Abstract
Respiratory illnesses due to respiratory virus infections disproportionately impact pregnant individuals and their infants, leading to significant morbidity and mortality globally. Data describing the incidence and impact of these infections in pregnancy is sparse and more common for influenza and now severe acute respiratory syndrome coronavirus 2 with less data available on other respiratory virus infections in pregnancy. This lack of data is a result of limited prospective surveillance and issues surrounding the calculations of seroprevalence, as well as disproportionately low funding for reproductive health research. In this review article, we aimed to summarize available data on respiratory virus infections in pregnancy and identify gaps in the published literature.
Plain language summary
Respiratory virus infections, like flu and COVID-19, cause serious illness and death in pregnant people and their babies, worldwide. However, there’s not much data on how common these infections are or their full impact during pregnancy, especially for viruses other than flu and COVID-19. This is due to limited ongoing studies, challenges in measuring past infections, and underfunding of reproductive health research. This article reviews what we know about these infections in pregnancy and points out where more research is needed.
Introduction
Respiratory illnesses disproportionately impact pregnant individuals and their infants, which can lead to adverse health events throughout the mother’s and child’s life.1,2 Pregnant people undergo substantial physiologic and immunologic changes over the course of their pregnancy increasing their risk for respiratory virus infections.1,3 More specifically, when individuals become pregnant their immune system adapts to provide a tolerogenic environment for non-host fetal antigens.4,5 Although respiratory virus infections commonly occur in pregnant persons, data describing the incidence and impact of these infections is sparse and more common for influenza and now severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) with less data available on other respiratory virus infections in pregnancy.6 –8 This lack of data is a result of limited prospective surveillance and issues surrounding the calculations of seroprevalence, as well as disproportionately low funding for reproductive health research. 2 One cross-sectional study performed in 2018, consisting of 155 pregnant women, found that the most common respiratory infections detected during pregnancy were rhinovirus (27%), seasonal coronaviruses (17%), and respiratory syncytial virus (RSV) (10%). 9 More recently SARS-CoV-2, the pathogen which causes COVID-19, has been shown to be a common cause of respiratory tract infections during pregnancy. A study conducted in 2021, consisting of 138 pregnant individuals, estimated the cumulative incidence of SARS-CoV-2 infection to be 10.1% in the first trimester alone. 10 The risk of severe symptoms and complications associated with respiratory virus infection during pregnancy drastically increases throughout pregnancy, with the greatest risk presenting in the third trimester.1,2 It has been estimated that pneumonia resulting from viral infection is diagnosed in 1.5 of every 1000 pregnancies and is associated with increased maternal morbidity and mortality.11,12 We aimed to summarize available data on common respiratory virus infections in pregnancy, including influenza, RSV, SARS-CoV-2, parainfluenza, human metapneumovirus, parvovirus B19 (fifth disease), rhinovirus, and bocavirus to identify and address gaps in the published literature (Table 1).
Overview of three principal viruses that commonly infect pregnant individuals and newborns.

RSV: respiratory syncytial virus; SARS-CoV-2: severe acute respiratory syndrome coronavirus 2.
