Abstract
In 2023, a breast cancer risk assessment and a subsequent positive test for the BRCA-2 genetic mutation brought me to the uncomfortable intersection of a longstanding career as an advocate for high-quality medical evidence to support shared patient-provider decision making and a new role as a high-risk patient. My search for studies of available risk-management options revealed that the most commonly recommended approach for women with a ⩾20% lifetime breast cancer risk, intensive screening including annual mammography and/or magnetic resonance imaging beginning at age 25–40 years, was supported only by cancer-detection statistics, with almost no evidence on patient-centered outcomes—mortality, physical and psychological morbidity, or quality of life—compared with standard screening or a surgical alternative, bilateral risk-reducing mastectomy. In this commentary, I explore parallels between the use of the intensive screening protocol and another longstanding women’s health recommendation based on limited evidence, the use of hormone therapy (HT) for postmenopausal chronic disease prevention, which was sharply curtailed after the publication of the groundbreaking Women’s Health Initiative trial in 2002. These declines in HT utilization were followed by marked decreases in breast cancer incidence, providing a compelling lesson on the critical importance of a solid evidentiary basis for women’s health decisions. Known harms accompanying the benefits of breast screening—overdiagnosis, psychological effects, and mammography-associated radiation-exposure risks—make empirical measurement of patient-centered outcomes essential. Yet, published research on intensive screening of women at high breast cancer risk has largely ignored these outcomes, leaving patients, providers, and guideline developers lacking the evidence needed for best practice. Outcomes research is both feasible and urgently needed to inform care decisions and health policy for this patient population.
Introduction
Two seemingly unrelated recent announcements on women’s health may have more in common than initially apparent. First, in November 2022, the U.S. Preventive Services Task Force (USPSTF) issued a “D” (do not perform) recommendation for postmenopausal hormone therapy (HT) to prevent chronic conditions. 1 This recommendation was consistent with its previous (2012, 2017) guidance on HT but differed markedly from earlier guidelines recommending long-term use.1 –3 Second, in May 2023, the American College of Radiology (ACR) updated its 2018 screening recommendations for women at “higher-than-average” risk of breast cancer. 4 In addition to reiterating 2018 guidance that annual digital mammography should begin before age 40 years, with “supplemental screening, depending on risk,” preferably using magnetic resonance imaging (MRI), the ACR 2023 guideline reduced the age for initial risk assessment from 30 to 25 years.4,5 The ACR also modified recommendations for the subset of high-risk women with known breast cancer gene (BRCA) mutation to delay mammography until age 40 years for those choosing annual MRI.4,5 Similar recommendations for earlier-age or more intensive screening of high-risk women date to a 1997 National Human Genome Research Institute (NHGRI) expert panel on BRCA mutation and have since been reaffirmed by the American Cancer Society (ACS) and National Comprehensive Cancer Network (NCCN; Figure 1).6 –11
