week; mg/L: milligrams per litre; mmHg: millimetres of mercury; mmol/L: millimoles per litre; MPA: moderate-intensity physical activity; nmol/L: nanomoles per litre; p: significance level; PA: physical activity; Screen time: summed values of computer usage and TV viewing; Sedentary time: screen time and driving time summed; SHBG: sex hormone-binding globulin; VPA: vigorous-intensity physical activity; WHR: waist-to-hip-ratio; WHtR: waist-to-height-ratio; χ2: chi-square statistic; ε2: epsilon square effect size; reported outcomes are based upon fasted data.
When pairwise comparisons were made, most differences were found when the PCOS cohorts were compared to the age-matched control group (Table 2). There were, however, four outcomes that were statistically different when the PCOS group was compared to the age- plus BMI-matched group; high-density lipoprotein cholesterol (HDL-C), triglycerides, glycated haemoglobin (HbA1c), and SHBG all had less favourable values in the PCOS cohort (Table 2).
DSCF pairwise comparisons of between group characteristics.
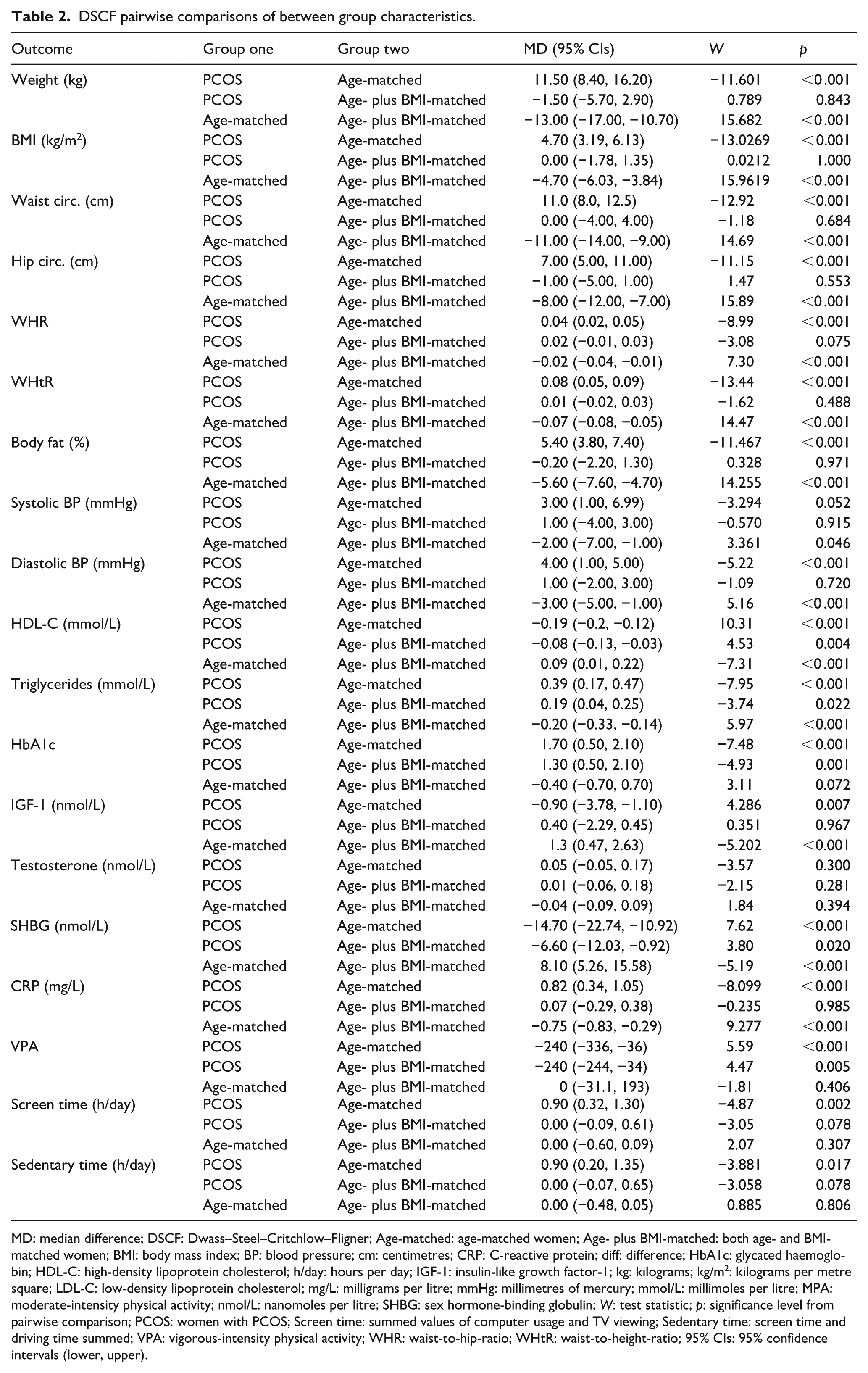
MD: median difference; DSCF: Dwass–Steel–Critchlow–Fligner; Age-matched: age-matched women; Age- plus BMI-matched: both age- and BMI-matched women; BMI: body mass index; BP: blood pressure; cm: centimetres; CRP: C-reactive protein; diff: difference; HbA1c: glycated haemoglobin; HDL-C: high-density lipoprotein cholesterol; h/day: hours per day; IGF-1: insulin-like growth factor-1; kg: kilograms; kg/m2: kilograms per metre square; LDL-C: low-density lipoprotein cholesterol; mg/L: milligrams per litre; mmHg: millimetres of mercury; mmol/L: millimoles per litre; MPA: moderate-intensity physical activity; nmol/L: nanomoles per litre; SHBG: sex hormone-binding globulin; W: test statistic; p: significance level from pairwise comparison; PCOS: women with PCOS; Screen time: summed values of computer usage and TV viewing; Sedentary time: screen time and driving time summed; VPA: vigorous-intensity physical activity; WHR: waist-to-hip-ratio; WHtR: waist-to-height-ratio; 95% CIs: 95% confidence intervals (lower, upper).
Physical activity and sedentary behaviour
There were no statistical between group differences for total MET-min/week, summed weekly minutes of physical activity, or MET-min/week of moderate-intensity physical activity (Table 1). However, a statistical difference (p < 0.001; ε2 = 0.01165) in MET-min/week achieved through vigorous-intensity physical activity was identified. Pairwise comparisons (Table 2) showed that these differences lay between the PCOS group and the age-matched cohort (median difference (MD): −240 MET-min/week; p < 0.001), and between the women with PCOS and the age- plus BMI-matched group (MD: −240 MET-min/week; p = 0.005); in both instances, women with PCOS performed significantly less vigorous-intensity physical activity per week. There were no statistical differences when the two non-PCOS groups were compared.
