Abstract
Background:
Unexplained recurrent pregnancy loss (URPL) is a significant obstetric challenge affecting maternal health and well-being. Genetic factors, including mutations in the methylenetetrahydrofolate reductase (MTHFR) gene and elevated homocysteine levels, are increasingly recognized as contributors to URPL, though their precise roles remain complex. This study aimed to comprehensively explore these factors.
Objectives:
This study examines the links between MTHFR gene polymorphisms (C677T and A1298C), plasma homocysteine levels, and URPL. It also aims to create a predictive model for URPL based on these factors.
Design:
A case-control study with Vietnamese women who had at least one pregnancy between January 2017 and June 2020, recruited from Medlatec Hospital and Hanoi Medical University. Participants included URPL cases (n = 128) and controls (n = 126).
Methods:
Participants were selected based on specific criteria. Main analyses identified the optimal multivariable logistic regression model for predicting URPL using Bayesian Model Averaging, with the optimal model chosen based on the highest Area Under the Curve (AUC) and posterior probability. A nomogram for clinical risk prediction was developed based on parameters from the optimal model.
Results:
URPL cases exhibited significantly higher plasma homocysteine levels (11.73 ± 6.08 µmol/L) compared to controls (7.64 ± 1.78 µmol/L), correlating with increased URPL risk (OR: 1.64, 95% confidence interval (CI): 1.41–1.96; p < 0.001). The MTHFR C677T variant showed strong associations with URPL, particularly the CT genotype (OR: 6.07, 95% CI: 3.00–12.93; p < 0.001) and TT genotype (OR: 14.62, 95% CI: 2.85–114.77; p = 0.003). Similarly, the A1298C variant demonstrated elevated URPL risk with the AC genotype (OR: 2.73, 95% CI: 1.34–5.78; p = 0.007) and CC genotype (OR: 12.43, 95% CI: 3.17–64.22; p = 0.001).
Conclusion:
This study provides insights into the complex interplay of MTHFR gene mutations, elevated homocysteine levels, and URPL. Genetic testing and biomarker assessment may play a crucial role in customizing risk assessment and management strategies for women at risk of URPL.
Introduction
Pregnancy loss (PL) is a significant complication during pregnancy that impacts maternal health, imposes an economic burden, and affects the mental well-being of the entire family.1 –3 Recurrent pregnancy loss (RPL), defined as having two or more miscarriages, has a prevalence of 2%–5% in women,4 –6 but is only 1%–2% among those with three or more consecutive miscarriages within the first 20 weeks of pregnancy. 7
In 2015, an estimated 2.6 million stillbirths occurred globally, with a projected rate of 12 stillbirths per 1000 births in each country by 2030. 8 Research by Magnus indicates a notable increase in miscarriages among women after age 30, particularly after age 35. 9 Similarly, Wie et al.’s research shows a significant association between maternal age and the frequency of stillbirth complications. 10
Several studies have identified factors associated with miscarriage and stillbirth, including maternal age, genetic abnormalities, genital abnormalities, hormonal disorders morphological, biochemical, vascular, and immune factors.11,12 Despite this, approximately 50% of cases remain unexplained, classified as unexplained recurrent pregnancy loss (URPL).12 –14
Recently, studies have highlighted the role of MTHFR (methylenetetrahydrofolate reductase) gene polymorphisms in URPL patients. The two most extensively studied mutations are MTHFR C677T and A1298C.15 –18 These mutations result in a recessive genetic disorder causing MTHFR deficiency, which leads to elevated homocysteine (Hcy) levels in the blood. 19 Emphasizing the necessity for further research to elucidate the underlying biomolecular mechanisms and to develop effective diagnostic and therapeutic interventions.
Based on this evidence, our study aims to investigate the association between MTHFR C677T and A1298C mutations and plasma Hcy concentrations in URPL. This research seeks to provide a further understanding of the genetic factors contributing to URPL.
Materials and methods
Study design
This study is a prospective case-control study involving Vietnamese women with a history of at least one pregnancy. The research was conducted from January 2017 to June 2020, at Medlatec Hospital—No. 42, Nghia Dung, Ba Dinh, Hanoi, Vietnam, and the Center for Clinical Genetics and Genomics at Hanoi Medical University—No. 1, Ton That Tung, Dong Da, Hanoi, Vietnam. The article for this case-control study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Guidelines, 20 with adherence details provided in Supplemental File 1, which contains the STROBE Checklist.