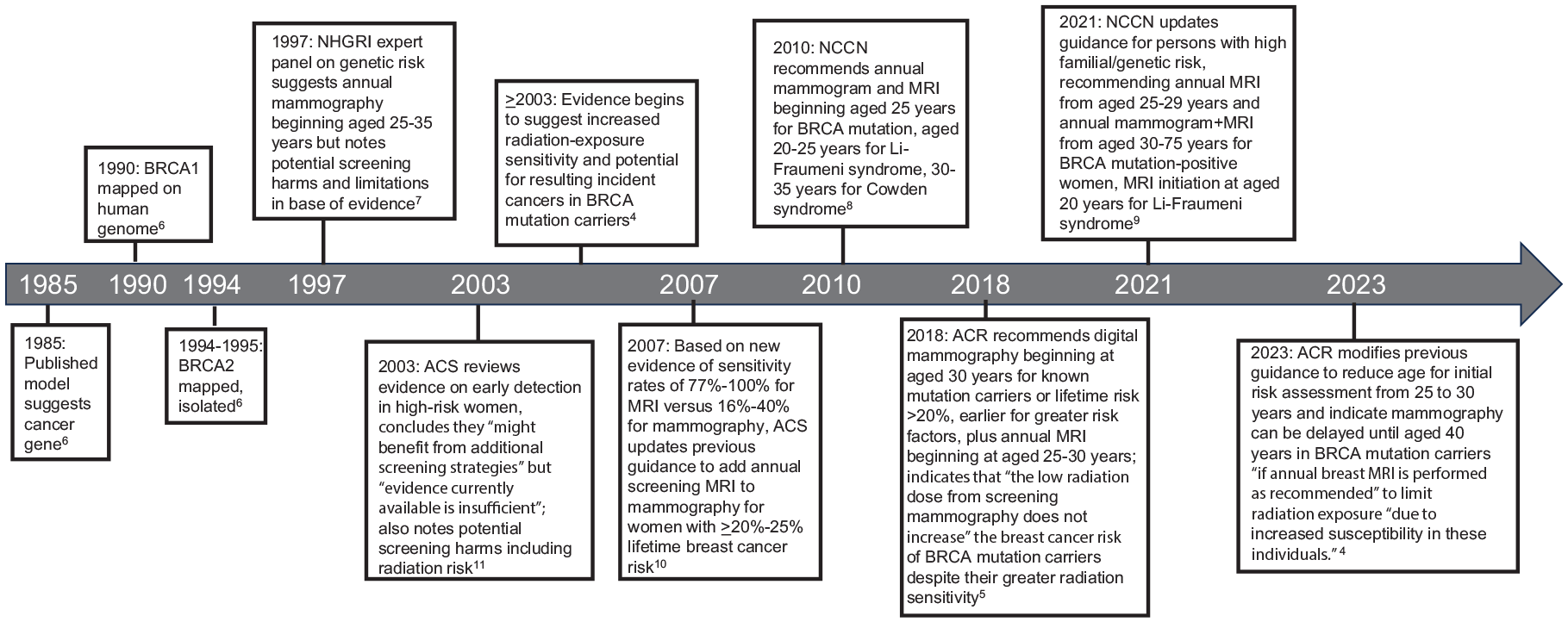
Timeline of key events, intensive breast screening for high-risk women in the United States.
The 2023 ACR publication was of particular interest to me because several of my family members had recently tested positive for the BRCA-2 mutation. I had been encouraged by my primary care provider to seek genetic counseling but wanted to ensure that, if I did test positive, the information would be actionable. As a longstanding advocate of high-quality medical evidence, and as a doctoral faculty member who teaches statistics, epidemiology, and evidence-based health care, I researched available risk-management options for women at elevated risk of breast cancer, focusing primarily on evidence reviews conducted by the ACR and USPSTF.4,12 I traced their citations and evaluated the specific findings of each study. Additionally, I conducted separate literature searches using terms representing patient-centered outcomes (e.g., morbidity, mortality, psychological distress). I completed this process and made my decision in June 2023.
This process revealed parallels between the intensive screening protocol and a topic on which I have taught for many years: the longstanding use of HT for prevention of cardiovascular disease (CVD) and other chronic conditions in the 1990s. Grounded only in epidemiological associations between HT and positive health outcomes, this practice was ultimately refuted by the groundbreaking Women’s Health Initiative (WHI) trial and other higher-quality evidence described in detail in the next section of this article.3,13 –21 Later called “a victory for women and their health,” the WHI trial was launched in 1991 because the first female director of the National Institutes of Health recognized the need to provide empirical information on previously understudied populations, including women and minorities. 22
As demonstrated herein, both the original HT guidelines for postmenopausal women and the longstanding intensive screening approach for high-risk women exemplify a tendency that is unfortunately common in epidemiological history: neither was supported by a reasonably rigorous base of evidence. Yet, even while untested in high-quality outcomes research, both were strongly promoted, widely viewed as evidence-based, and considered by many to be the only ethically appropriate approach to care.
This tendency raises important questions about why and how care protocols so crucial to women’s health go relatively untested for long periods of time—all the while they are in widespread use. For HT, the improvements in patient safety outcomes that occurred when a weak evidence base was replaced with scientifically rigorous data are known. For the intensive screening protocol, however, these outcomes remain a crucial but unexplored chapter in epidemiological history. This commentary offers research-based explanations for these patterns, what they might tell us about our culture, and implications for future research.
