Abstract
Background:
Symptomatic hand osteoarthritis frequently affects perimenopausal women and is believed to be associated with estrogen deficiency. However, effective medical therapies for symptomatic relief remain limited.
Objective:
To compare the effectiveness of menopausal hormone therapy and complementary and alternative medicine in treating symptomatic hand osteoarthritis in perimenopausal women.
Design:
Retrospective observational study.
Data sources and methods:
We retrospectively reviewed the medical records of 73 perimenopausal Japanese women treated for symptomatic hand osteoarthritis at Kagoshima University Hospital between 2019 and 2022. Fifty-four patients received menopausal hormone therapy, and 19 received complementary and alternative medicine (Kampo formula and/or S-equol supplementation). No patients received additional orthopedic treatments for hand osteoarthritis, such as analgesics, splinting, rehabilitation, or injections. The outcomes included the disability of the arm, shoulder, and hand score and visual analog scale score for hand pain, which were assessed at baseline and 3 months. Two-factor repeated-measures analysis of variance was used to assess condition-by-time interactions, and effect sizes were calculated. For outcomes with significant interactions, post hoc Mann–Whitney U tests were used to compare between-group changes.
Results:
Significant condition-by-time interactions were found for the disability of the arm, shoulder, and hand score (F = 12.85, p = 0.0007, partial η2 = 0.17) and visual analog scale score (F = 7.39, p = 0.008, partial η2 = 0.02), indicating that treatment effects differed between groups over time. Post hoc analyses revealed that the menopausal hormone therapy group showed significantly greater improvements than the complementary and alternative medicine group in both the disability of the arm, shoulder, and hand (10.85 versus −1.75 points, p = 0.003) and visual analog scale scores (27.93 versus 9.17 mm, p = 0.02). The mean improvement in disability of the arm, shoulder, and hand scores in the menopausal hormone therapy group exceeded the minimal clinically important differences, supporting both statistical and clinical significance.
Conclusions:
Compared with complementary and alternative medicine, menopausal hormone therapy showed superior efficacy in improving hand pain and dysfunction associated with symptomatic hand osteoarthritis in perimenopausal Japanese women.
Plain language summary
This study looked at Japanese women in their 50s with hand pain caused by arthritis, which can be linked to low estrogen levels around menopause. We compared two treatments: menopausal hormone therapy that adds back estrogen and a group of traditional and natural therapies called complementary and alternative medicine. We reviewed medical records from 73 women treated at a university hospital over 3 years. We found that women who received menopausal hormone therapy had much greater improvement in hand pain and daily hand use compared to women who used complementary and alternative medicine. Importantly, these improvements were not only statistically significant (meaning unlikely due to chance) but also big enough to be meaningful for real life. Menopausal hormone therapy works by restoring estrogen levels, which helps protect joints and reduce pain sensitivity. In contrast, complementary and alternative medicine treatments had only minor effects. Our results suggest that for women around menopause who have arthritis in their hands, hormone therapy may be a very effective treatment to improve their quality of life. We recommend doctors consider hormone therapy as part of care for these patients, while recognizing that more research is still needed.
Keywords
Introduction
Hand osteoarthritis (HOA) is a heterogeneous disorder involving lesions in the joints of the thumb and fingers, with symptoms ranging from asymptomatic to varying degrees of pain, stiffness, and functional limitations.1,2 Although HOA has historically received less attention than knee or hip osteoarthritis (OA), recent epidemiological data indicate that its prevalence is comparable to or even higher than that of these other joint sites. According to population-based studies, HOA affects 14%–22% of individuals aged 45 years and older, compared to 17%–35% for knee OA and 4%–10% for hip OA, depending on the population and diagnostic criteria used.3 –5 HOA can be broadly categorized into two clinical entities: radiographic HOA, defined by imaging findings, and symptomatic HOA, characterized by pain and disability. The latter is of particular clinical importance because it interferes with daily activities and significantly reduces quality of life. Notably, the severity of symptoms in patients with symptomatic HOA does not necessarily correlate with radiographic changes.
