Abstract
Background:
Overactive pelvic floor muscles (PFMs) are a source of pain in patients with vulvodynia. Peripheral sensitization may also lead to central sensitization (CS).
Objectives:
This study aimed to validate the Vulva, Anus, Muscle, and Paraurethra (VAMP) protocol as a diagnostic tool for assessing PFM overactivity in vulvodynia in correlation with the Central Sensitization Inventory (CSI).
Design:
This research was the secondary outcome of an ongoing randomized clinical trial reported in accordance with the CONSORT guidelines. Inclusion criteria and research goals were established using the Population Intervention Comparison Outcome model for clinical questions.
Methods:
The VAMP protocol includes the examination of the vulva (V), anus (A) using a cotton swab test and pelvic muscle (M) and paraurethra (P) examinations during the internal exam with pain evaluation. VAMP was validated by comparison with a swab test and correlation with the CSI score.
Results:
Of the 152 premenopausal participants, 91.6% suffered from provoked/mixed vulvodynia versus 8.4% spontaneous, and 61.3% had localized versus 38.7% generalized subtypes. Mean VAMP scores were: V = 6.49; A = 0.2; M = 6.57; P = 3.63. The VAMP protocol showed a high degree of internal consistency and test–retest reliability for the V, M, P domains (Spearman’s correlation coefficient = 0.916–0.646; p < 0.05) and 98.7% of participants fulfilled the VAMP criteria (V, M, P score ⩾ 3) for PFM overactivity. The swab test was negative in 3.9%, a CSI score > 40 was found in 53.7% of participants with a significant correlation for M (Spearman’s correlation coefficient = 0.210; p < 0.05) and P (Spearman’s correlation coefficient = 0.209; p < 0.05) in VAMP.
Conclusions:
The VAMP confirmed the presence of a painful vestibule, PFM, and paraurethra under pressure. The VAMP protocol was successfully cross-validated and effective in discriminating women with PFM overactivity, confirmed in almost all patients with vulvodynia. Half of the participants met the criteria for CS. The correlation between CSI and PFM pain on pressure suggests a central pain mechanism. The VAMP is a simple, rapid quantitative diagnostic tool.
Registration:
ClinicalTrials.gov ID NCT05955313 Registered on June 6, 2023.
Introduction
This study aimed to validate a diagnostic tool for pelvic floor muscle (PFM) overactivity assessment in women with vulvodynia in correlation with central sensitization (CS) features.
Vulvodynia
The consensus terminology and classification of persistent vulvar pain and vulvodynia from 2015, define vulvodynia as vulvar pain:1 –4
Without visible or known, identifiable cause of the pain: there is nothing to hurt but hurts
Is characterized by wide spectrum of vulvar pain, burning sensation, stabbing, pressure, pulling pain and the itching,2,3 dryness or other discomfort, and oversensitivity
Occurs for >3 months, constantly, intermittently or related to provocation
Has potential associated factors: musculoskeletal with PFM overactivity involvement, neurological, psychosocial, and comorbid pain syndromes (e.g. irritable bowel syndrome (IBS), dysuria, or fibromyalgia)
In different vulvar region: generalized (pain extending beyond Hart’s line onto the labia minora or majora, perineum, perianal area, upper legs, or lower abdomen) or localized (vestibulodynia or clitorodynia)
Either spontaneous or upon touch or vaginal penetration (spontaneous or provoked).
Vulvodynia, primarily as vestibulodynia, represents a summation and overlapping of various trigger factors (infections, hormonal disturbances, allergies, genetic aspects, psychological vulnerability, and others) with weight and predominance varying from patient to patient. 5 Its prevalence is approximately 1 in 10 women over their lifetime, and 45% of women with vulvodynia report adverse effects on their sexual life. 6 Provoked vulvodynia is the cause of superficial dyspareunia; in the most severe form, vaginal intercourse is frequently or completely impossible because of vulvar pain (Marinoff dyspareunia scale: 2–3). 7 Vulvodynia is a growing problem, and determining the correct treatment is important. 8
Women with vulvodynia are usually diagnosed late, often after several years of treatment for vulvovaginal infections or psychiatric diseases. A correct diagnosis is essential to guide the treatment of the disease.
PFM overactivity
The etiology of vulvodynia remains unclear, with multiple theories regarding its possible causes. 8 Dysfunctional, overactive PFMs are suspected to be a source of chronic pain in almost all women with vulvodynia.9,10 Findings suggesting hypertonic (overactive) PFMs in women with vestibulodynia include elevated overall PFM tension. 11 Other factors of vulvar pain are also emphasized, such as systemic, inflammatory, genetic, psychosocial, neuroproliferative, and central nervous system-related components. 1 Peripheral sensitization and psychological predisposition can lead to CS and chronic pain in some patients. 2
According to the recent guidelines of the International Continence Society report on the terminology for PFM assessment, “pelvic floor muscle dysfunction may appear as hypotonic/decreased tone presenting primarily as an overactive state.” 12 Classification of areas of interest 12 :
“Disorder of increased PFM tone” in the form of either “Pelvic floor tension myalgia” or “Pelvic floor myofascial syndrome.”
“Disorder of PFM pain,” in which case “Pelvic floor myalgia” or “Disorder of decreased PFM tone” should be differentiated as a potential PFM-related source of pain.
In gynecological practice, this new classification is difficult and not sufficiently clear to apply. From a practical point of view, the classification of PFM increased tone is described as PFM overactivity, commonly referred to in the literature13,14 where “Pelvic floor overactivity” occurs when PFMs are not relaxing or even contracting, when relaxation is necessary, for example, during defecation or micturition. Women with PFM overactivity usually present with gynecological, gastrointestinal, urological, sexual, and musculoskeletal symptoms caused by either peripheral or CS mechanisms.13,15,16 A wide spectrum of chronic pelvic pain (CPP) is associated with increased PFM tone, as assessed by digital palpation. 17 The most important feature associated with PFM overactivity is pressure pain in the vulva, a form of localized pain sometimes referred to as referral pain. Deep muscles also reproduce pain when the examination is performed internally through the vagina or rectum. 18
Several protocols for PFM examination have been proposed.11,18 –20 Harm-Ernandes et al., in their systematic review, failed to prove the superiority of any diagnostic method over a predefined reference test for PFM overactivity, although a theoretical basis was available for healthcare practitioners. 18 There is also a lack of consensus on the definition of pelvic floor myofascial pain and diagnostic criteria. This problem must be resolved before meaningful research in this area can proceed.19,20 Simple and standardized procedures for PFM overactivity examination are not available.21,22 Pelvic floor examination is recommended in gynecological patients with persistent vulvar pain or other discomfort, whether provoked (a leading cause of superficial dyspareunia) or spontaneous (usually perceived as “recurrent intimate infections”), and when vulvodynia is diagnosed, similarly to the cases of CPP.