Participant selection criteria
Participants were selected based on the following inclusion criteria: (1) URPL cases—females who had experienced at least two miscarriages and/or stillbirths; (2) control cases—females with no history of miscarriage or stillbirth and at least one normal birth. Eligible participants were women of childbearing age (18–45 years old), with a height above 150 cm and weighing over 40 kg. Exclusion criteria included: (1) positive antiphospholipid antibody syndrome; (2) uterine and/or ovarian abnormalities; (3) abnormal FSH and LH levels; (4) chromosomal abnormalities in self or spouse; (5) RH negative blood type; (6) abnormal semen parameters in spouses.
Sample size
Our study is based on the 50 events per variable criterion for sample size considerations in logistic regression analysis, which is optimal for achieving higher accuracy. 21 Accordingly, with five variables, the minimum sample size required for our study is 250. In line with this requirement, 128 subjects were enrolled in the study group and 126 in the control group, adhering to the study’s defined inclusion and exclusion criteria.
Statistical analysis
Descriptive statistics were performed using the CompareGroups package, employing Student’s t-test for comparing continuous variables between groups, and the Chi-square test for categorical variables. Fisher’s Exact test was utilized if more than 25% of the expected cell frequencies were below 5. 22 The Bayesian Model Averaging approach implemented with the “BMA” package identified the optimal model associated with URPL. 23 The “rms” package’s Nomogram method was employed to construct a probability prediction figure for URPL cases. 24 All analyses were conducted using R version 4.3.1 on the Windows 11 platform. 25
Results
This study analyzed participant characteristics, focusing on biochemical markers and genetic polymorphisms associated with URPL. The cohort included 254 participants: 126 controls and 128 URPL cases. We compared homocysteine and folate levels, along with the distribution of MTHFR C677T and A1298C alleles, between the groups to identify significant differences and potential risk factors for URPL. Detailed findings are presented below.
Table 1 presents the characteristics of the study participants, focusing on the statistically significant differences between the control group and the URPL cases.
Characteristics of the research object.

URPL, unexplained recurrent pregnancy loss.
t-Test was used for quantitative variables and Chi-square was used for categorical variables.
Notably, homocysteine levels were significantly higher in the URPL group (11.73 ± 6.08 µmol/L) compared to the control group (7.64 ± 1.78 µmol/L), with a p-value of
Regarding genetic factors, significant differences were observed in the distribution of MTHFR C677T and A1298C alleles. For the C677T allele, the CC genotype was more prevalent in the control group (73.02%) than in the URPL group (43.75%), while the CT and TT genotypes were more common in the URPL group (46.88% and 9.38%, respectively) compared to the control group (25.40% and 1.59%, respectively), with a p-value of <0.001. Similarly, for the A1298C allele, the AA genotype was more frequent in the control group (66.67%) than in the URPL group (44.53%). Conversely, the AC and CC genotypes were more prevalent in the URPL group (42.97% and 12.50%, respectively) compared to the control group (30.95% and 2.38%, respectively), also with a p-value of <0.001.
Table 2 displays the outcomes of a multivariable logistic regression model designed to predict URPL, utilizing homocysteine levels and MTHFR gene polymorphisms (C677T and A1298C alleles) as predictive factors. This analysis, based on 254 observations, demonstrates a robust discriminatory ability with an AUC of 0.862, indicating excellent accuracy in distinguishing individuals at varying risks of URPL.
Multivariable logistic regression model predicts consecutive URPL.