For both the screen time outcome, derived from the summation of TV viewing time and computer usage (h/day), and the sedentary time outcome, derived from screen time summed with driving time, statistically significant differences were identified (p = 0.003; ε2 = 0.007, and p = 0.022; ε2 = 0.005, respectively) (Table 1). Post-hoc tests for both these outcomes (Table 2) revealed that between group differences lay between the PCOS group and the age-matched group only (MD screen time: 0.9 h/day; p = 0.002, and MD sedentary behaviours: 0.9 h/day; p = 0.017).
When activity levels were assessed via the IPAQ categorical scores, a statistical effect was found (χ2 = 10.7; p = 0.031), indicating that there is a statistical difference between the expected and the observed frequencies in one or more categories (Table 3). The frequencies reported indicate that a greater percentage of women with PCOS (30.4%) are in the lowest level activity group when compared to the other groups (age-matched: 20.7%, and age- plus BMI-matched: 21.9%). Furthermore, a smaller percentage of women with PCOS are categorised as achieving the highest activity levels (30.8% versus 38.2% and 37.7%, respectively).
Frequencies of IPAQ activity categorisation, self-rated health, and risk group (based upon behaviour) for each study group.

Data are presented as number of participants in each categorical group (percentage of study group); all percentage data rounded to one decimal place.
age-matched: control group of women age-matched only to case; age- plus BMI-matched: control group of women both age- and BMI-matched to case; BMI: body mass index; IPAQ: International Physical Activity Questionnaire; PCOS: polycystic ovary syndrome; PA: physical activity; SB: sedentary behaviour; Risk group: categorical variable based upon reported physical activity and sedentary behaviours (lowest risk = high PA plus low SB; and highest risk = low PA plus high SB).
Self-rated health and comorbidity
All UK Biobank participants were asked to self-rate their perceived health. A chi-squared analysis revealed a statistical effect (χ2 = 80.1; p < 0.001), indicating a difference between the observed and expected frequencies (Table 3). Compared to the age-matched group and the age- plus BMI-matched group, a lower percentage of women with PCOS rated their health as either “Excellent” or “Good.” Furthermore, a higher percentage (18.2%) also self-rated their health as “Poor.”