The management of symptomatic HOA includes several nonpharmacological and pharmacological options. Nonpharmacological therapies include patient education, exercise, and splinting. 1 Pharmacological interventions include topical or limited-use oral nonsteroidal anti-inflammatory drugs, intra-articular corticosteroids, and surgery in advanced cases. Despite these options, no standard treatment strategy has been established because of the lack of comparative clinical studies. 1
Approximately 90% of patients seeking care for hand symptoms are women, most of whom develop symptoms in their 50s, the typical age of perimenopause.6,7 This epidemiological pattern suggests a potential link between declining estrogen levels and the onset or exacerbation of HOA symptoms. Menopausal hormone therapy (MHT) and complementary and alternative medicine (CAM), such as the Kampo formula and S-equol supplementation, have been used to alleviate menopausal symptoms.8 –13 MHT involves the direct replacement of systemic estrogen using preparations such as estradiol and progesterone, typically in combination, for women with an intact uterus. 14 In contrast, CAM therapies aim to indirectly modulate the hormonal balance or mimic estrogenic activity through nonhormonal mechanisms. Kampo formulas are traditional Japanese herbal medicines believed to act via autonomic and inflammatory modulation, although high-quality evidence remains limited. S-equol, a soy isoflavone metabolite with selective affinity for estrogen receptor β (ERβ), exerts anti-inflammatory and antioxidant effects.15,16
Because of these mechanistic differences, the therapeutic intensity, onset of action, and symptom relief profiles may differ between MHT and CAM. Although both have been reported to improve musculoskeletal symptoms during menopause, such as body aches and stiffness, no studies have focused specifically on their effects on hand joint pain.2,12,17 Therefore, this study aimed to investigate whether MHT or CAM, both commonly used for menopausal symptoms, are effective at relieving symptomatic HOA.
Methods
Study design and reporting guideline
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 18
Patient data
This study retrospectively analyzed the medical records of 169 ethnically Japanese women treated for menopausal disorders, including hand pain, at our institute between April 2019 and December 2022. The inclusion criteria included an age under 60 years and availability for at least 3 months of follow-up. Patients with carpal tunnel syndrome, tendinopathy (e.g. trigger digit or de Quervain disease), and rheumatoid arthritis were excluded. A diagnosis of symptomatic HOA was established according to the European League Against Rheumatism classification criteria. 19 During the study period, patients were offered only gynecological treatments (MHT, Kampo formula, or S-equol supplementation), and their preferred treatment was selected after receiving a standardized explanation of the available options. No participants received analgesics, splints, rehabilitation, or intra-articular injections.
Patients’ clinical characteristics, including age; disease duration; body mass index (BMI); the simplified menopausal index (SMI); the self-rating questionnaire for depression (SRQ-D); menopausal status; disability of the arm, shoulder, and hand (DASH) score; visual analog scale (VAS) score; and hand grip (HG) strength (dominant/nondominant), were collected from the medical records. The DASH and VAS scores were recorded before and 3 months after treatment. The differences between the baseline and 3-month posttreatment values were calculated. All the questionnaires were administered in a traditional article-and-pencil format, in which participants completed printed forms by hand during their outpatient visit. This method ensured accessibility for all age groups and allowed immediate review by clinical staff.
Intervention details
MHT was administered based on menopausal status and the presence or absence of a uterus.
Estrogen therapy (ET) was prescribed for women without a uterus. These patients received transdermal estradiol at 0.72 mg every 2 days.
Estrogen–progestogen therapy (EPT) was used for women with an intact uterus.
In premenopausal women, a cyclic regimen was employed, combining transdermal estradiol (0.72 mg every 2 days) with intermittent oral progestogen—either dydrogesterone (10 mg/day) or micronized progesterone (200 mg/day).
In postmenopausal women, a continuous regimen was used, with two options: a. Transdermal estradiol (0.72 mg every 2 days) plus continuous oral progestogen—either dydrogesterone (5 mg/day) or micronized progesterone (100 mg/day) or b. A transdermal combination patch that delivers 0.62 mg estradiol and 2.7 mg norethisterone acetate is applied twice weekly.
All treatments were continued for 12 weeks.
The CAM treatments included various Kampo formulas and/or S-equol supplementation (10 mg/day orally). Kampo prescriptions were individualized by gynecologists based on the symptom patterns and were typically taken two to three times per day according to the manufacturer’s guidelines. All CAM treatments were continued for 12 weeks.