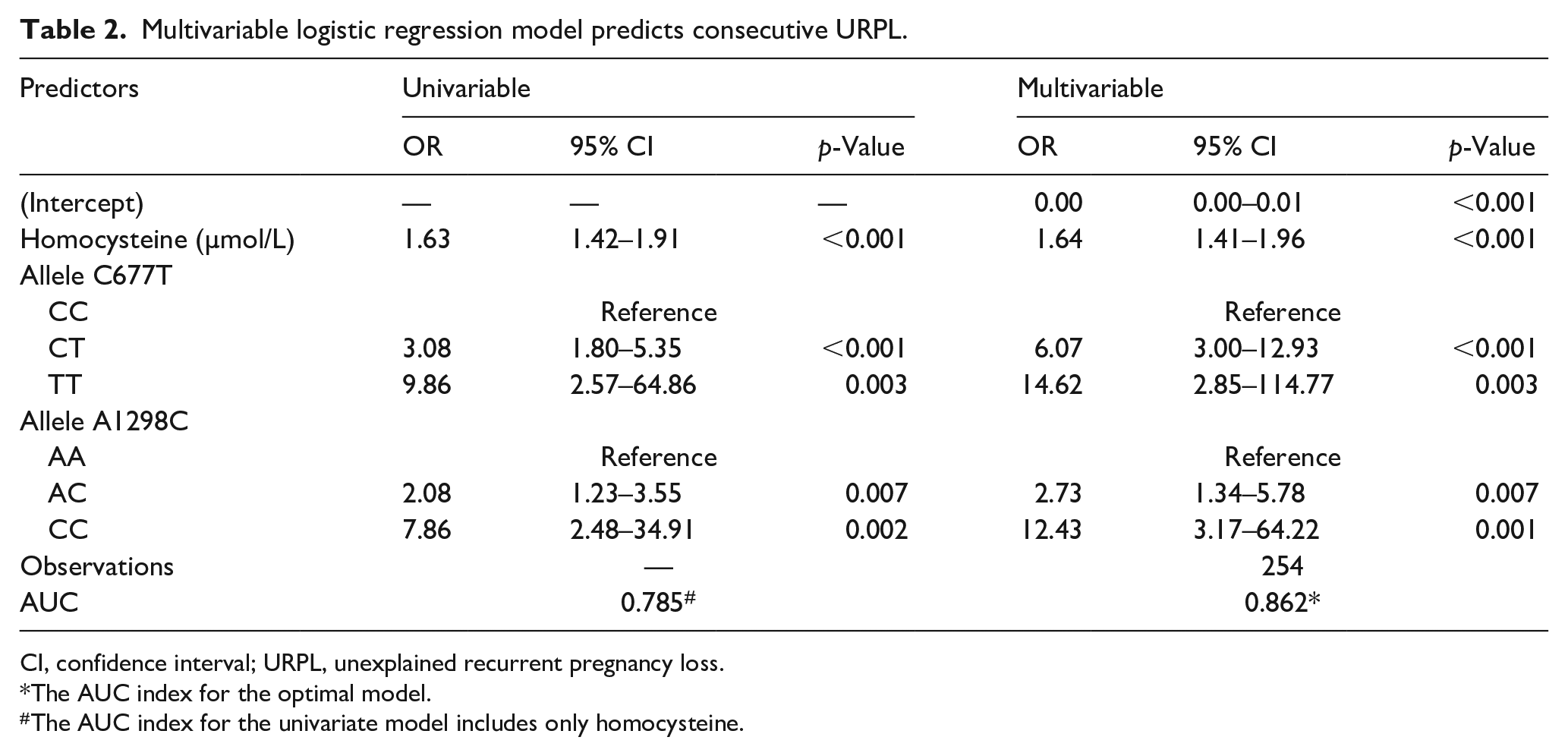
CI, confidence interval; URPL, unexplained recurrent pregnancy loss.
The AUC index for the optimal model.
The AUC index for the univariate model includes only homocysteine.
Elevated homocysteine levels independently increase the odds of URPL in both univariable (OR = 1.63, 95% confidence interval (CI): 1.42–1.91, p < 0.001) and multivariable models (OR = 1.64, 95% CI: 1.41–1.96, p < 0.001), highlighting their significant association with heightened URPL risk.
Regarding the C677T polymorphism, the CT genotype significantly raises the odds of URPL in both univariable (OR = 3.08, 95% CI: 1.80–5.35, p < 0.001) and multivariable models (OR = 6.07, 95% CI: 3.00–12.93, p < 0.001), underscoring its substantial impact on URPL risk. Similarly, the TT genotype shows a significant association with URPL in both models (univariable: OR = 9.86, 95% CI: 2.57 – 64.86, p = 0.003; multivariable: OR = 14.62, 95% CI: 2.85–114.77, p = 0.003), indicating a dose-dependent effect on URPL susceptibility.
Regarding the A1298C polymorphism, the AC genotype demonstrates increased odds of URPL in both univariable (OR = 2.08, 95% CI: 1.23–3.55, p = 0.007) and multivariable models (OR = 2.73, 95% CI: 1.34–5.78, p = 0.007). Furthermore, the CC genotype exhibits a strong association with URPL in both models (univariable: OR = 7.86, 95% CI: 2.48–34.91, p = 0.002; multivariable: OR = 12.43, 95% CI: 3.17–64.22, p = 0.001), emphasizing its substantial impact on URPL risk, particularly the homozygous CC genotype.
Figure 1 illustrates the interpretation of the multivariable logistic regression model presented in Table 2. The optimized model includes variables such as Hcy levels and the presence of C677T and A1298C alleles, which are used to predict URPL status.

Nomogram chart predicts the risk of URPL for women.