When tests of frequencies were analysed for comorbidities (Table 4), statistically significant chi-squared tests were returned for type 2 diabetes, hypertension, hypercholesterolaemia, obesity, kidney disease, chronic lung disease, and benign cancers. Without exception, prevalence was higher for individuals with PCOS than the other two groups.
Frequencies and χ2 tests of frequencies for comorbidities, based on ICD codes, between the three study groups.
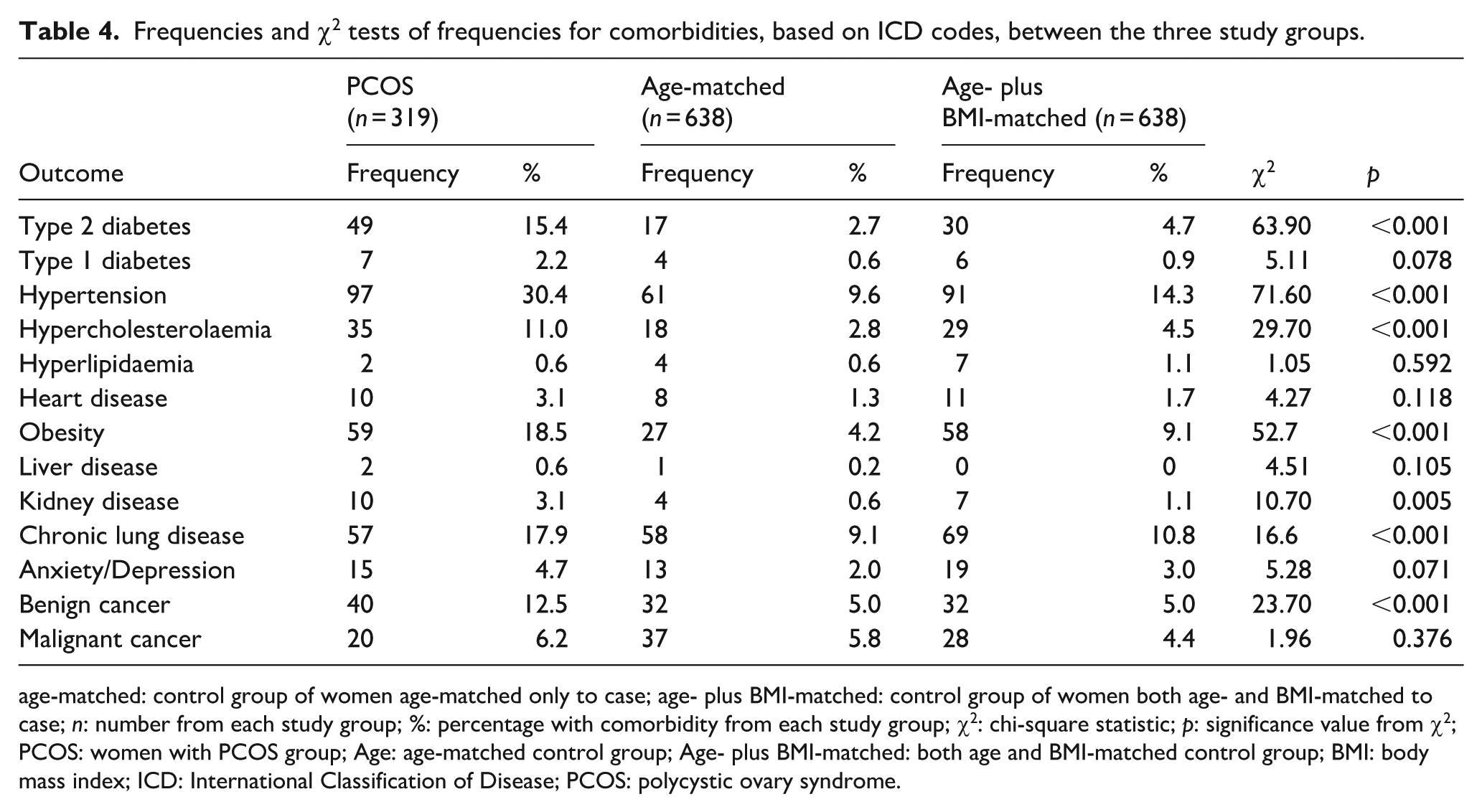
age-matched: control group of women age-matched only to case; age- plus BMI-matched: control group of women both age- and BMI-matched to case; n: number from each study group; %: percentage with comorbidity from each study group; χ2: chi-square statistic; p: significance value from χ2; PCOS: women with PCOS group; Age: age-matched control group; Age- plus BMI-matched: both age and BMI-matched control group; BMI: body mass index; ICD: International Classification of Disease; PCOS: polycystic ovary syndrome.