HT: improved outcomes follow improved evidence
An instructive sequence of events underpinned changes in the USPSTF guidance on HT use (Figure 2).3,13 –20 In the 1990s, extensive nonexperimental evidence linked postmenopausal HT to reduced rates of cardiovascular events (CVEs) and several chronic conditions.3,16 Despite nonrandomized designs, study results were readily accepted by the medical community and an enthusiastic public.17,18 Guidelines from influential medical organizations recommended consideration of long-term postmenopausal HT to prevent chronic disease and increase life expectancy.2,3 Remarkably, one guideline observed that although most studies of HT had been conducted in white women, they should nonetheless “also be applied to nonwhite women” because “coronary heart disease is the most common disease of women regardless of race.” 2 A series of “educational” advertisements, funded by the HT manufacturer, featured a popular super-model advising women that the “time to protect your future [with estrogen replacement] is now.” 18

Timeline of key events, HT use in the United States.
Yet, by 2002, several randomized controlled trials (RCTs) including the first large-sample assessment of HT in chronic disease prevention, the WHI trial, indicated benefits were outweighed by harms, particularly increased risks of breast cancer, CVEs, and pulmonary emboli. 3 The earlier favorable nonexperimental results had been confounded, in a classic selection bias, by the better health habits and socioeconomic status of women who chose HT.19,20 This “shock to the medical system” 17 was quickly followed by markedly curtailed HT use and, not surprisingly, a 9%–23% decline in breast cancer incidence in the United States, with similar trends in other countries.13,14 A recent clinical practice update to the WHI reaffirmed its findings that use of HT “to prevent cardiovascular disease, dementia, or other chronic diseases” remains unsupported by evidence. 21
Examination of root causes underlying guideline recommendations for long-term HT use based only on weak correlational evidence reveals a combination of commercial interest, ethical breaches, and sentiments that today would be recognized as blatant misogyny. Enthusiasm for estrogen replacement as a remedy for the “endocrine disease” of menopause dates from the 1960s to 1970s, rooted in a “philosophy of estrogen maintenance” promoted by physician Robert Wilson.13,15 Unlike men, whose “androgen capital is an annuity which lasts for life,” wrote Wilson, postmenopausal women face loss of “the allure which attracts the male,” becoming “dull and unattractive.” This “galloping catastrophe” was treatable with estrogen, he argued. 15
Supplementing popular interest in HT with “evidence” of its health benefits, the HT manufacturer paid a medical marketing firm to ghostwrite more than 50 medical journal publications and to recruit “authors” to attach their names to the works. 13 These publications and other “scientific” communications downplayed the risks of HT, especially breast cancer; promoted off-label uses with, according to an internal email, “messages that fill the gaps not addressed by current studies”; highlighted HT’s potential benefits in preventing numerous health ills, including CVE, neurological diseases, and impairment of cognition or mood; and suggested that HT was superior to other treatments for postmenopausal symptoms. 13
Intensive breast screening: HT redux?
The history of HT may be repeating itself in the intensive screening protocol. Although a large body of research, some randomized, clearly indicates that adding MRI to screening mammography yields earlier cancer detection with smaller tumors and fewer node-positive and interval (i.e., diagnosed after screening) cancers (Table 1),4,5,9,10,23 –26 information about superior MRI screening performance alone is insufficient to support the protocol. Core principles of evidence-based practice, including seeking the best available evidence to address the patient’s clinical condition and incorporating patient preferences into shared decision making, demand high-quality evidence on outcomes important to patients. 27 Such outcomes include all-cause and cancer-specific mortality and morbidity, psychological well-being, and quality of life (e.g., avoidance of chemotherapy, ability to engage in daily activities).
Key studies of MRI and/or mammography performance in breast cancer detection.

ACR, American College of Radiology; ATM, ataxia-telangiectasia mutated; BRCA, breast cancer gene; BRRM, bilateral risk-reducing mastectomy; CBE, clinical breast examination; CHEK2, checkpoint kinase 2; CI, confidence interval; HR, hazard ratio; MRI, magnetic resonance imaging; NCCN, National Comprehensive Cancer Network; PALB2, partner and localizer of BRCA2; QoL, quality of life; RCT, randomized controlled trial; TP53, tumor protein p53; USPSTF, U.S. Preventive Services Task Force.