The diagnostic criteria for vulvodynia and examination recommendations have focused on the vestibular mucosa (as a cotton swab test) since the Friedrich vulvar vestibulitis syndrome (previous description of vulvodynia) diagnostic criteria were established. 23 In a study by Reed et al., 13.8% of patients with vulvodynia showed no increase in sensitivity to cotton swab pressure, suggesting that testing vulvar sensitivity may not be an essential part of the vulvodynia diagnostic algorithm. 24 Pain associated with pressure on the vestibular mucosa is related to the neuroproliferation caused by mast cell activation in the epithelium. The cause, response to hormonal changes, or trigger in the form of a vulvovaginal yeast infection is unclear.2,8,25
The cascade of pathophysiological changes may be caused by tension in the superficial muscles (muscle bulbocavernosus) and may not necessarily involve the deep pelvic muscles. Based on scientific reports, it should be considered that activation of the deep PFM may represent a distinct mechanism of pain in vulvodynia, including a lower pain threshold and lower pain tolerance, increased tone at rest, decreased ability to voluntary contract and abnormal muscle morphology. Studies have not shown whether these muscle abnormalities are a cause or consequence of vestibulodynia or whether they are due to other central factors, such as anxiety and fear-avoidance response, which are psychological features associated with increased muscle tone and pain.8,26
Some authors have proven that the severity of muscular pain (such as PFM pain during pelvic palpation), but not mucosal pain (such as vestibular pain in the cotton swab test), had a detrimental impact on sexual function. PFM overactivity greater than the positive cotton swab test (mucosal pain) is associated with comorbid pain. 27
Two potential mechanisms are suspected to be involved in PFM overactivation or increased tone: ischemia and reduced blood perfusion with hypoxia. 28 Pain arising from muscle overactivation is mediated by myofascial trigger points (TrPs), which can cause myofascial pain syndrome. 29
Based on the literature and our clinical experience, dysfunctional pelvic muscles and fascia act as pain generators. According to Jantos et al.,3,4,15 pain in the “end organs,” such as the vulva or bladder, appears to be “innocent bystanders.” The literature review, based on scientific articles published between 1997 and 2022, emphasized that psychotherapy, physical therapy, and surgery were the most definite treatments when non-pharmacological therapy was considered; however, the etiology of vulvodynia should be taken into account. 8
Vulvodynia as nociplastic pain
The revised definition of pain accepted by the International Association for the Study of Pain (IASP) in 2020 16 describes it as “an unpleasant sensory and emotional experience associated with, or resembling, actual or potential tissue damage, where pain is a personal, subjective experience with influence on biological (tissue or nerve pathology), psychological, and social factors.” 30 Vulvodynia (ICD-11, code GA34.02) meets the IASP clinical criteria for nociplastic pain of the musculoskeletal system. Referred pain, which is the main symptom of TrPs, is a central phenomenon caused by peripheral stimuli. 31 Chronic pain (lasting >3 months) is classified into 32 nociceptive pain, neuropathic pain, and nociplastic pain. Different types of Chronic Primary Pain (ICD-11) are described as vulvodynia, chronic primary visceral pain (CPP), and comorbid conditions such as fibromyalgia, IBS, bladder pain, headaches, and other complex regional pain syndrome type 1.33,34 The IASP has defined nociplastic pain as “pain that arises from altered nociception despite no clear evidence of actual or threatened tissue damage causing the activation of peripheral nociceptors or evidence for disease or lesion of the somatosensory system causing the pain.” 16 For clinical purposes, CS should be considered. CS is defined as an enhancement of neuronal signaling in the central nervous system that causes hypersensitivity to pain.35,36 According to recent literature (IASP 2020), the term CS (from IASP 2014) has been gradually replaced by nociplastic pain, although they are not synonyms.
The Central Sensitization Inventory (CSI) is an established diagnostic tool for CS assessment. 37 In line with the IASP statement, the current literature provides evidence that the diagnosis of nociplastic pain is a highly complex process and that a gold standard has not yet been developed. 38 A literature review suggests that CSI is reliable and provides valid data that quantifies the severity of CS symptoms in patients with non-specific, non-cancer pain. 39
Creating a diagnostic procedure
To make the PFM assessment a diagnostic tool, steps must be taken, as in the case of measuring health-related patient-reported outcomes.40,41
The Quality Assessment of Diagnostic Accuracy Studies (QUADAS) or its revised version (QUADAS-2) tool for systematic reviews of diagnostic accuracy studies was developed. QUADAS has been widely used since its publication in 2003. 42 The QUADAS-2 has been suggested to detect pelvic floor myofascial pain. 19 In an international Delphi study, the COnsensus-based Standards for the selection of health status Measurement INstruments (COSMIN) checklist has been developed for assessment of the methodological quality of the measurement properties. Regarding the Vulva, Anus, Muscle, and Paraurethra (VAMP) protocol, four principles were followed alongside the COSMIN checklist. 43
Proper definition of the problem under test (diagnostic procedure)
Careful test construction, determination of the format of responses
Development of the test (standardization)
Carrying out the validation and standardization process.
The proper definition of the research problem based on theoretical foundations is a key issue in constructing a new tool. The reason for the execution of the VAMP protocol by the investigator (physician, mainly gynecologist, urologist, and physiotherapist) in the diagnosis of vulvodynia or other comorbidities is that PFM overactivity is seen as the cause and not simply comorbid with vulvar pain. Confirmation (or exclusion) of PFM involvement guides further management, as pelvic muscle therapy is indicated in patients with overactive, non-relaxing pelvic floor dysfunction. 44
A careful test construction was built on a two-center study of the integrated mapping and assessment protocol (IMAP) developed by Jantos and Johns, aimed at systematically locating the sources of pain in chronic urogenital pain (CUP) syndromes. This mapping has enabled the development of evidence-based diagnostic interventions. The three pain maps (54 examined points) were based on clinical work, research studies, and cadaver dissection.3,4,15 This IMAP study provides a scientific basis for the VAMP protocol, which is a practical and simple test for clinical applications.3,4
Determination of the format of responses is based on the Numerical Pain Rating Scale (NRS), which has been the preferred instrument for the assessment of pain, used in 68% of studies where PFMs were examined in women presenting with pain.45,46
For a test protocol to be a high-quality diagnostic tool, the relevance and reliability of the test method must be confirmed. 47 According to pioneering research toward developing a method to test the PFM status and vestibular mucosal pressure pain detection threshold, validation of the test should include the reliability (reproducibility) and validity (accuracy) of the outcome measures. The reliability score is the sum of four domains: intra- and inter-examiner reliability, stability of the phenomenon being measured over time (test–retest), and reliability of the participant’s pain report. 48
Methods
This research is the secondary outcome of a quadruple-blind, parallel-group, single-center, ongoing randomized clinical trial (RCT) conducted in the TERPA outpatient clinic in Lublin, Poland, in partnership with the Medical University of Lublin, Lublin, Poland. The TERPA is a clinical center that focuses primarily on vulvar diseases. Approval for this study was obtained from the appropriate IRB at the Medical University of Lublin, Poland (Ethics Committee approval KE-0254/210/2021). The RCT was registered at ClinicalTrials.gov ID NCT05955313 on June 6, 2023. The secondary outcome was the validation of the diagnostic tool (VAMP protocol) for PFM assessment in women with vulvodynia. All participants signed an informed consent form before participation. The RCT was reported in accordance with the CONSORT guidelines. 49 Inclusion criteria and research goals were established using a Population Intervention Comparison Outcome (PICO) model for clinical questions.50,51
Population
According to the latest version (version 4.0) of the study protocol, the target population was non-pregnant, premenopausal women between 18 and 50 years of age with vulvodynia diagnosed according to the 2015 Consensus Terminology and Classification of Persistent Vulvar Pain and Vulvodynia. 1
Patients with vulvodynia were classified into subtypes:
(a) Localized as vestibulodynia (pain in the vaginal vestibule), clitorodynia (pain in the clitoris), or other locations (generalized).