When the total number of comorbidities, and only those linked to metabolic health (i.e. type 2 diabetes, hypertension, hypercholesterolaemia, hyperlipidaemia, heart disease, and obesity) were summed (Supplemental Table 2), a Kruskal–Wallis test revealed differences for both total comorbidities (χ2 = 109; p < 0.001) and metabolic morbidity (χ2 = 116; p < 0.001). Pairwise analyses revealed that women with PCOS had significantly more total comorbidities (PCOS versus age-matched: W = −14.26; p < 0.001; PCOS versus age- plus BMI-matched: W = −11.12; p < 0.001), and metabolic comorbidities (PCOS versus age-matched: W = −14.83; p < 0.001; PCOS versus age- plus BMI-matched: W = −10.45; p < 0.001) compared to both comparator groups.
Risk profiles of physical activity and sitting
When the physical activity and sitting data were used to create a new categorical variable intended to reflect risk of ill health, 207 participants were excluded because data from at least one of these two variables were missing (i.e. physical activity or sitting time). Most missing cases (n = 199) came from missing data on MET-min/week performed. The mean ± SD of total physical activity (MET-min/week) and sedentary behaviour (h/day), which were used to define groups are presented in Supplemental Table 3, whilst the descriptive characteristics (median (IQR)) of key study outcomes, and results of Kruskal–Wallis tests are presented in Supplemental Table 4. Statistical significance was identified for waist circumference, BMI, systolic and diastolic BP, CRP, HbA1c, HDL-C, insulin-like growth factor-1 (IGF-1), triglycerides, and SHBG. Pairwise comparisons (Supplemental Table 5, and Supplemental Figures 1 and 2), showed that the greatest number of statistical differences (n = 11), and the magnitude of those differences, were between the low risk (i.e. High physical activity, low sedentary behaviour) and the high-risk (i.e. Low physical activity, high sedentary behaviour) groups. Without exception, outcomes favoured the risk groups with more physical activity and less sitting time.
Finally, morbidity prevalence was reported for the lowest risk group, and comparisons were made to the highest risk group, showing that there was a 63% greater chance of having PCOS if in the high-risk group. Furthermore, for all other reported morbidities, prevalence was greater, and risk increased if in the high-risk group (Figure 1).

Prevalence of morbidity for those with low-risk behaviours (high physical activity and low sedentary/sitting time) and relative risk of disease incidence for those in the high-risk group (low physical activity and high sedentary/sitting time).
Discussion
The findings of the present study offer novel insight based on UK Biobank data regarding risk/disease characteristics of individuals with PCOS, which may be attributed to the PCOS diagnosis per se and/or to obesity as a key comorbidity of PCOS. As such, the present study showed that the identified statistical differences relating to anthropometry/body composition between the participants with PCOS who are included in the UK Biobank, and an age-matched control group were not present when the group with PCOS was compared to a control group which was both age- and BMI-matched. This suggests that no significant differences exist in common obesity indices and the distribution of adiposity in women with PCOS when compared to counterparts of similar age and BMI without PCOS. Whilst it has been widely reported that those with PCOS tend to be more overweight than their non-PCOS counterparts,13,36,37 more data are still required which compare the body compositional outcomes of women with PCOS to (age and) BMI-matched women without PCOS. Within the current study, the control women in the age- plus BMI-matched cohort were selected so that their BMI closely matched to the women with PCOS, but their waist circumference or their body fat percentage were not factored into their inclusion for matching. Previous studies report that the distribution of adipose tissue in women with PCOS tends to be more central,38 –41 [resulting in greater metabolic disruption related to central obesity. However, this does not appear to be evident across the current UK Biobank sample since there are negligible differences for waist circumference (MD: 1 cm) and body fat (MD: 0.01%) between the PCOS and the (age and) BMI-matched control group without PCOS. These findings tend to agree with some previous studies albeit in much smaller samples. Although individuals with PCOS have increased risk of higher BMI than women without PCOS, when BMI is matched, the differences in the volume and distribution of adipose tissue appears to be less prominent. More studies are needed to further address this research question, employing more precise assessments of adipose tissue accumulation/distribution [e.g. Magnetic Resonance Imaging (MRI)-assessments of visceral fat]; however, at the population level, common obesity/adiposity indices (e.g. waist circumference, waist-to-hip-ratio, and waist-to-height-ratio) and/or less accurate body composition methods (e.g. bioelectrical impedance analysis) are typically utilised.