Disability of the arm, shoulder, and hand score
The DASH score is a validated, patient-reported outcome measure used to assess upper extremity disability and symptoms. It consists of 30 items that evaluate physical function and symptom severity in the arm, shoulder, and hand. Each item is scored on a five-point Likert scale, and the total score ranges from 0 to 100, with higher scores indicating greater disability. The DASH score has been widely used in clinical research and practice to evaluate treatment outcomes for musculoskeletal disorders affecting the upper limb. 20
Simplified menopausal index
The SMI is a 10-item self-administered questionnaire commonly used in Japan to assess menopausal symptoms. It includes vasomotor (e.g. hot flashes), somatic (e.g. joint pain), and psychological (e.g. irritability) components. The total score reflects the overall symptom burden. 21
Self-rating questionnaire for depression
The SRQ-D is a validated screening tool designed to identify depressive symptoms in clinical populations. It consists of 18 yes/no items covering the emotional, physical, and behavioral symptoms associated with depression. The total score ranges from 0 to 18. A score of 10 or below indicates no depressive tendency, scores between 11 and 15 suggest borderline depression, and scores of 16 or higher indicate a depressive tendency. This tool has been widely used in Japanese clinical settings for screening masked depression, particularly in primary care and psychosomatic medicine. 22
Sample size
The sample size was determined through an a priori analysis via G*Power version 3.1.9.6 (Franz Faul, Kiel University, Kiel, Germany). The primary endpoint was the change in the DASH score before and after treatment. Based on a two-factor repeated-measures analysis of variance (ANOVA) model (condition × time), a moderate effect size (partial η2 = 0.06, corresponding to f = 0.25), a significance level of α = 0.05, and a desired power of 0.80 were assumed.
The effect size was chosen based on previous studies evaluating functional improvement in patients with upper limb disorders and considering a clinically relevant moderate change. As a result, the minimum required total sample size was calculated to be 70 participants. Our study included 73 participants, who met the calculated sample size requirement. Although the total sample size met the required threshold, the unequal group sizes (MHT n = 54, CAM n = 19) may have affected the statistical power for between-group comparisons. This limitation was addressed in the “Discussion” section.
Statistical analysis
Baseline characteristics (age, disease duration, BMI, SMI, SRQ-D, menopausal status, DASH score, VAS score, and HG) were compared using the Mann‒Whitney U test and chi-square test, as appropriate. Since no significant between-group differences were found, covariate adjustment was not performed.
Two-factor repeated-measures ANOVA was performed to examine the effects of the treatment group (MHT versus CAM) and time (pre- versus posttreatment) on functional outcomes. When a significant interaction was observed, post hoc comparisons using the Mann‒Whitney U test were performed to compare change scores (i.e. post-pre) between the two groups. These Mann–Whitney U tests were not considered multiple comparisons but rather exploratory post hoc analyses, each performed only once per outcome variable (e.g. DASH or VAS score). Therefore, correction for multiple comparisons (e.g. the Bonferroni adjustment) was not applied. Effect sizes (r for nonparametric tests and partial η2 for ANOVA) were calculated, and a two-tailed p < 0.05 was considered statistically significant. 23
Results
Of the 169 patients initially reviewed, 96 were excluded because of missing data, diseases other than HOA, or an age of over 60 years. Thus, 73 patients were included in the analysis. Fifty-four patients received MHT, and 19 received CAM. The MHT group consisted of 53 patients treated with EPT and one with ET. The CAM group included 14 patients receiving the Kampo formula, two receiving S-equol supplementation, and three receiving a combination of the Kampo formula and S-equol supplementation (Figure 1). The following Kampo formulas were used: Tokishakuyakusan, Keishikajutsubuto, Kamishoyosan, Sokeikakketsuto, Eppikajutsuto, Nyoshinsan, Maobuyokanto, Kakkonto, Hachimijiogan, Unkeito, and Hangekobokuto.

Flow diagram of patient selection and treatment allocation. A total of 169 women with menopausal disorders, including hand pain, were screened. Ninety-six patients were excluded due to conditions other than HOA (e.g. trigger finger, de Quervain disease, carpal tunnel syndrome, or rheumatoid arthritis), missing data, or an age over 60 years. The final cohort included 73 patients, of whom 54 received MHT and 19 received CAM. The MHT group comprised 53 patients treated with EPT and one treated with ET. The CAM group included 14 patients treated with the Kampo formula alone, two with S-equol supplementation alone, and three with a combination of the Kampo formula and S-equol.