(b) Provoked with insertional (pain during penetration alone or with a spontaneous appearance, also referred to as mixed subtype) or spontaneous (without provocation) according to recent attitudes. 52
(c) Onset according to the provoked subtype: primary (present from first sexual contact or tampon insertion) or secondary (present after a period of asymptomatic sexual contact).
The detailed inclusion and exclusion criteria are provided in the SPIRIT checklist. The participants were assessed for age, educational level, residency, parity, contraception, period and type of vulvar pain, weight, height, and blood pressure at visit 1.
Recruitment began on May 29, 2023, and ended on November 18, 2023, when 155 participants were assessed for eligibility, and 152 women were included in the study. The study is ongoing when the article was submitted to the publisher. The overall design is illustrated in Figure 1.

CONSORT 2010 study diagram.
Interventions
According to the RCT protocol (as in the CONSORT and SPIRIT checklists, in the supplemental material), as the informed consent form was signed, the inclusion and exclusion criteria were confirmed in women with vulvodynia in the intention-to-treat population. The intention-to-treat principle involves analyzing the data of all study participants according to their original group assignment, regardless of whether they completed the intervention according to the protocol. 53 The study design, according to the VAMP examination, was as follows.
At Visit 1, a bimanual examination (BME) was performed according to the VAMP protocol (VAMP I by gynecologist first). Any visible or detectable vulvovaginal pathology was excluded from the study. At an interval of no less than 2 h, the VAMP protocol was performed a second time (VAMP II by a gynecologist second).
At Visit 2, the VAMP protocol was conducted for the third time (VAMP III by the first gynecologist), and all participants completed the CSI.
The same examination was conducted twice by the same gynecologist, 4 weeks apart, to determine the intra-observer reliability. To assess inter-observer reliability, all women were examined by a second gynecologist. Each examiner was blinded to the first examiner’s results.
PFM examination: VAMP protocol
In the lithotomy position, PFM assessment was performed according to the VAMP protocol developed by the authors as described in their previous publications.21,22
During BME, “four anatomical regions were assessed: the vulva (V) and anus (A) with a cotton swab test, the internal pelvic muscles (M) with digital examination of the levator ani muscle, and the paraurethral (P) area with digital pressure” (Figure 2). The pain experienced by the participant was graded using an 11-point numeric scale from 0 to 10, with 0 representing “no pain at all” and 10 “the worst possible pain,” according to the NRS.

VAMP examination scheme.
The VAMP protocol, as described by the study authors in previous publications,21,22 consists of the following steps:
Verbal consent was obtained after an explanation of the examination scheme.
After explaining the NRS method, patients were asked to rate the pain experienced at each examined point.
Gentle pressure was applied by the dry cotton swab to 5 points at the vestibular base of the hymen remnant (from position 2 to 10 according to the “vestibular clock”). The applied pressure was adjusted to the level tolerated by the patient (PPTol). 12 Only the maximum NRS rating was noted (the V module in the VAMP acronym).
A cotton swab test, as described above, was then repeated at two points around the anus using pressure similar to that of the vulva, and the maximum NRS score was recorded (module A).
Bimanual vaginal examination was performed using a single lubricated, gloved index finger.
To palpate the PFM, the palmar side of the index finger was rotated backwards, and a sliding motion was performed by bypassing the rectum with pronounced pressure on the muscles along the iliococcygeus muscle to the anterior part of the puborectalis muscle (mid-muscle belly technique). The PPTol technique was performed bilaterally. This technique allows the assessment and differentiation of pain intensity in superficial PFM (bulbospongiosus, ischiocavernosus, and painful pressure most externally) and deep PFM (levator ani). The maximum NRS score (M module) was recorded.
To examine the paraurethral region, the palm of the index finger was rotated upward and lateral to the urethra, and a maneuver similar to detaching the urethra from the pubic bone was performed. This movement was performed from the outside to the inside of the pubic bone on both the right and left sides. The pressure was gradually increased using the PPTol technique, and the maximum NRS score (P module) was recorded.
VAMP scores were evaluated for each patient. Physical examination results were recorded according to the VAMP acronym (e.g. VAMP 6068), including the maximum NRS scores in four areas: the Vulva, Anus, Muscles, and Paraurethra. 22 The time required for the VAMP protocol was monitored for each exam.
Central Sensitization Inventory
The CSI is a self-report screening instrument that identifies patients with central sensitivity syndrome. It consists of two parts, A and B. Part A of the original CSI consists of 25 items. Each item is scored on a scale from 0 to 4, with higher total scores reflecting more severe CS symptoms. Part B (not scored) was designed to determine whether one or more specific CS disorders were associated with a specific pathology, such as restless legs syndrome, chronic fatigue syndrome, fibromyalgia, temporomandibular joint disorder, migraine or tension headaches, IBS, chemical sensitivity, neck trauma, anxiety or panic attacks, and depression. A cut-off score of at least 40 out of 100 points was established as CS confirmation, as in the original publication. 54 The higher the score, the more severe the somatic and emotional symptoms common to CS. A validated Polish version of the CSI was administered. 55 The convergent accuracy was assessed between vulvar and pelvic floor tenderness with respect to pressure and CSI.
Control
Hypothesis testing: (H0) The cotton swab test (V module testing in the VAMP protocol) is sufficient for the study of vulvodynia, and index test (Ha) the VAMP protocol (evaluation of four modules) has an advantage in the diagnosis of vulvodynia. 41 Symptoms of vulvar tenderness on pressure were considered positive when pain was noted (NRS score, >0).
Outcomes
For the main study outcomes, the following data were extracted:
Characteristic of women with vulvodynia.
Confirmation of PFM overactivity based on the VAMP protocol score (VAMP + group: V ⩾ 3 and/or M ⩾ 3 and/or P ⩾ 3) in correlation with vulvodynia subtypes and other values.
VAMP protocol validation (reproducibility with intra-observer reliability).
Comparison of cotton swab test results for area V in the VAMP protocol as H0 with the VAMP protocol (Ha).
Convergent validity between V(A)MP scores and the CSI score.