During pregnancy, the immune system plays two major roles: to adapt to multiple pregnancy stages to ensure a successful pregnancy and to protect the pregnant individual from pathogens. 5 Upon exposure to a respiratory virus, the immune system induces humoral and cellular responses to prevent or clear the infection. Importantly, the production of protective antibodies occurs. These antibodies work by preventing future virions from entering cells and replicating to establish an infection. 5 In addition to responding to pathogens, the maternal immune system must dynamically and robustly respond to the four stages of pregnancy to have a successful delivery of a viable fetus. 5 First, the immune system provides a pro-inflammatory response during the implantation and placentation stages. 13 To establish a viable placenta for a successful pregnancy, trophoblast cells invade and attach to the surface epithelium of the receptive maternal decidua, which involves the active breakdown and reconstruction of the decidua. Thus, during implantation and early placentation, the immune system responds by releasing pro-inflammatory cytokines, growth factors, chemokines, proteins, and tumor necrosis factors, which also occur when the immune system responds to tissue injury and repair. 5 While the development of the placenta modulates much of this response, it is also a target for viral infections. 14 It is still unclear how the placenta is directly impacted by infection, but we do know that it has a unique capacity to prevent further replication of viruses and subsequent transmission of infections to the fetus. 14 Next, the immune system provides an anti-inflammatory response utilizing macrophages, decidual natural killer cells, and regulatory T cells during the fetal growth stage. 5 Pro-inflammatory responses during this stage of the pregnancy, such as those induced by infection, have been shown to result in preterm birth.15,16 Lastly, the immune system provides a second pro-inflammatory response, which is necessary for the initiation of parturition. The switch to this pro-inflammatory response involves a nuclear factor signaling pathway, which is responsible for initiating and continuing the progress of labor and delivery. Contraction of the uterus, delivery of the infant, and detachment of the placenta are initiated when the myometrium is saturated with immune cells. 5 Therefore, the immune system not only protects pregnant persons from pathogens but also plays a role in establishing the pregnancy, growing the fetus, and delivery of the fetus.
In addition, in pregnant individuals, a mechanism known as transplacental antibody transfer occurs where antibodies, specifically immunoglobulin G (IgG), from previous exposures and infections, are actively transferred from the mother to fetus across syncytiotrophoblast cells of chorionic villi in the placenta. 17 Other Igs including IgM and IgA are not transferred. The transfer of maternal IgG across the syncytiotrophoblast involves binding to neonatal Fc receptors (FcRn), which is highly dependent on pH. IgG is first endocytosed from the maternal side into the syncytiotrophoblast. The endosomes provide an acidic environment that facilitates the binding of IgG to FcRn. The FcRn–IgG complex is then transcytosed to the fetal side of the syncytiotrophoblast, where IgG dissociates from FcRn due to the return to physiologic pH. 17 The maternally derived IgG obtained via transplacental transfer and present in infants at birth have been shown to be protective against respiratory virus infections and is a primary source of passive immunity in the neonate.18 –20 The range of time of protection is dependent on the quantity of IgG transferred to the infant and the rate at which the antibodies wane. 21 For example, one study investigating the transplacental transfer of Influenza (H1N1) binding antibodies found that within 2 months of birth, antibodies waned to below the accepted protective threshold. 22 In contrast, studies used to determine the optimal timing of RSV maternal immunization found that maternally derived RSV antibodies waned to a non-protective level in infants between 6 and 24 months after birth. 23 Factors that have been shown to decrease the amount of maternally derived IgG include birth gestation, birth weight, maternal vaccination timing, hypergammaglobulinemia, antibody glycosylation state, IgG subclass, neonatal FcRn efficiency, and antigenic characteristics.17,24 –28 Protective thresholds are highly variable across different viruses due to the use of different laboratory assays and individual-level exposure patterns. Therefore, more research must be done to get a better understanding of how protective thresholds compare across viruses.
Pregnant persons are at a higher risk for severe signs, symptoms, and complications from respiratory virus infections, with the leading cause of complications due to subsequent pneumonia-inducing respiratory failure. 29 These viruses increase the risk of bacterial pneumonias from damage to the respiratory epithelium. 30 In addition, physiologic changes during pregnancy, such as decreased total lung volume due to the growing fundus, increase risks associated with respiratory virus infections. 31 Respiratory virus infection during pregnancy also increases the risk of adverse pregnancy and neonatal outcomes. These include maternal death, pregnancy loss, preterm delivery, and infants born with low birthweight, in addition to other adverse events such as fetal demise.1,32 –34 Preterm and low birthweight can have lasting impacts on infants throughout their lifetime and potentially increase the risk of comorbidities in adulthood.1,35 In addition, maternal fever can have a detrimental impact on the growing fetus, and sustained high maternal core temperatures have been associated with an increased risk of developing neural tube defects, congenital heart defects, and oral clefts. 36 RSV and SARS-CoV-2 infections during pregnancy can potentially cause vertical transmission, although this is a rare occurrence, and many have speculated on the nature in which the observed vertical transmission cases have been investigated and documented.37 –39 Therefore, there is an increased focus on preventing respiratory virus infections during pregnancy to mitigate morbidity and mortality of the pregnant person as well as stillbirth and other adverse pregnancy and neonatal outcomes that could potentially have long-lasting effects in infants.