Baseline clinical and demographic characteristics, including age, disease duration, BMI, SMI score, SRQ-D score, menopausal status, DASH score, VAS score, and HG strength, did not differ significantly between the MHT and CAM groups (all p > 0.13; Table 1).
Clinical and demographic characteristics of 73 patients.
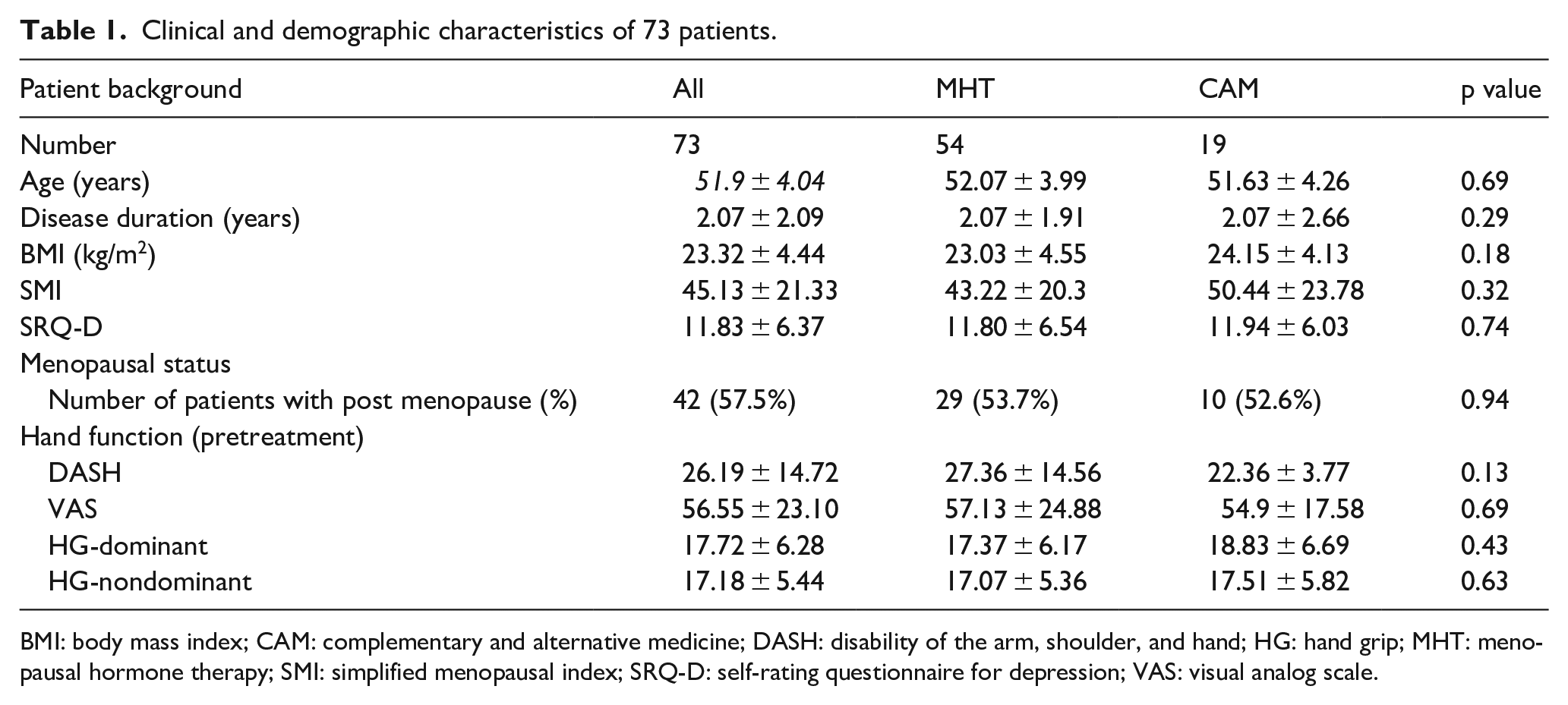
BMI: body mass index; CAM: complementary and alternative medicine; DASH: disability of the arm, shoulder, and hand; HG: hand grip; MHT: menopausal hormone therapy; SMI: simplified menopausal index; SRQ-D: self-rating questionnaire for depression; VAS: visual analog scale.