Statistical analysis
The following descriptive statistics were calculated for the analyzed variables: the number and percentage of participants for qualitative variables, minimum and maximum values, mean, median, confidence interval (±95%), standard deviation, and standard error. Normality of the distribution of variables in the study groups was verified using histograms and the Shapiro–Wilk normality test. 56 The analysis showed that the data distributions differed significantly from normal distributions (the significance levels of the Shapiro–Wilk test were less than 0.05). Therefore, non-parametric statistical methods were used to analyze the data. The most common method of assessing the reliability of a test is to test its internal consistency. The premise of this method is to divide the test (VAMP protocol) into four components (V, A, M, and P) and calculate Cronbach’s alpha coefficient. 57 The chi-squared test for binary outcomes 58 and the Mann–Whitney U-test 59 for continuous outcomes were applied. The Kruskal–Wallis test 60 was used to assess differences between three or more groups. p-Values were reported to four decimal places with p-values less than 0.001 and reported as p < 0.001. Spearman’s rank order correlation coefficients (Spearman’s correlation coefficient) 61 was used to analyze the correlation between variables. Sample size required to achieve adequate power for primary analyses was sufficient. All analyses were performed using Statistica 13.3, GraphPad Prism, and Microsoft Excel, Microsoft Corporation. A Statistical Analysis Plan was included in this study.
Results
Of the 155 participants in the RCT, 91.6% (N = 142) had provoked (including mixed) vulvodynia, localized to the vaginal vestibule and/or clitoral area (61.3%; N = 95). The spontaneous subtype was diagnosed in 13 patients (8.4%). The mean age of the participants was 30.1 years (range 18–46 years), and their body mass index was 22.9 kg (16.4–36.2). Almost all participants had a high school or college (secondary) education (96.8%; N = 150), and the majority lived in an urban area (69.1%; N = 107). The characteristics of the women with vulvodynia are presented in Table 1.
Characteristics of the participating cohort.
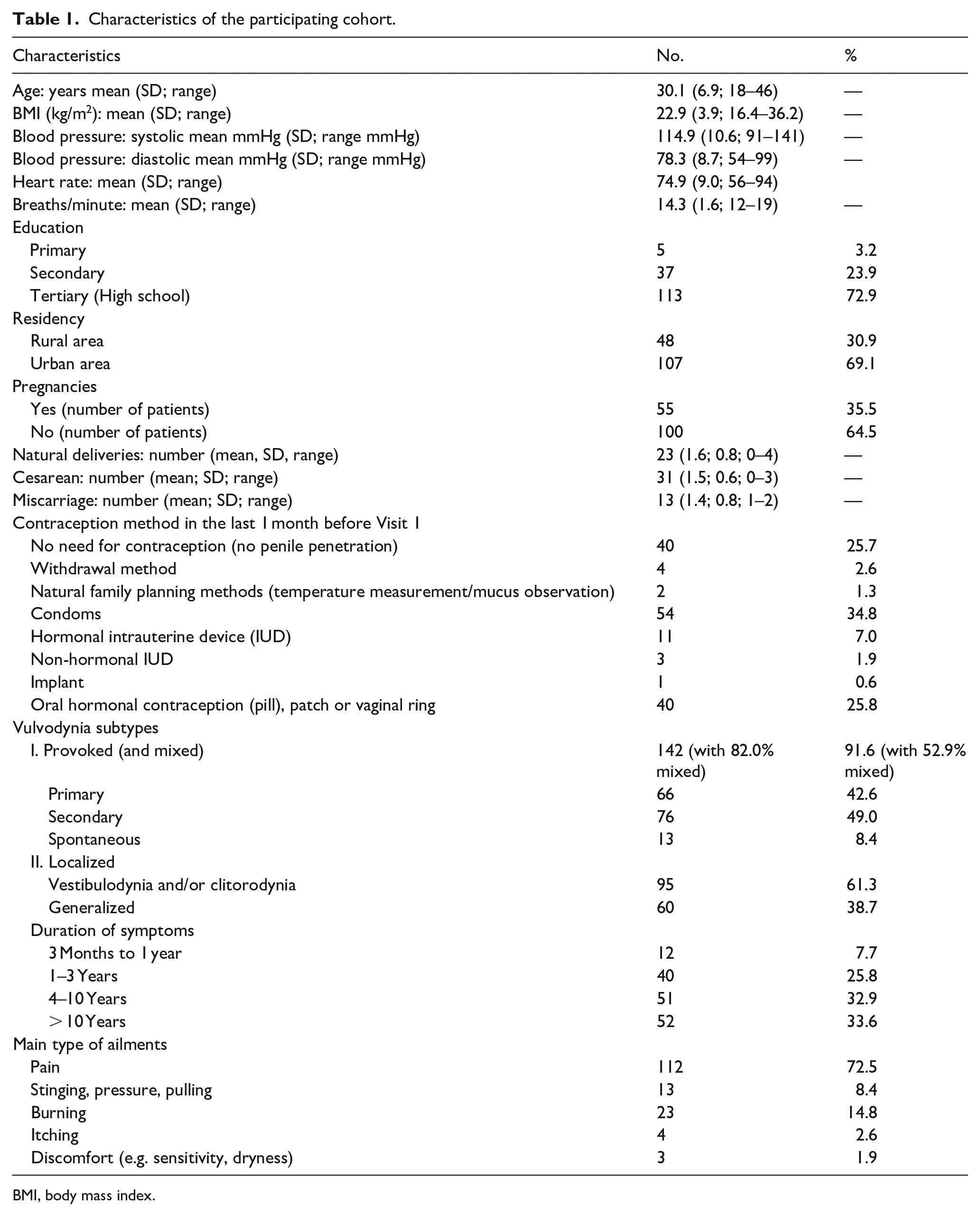
BMI, body mass index.
VAMP modules
The mean total scores for VAMP, according to the four scale areas examined, were high in the population of women with vulvodynia: V = 6.49 (SD = 2.87); A = 0.2 (SD = 1.07); M = 6.57 (SD = 2.62); P = 3.63 (SD = 3.52) (Table 2), and the V, M, and P scores by patient (the A module was omitted because it was 0 in almost all cases) are shown in Figure 3. Detailed descriptive statistics are also included.
Descriptive statistics regarding four areas of examination in VAMP II.

VAMP, Vulva, Anus, Muscle, and Paraurethra.

Patient-by-patient V, M, P scores results (the cut-off ⩾ 3 for positive results).
By comparing the results of modules V, A, M, and P with the variables of vulvodynia (subtypes: localized, generalized, primary and secondary provoked, mixed, and spontaneous, and duration of symptoms), a difference in the level of pain in the swab test between mixed and spontaneous vulvodynia was confirmed. Significant p-value (p = 0.009) for multiple (two-sided) comparisons according to the V module (in VAMP II) and the independent (grouping) variables mixed and spontaneous vulvodynia using the Kruskal–Wallis test (p = 0.0096) (Figure 4) were confirmed.

Correlation between V score and vulvodynia subtype: provoked, mixed, and spontaneous.
The average time required to perform a pelvic examination using the VAMP protocol was 1 min in all cases (counting full and completed minutes).