Comparing the PCOS and age-matched groups, we also found statistical differences for diastolic BP, HDL-C, triglycerides, HbA1c, IGF-1, total testosterone, SHBG, and CRP; six of those differences (not HbA1c or testosterone) were also reported when comparing the age-matched to the age- plus BMI-matched group. When comparing the PCOS to the age- plus BMI-matched group, statistical differences for HDL-C, HbA1c, triglycerides, and SHBG were also found. For each of these observed differences, the group with PCOS had less favourable values compared to the other control groups.
In addition to the measured/objective outcomes noted above, those with PCOS tended to rate their own health less favourably (i.e. perceived health rating) than either comparator group. This was in line with the findings regarding the prevalence of diagnosed type 2 diabetes, hypertension, hypercholesterolemia, obesity, kidney disease, chronic lung conditions, and non-invasive cancers, which were also statistically higher in individuals with PCOS compared to the two control groups. Indeed, when both total and metabolic-related morbidities were compared, they were statistically higher in PCOS than either control group.
In agreement with previous studies,13,15 –17 there were no statistical differences in the total amount of physical activity performed per week (either MET-min/week or summed min/week), but individuals with PCOS performed less vigorous-intensity physical activity than the comparators. Differences were also identified for sedentary behaviours; screen time and summed sitting time were statistically higher in the PCOS group than the age-matched group. To further support these findings, when the IPAQ categorisation data were assessed for each group, a greater proportion (~9% more) of women with PCOS were in the low activity category, and fewer in the high-activity category, than either comparator control group. Engaging in regular physical activity can help to minimise weight and fat mass gain, 42 whilst maintenance of a physically active lifestyle is an effective strategy to improve obesity-related health outcomes irrespective of any concomitant weight loss. 43 However, when the physical activity levels of these women are compared, there are no statistical differences between each study group for the MET-min/week performed or the weekly minutes spent engaged in physical activity. These findings agree with previous studies which included measures of self-reported physical activity.13,15,16,44 –46 Lin et al. 17 further supported these findings by incorporating device-measured physical activity and physical activity intensity levels; they reported no statistical differences in duration, type, or intensity of activity. Whilst the UK Biobank did not capture the type of physical activity (beyond walking) that participants engaged in, the relative intensity was captured and this is where, in contrast to Lin et al., 17 significant differences were identified. Women with PCOS achieved fewer MET-min/week through vigorous-intensity physical activity than either the age-matched (MD: 185.8 MET-min/week; p < 0.001) or age- plus BMI-matched (MD: 104.8 MET-min/week; p = 0.005) groups. No such differences were observed between the age-matched and the age- plus BMI-matched groups despite the differences in BMI, body fat, and waist circumference. Whilst evidence supporting the role of vigorous-intensity physical activity for reducing morbidity47,48 and mortality49,50 risk is promising, its relevance for body composition management in the general population is less certain.51,52 However, current evidence in those living with PCOS53,54 indicates that engaging in vigorous aerobic exercise can have a beneficial effect on body composition, waist circumference, and BMI. This may be particularly pertinent, since those with PCOS and increased BMI who achieve reductions in central adiposity 55 or a 5–10% decrease in body weight 9 typically demonstrate improvements across a range of symptoms. It is likely that vigorous-intensity physical activity may represent a time-efficient benefit to those with PCOS, but uncertainties around how higher intensity exercise may affect adherence, 56 or indeed what the optimal frequency, time, and type should be, suggest that future studies should aim to answer these questions.
Another contrasting factor is time spent engaged in sedentary behaviours; compared to the age-matched cohort, those with PCOS reported more screen time and more total sedentary time (0.4 h/day). Sedentary time is a known risk factor, independent of physical activity, 57 for a multitude of undesirable health outcomes, and, although sedentary behaviours may be a contributing factor to less favourable body composition, similar differences were not evident between the age- plus BMI-matched and the age-matched cohort. This could suggest that sedentary time may not be a factor in outcomes relating to body composition for this study population and is likely due to metabolic dysregulation associated with PCOS. However, the chronic effects of longer-term increased sedentary time may further exacerbate associated health complications.