For example, a participant exhibiting the heterozygous C677T allele (CT form) accrues a score of 7 points, and the heterozygous A1298C allele (AC form) adds 3 points. Additionally, a measured plasma Hcy concentration of 10.0 µmol/L contributes 20 points.
Thus, the cumulative score for this participant totals 30 points, reflecting a significant combination of genetic and biochemical factors. Applying this scoring system places the participant in a category where over 80% of individuals are classified as at elevated risk for URPL. This observation underscores the potential importance of composite genetic and biochemical markers in understanding and predicting susceptibility to URPL.
Discussion
URPL represents a common occurrence in pregnancies, often stemming from diverse factors, including genetic mutations that exert significant impacts on maternal well-being and long-term psychological health. This case-control study, encompassing 254 participants (128 cases and 126 controls), aimed to investigate the correlation between allele polymorphisms C677T and A1298C, Hcy levels, maternal age, and URPL. The Bayesian Model Averaging method was utilized to discern the optimal model integrating Hcy levels, and variations in C677T and A1298C alleles, with posterior probability and an AUC of 86.2%. In particular, the nomogram prediction model was constructed based on the optimal model to easily determine the risk of URPL in women, making it clinically applicable.
Di Simone’s study explores the impact of Hcy and folic acid on trophoblast cells and their roles in miscarriage. Elevated Hcy levels induce trophoblast apoptosis, as indicated by increased M30 and Terminal deoxynucleotidyl transferase-mediated dUDP nick-end labeling (TUNEL) staining, DNA fragmentation, and cytochrome c release. Administration of folic acid prior to Hcy exposure significantly mitigates these effects by reducing cell death, DNA fragmentation, and restoring hCG secretion, which is crucial for pregnancy maintenance. 26 Our findings corroborate this, demonstrating that elevated Hcy levels independently increase the risk of URPL in both univariate (OR = 1.63, 95% CI: 1.42–1.91, p < 0.001) and multivariate models (OR = 1.64, 95% CI: 1.41–1.96, p < 0.001). However, no significant association between folate levels and URPL was found, potentially due to folic acid supplementation by participants prior to the study.
The MTHFR gene plays a crucial role in synthesizing the MTHFR enzyme, responsible for remethylating Hcy to methionine. Genetic variations in the MTHFR gene can lead to reduced Hcy degradation, resulting in elevated Hcy levels, which can be detrimental to cells and overall health. This elevation, especially when linked to thrombus formation can contribute to reproductive complications such as miscarriages or stillbirths as discussed earlier. One of the most prevalent genetic variations in the MTHFR gene is position 677C→T. Our multivariable regression model further highlighted the significance of genotypic abnormalities in the C677T allele, showcasing a notable association with an elevated rate of URPL. Specifically, both the CT heterozygous genotype and the homozygous TT genotype were associated with a substantially heightened occurrence of URPL, with rates 6.0 and 14.6 times higher, respectively.
The results of meta-analysis conducted by Chen et al. on the association between MTHFR C677T and RPL revealed a statistically significant connection between the MTHFR C677T and RPL (TT and CT versus CC; OR: 2.36; 95% CI: 1.92–2.90). 15 However, our study’s findings differ notably from Chen’s in both univariable models (CT versus CC; OR 3.08; 95% CI: 1.80–4.35; p < 0.001 and TT versus CC; OR 9.86; 95% CI: 2.57–64.86; p = 0.003) and multivariable model (CT versus CC; OR 6.07; 95% CI: 3.00–12.93; p < 0.001 and TT versus CC; OR 14.62; 95% CI: 2.85–114.77; p = 0.003). Interestingly, these results also differ from the study conducted by Yang et al. reported an association between the MTHFR C677T polymorphism with RPL (TT and CT versus CC; OR: 2.36; 95% CI: 1.92–2.90). 16 Additionally, the study by Wang et al. demonstrated a statistically significant association between the MTHFR C677T polymorphism and a specific form of URPL (TT versus CC; OR 3.06; 95% CI: 2.56–3.66 and CT versus CC; OR 1.59; 95% CI: 1.40–1.80). 14 These discrepancies highlight the complexity and potential variations in the relationship between the MTHFR C677T polymorphism and RPL or URPL across different studies.