Estimates of influenza virus infection rates in pregnant individuals vary due to differences in antigenic properties and influenza vaccine mismatch across seasons (Figure 1). A systematic review estimated the incidence of influenza infections in pregnant persons to be within the range of 483 and 1097/10,000 pregnancies. 40 The risk of influenza infection and associated morbidity can be exacerbated if an individual is pregnant during the peak of influenza virus circulation or has other comorbidities.1,41 One study estimated that over nine influenza seasons, 33% of reproductive-aged women hospitalized for complications due to influenza infection were pregnant. 42

Respiratory viruses detected at Seattle Children’s Hospital, 2020–2024.
Influenza virus is commonly transmitted through exposure to respiratory droplets carrying influenza virions. The immune system responds in multiple ways to neutralize the infection which commonly causes fever, chills, fatigue, malaise, runny nose, and cough.1,43 This clinical presentation is similar for both influenza A and B, with the most common seasonal infections occurring from Influenza A H3N2 and H1N1. 43 Antibodies for influenza have shown to be protective for multiple subtypes and strains; however, the level of cross-protection varies by how genetically similar previous and subsequent exposure viruses are. 43 Influenza infection during pregnancy generally requires diagnostic testing to verify infection, and treatment is limited by the timing of clinical presentation. Common recommendations include home and over-the-counter interventions such as rest, increased fluid intake, and fever-reducing medication. In addition, the American College of Obstetricians and Gynecologists (ACOG) recommends starting empiric antiviral treatment within 48 h of symptom onset without waiting for confirmation from respiratory infection test results. 44 Oseltamivir is the preferred antiviral medication in pregnancy, with a treatment regimen of 75 mg orally twice daily for 5 days. In addition, Individuals who are pregnant with a moderate risk of severe illness and those with a high risk of severe illness should be seen in the ambulatory clinic and emergency department, respectively, for evaluation.44,45
The most effective method used to prevent influenza infection during pregnancy is maternal immunization. The seasonal influenza vaccine is recommended for all pregnant individuals as soon as it is available during flu season. 46 A multiyear meta-analysis estimated that the maternal influenza vaccine was associated with a 64% decrease in risk of laboratory-confirmed infection, with variability by year due to differences in antigenic properties and vaccine mismatch by season. 47 These vaccines protect the pregnant individual by inducing immune responses. The humoral immune response produces antibodies specific to the vaccine antigen, which prevent the binding of the influenza surface proteins and host-cell receptors, ultimately inhibiting infection.43,48 Not only do these vaccines protect individuals during pregnancy, but they also result in protection in infants after birth. During pregnancy, transplacental transfer of influenza antibodies from mother to fetus takes place.22,49,50 These antibodies work similarly immunogenically in infants as they do in their mothers, by blocking the binding of the virion and host-cell and inhibiting infection.43,50 Meta-analyses have estimated a 72% decrease in the risk of influenza infection during the first 6 months of life in infants whose mothers received the seasonal influenza vaccine. 47
Individuals are repeatedly exposed to and infected with RSV throughout their lifetimes, with most people experiencing their first infection before the age of 3. 51 Transmission of RSV is typically seasonal with the most circulation occurring between January and April in the USA with the highest burden of disease in children <5 years of age (Figure 2). 52 A meta-analysis including 11 studies between 2010 and 2022 estimated the incidence rate of RSV infections in pregnant individuals was 26 episodes/1000 person-years. 53 However, the exact number of infections in pregnant persons is likely underestimated due to a lack of prospective surveillance and asymptomatic case presentation, which is common in reproductive-age individuals. 51 One study tested asymptomatic and symptomatic pregnant individuals in their second and third trimesters and found 10% were infected with RSV. 9

Country-specific RSV epidemiology.