Collectively, the findings of this study, alongside previous studies, indicate that women with PCOS have increased body weight and BMI compared to age-matched women without PCOS, but it appears that total self-reported physical activity and sedentary behaviour levels may not be predictors of this phenomenon. It is likely that the magnitude of differences in physical activity and sedentary time reported here are not responsible for observed differences in BMI, but, as in any population, they may be contributing factors. These findings highlight the need for a comprehensive study that incorporates device-measured physical activity and sedentary time alongside a detailed dietary analysis to gain a fuller understanding of their respective impact.
If factors pertaining to lifestyle are not fully implicated in overweightness and obesity in PCOS, the findings of this study may be able to implicate some additional mechanisms. Teede et al. 58 state that PCOS is an independent risk factor for overweightness/obesity, but that a bidirectional interaction exists, where PCOS causes a physiological and/or psychological environment which promotes weight gain, and weight that is gained leads to increased prevalence and severity of PCOS symptoms. It is apparent from the study results that the less-favourable values observed for most outcomes are likely due to excess body weight and/or increased adiposity. Indeed, for women with PCOS and the age- plus BMI-matched cohort, most circulating biomarkers and physical measurements are significantly worse than the age-matched (and lower weight) group. However, four outcomes are statistically less favourable in women with PCOS than the age- plus BMI-matched study group, namely differences in HbA1c, HDL-C, triglycerides, and SHBG levels.
When participants were classified into a group based upon their health behaviours (i.e. physical activity and sedentary behaviour), statistical differences for waist circumference, BMI, diastolic and systolic BP, HbA1c, CRP, HDL-C, IGF-1, SHBG, and triglycerides were identified, which, without exception, favoured the low-risk group (i.e. high physical activity, low sedentary behaviour). When considering morbidity, those in the highest risk group (i.e. low physical activity, high sedentary time) were 23% more likely to have type 2 diabetes or heart disease, 22% more likely to have hypertension, 21% more likely to have obesity, and 31% more likely to have anxiety or depression, when compared to the lowest risk group. In addition, there was also a 62% increased risk of having PCOS in the high-risk group. However, in order to establish causal links and potential bidirectional links and causal effects between PCOS symptoms/severity, lifestyle behaviours, and cardio-metabolic risk, large prospective studies are required.
The results from the current study also provide additional information about the association of physical activity to a range of health-related parameters in those with PCOS (and indeed the control groups). Whilst there were only small differences between study groups for physical activity and sedentary behaviours, when all participants were grouped according to those health risk behaviours, there were some notable differences for a range of outcomes between risk groups. When the lowest risk group (i.e. the most active and least sedentary) were compared to the highest risk group (i.e. the least active and most sedentary), there are statistical differences for a range of markers/indices linked to body weight/composition (i.e. waist circumference and BMI), cardio-metabolic health (i.e. diastolic and systolic BP, HbA1c, HDL-C, triglycerides, IGF-1, and CRP), and androgenic profile (i.e. SHBG). The benefits of being active are further indicated by a reduced risk for key non-communicable diseases (i.e. type 2 diabetes, heart disease, hypertension, hypercholesterolaemia/hyperlipidaemia, anxiety/depression, and obesity) in the low-risk group.
Current physical activity guidelines 59 recommend that activities equivalent to 600 MET-min/week are beneficial for good mental and physical health. In this study, the median values reported by all study groups (i.e. PCOS or controls), far exceed this recommendation, whilst the average values for even those in the high-risk group are almost equivalent. The all-cause mortality benefits of physical activity are based upon an inverse linear dose-response relationship; 60 it has previously been reported that, when compared to those engaging in no physical activity, those performing three to five times the recommendations have a 39% lower risk of premature all-cause mortality. 61 Furthermore, an additional 4.5 years in life expectancy can be gained when weekly physical activity exceeds 1350 MET-min/week. 62 When considering morbidity rather than mortality, it is those completing the greatest volume of physical activity that have the greatest risk reduction for chronic disease. For example, those achieving five to six times the physical activity guidelines (as in the low-risk group) have relative risk reductions of 28% for type 2 diabetes, 26% for ischaemic stroke, and 26% for coronary artery disease compared to those falling below the guidelines. 63
Time spent in sedentary behaviours may offer another possible explanation for health variability. Sedentary behaviour is an independent risk factor for chronic disease and premature mortality. 64 In fact, it has been demonstrated that individuals must complete high levels (>35.5 MET-h/week) of physical activity to protect against prolonged bouts of sitting. 65 In a longitudinal study, 66 prolonged daily sitting has been demonstrated to be a predictor of fasting insulin, maximum oxygen consumption (VO2 max), and waist circumference, independent of physical activity.