Remarkably, although these outcomes have been reported for standard mammography screening, almost no comparative studies of the effects of either earlier-age screening or MRI screening on these outcomes have been published (Table 1).4,12,28 –34 In its 2019 recommendations for risk assessment and genetic testing for BRCA-related cancer, the USPSTF reported that its systematic literature review had identified “no studies on the benefits of intensive screening for BRCA-related cancer on clinical outcomes.”12,35 Similarly, the ACR’s 2023 “robust strength-of-evidence analysis” supporting intensive over standard screening included only 3 comparative studies of patient outcomes, none with strong research designs: 2 modeling studies31,36 and a cohort comparison of BRCA2-positive women in 2 different European countries, of whom only 34 received mammography + MRI. 28 Notably, although the European study’s title referred to “mortality,” the statistic actually reported was survival, a commonly misunderstood measure known to be affected by the detection and lead-time biases typical of many analyses of intensive cancer screening protocols.37,38
A handful of comparative studies, with mixed findings, were not included in the ACR recommendation statement, one because it was published after 2023 (Table 1).29,30,32 –34 Highlighting the persistent lack of evidence about intensive surveillance, the report of a mortality study comparing screening with versus without MRI, published online 8 months after I made my risk-management decision, observed that although tumor stage and survival are important, “ultimately it is necessary to show that the [surveillance] intervention is associated with a decline in breast cancer mortality.” 32
This deficit of information about intensive screening outcomes is especially remarkable given the availability of evidence about the surgical alternative, bilateral risk-reducing mastectomy (BRRM). A 2018 Cochrane review identified 21 studies, 9 with comparison groups, documenting post-BRRM reductions in breast cancer incidence, morbidity, and/or mortality in high-risk women, particularly those with BRCA mutations. 39 Psychosocial outcomes included satisfaction with the decision and reduced worry about breast cancer, but increased concerns about body image. Adverse events reported in case series included post-surgical complications requiring new surgery at a low rate (4%) without reconstruction and higher rate (64%) with implant reconstruction. Although not mentioned by the ACR, BRRM was described in both NCCN guidance on high-risk patients and the USPSTF policy statement on BRCA-mutation testing as an evidence-based option associated with large (90%–100%) reductions in breast cancer incidence.9,12
The current lack of similar information about intensive screening leaves patients, providers, and guideline developers with an extremely limited evidentiary basis for comparison of management options. Lacking such evidence, the assertion that earlier cancer detection translates into patient benefit for high-risk women represents an assumption similar to that made regarding the effects of exogenous estrogen on women’s health, which, although biologically plausible, was ultimately refuted by stronger research. 3 This reliance on assumption is a critical flaw, partly because the benefits of screening are accompanied by known harms.11,12,40,41
First, the risk of radiation-induced breast cancer is increased in women who receive more intensive or longer-term mammography screening, including follow-up of initially positive results. 42 In one modeling study, the benefit-to-harm ratio (deaths prevented per incident radiation-caused case) diminished with radiation exposure in dose-response fashion, from 26 (lowest category estimated: biennial screening, aged 50–74 years, nonlarge breasts) to 4 (highest category estimated, annual screening, aged 40–74 years, large breasts). 42 Although even that smallest ratio is clearly favorable, the benefit-to-harm ratio for mammography beginning as early as age 25 years is unknown.
Amplifying this concern is another unknown: potentially increased breast cancer incidence from exposing women with a known or undetected genetic mutation to up to 50 years of mammography-associated radiation. This concern is supported by several studies of BRCA mutation-positive women: biological evidence of impaired genomic integrity after radiation exposure; 43 a large cohort analysis documenting cumulative radiation dose-dependent increases in breast cancer incidence; 44 and a modeling study suggesting that the percentage reductions in breast cancer mortality needed to offset radiation-associated risk would range from 45% to 51% with screening from ages 25 to 29 years. 45 Notably, that study assumed only two mammogram views per screening episode. Because of dose dependency, exacerbated risk would be expected in the subpopulation of women with large breasts or dense tissue requiring additional views, likely further decreasing the benefit-to-harm ratio.