VAMP protocol validation
Measurements of VAMP scores by 2 examiners at the same visit to assess inter-examiner reproducibility were collected from 152 female participants with vulvodynia. Spearman’s rank order correlation coefficients (Spearman’s correlation coefficient) for significant correlations (p < 0.05) between each pair of measurements are shown in Table 3. The correlation between the first (VAMP I) and third (VAMP III at visit 2) measurements was high for all four pain measurement areas (V, A, M, and P).
Spearman’s rank order correlation between V, A, M, P scores between VAMP I, VAMP II, and VAMP III.
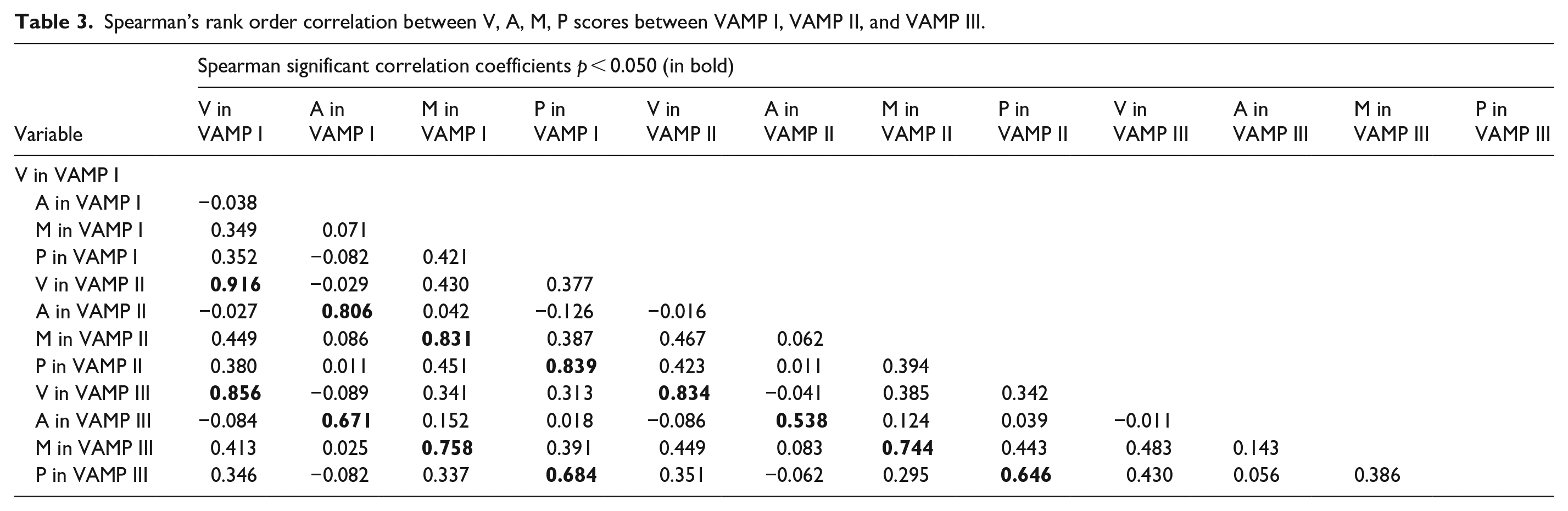
However, correlation analysis suggested that module A was not diagnostic in the VAMP protocol (the anal area was not painful in almost all patients, mean A score = 0.2). Therefore, the test validation has not been confirmed. To ensure consistency across all modules, Module A was removed. Validation was performed for the remaining modules (V, M, and P), and the test was confirmed to be reliable (Table 4) for calculating inter- and intra-individual reliability.
Spearman’s rank order correlation between V, M, and P scores (module A excluded) between VAMP I, VAMP II, and VAMP III.

VAMP, Vulva, Anus, Muscle, and Paraurethra.
In the ranges of the V, M, and P modules, all measured correlations (reliability and consistency) were >0.6, confirming the validity of the VAMP diagnostic protocol. The smallest differences (i.e. superior reliability and consistency) were observed for mucosal pain measurements (V-score p = 0.916 and p = 0.856, respectively), and the largest for paraurethral pressure pain (P-score p = 0.684 and p = 0.646, respectively). The Spearman’s rank order correlation coefficient was very high for both mucosal measurements (V) and moderate-to-good reliability for pelvic measurements (M, P). Results of reliability analysis according to the VAMP III with A module exclusion mean = 15.8931, SD = 6.8379, N = 131, Cronbach’s alpha = 0.667, and the VAMP I and VAMP II (also according to the V, M, and P modules) mean = 15.0719, SD = 7.0655, N = 153, Cronbach’s alpha = 0.635. The calculated Cronbach’s alpha coefficient was satisfactory for assessing the test reliability. Although the interpretation of Cronbach’s alpha values can be contextualized as “questionable,” 62 may still be usable depending on the study context.
VAMP protocol score as confirmation of PFM overactivity
Patients with vulvodynia were divided into two groups based on the results of the VAMP protocol (according to the cut-off point for differential PFM overactivity):
VAMP + (positive with V ⩾ 3 and/or M ⩾ 3 and/or P ⩾ 3) with confirmation of high PFM tone in 150 patients (98.7%)
VAMP − (negative) with each module V, M, and P < 2, which was considered a normal PFM state in two women (1.3%).
Since PFM overactivity was confirmed in almost all women with vulvodynia, statistical analysis was not performed for either group (VAMP + versus VAMP −).
Hypothesis testing
In the case of no pain on cotton swab pressure (V score = 0), six patients (3.9%) with vulvodynia met this criterion. Four of the six patients with a V score of 0 had a very high muscle pain score (M module): Pearson chi-square test = 6, df 1, s = 0.014, with Yates correction (for small groups) chi-square = 3.375, df = 1, s = 0.066; the exceedance of the critical value of the significance level was not large (Table 5).
Patients with no pain on cotton swab test vulvar hymen remnant pressure (V = 0 in VAMP) with other VAMP II modules scores (M, P).

VAMP, Vulva, Anus, Muscle, and Paraurethra.
Correlation between V(A)MP score and the CSI score
The mean CSI score in women with vulvodynia was 42.1 (range 12.0–76.0; SD 14.7) (Table 6). A CSI cut-off value of >40 indicates confirmation of CS.
Descriptive statistics regarding CSI in vulvodynia cohort.

CSI, Central Sensitization Inventory.
The percentage of patients with a CSI score of up to 39 was 46.3% (N = 62), whereas a score >40 was achieved in slightly more patients with vulvodynia (53.7%; N = 72). The Spearman rank order correlation coefficient (Spearman’s correlation coefficient) between the CSI score and VAMP modules (V, M, P) was significant for modules M (Spearman’s correlation coefficient = 0.210, p < 0.05) and P (Spearman’s correlation coefficient = 0.209, p < 0.05), but not for module V (r = 0.147). A detailed analysis using the Mann–Whitney U-test also showed that participants with CSI scores >40 demonstrated higher M and P scores (but not V) (Table 7).