Some potential mechanisms to explain the independent association of sedentary behaviours with cardio-metabolic health have been purported; lipid and glucose metabolism are severely impaired during prolonged sedentary periods, due to the inactivity of large skeletal muscles which are usually engaged when standing/moving.67,68 Another suggested mechanism relates to the dysregulation of hemodynamic vascular signalling: that is an absence of exercise-induced vascular dilation which promotes inflammatory-mediated atherogenesis,69,70 leading to a narrowing of blood vessels. Finally, the timings of sedentary behaviour may also be key in determining the degree of harm. For example, Almoosawi et al. 71 suggest that prolonged screen time is more likely to occur in the evening, following many people’s main meal of the day, with the implication being that postprandial glucose and lipids are elevated during that sedentary period with a potentially harmful effect to cardiovascular health.
Overall, the findings of the present study indicate that, compared to the most active individuals, those who are the least physically active and spend the most amount of time in sedentary behaviours have the least favourable markers of metabolic health, regardless of their health status (i.e. PCOS or control). Moreover, it appears that women who have PCOS are more likely to be classified as high risk based on their activity levels; this categorisation means that they have an increased risk of developing additional chronic health conditions. In this context, it is reasonable to assume that unless physical activity is increased with a parallel decrease in time spent sitting, the metabolic health of this population is at greater risk to further deteriorate and the sequelae of PCOS could be further exacerbated.
Limitations
Whilst one of the strengths of the current study is the inclusion of a large national sample of participants with PCOS and two large comparator groups of matched participants, certain study limitations should be acknowledged. Indeed, the UK Biobank project reports participants’ health conditions and ICD codes which allow for the women with PCOS to be identified within this UK national population, but it does not report any further diagnostic criteria or phenotypic subgroups for PCOS. It has previously been reported 72 that there are variations in the biochemical profiles of women with PCOS dependent upon the specific phenotype/criteria with which they have been diagnosed. Similarly, there are reportedly variations in how PCOS manifests 73 and indeed the levels of physical activity and sedentary behaviour performed,74,75 depending upon ethnic background. Whilst most participants in the UK Biobank are from a white European background (94.6%), 76 unfortunately, data on ethnic background were not captured during the present study. It is likely that most participants were from a white background, but future studies should report ethnicity and seek to evaluate whether it is a contributing factor to health and health behaviours.
Since the study objective was to identify all participants who were diagnosed with PCOS, an a-priori power calculation was not performed. However, to increase statistical power, we recruited participants in the control groups on a 2:1 ratio.
There were also fewer participants with PCOS identified than anticipated, fewer than 0.5% of female participants had a documented diagnosis of PCOS, compared to up to 15% in the general population. 2 The reason for this is unclear but could be due to a high rate of women with PCOS who remain undiagnosed, and/or due to errors/omissions in precisely classifying/reporting the PCOS diagnosis of participants. Thus, there is potential for contamination (i.e. women with PCOS incorrectly classified as not having PCOS and vice versa), which may have influenced the results of the present study. Moreover, data on the menopausal status of these participants were not available from the UK Biobank and, thus, it is also possible that the relatively older age of these women, and their menopausal status could have impacted upon the present findings.
Another limitation is the lack of certain data/variables, such as circulating hormones, including luteinising hormone, 77 follicle stimulating hormone, 78 anti-Mullerian hormone, 79 and progesterone 80 levels, which are implicated in the pathophysiology of PCOS and/or the severity of its symptoms. However, these hormones were not available in the UK Biobank data set. Furthermore, PCOS-specific quality of life measures, such as the PCOSQ (a disease-specific quality of life measure for PCOS), are also not available in the UK Biobank dataset.
The age of included participants can also be considered a limitation of this study. UK Biobank participants are aged between 40 and 69 years; thus, generalising the present findings for all PCOS age groups requires caution.