Second is psychological harm from false-positive results, which occur at a cumulative 10-year rate of 61% with annual mammography. 40 These include negative psychosocial effects lasting up to 12–14 years, transient or persistent anxiety, worry affecting mood or activities, impaired mental functioning, and a modest increase in screening nonadherence.40,46,47 Suggesting possible increases in these harms with more intensive screening, false-positive results are somewhat more common with MRI than mammography, for example, at rates per 1000 screening rounds of 159.7 with MRI and 89.8 with mammography in one high-quality randomized trial cited by the ACR.9,12,25
Third is overdiagnosis: a portion of cancers identified by screening would not have progressed to life-threatening invasive cancer if left untreated.40,41,48 Estimated rates of mammography overdiagnosis vary widely depending on methodology, ranging from 11% to 22% in RCTs and 0% to 54% overall. 40 Regardless of the exact rate, the assumption that every early detection equals a life saved is questionable, as widespread use of screening mammography in the United States was accompanied by a 30% increase in breast cancer diagnosis but no change in the rate of metastasis. 48 In addition, despite population-level evidence of mammography-associated reductions in breast cancer mortality, 4 a recent systematic review and meta-analysis of cancer screening trials found no mammography-associated all-cause mortality benefit after a median 13-year follow-up, partly because competing causes of mortality (e.g., CVD), such as cancer, become increasingly common with older age. 49 The rate of overdiagnosis in a population that is higher-risk on the one hand, but more frequently screened on the other, is unknown.
Considered together with improvements in treatment that may diminish the clinical value of early detection,48,50 these findings cast doubt on the assumption that earlier detection will inevitably result in fewer deaths. Even the ACR’s assertion that decreased interval cancer rate (confirmed diagnosis of cancer after a negative screen) is “a surrogate for mortality reduction” 4 is in question until mortality rates are empirically measured, rather than simply assumed.
Finally, socioeconomic advantage may be positively associated with MRI use, as it was with HT use,19,20 especially given the cost differential between MRI and mammography. 51 If so, nonexperimental studies of intensive screening will likely be confounded by socioeconomic status. More seriously, the option for BRCA mutation-positive women to use MRI instead of mammography up to age 40 years, intended to reduce their radiation exposure, 4 has the potential to exacerbate existing socioeconomic disparities in breast cancer outcomes52,53 by disproportionately reducing cumulative mammography-associated radiation exposure in higher-income women.
Sociocultural factors underlying intensive screening
Guideline recommendations for both HT use and intensive screening date to the 1990s; yet only the former has been subjected to rigorous outcomes research. We should consider hard questions about the willingness to accept intensive screening based on such limited evidence for such a long period of time, especially when an evidence-based alternative exists in BRRM. First, the intensive screening protocol may reflect an unstated and untested assumption that women want to preserve their breasts at all costs, possibly due to mastectomy-associated stigma. 54 Second, given the revenue implications of up to 50 years of imaging and follow-up compared with a surgical strategy, the possibility of commercial influence on guideline development should not be overlooked, as the problem of vested interests in cancer screening recommendations has been noted elsewhere. 55 Recent analyses of U.S. Food and Drug Administration approvals indicate that commercial influence often takes the form of reliance on evidence lacking scientific rigor. 56
Finally, the intensive screening recommendations may be driven partly by what has been described as an “irrational exuberance for early detection” in U.S. culture and medicine. 57 Although controversial, this view is supported by cross-national evidence of higher patient participation rates and lower positive predictive value for mammography in the United States than in European countries.58,59 U.S. patients may overestimate the benefits of screening. 38 As the ACS 2003 guidelines noted, some mammography-related harms may occur because patients desire definitive investigation of even low-suspicion lesions. 11
Critical research needs
The all-important patient-centered question of what the balance of increased cancer-detection sensitivity and screening-related harms means for patient morbidity or mortality remains unaddressed. Rigorously designed outcomes studies of management protocols for high-risk women are needed. This concern is not new; it was raised by the ACS in 2003 11 and the USPSTF in 2019. 12 Well-controlled comparative studies using large databases could provide much, albeit not all, of this evidence in a reasonable time frame (Table 2).