After 3 months of treatment, the MHT group showed substantial improvements in both the DASH and VAS scores. The mean DASH score improved from 27.36 ± 14.56 to 15.84 ± 10.38, and the VAS score decreased from 57.13 ± 24.88 to 29.20 ± 24.63 mm. In contrast, the CAM group showed minimal or no improvement, with the DASH score slightly worsening from 22.36 ± 3.77 to 24.93 ± 17.84 mm and the VAS score decreasing only modestly from 54.90 ± 17.58 to 47.22 ± 21.93 mm. Two-factor repeated-measures ANOVA revealed significant condition-by-time interactions for both the DASH (F = 12.85, p = 0.007, partial η2 = 0.17) and VAS scores (F = 7.39, p = 0.008, partial η2 = 0.02), indicating that changes over time differed between the treatment groups (Table 2).
Effects of medications (MHT and CAM) and time (pre- and posttreatment) on hand functions, such as DASH and VAS scores.

CAM: complementary and alternative medicine; DASH: disability of the arm, shoulder, and hand; MHT: menopausal hormone therapy; VAS: visual analog scale.
Statistically significant difference between groups (p < 0.05).
Post hoc comparisons confirmed that the improvements in the MHT group were significantly greater than those in the CAM group. The mean difference in the DASH score was 10.85 ± 11.5 points in the MHT group compared with −1.75 ± 13.01 points in the CAM group (p = 0.003), and the mean difference in the VAS score was 27.93 ± 25.6 mm in the MHT group compared with 9.17 ± 23.36 in the CAM group (p = 0.02; Table 2 and Figure 2).

Differences in the DASH and VAS scores before and after treatment in the MHT and CAM groups. Mean changes in the DASH and VAS scores after treatment in the MHT and CAM groups. Bar graphs show the mean differences (posttreatment minus pretreatment) in the DASH score (left panel) and VAS score (right panel) over the 12-week period. The MHT group presented significantly greater improvements in both the DASH and VAS scores compared to the CAM group (*p = 0.003 and *p = 0.02, respectively). Error bars represent the standard deviations.
Notably, the mean improvements in both the DASH and VAS scores in the MHT group exceeded the established minimal clinically important differences (MCIDs), which are generally considered 10–15 points for the DASH score and 15–20 mm for the VAS score. These findings suggest that the treatment effects observed in the MHT group were not only statistically significant but also clinically meaningful.
Discussion
This study examined the effects of MHT and CAM—including the Kampo formula and S-equol supplementation—on symptomatic HOA in Japanese perimenopausal women. Our findings indicated that compared with CAM, MHT led to significantly greater improvements in pain and upper limb function. Notably, the mean changes in DASH and VAS scores in the MHT group exceeded the established MCIDs, suggesting that the observed improvements were not only statistically significant but also clinically meaningful.24,25
Estrogen has been shown to exert multifaceted protective effects on joint tissues through ERs (ERα and ERβ), which are expressed in articular cartilage, subchondral bone, and the synovium. 26 Experimental studies have demonstrated that estrogen promotes matrix synthesis (e.g. proteoglycans and type II collagen) and suppresses cartilage-degrading enzymes such as matrix metalloproteinase-3 (MMP-3) and MMP-13.27,28 Estrogen also inhibits inflammatory cytokine production via the nuclear factor-kappa B pathway and reduces oxidative stress within synovial tissue, thereby maintaining intra-articular homeostasis. 29 Furthermore, estrogen modulates central pain processing by enhancing the endogenous opioid and monoaminergic systems, potentially attenuating central sensitization. 30 Consequently, the decrease in estrogen levels during menopause may not only promote the structural progression of OA but also exacerbate pain sensitivity, resulting in symptomatic HOA even in the absence of advanced radiographic changes.