There are also potential limitations in the sedentary behaviour data, since the UK Biobank data do not explicitly ask about total daily sitting time. Indeed, the captured data sums total minutes driving, using a computer and watching TV; hence, this may miss other sedentary activities from participants’ day-to-day routines. However, there is some evidence 81 that multi-item questionnaires may result in more accurate reporting of self-report sedentary time. In addition to questionnaire format, there is no evaluation of when these sedentary periods occurred making it difficult to establish the role of the aforementioned mechanisms in metabolic health. Finally, it is unclear whether the sedentary bouts were continuous or accumulated throughout the day; Rutten et al. 82 found that interrupting prolonged bouts of sedentary time with short activity breaks was beneficial for a range of CVD risk factors.
Conclusion
In the current study, more women with PCOS were classified as having increased BMI (overweightness/obesity) with poorer markers of metabolic health compared to an age-matched group. The findings that women with PCOS are at a greater risk of poor cardio-metabolic health are generally supported by the existing relevant research. Moreover, the current study benefitted from a large comparator group which was matched both for BMI and age, thus removing a variable that potentially has a large mediating role in an individual’s health. Indeed, the current study revealed that a higher proportion of women with PCOS self-rated their health as “poor” (+10.4%), compared to their age- plus BMI-matched counterparts. This may be a fairly crude indicator that PCOS per se, as opposed to elevated BMI, is having a negative effect upon their own health perceptions. Moreover, the objectively measured metabolic and hormonal biomarkers also indicate that women with PCOS have worse health profiles, or increased risk of worse health, than their counterparts without PCOS. It is true that the greatest observed differences were between women with PCOS and the age-matched cohort only, whilst it is also pertinent that some statistical differences were lacking when women with PCOS were compared to the BMI-matched group. This is indicative that increased body weight/adiposity has a significant role in metabolic dysregulation, and clearly, if women with PCOS are more susceptible to overweightness and obesity, this should be a treatment priority.
The current study also agrees with previous studies in that there are no differences in the amount of physical activity performed between women with PCOS and the control groups. However, in contrast to findings from previous studies, herein, it was apparent that women with PCOS performed less vigorous-intensity physical activity each week and spent more time in sedentary behaviours. The cumulative effect of this may explain some of the variability, but further research is required to assess the true magnitude of the impact of these parameters on health-related outcomes in PCOS.
Overall, considering both the findings and the limitations of the current study, a need for further population-based and prospective studies in individuals living with PCOS is also highlighted. The UK Biobank is a valuable resource for population level inquiry, but large prospective studies are essential to elucidate causal links particularly for certain complex conditions such as PCOS. Indeed, studies such as this one offer further insight which can generate subsequent research ideas and informed hypotheses, but are also limited in their ability to make definitive statements/conclusions (e.g. on causal links within the PCOS pathophysiology). This reiterates the need for robustly designed and well-conducted studies in the PCOS population, which is characterised by markedly increased risk for cardio-metabolic diseases.
Supplemental Material
sj-docx-1-whe-10.1177_17455057251385800 – Supplemental material for Metabolic, androgenic, and physical activity profiles in women aged over 40 years with polycystic ovary syndrome: A comparative analysis using UK Biobank data
Supplemental material, sj-docx-1-whe-10.1177_17455057251385800 for Metabolic, androgenic, and physical activity profiles in women aged over 40 years with polycystic ovary syndrome: A comparative analysis using UK Biobank data by Chris Kite, Ioannis Kyrou, Harpal S. Randeva, Ian M. Lahart and James E. P. Brown in Women's Health
Footnotes
Acknowledgements
All authors acknowledge the UK Biobank for their support and guidance during the design and implementation of this study. Also, we thank the participants of the UK Biobank for their involvement in bettering population health.
Ethical considerations
UK Biobank has approval from the Northwest Multi-centre Research Ethics Committee as a Research Tissue Bank (RTB). This approval means that researchers do not require separate ethical clearance and can operate under the RTB approval.
Consent to participate
When they first joined the study, all participants in the UK Biobank gave explicit consent for UK Biobank to access all their medical and health-related records, and for the long-term storage and use of any samples (i.e. blood and urine).
Consent for publication
Approval of reports is not required by the UK Biobank. All participant data are anonymised, and consent to publish reports is implicit in the participant consent process.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was completed as part of a funded PhD project (CK) through Aston University. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available upon request from the UK Biobank. The authors of this paper are not authorised to directly share the dataset.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