Study designs a to address critical evidentiary gaps in breast screening protocols for high-risk women.

BRRM, bilateral risk-reducing mastectomy; DMIST, Digital Mammographic Imaging Screening Trial; MRI, magnetic resonance imaging; SES, socioeconomic status.
Except for Study D, all studies should measure as many of the following as possible: breast cancer incidence; breast cancer-related and all-cause mortality and morbidity; and, if feasible, psychological well-being and health-related quality of life.
Retrospective cohort studies could be conducted using electronic medical records or claims database sources for adequate statistical power.
In their modeling study of radiation-induced breast cancer incidence and mortality from digital mammography screening, Miglioretti et al. used mean radiation dosage information from the DMIST because exposure data is considered proprietary information. 42 An optimal study would directly measure radiation exposure.
An important step in the right direction, the 2024 comparison of mortality with MRI versus no-MRI surveillance identified a significant benefit in BRCA1 mutation (hazard ratio (HR) = 0.20, 95% confidence interval (CI) = 0.10–0.43), but only a nonsignificant trend toward benefit in BRCA2 mutation (HR = 0.87, 95% CI = 0.10–17.25). 32 Its methodological limitations—a sample consisting almost entirely (97%) of White women, small number of mortality events, and no examination of earlier-age screening—could be addressed with rigorous population-level database analysis.
Participation in RCTs, the most rigorous design, could be facilitated by transparent disclosure of screening benefits and harms. For example, the Harding Center for Risk Literacy summarizes the risks and benefits of mammography, based on a Cochrane review, into a graphic “Fact Box” that presents quantitative information about breast cancer death, any cancer death, “false alarms,” and unnecessary testing and surgeries. 60 For a trial of intensive screening for high-risk women, adding information about potential radiation-related cancer risks owing to earlier-age mammography screening would be appropriate. This approach is consistent with recent calls for greater transparency in communication about the benefits and harms of cancer screening.40,49,55
Unfortunately, plans for such studies may face tough challenges from those who believe strongly in the benefits of intensive screening despite the lack of supporting evidence. Such objections are remarkably common when accepted protocols are questioned27,61,62 and, indeed, were expressed regarding HT. 17 Even as some epidemiologists were appropriately asking questions about whether selection bias partly explained the association between HT use and reduced CVE incidence, 19 some physicians and researchers were arguing that the benefits of HT were so clear that a placebo-controlled RCT would violate ethical standards. 17
Ultimately, though, HT became just one more example in a sequence of events that has been repeated many times in epidemiological history: a treatment strategy once thought so beneficial that a placebo group is considered unethical is found to be harmful when finally tested using a rigorous research design.27,61,62 It is unknown whether this pattern will be repeated for the intensive screening protocol for high-risk women. It is past time to find out.
Conclusions: living at the intersection of the personal and the professional
Informed consent requires information. Evidence-based medicine requires evidence. Assumptions are not evidence. I have taught these vital lessons, learned time and again in epidemiological history, for years. How ironic that I experienced them firsthand when I discovered that more than a quarter-century after the NHGRI task force suggested earlier-age mammography screening for BRCA mutation-positive women and 16 years after the ACS recommended MRI for high-risk women, almost no evidence exists on the balance of benefits and harms from these strategies.
The intersectionality of my dual roles—both longstanding advocate for evidentiary quality in medical decision making and patient in need of high-quality evidence to inform my own decisions—was unwelcome but instructive. Ultimately, I chose BRRM based on my personal risk factors and, especially, my strong preference for evidence-based treatments. I am grateful for my decision and for the career that helped me to make it.
That same passion for evidence was notably exemplified in the WHI trial. By providing the first methodologically rigorous, large-sample evidence on the benefits and risks of HT for chronic disease prevention, the trial fundamentally altered preventive medical care after menopause. Just as postmenopausal women and the physicians who treated them deserved high-quality evidence in 1991, so do high-risk women and their physicians deserve it now.