Results of the Mann–Whitney U-test significance of differences in performance between CSI score >40 and V, M, P modules in vulvodynia patients.

CSI, Central Sensitization Inventory; VAMP, Vulva, Anus, Muscle, and Paraurethra.
The mean CSI scores were 38.08 in generalized vulvodynia, 44.30 in localized subtype (Table 8), 42.50 in primary, and 42.57 in secondary vulvodynia.
Descriptive statistics regarding CSI scores in vulvodynia subgroups: generalized and localized.
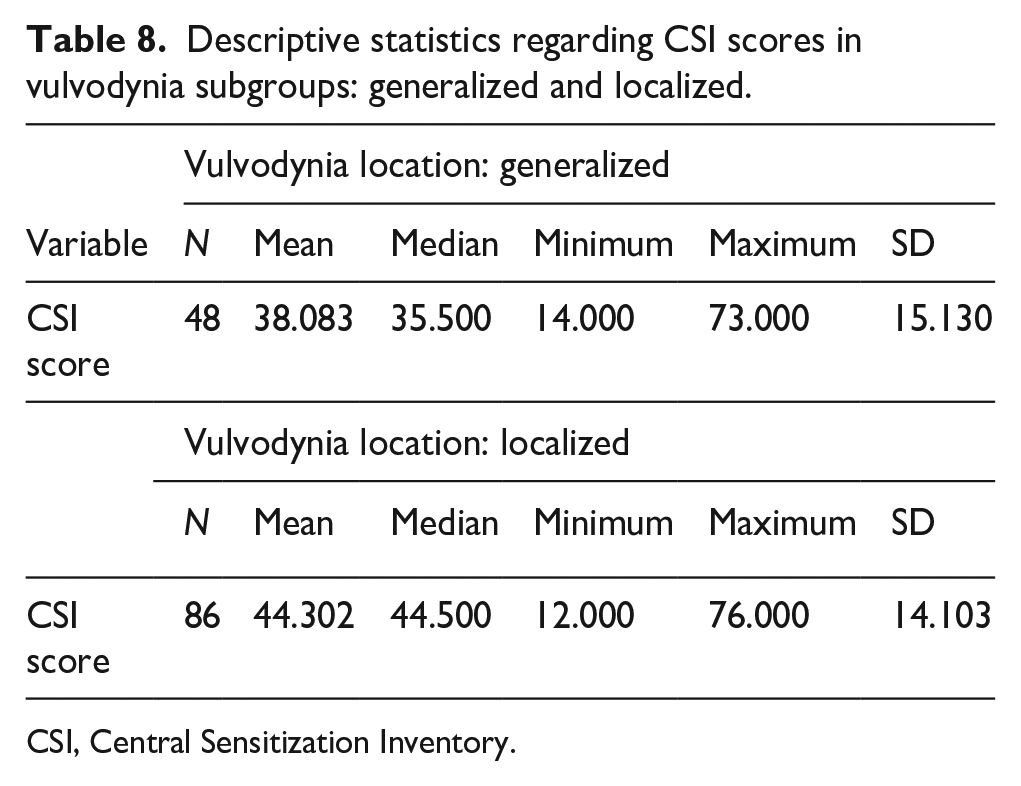
CSI, Central Sensitization Inventory.
Using the Mann–Whitney U-test, the localized vulvodynia subgroup showed higher CSI scores (Table 9). There were no differences between the CSI score and primary or secondary vulvodynia (Mann–Whitney U-test, p = 0.941) or duration of pain experienced (Kruskal–Wallis test; p = 0.144).
Results of the Mann–Whitney test showed significant differences in performance between vulvodynia subgroups (localized and generalized) with CSI results.

CSI, Central Sensitization Inventory.
Discussion
The results of this study showed that the most common manifestation of chronic vulvar pain was provoked (and mixed) vulvodynia (91.6%), and spontaneous vulvodynia was rare. Vestibulodynia was assessed in 61.3%. As many women report vestibular pain or variable pain at other sites, the division (localized versus generalized) is not precise.
Because, to date, there is no “gold standard” testing strategy for evaluating pelvic floor myofascial pain, 20 the study authors described and validated a practical tool for testing PFM overactive, increased tone in women with vulvodynia.
The mean total scores (in NRS) for the VAMP protocol, according to the four areas examined, were high in the population of women with vulvodynia according to modules V, M, and P (V = 6.49; M = 6.57; P = 3.63), but not for module A (A = 0.2), which confirmed the pressure pain of the vestibule, pelvic muscles, and paraurethral area in women with vulvodynia. The anal area was not tender in the cotton swab test, although it should not be omitted during examination because it provides an opportunity to visualize the anus to exclude pathological lesions (varices, fissures, and anal cancer) and indicates an overestimation of pain during the examination of other modules of the VAMP protocol.
In a previous publication of the study, Authors, who included 300 healthy women, V, M, and P scores were very low (the anal area in the swab test was not painful either) in the asymptomatic population. 63 A comparison of the different modules is shown in Figure 5.

VAMP protocol modules scores in vulvodynia and asymptomatic 63 cohort.
These results are consistent with a pilot study evaluating the VAMP protocol, 22 where differences in the results of the VAMP protocol were identified between patients with vulvodynia and the control group. In addition, in that publication, it was proved that pain on examination of three modules (at least one of V, M, P ⩾ 3) indicates PFM overactivity. 22 Positive results of the VAMP protocol confirmed pelvic muscle overactivity in almost all women with vulvodynia (98.7%).
The diagnosis of PFM is a key issue in patients with vulvodynia. Confirmation of PFM overactivity is an indication for multidisciplinary therapy that includes pelvic muscle and general physiotherapy, which is the authors’ daily practice at the center where they work with patients with vulvar diseases. According to the literature, the severity of pain on PFM examination in provoked vestibulodynia is a significant predictor of the treatment response to pain intensity reduction. Lower PFM tone at baseline was associated with a greater reduction in pain intensity after physiotherapy and at follow-up. 64
The study demonstrated acceptable and satisfactory internal consistency based on Spearman’s correlation coefficient test (coefficients > 0.60) for each of the three modules (excluding Module A). The VAMP protocol showed a high degree of internal consistency and test–retest reliability for the three (V, M, and P) domains (r = 0.916 – 0.646). While the Cronbach’s alpha values of 0.635 and 0.667 are borderline for reliability, 62 they are acceptable in the context of exploratory or developmental research, especially when considering the subjective perception and reporting of pain (context-specific acceptability). As in the publication evaluated the reliability and validity of the Chronic Pain Coping Inventory in a sample of chronic pain patients, the subscales of this inventory demonstrated Cronbach’s alpha coefficients ranging from 0.60 to 0.85, indicating varying levels of internal consistency. Despite some subscales exhibiting alpha values around 0.6, the instrument was deemed acceptable for research purposes in this population. 65
The scales may require refinement in future studies to improve internal consistency, but they provide sufficient reliability for the study’s current purpose.