MHT directly targets this hormonal mechanism. Prior studies investigating the preventive role of MHT in HOA have yielded mixed results, likely due to differences in study populations, the timing of hormone initiation, and the definitions of HOA (radiographic versus symptomatic).3,31–35 While randomized controlled trials (RCTs) remain limited, the present study adds novel clinical data suggesting that the timely administration of MHT may alleviate the symptoms of symptomatic HOA in perimenopausal women. Our findings align with previous evidence indicating that MHT can alleviate musculoskeletal symptoms in menopausal women. Previous studies have shown that ET may improve muscle mass and strength, reduce joint pain and stiffness, and enhance overall physical function in this population.2,17,36
In contrast, evidence for CAM—including the Kampo formula and S-equol supplementation—remains limited, particularly in relation to HOA. Kampo formulas are empirically prescribed for menopausal symptoms in Japan and are believed to modulate the autonomic imbalance and inflammation; however, their efficacy for treating joint-specific symptoms such as symptomatic HOA has not been established in clinical trials. Similarly, while no studies have directly investigated the efficacy of S-equol supplementation for symptomatic HOA, its beneficial effects on joint and muscle pain in postmenopausal women have been reported. An RCT demonstrated that 10 or 20 mg/day S-equol significantly improved self-reported muscle and joint pain in postmenopausal women. 12 Furthermore, an animal study in ovariectomized rats indicated that S-equol ameliorated osteoarthritic changes by suppressing oxidative stress and cartilage degradation. 37
In the present study, CAM was associated with only modest improvements in VAS scores and no measurable improvement in upper limb function, as assessed by the DASH score. However, some components of CAM, such as S-equol supplementation, have shown beneficial effects on musculoskeletal discomfort in postmenopausal women in previous studies. Given the favorable safety profiles and accessibility of these interventions, CAM may still be a viable treatment option for patients who are ineligible for or unwilling to undergo MHT.
These findings support the growing perspective that symptomatic HOA in perimenopausal women should be recognized not only as an age-related degenerative condition but also as a sex-specific disorder triggered by hormonal changes. Conventional orthopedic approaches alone may be insufficient to manage joint pain in this population. Introducing gynecologic interventions, such as MHT, could provide significant therapeutic benefits. Future treatment strategies should aim to integrate gynecologic and orthopedic insights, advancing toward more individualized, life stage- and sex-specific care in musculoskeletal medicine.
Limitations
This study has several limitations. First, it was a retrospective observational study with a convenience sample, which limits causal inference and introduces the potential for selection bias. Treatment allocation was not randomized but was based on patient preference, and although no significant baseline differences were observed, unmeasured confounders—such as symptom severity, health beliefs, or treatment expectations—may have influenced the results.
Second, the sample size was relatively small and imbalanced (MHT n = 54 versus CAM n = 19), particularly limiting the power to detect treatment effects in the CAM group. Third, although Mann–Whitney U tests were used only post hoc after significant interactions in ANOVA and one comparison per outcome was conducted, the risk of type I error should still be acknowledged.
Finally, all participants were ethnically Japanese women. The generalizability of these findings to other populations may be limited due to potential differences in menopausal symptom expression, the equol-producing capacity, and the treatment response.
Conclusions
MHT resulted in clinically meaningful improvements in pain and hand function in perimenopausal Japanese women with symptomatic HOA, whereas CAM showed a limited benefit. These findings suggest that MHT, when appropriately indicated and initiated in a timely manner, may represent a viable treatment strategy for menopausal joint symptoms. A multidisciplinary approach involving both gynecology and orthopedics may be valuable. Future prospective randomized studies are warranted to confirm the effectiveness of both hormonal and nonhormonal therapies and to establish evidence-based treatment pathways for symptomatic HOA in middle-aged women.
Supplemental Material
sj-docx-1-whe-10.1177_17455057251359384 – Supplemental material for Menopausal hormone therapy shows superior efficacy to complementary and alternative medicine in treating symptomatic hand osteoarthritis in Japanese women during perimenopause
Supplemental material, sj-docx-1-whe-10.1177_17455057251359384 for Menopausal hormone therapy shows superior efficacy to complementary and alternative medicine in treating symptomatic hand osteoarthritis in Japanese women during perimenopause by Hiromi Sasaki, Mika Sakihama, Noriko Karakida, Takasuke Miyazaki, Hiroaki Kobayashi and Noboru Taniguchi in Women’s Health
Footnotes
Acknowledgements
Ethical considerations
This study was reviewed and approved by the Ethics Committee of Kagoshima University (reference number 190018). All procedures complied with the Ethical Guidelines for Medical Research Involving Human Subjects and the Declaration of Helsinki.
Consent to participate
Written informed consent was obtained from all participants.
Author contributions
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