Similar to influenza, RSV is most commonly transmitted through exposure to respiratory droplets carrying RSV virions. The immune system responds by inducing humoral and cellular responses, which can present as upper respiratory tract cold-like symptoms with persistent cough and, less commonly, as respiratory distress in adults. 30 Individuals can be infected and present without symptoms, increasing the risk of transmission due to normal day-to-day activities, which increases interactions with others. 51 There are two types of RSV (A and B); studies have shown that infection from one strain usually results in the production of cross-reactive antibodies, which provide protection to both strains upon future exposure. 54 Antibodies are developed against RSV antigens, including surface glycoproteins F and G. 55 There is a fourfold rise in antibodies after natural infection in adults, which return to baseline concentrations within 2 years. 56 Higher levels of antibodies are generally thought to correlate with higher resistance to infection, however, there is no defined serologic threshold for protection in children or adults.57,58
Treatment for RSV in pregnant individuals combines multiple supportive care interventions such as the use of bronchodilators, supplemental oxygen, intravenous fluid, and antipyretics. 59 Ribavirin, a nucleoside analog, is currently the only Food and Drug Administration (FDA)-approved antiviral therapy for use against RSV for infants and young children, but is not recommended for pregnant persons or their male partners, given teratogenicity seen in animal studies.60,61 Results of a Ribavirin Pregnancy Registry (NCT00114712) that was established in 2003 to evaluate pregnancy outcomes after Ribavirin exposure showed congenital anomaly rates that exceeded the Metropolitan Atlanta Congenital Defects Program, however, no clear pattern of pathophysiology for teratogenicity was found. 61
In the past, mitigation methods against RSV typically included normal measures against respiratory infections such as hand hygiene and masking. Recently in 2023, the FDA approved a bivalent pre-fusion F protein RSV vaccine, Abrysvo™ (Pfizer Inc., New York, NY, USA), for pregnant persons. This vaccine provides some maternal protection in the mid to late third trimester of pregnancy but is mainly used for infant protection from RSV via transplacental antibody transfer to the fetus. Phase 3 clinical trial results showed that in infants, the vaccine reduced the risk of severe lower respiratory tract disease from RSV infection by 82% within 90 days of birth and 69% within 180 days of birth. 