The VAMP protocol was successfully cross-validated and shown to be highly effective in discriminating between women with PFM overactivity. Validation showed that the anal examination (module A) should be omitted because it differs significantly from the other modules of the protocol (the score is 0 in almost all women tested). Literature indicates that the rectal area is a non-significant area for assessing PFM pain in conjunction with an overactive increased tone. The authors suggested that touching this area with a cotton swab may help identify patients who overestimate their pain levels, which may be the case in catastrophic patients. For clinicians, this may be an opportunity to assess the anal area, rule out concurrent anal pathology (anal fissure or hemorrhoidal disease), and assess the reliability of pain scores reported by the subjects.
Reliability was not affected by performing the test twice at visit 1, which was described as a source of bias.10,66
Differentiating women with pelvic pain from those with overactive PFM dysfunction remains challenging for researchers and practitioners. The VAMP protocol was proposed as a simple tool for physical examination. In total, gynecologists should examine four anatomical areas to make the assessment more reliable than the swab test, which is commonly used in the evaluation of women with vulvodynia.
The results of this study showed that, in the vast majority of women with vulvodynia, the vulvar pain score (V module > 0 in 96.1%) was mostly consistent with pain in the pelvic muscle and paraurethral area (M and P modules). The mean score on the cotton swab test (V module) was significantly lower in the spontaneous vulvodynia subtype group, although the spontaneous vulvodynia cohort was much smaller than the provoked (and mixed) subgroups. These results indicate different pain mechanisms in patients in these vulvodynia subgroups.
Although the number of women with vulvodynia with no pain on cotton swab pressure was very small (3.9% of the study cohort), it has been shown that 2/3 of these women with PFM overactivity (diagnosis based on modules M and P) would have been missed using only the cotton test during gynecological examination. Therefore, assessing women with vulvodynia using only the cotton test is not a sufficiently sensitive diagnostic test.
Other authors 24 hypothesized that the “subgroup of vulvodynia cases without increased vulvar sensitivity differ little from those with greater sensitivity.” Vulvodynia cases with minimal or no increase in vulvar sensitivity were similar to those with higher sensitivity for most of the characteristics assessed. Although the group with negative hymenal base sensitivity (V-score = 0) was not large in this study, these women may not have received a proper diagnosis or may have been excluded from participating in clinical trials, making treatment more difficult. 67
According to Lo et al., 27 patients with higher PFM pain scores (similar to the P and M modules in the VAMP protocol) than the mucosal pain score (the V module in VAMP) may have different types of provoked vestibulodynia. This study 27 highlights the importance of PFM assessments, in addition to vestibular swab tests. Analyzing a multisite database of patients presenting for provoked vulvodynia treatment, several factors can help distinguish the distinct phenotypes of mucosal-predominant vestibulodynia and muscle-predominant vestibular pain. Consistent with the literature, pain duration was more strongly associated with mucosal pain (which was not confirmed in this study), whereas anxiety was more strongly associated with muscular pain. 28
According to a study by Meister et al., 45 based on a systematic review of the literature (55 studies conducted since 1946), eight recommendations were made for the study of pelvic floor myofascial pain in women. The VAMP protocol (in terms of the V, M, and P modules) meets the requirements outlined in the study authors’ previous publications. 21 The last recommendation mentioned in the above-cited review concerns the area of examination: cotton swab pressure on the vestibular area is obligatory in vulvodynia,20,48 and in 41.8% of the studies, other areas (the anus, abdomen, and urethra) were incorporated into the pelvic examination. Paraurethral area examination indicated the lowest correlation in Spearman’s correlation coefficient test for test validation (p = 0.646), which indicates an emphasis on how to palpate the area in the process of training of healthcare practitioners. According to Jantos, 68 the periurethral area is particularly important in the evaluation of PFM overactivity. The inventor of the IMAP protocol (the scientific basis of the abbreviated VAMP protocol) emphasized that examination of the paraurethral region “appears to be the primary generator of chronic urogenital pain (CUP) symptoms and is diagnostically the most reliable method for differentiating between CUP cases and asymptomatic controls. As an anatomical region, the paraurethral area is an overlooked source of pain and rarely tested during diagnostic assessments.” 68 Paraurethral area (the urogenital diaphragm muscle and fascia) was also included in the examination protocol proposed as the Multidisciplinary Approach to the Study of Chronic Pelvic Pain Research Network, established to provide new perspectives on the clinical features and pathogenesis of urological CPP syndrome. 69
The validated VAMP protocol is similar in many ways to the pelvic examination methods described by other authors20,48,64,66,70 rather than a comprehensive study to map all possible muscles associated with pelvic pain. The study Authors and other researchers emphasized that identifying TrPs within muscles (without identifying which muscle, there is no need to examine the entire pelvis) is sufficient for the assessment of pelvic floor myofascial pain. The procedure should be simple enough so that even a gynecologist or urologist with less experience in pelvic assessment can perform a diagnostic test in patients with urogenital pain. The advantage of the VAMP protocol over other PFM testing protocols in patients with vulvodynia is quantitative evaluation (not only qualitative confirmation of overactive status). The NRS score is noted using the VAMP acronym in medical reports for comparison during subsequent visits or clinical trials to objectify the results, which can be beneficial in comparative studies. Although the NRS pain severity score has been described in some studies, 34 the cut-off value has not been specified (as positive circled as greater than 0). Moreover, to perform a BME according to the VAMP protocol, it is not necessary to use additional devices (e.g. manometer, algometer, ultrasound, or electromyography (sEMG)). Once an evidence-based standardized examination is established, efforts can turn to promoting physician education.