The study by Babker et al. on Sudanese women found no significant association between the MTHFR C677T variant and recurrent spontaneous abortion (p = 0.091). 27 In contrast, our study demonstrated a strong association, with odds ratios of 6.07 for the CT genotype and 14.62 for the TT genotype (p < 0.001 and 0.003, respectively). Additionally, we observed elevated homocysteine levels in the URPL group (p < 0.001), suggesting a link between high homocysteine and URPL. Similarly, Nguyen Ngoc et al.’s study on Vietnamese women identified a significant association between the MTHFR C677T polymorphism and URPL, with a 9-fold increased risk linked to the 677CT-1298AC genotype and a 10-fold increase with multiple loci variations (p < 0.05). 28 Unlike the Sudanese study, which found no significant connection between the MTHFR C677T, FII G20210A, and FVL G1691A variants and RPL, 29 our study emphasizes the relevance of MTHFR variants and homocysteine levels in URPL risk. These findings highlight the importance of biochemical factors, especially homocysteine, in predicting URPL. Further research is needed to investigate these factors across different populations.
In addition, the MTHRF polymorphism 1298A→C leads to a reduction of 30%–60% in the normal function of the MTHRF enzyme. Our study’s multivariable regression model indicated that genotype abnormalities of the A1298C allele were also significantly associated with a heightened incidence of URPL. Specifically, the AC heterozygous genotype and the CC homozygous genotype were associated with the occurrence of URPL 2.7-fold and 12.4-fold higher, respectively. The findings from Wang et al.’s meta-analysis in 2021 revealed a significant connection between the MTHFR A1298C polymorphism and URPL (CC versus AA; OR 1.53; 95% CI: 1.22–1.91). 14
However, this observed association contrasts markedly with our study’s outcomes, as evidenced by the distinct OR calculated from both univariable and multivariable models, measuring 7.68 (95% CI: 2.48–34.91; p = 0.002) and 12.43 (95% CI: 3.17–64.22; p = 0.001), respectively. Our results stand in clear discrepancy with those of Chen et al., and Du et al. which both indicated the absence of a relationship between the A1298C polymorphism and RPL.15,17
Our study findings indicate a statistically significant association between MTHFR polymorphisms in both C677T and A1298C and URPL, aligning with the conclusions drawn by (Wang, Lin and Wang, 2021). 14 In contrast, certain other investigations conducted on RPL patients exhibit a similar trend concerning the C677T polymorphism but diverge from our observations in the case of A1298C.15,17
Based on the findings from our study, we propose the integration of genetic testing for MTHFR polymorphisms and the measurement of homocysteine levels into clinical practice to evaluate the risk of URPL in women with a history of unexplained PL. Furthermore, our prediction tool has the potential to serve as an additional resource for risk assessment and may support the identification of prospective treatment strategies aimed at improving pregnancy outcomes in patients with unexplained PL.
Limitations
This study has several limitations. First, while the sample size was adequate for detecting key associations, it may not be large enough across the two research sites in Hanoi, Vietnam, to generalize the findings to all populations and ethnic groups. Second, our study is limited by its focus on the C677T and A1298C alleles, which have been proven to be associated with URPL, while many other potential alleles were not considered. Finally, although we utilized multivariate logistic regression and predictive modeling, further studies are needed to confirm our results and explore the role of additional factors in URPL risk.
Future longitudinal studies with larger and more diverse populations are needed to validate these findings, explore the mechanisms underlying the observed associations, and incorporate additional genetic markers to investigate gene-environment interactions for a more comprehensive understanding of URPL.
Conclusion
This study explored the relationship between MTHFR gene polymorphisms (C677T and A1298C), plasma homocysteine levels, and URPL in Vietnamese women. Our findings indicate significant associations between these genetic factors and URPL risk. Specifically, the CT and TT genotypes of the C677T allele, along with the AC and CC genotypes of the A1298C allele, showed elevated odds ratios for URPL, highlighting their role as genetic risk factors.
The nomogram developed from our optimized predictive model integrates homocysteine levels and MTHFR gene polymorphisms, offering clinicians a practical tool for assessing URPL risk based on patients’ genetic and biochemical profiles. This approach supports personalized risk assessment and management strategies in reproductive health.
Supplemental Material
sj-doc-1-whe-10.1177_17455057241296608 – Supplemental material for Exploring the sophistications of unexplained recurrent pregnancy loss: A case-control study
Supplemental material, sj-doc-1-whe-10.1177_17455057241296608 for Exploring the sophistications of unexplained recurrent pregnancy loss: A case-control study by Trinh Thi Que, Nguyen Van The, Vu Thi Thu Trang, Le Van Truong, Ta Thanh Van and Doan Thi Kim Phuong in Women’s Health
Footnotes
Acknowledgements
Sincere thanks to the leadership of Medlatec Hospital for creating favorable conditions for the completion of this research.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