62 The FDA approved this vaccine to be administered between 32 and 36 weeks gestation, given a numerical imbalance in preterm births in Abrysvo recipients (5.7%) compared to placebo recipients (4.7%), which was only seen in one participating middle-income country. 63 In addition, a new RSV monoclonal antibody directed against the same F protein used in the RSV vaccine has been licensed and recommended by the Centers for Disease Control for administration to infants for the prevention of RSV in the first year of life for both healthy and at-risk children, with good effectiveness already demonstrated in the United States.64,65
SARS-CoV-2 is a pathogen that emerged in the last 5 years, sparking a global COVID-19 pandemic. Since the pandemic, the SARS-CoV-2 virus has become endemic, and in the past 2 years, patterns show that it has seasonally circulated with other respiratory viruses in the USA (Figure 3). 66 Although there has been an influx of research surrounding this pathogen, we still know very little about the burden this virus has on pregnant individuals and their infants, in part because many infections may be asymptomatic and partially due to the complex nature of the immune response to SARS-CoV-2 infection. One study found that ~10% of individuals who became pregnant just before or during the first wave of the COVID-19 pandemic tested positive for SARS-CoV-2 within their first trimester of pregnancy. A moderate proportion (42%) of these individuals were asymptomatic. 10 One large meta-analysis and another cohort study found that individuals who were diagnosed with SARS-CoV-2 during pregnancy were more likely to be hospitalized, admitted to the intensive care unit, delivered preterm, and required cesarean sections.67,68 Although infected pregnant persons were more likely to experience severe illness and adverse events, there was a low incidence (1.4%) of SARS-CoV-2 documented in their newborns. 67
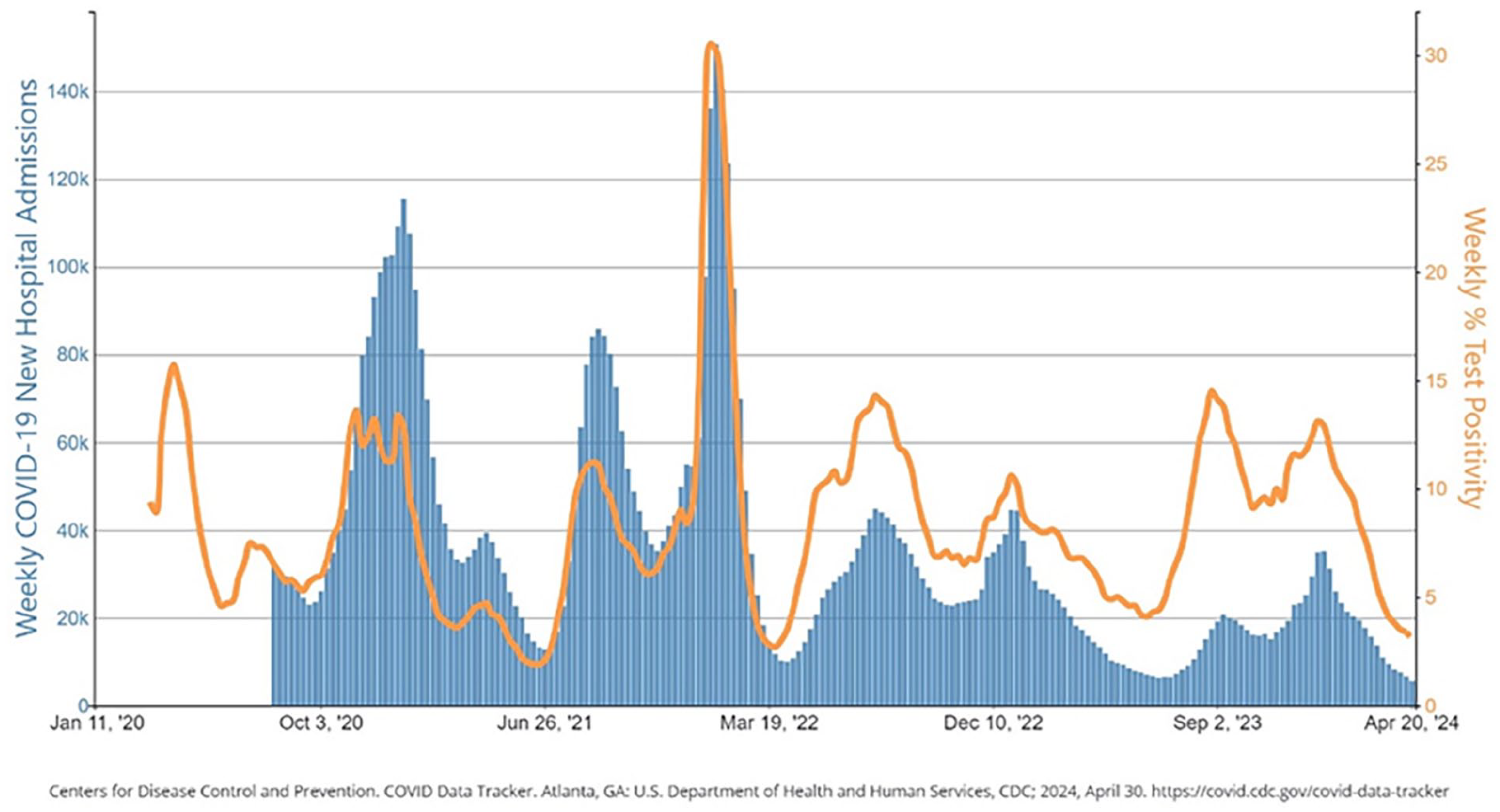
COVID-19 new hospital admissions and NAAT percent positivity, by week, in the USA, reported to CDC.