The assessment of the symptoms of CS in patients with complex diseases is gaining importance. The final aspect of this study was the validation of the VAMP protocol in correlation with the CSI score. Half (53.7%) of the women with vulvodynia met the criterion of CS (CSI score > 40) for pain stimuli as a feature of chronic pain. Other authors have confirmed that 33% of women with vulvodynia have CS, which is associated with comorbidities, dyspareunia, pain during micturition, and pain during defecation. Patients with CS had more comorbidities and responded poorly to treatment. More treatment attempts were required, with longer response time (>2 months). 71 Some controversy exists about whether peripheral or central mechanisms cause myofascial pain syndrome and chronic regional pain syndrome. Peripheral pain mechanisms are involved in myofascial pain syndrome. 54 Future studies should consider how a high or low CSI score may affect the differential diagnosis, prognosis, and response to treatment in patients with chronic pain. 72 Patients with CSI scores above the cut-off value had higher M and P values (but not V) in the VAMP protocol. Other authors also found convergent validity of PFM tenderness on palpation (as an M module in the VAMP protocol) and a CSI score of >40 (p = 0.04), suggesting central pain mechanisms in such patients. 73 In this study, patients with localized vulvodynia showed higher CSI scores than those with generalized vulvodynia. Another study failed to demonstrate the clinical utility of combined CSI and pressure pain threshold analysis. 74
The question of whether vulvodynia is an example of nociplastic pain remains open, with arguments in favor of this hypothesis. Although the features of CS (CSI score above the cut-off) are not the same as those of nociplastic pain, it can be inferred that they leave aside some details. The severity of nociplastic pain in patients with myofascial pain appears to be inversely related to improvement resulting from peripheral treatment; that is, a reduction in pelvic tension may increase perceived discomfort. This does not mean that people with moderate or high levels of nociplastic pain do not benefit from physical therapy; rather, they benefit proportionally less. 75 The prevalence of pelvic floor myofascial pain ranges from 13% to 22% in women with persistent pelvic pain and is as high as 78% in women with interstitial cystitis, depending on the diagnostic criteria used and the method of assessment.76,77
An overactive state of PFM is considered the source of chronic pain in almost all women with vulvodynia,9,10 which was also confirmed in the study using VAMP protocol (98.7% vulvodynia cohort with V, M, P ⩾ 3). As pelvic floor overactivity is often overlooked in bimanual gynecological examinations, the actual incidence of PFM overactivity is unknown. 19
The index test (VAMP), which allows replication, is simple to teach healthcare practitioners, can be performed during gynecological examination in symptomatic women, and is not time-consuming (requires app. 1 min). Quantitative evaluation allows the comparison of research results in scientific studies and clinical trials and indicates physical therapy for chronic pain conditions.78,79
According to the conclusion of the literature review (151 studies), “despite the large literature, few studies provide convincing evidence for increased tone/overactivity of pelvic floor muscles in pelvic health conditions. Interpretation is hampered by design and measurement issues. Terminology was often inaccurate.” 14 Therefore, there is a need to develop a simple tool for clinical practice, which has been described and validated in this study. Further studies are required to quantitatively assess the effectiveness of vulvodynia treatment using the VAMP protocol as the core outcome set.
A clinical limitation of the VAMP protocol is the inability to access the vagina for pelvic palpation. A comparison of transvaginal and rectal examinations was not performed. Another limitation to the widespread adoption of the VAMP test, especially in clinical trials, is the anxiety associated with performing a painful test, and different types of emotional attitudes can be a source of bias. 80
This study had some limitations. First, the sample size was not large, which could have resulted in borderline results. Increasing the group size improved the power of the statistical analyses. A larger study would help confirm the robustness of the findings. The authors encourage physicians to perform pelvic examinations according to the VAMP protocol and share their opinions. In addition, the pelvic examination was based on palpation and cotton swab pressure (in the subjective NRS assessment), without the use of calibrated instruments, to simplify the diagnostic protocol and facilitate clinical practice.
Conclusions
The results of this study showed that the most common manifestation of chronic vulvar pain was provoked/mixed vulvodynia (91.6%), and spontaneous vulvodynia was rare. Vestibulodynia was assessed at 61.3%. Nevertheless, a large number of women reported vestibular pain and pain elsewhere; it seems that the division (localized versus generalized) is not precise for the researchers.
The mean total scores (in NRS) for the VAMP protocol according to the four areas examined were high in the population of women with vulvodynia according to modules V, M, and P (V = 6.49; M = 6.57; P = 3.63) but not for module A (A = 0.2), which confirmed the pain of the vestibule (V), pelvic muscle (M), and paraurethral (P) area to pressure. The anal (A) area was not tender in the cotton swab test, although it should not be omitted from the examination. The VAMP protocol was successfully cross-validated (according to the V, M, and P, Spearman’s correlation coefficient value =0.916–0.646) and was shown to be highly effective in discriminating women with PFM overactivity. The VAMP protocol’s positive results (V, M, P score ⩾ 3) confirmed PFM overactivity in almost all women with vulvodynia (98.7%), although differentiating women with pelvic pain arising from overactive PFM dysfunction remains a challenge for researchers and practitioners. The VAMP protocol was proposed as a simple tool for physical examination. In total, gynecologists should examine four anatomical areas to make the assessment more reliable than the swab test, which is commonly used in the evaluation of women with vulvodynia. The results showed that the vast majority of women with vulvodynia had positive vulvar pain scores (V module > 0 in 96.1% of the study cohort), which were mostly consistent with pain in the pelvic muscle and paraurethral area (M and P modules). The mean score of the cotton swab test was significantly lower in the spontaneous subtype, though the group was much smaller than that of the provoked subtype, which may indicate different mechanisms of pain in patients in the vulvodynia subgroups. Although the group of women with vulvodynia with no pain on cotton swab pressure was very small (3.9% of the study cohort), it has been shown that 2/3 of these women exhibit pelvic muscle dysfunction that would be overlooked using only the cotton test in a gynecological examination. Analyzing a multisite database of patients with provoked vulvodynia, several factors can help distinguish the distinct phenotypes of mucosal-predominant vestibulodynia and muscle-predominant vestibular pain.
As much as half of the study cohort (53.7%) met the criterion of CS to pain stimuli as a feature of chronic pain (CSI score > 40), and higher PFM pain on pressure (M and P scores) was observed in women with CS. Localized versus generalized vulvodynia showed higher CSI scores (CSI = 44.30 versus 38.08; p = 0.01), suggesting central pain mechanisms. The question of whether vulvodynia is an example of nociplastic pain remains open, with arguments in favor of this hypothesis.
The index test was described in a way that makes its replication seem simple to teach healthcare practitioners. It can be performed during gynecological examinations in symptomatic women; it is not time-consuming (requires an app. 1 min). Further studies are needed to quantitatively assess the effectiveness of the treatment of vulvodynia based on the VAMP protocol as the core outcome set.
Supplemental Material
sj-docx-1-whe-10.1177_17455057251338410 – Supplemental material for Pelvic floor examination in vulvodynia: VAMP protocol validation in correlation with central sensitization
Supplemental material, sj-docx-1-whe-10.1177_17455057251338410 for Pelvic floor examination in vulvodynia: VAMP protocol validation in correlation with central sensitization by Ewa Baszak-Radomańska, Jadwiga Wańczyk-Baszak and Tomasz Paszkowski in Women’s Health
Supplemental Material
sj-docx-2-whe-10.1177_17455057251338410 – Supplemental material for Pelvic floor examination in vulvodynia: VAMP protocol validation in correlation with central sensitization
Supplemental material, sj-docx-2-whe-10.1177_17455057251338410 for Pelvic floor examination in vulvodynia: VAMP protocol validation in correlation with central sensitization by Ewa Baszak-Radomańska, Jadwiga Wańczyk-Baszak and Tomasz Paszkowski in Women’s Health
Footnotes
Acknowledgements
We thank all women who participated in this study.
Ethical considerations
Approval for the study was obtained from the appropriate IRB of the Medical University of Lublin, Poland; Ethics Committee approval KE-0254/210/2021 for RCT registered on ClinicalTrials.gov ID NCT05955313 on June 6, 2023.
Consent to participate
Written informed consent was obtained from all individual participants included in the study.
Consent for publication
Not applicable.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: A Clinical Trial was granted by the Medical Research Agency in Poland: 2020/ABM/01/00114-00; Effectiveness of low-dose naltrexone (LDN) on pain perception and quality of life in women with vulvodynia.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data supporting the findings of this study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