Like other respiratory viruses, SARS-CoV-2 is also commonly transmitted via contact with respiratory droplets expelled by an infected individual. Compared to nonpregnant reproductive-aged individuals, pregnant people are at a significantly higher risk for severe adverse pregnancy and fetal outcomes. 69 The pathophysiology for more severe outcomes in the pregnant population may be due to physiologic changes in pregnancy, including decreased lung capacity due to the growing fundus, increased heart rate and oxygen consumption, alterations in the immune system, and higher risk for a thromboembolic event.69 –71 In general, pregnant individuals with SARS-CoV-2 infection are at higher risk for ICU admission, need for invasive ventilation, need for ECMO, or death compared to non-pregnant individuals. 69 In addition, there is also a higher risk for preeclampsia, need for preterm delivery, and stillbirth.72,73 SARS-CoV-2 infections can significantly affect the placenta and result in fibrin deposition, and chronic placental insufficiency with outcomes as described above. 74 Vertical transmission of the SARS-CoV-2 virus resulting in congenital SARS-CoV-2 infection has been demonstrated but is thought to be rare. 75
Treatment for symptomatic COVID-19 illness in pregnant persons is similar to that of non-pregnant adults.76,77 Antiviral medications including ritonavir-boosted nirmatrelvir (Paxlovid; Pfizer Inc., New York, NY, USA) and remdesivir have been studied in pregnancy and are considered safe. 77 Other supportive measures, as well as monoclonal antibodies, if indicated, are also acceptable in pregnancy. 77
SARS-CoV-2 vaccination is the most efficient way to reduce the risk of excess negative maternal and fetal outcomes associated with infection during pregnancy. Although pregnancy was an excluding factor in the initial COVID-19 vaccine clinical trials, observational studies have provided reassuring data regarding the safety and efficacy of COVID-19 vaccination in pregnancy.78,79 In fact, multiple studies have shown that the overall rates of adverse perinatal outcomes, such as preterm birth and fetal growth restriction, did not increase after maternal COVID-19 vaccination.80 –82 Instead, COVID-19 vaccines decrease the risk of adverse maternal and neonatal outcomes seen with SARS-CoV-2 infections in pregnancy.83 –85 The current recommendations from the World Health Organization, Centers for Disease Control and Prevention, the Society for Maternal-Fetal Medicine, and the ACOG recommend the COVID-19 vaccine for pregnant persons.86 –88 Four COVID-19 vaccines (1. Pfizer Inc., New York, NY, USA & BioNTech SE, Mainz, Germany; 2. Moderna, Inc., Cambridge, MA, USA; 3. Johnsen and Johnsen Services, Inc., New Brunswick, NJ, USA; 4. University of Oxford, Oxford, UK & AstraZeneca, Cambridge, UK) have published data regarding their use in pregnant persons. This research has shown that pregnant individuals mount a robust maternal antibody response post-vaccination.28,89 However, studies have found that significant waning in antibodies occurs when vaccinated during the third trimester, but antibodies increase again after a booster dose further into the pregnancy.28,89 In the USA, COVID-19 vaccination is currently recommended at any time during pregnancy.87,88 COVID-19 vaccine has also been shown to be beneficial for newborns with maternal immunization, decreasing the risk of hospitalization due to COVID-19 illness in infants in the first 6 months of life. 90
Pregnant individuals are at increased risk of respiratory virus infection. Although much of the available research has been conducted on Influenza, RSV, and SARS-CoV-2, there is some research present regarding other respiratory viruses during pregnancy, such as parainfluenza, metapneumovirus, parvovirus B19 (Fifth Disease), and bocavirus. A study investigated the prevalence of symptomatic respiratory viruses during pregnancy in 2556 women between 2014 and 2017 and identified 17 cases of parainfluenza and four cases of human metapneumovirus. 91 Although parainfluenza normally produces symptomatic illness in young children, there have been documented cases of adverse fetal outcomes, including fetal ventriculomegaly and hydrocephalus.92,93 Another study was conducted to determine the incidence of human metapneumovirus among pregnant individuals and found that 17.2% tested positive.93,94 In addition, a study performed in Nepal investigating birth outcomes associated with human metapneumovirus infection during pregnancy found that human metapneumovirus infection during pregnancy was associated with an increased risk of having a small for gestational age newborn, but no increased risk of premature birth or significant differences in birth weight.93,95 Parvovirus B19 has been estimated to infect 1%–5% of pregnant women but can infect between 3% and 20% during epidemics.96 –98 In most cases, infection with B19 has no impact on pregnancy outcomes. 96 However, as gestation continues, the likelihood of fetal anemia, non-immune hydrops, and mortality due to maternal infection increases. 96 Rhinovirus is a common pathogen that infects pregnant persons and causes symptomatic respiratory illness, with one study estimating that 27% of their cohort was infected at some point during pregnancy. 9 One study conducted in Nepal, consisting of 3693 pregnant individuals, found that pregnant persons infected with rhinovirus during pregnancy were more likely to deliver low birthweight babies. 99 Although rhinovirus seems to pose little risk of severe maternal and fetal outcomes, one study did find that infants born to mothers with documented rhinovirus infections during pregnancy were more likely to experience wheezing at 12 months old. 100 Although active bocavirus infection is rarely detected in healthy adults due to prior long-lasting immunity from infections in early life, there have been a small number of bocavirus infections documented in pregnant individuals. 101 Because bocavirus is rarely detected in adults, the clinical implications for pregnant persons and their infants are still unclear. However, there have been case studies in which fetal demise has been documented in pregnant women with bocavirus infections. In addition, bocavirus has been detected in human placental tissues from aborted pregnancies. 102 There is little information available on the pathogenesis of these viruses in pregnant persons, and no vaccines or clinical treatments are available for pregnant individuals.
Conclusion
In this work, we have summarized the burden, pathogenesis, clinical outcomes, and treatment of influenza, RSV, SARS-CoV-2, parainfluenza, human metapneumovirus, parvovirus B19 (fifth disease), rhinovirus, and bocavirus during pregnancy. Our study is not without limits, as it only relies on previously published findings, which can be inconsistent or inaccurate. However, we feel confident in our assessment of the existing literature and feel that we have accurately summarized the available information. As interest in respiratory viruses increases, due to the COVID-19 pandemic and the development of new vaccines, significant knowledge gaps on the impact of respiratory virus infections and vaccines in pregnancy are becoming more apparent. 103 More research is necessary to properly understand the nuances of respiratory virus infections during pregnancy to better protect these individuals from these potentially dangerous pathogens. Unfortunately, research studies that comprise pregnant individuals and their infants are disproportionately underfunded compared to other high-risk groups. However, the studies that have been conducted have allowed researchers to develop safe and effective respiratory virus vaccines, as is the case with the RSV and Influenza vaccines. These promising developments highlight the need for more resources to be devoted to maternal and reproductive health research. Future discoveries can help us better prevent respiratory virus infections and aid in the application of new treatment methods.
Footnotes
Author contributions
Kalee E. Rumfelt: conceptualization; resources; writing-original draft; writing—review and editing; visualization. Janet A. Englund: conceptualization; resources; writing—review and editing. Alisa Kachikis: conceptualization; resources; writing—review and editing; supervision; project administration; funding administration.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by an NIAID K23 AI153390 grant. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors do not have competing interests to disclose related to this publication. Outside of this work, A.K. was an unpaid consultant for Pfizer and GlaxoSmithKline and is a co-investigator on studies funded by Merck and Pfizer. J.A.E. also receives grant support to her institution from Merck, GlaxoSmithKline, AstraZeneca, and Pfizer, and is a consultant for AbbVie, Ark Biopharma, AstraZeneca, GlaxoSmithKline, Moderna, Pfizer, Sanofi Pasteur, and Meissa Vaccines, Inc. outside of the described work.
