Abstract
Background:
Intimate partner violence (IPV) is a prevalent form of violence against women that encompasses physical, sexual, and emotional abuse, as well as controlling behaviors by intimate partners, and predisposes the victims to multiple diseases.
Objective:
This systematic review aims to identify epigenetic marks associated with IPV and the resultant stress experienced by victims.
Design:
This study is a systematic review conducted according to the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) 2020 guidelines. The review includes a comprehensive search and analysis of relevant literature to identify epigenetic changes associated with IPV.
Data Sources and Methods:
A systematic search was conducted across four databases: PubMed, Scopus, Web of Science, and ProQuest, using keywords related to IPV and epigenetics. The inclusion criteria were studies published in scientific journals with an experimental approach, focused on female survivors of gender-based violence, and providing information on epigenetic changes. The review included studies published up to June 15, 2024, with no time limits imposed, focusing on female victims of IPV. The inclusion criteria were studies published in scientific journals with an experimental approach, focused on female survivors of gender-based violence, and providing information on epigenetic changes.
Results:
The results revealed that epigenetic changes associated with IPV predominantly affect genes related to the glucocorticoid receptor, insulin-like growth factors, BDNF, and CPLX genes. These observations suggest that IPV is linked to significant epigenetic modifications in both victims and their offspring.
Conclusion:
It is concluded that IPV is associated with epigenetic changes both in the woman and in her offspring. These findings underscore the importance of understanding the biological embedding of IPV through epigenetic research to better address the long-term health consequences for women. However, more studies are necessary to validate these results.
Plain language summary
Background:
Intimate partner violence (IPV) involves physical, sexual, and emotional abuse, as well as controlling behaviors by partners, and it can lead to various health problems for the victims.
Objective:
This review aims to find out how IPV affects genes through changes called epigenetic marks, which can be caused by the stress experienced by victims.
Design:
This study is a thorough review following PRISMA 2020 guidelines, which includes searching and analyzing past studies to find epigenetic changes linked to IPV.
Data Sources and Methods:
We searched four major databases (PubMed, Scopus, Web of Science, and ProQuest) using terms related to IPV and gene changes. We looked for studies that focused on female survivors of IPV and provided information on gene changes. The studies included were published up to June 15, 2024.
Results:
We found that IPV is linked to changes in genes that are important for stress response and brain function, such as the glucocorticoid receptor, insulin-like growth factors, BDNF, and CPLX genes. These changes were seen in both the victims and their children.
Conclusion:
IPV causes changes in genes in both women and their children, which can have long-term health effects. Understanding these changes is important for addressing the health consequences of IPV, but more research is needed to confirm these findings.
Introduction
Intimate partner violence (IPV) is one of the most prevalent forms of violence against women, encompassing physical, sexual, and emotional abuse, as well as controlling behaviors by intimate partners. 1 The United Nations defines IPV as “a pattern of behavior in any relationship that is used to gain or maintain power and control over an intimate partner.” 2 This pattern can occur in various types of relationships, including married, cohabiting, or dating couples, and it affects individuals across all socioeconomic backgrounds, educational levels, ages, ethnicities, and genders. 2 The most recent World Health Organization estimates show that 30% of women globally have been subjected to physical or sexual, or both, violence by an intimate partner or sexual violence by a non-partner. Over a quarter (27%) of ever-partnered women (globally) over 15 years of age have been subjected to physical or sexual IPV, but many regions and countries have prevalence much higher than this. 3 Furthermore, up to 38% of all murders of women globally are committed by their intimate partners. 4 The United Nations Office on Drugs and Crime estimates that women constitute approximately 53% of all victims of killings in the home and 66% of all victims of intimate partner killings. 5
There are several risk factors for intimate IPV, including lower levels of education, exposure to abuse/violence during childhood, witnessing family violence, antisocial personality disorder, male controlling behaviors toward a partner, community norms that attribute higher status to men, limited opportunities for women to access paid employment, and the use of alcohol. In a study conducted in Spain by Ruiz-Pérez et al., 6 almost 25% of women had experienced some form of IPV, with a lifetime prevalence rate of 16% for physical IPV and 21.1% for psychological IPV. These prevalence rates are consistent with findings from other global data, where 30% of women have been subjected to physical and/or sexual violence by an intimate partner or non-partner sexual violence or both.7–9
Women who experience IPV have been shown to have increased rates of mental health conditions such as depression, anxiety, post-traumatic stress disorder (PTSD), sleeping and eating disorders, compared to women not exposed to IPV.10–13 IPV is also associated with physical conditions, including fibromyalgia, 14 hypertension, asthma, 15 and type 2 diabetes. 16 Additionally, women who have experienced IPV report a higher incidence of somatic symptoms compared to those who have not suffered abuse. 17 Victims of IPV also tend to consume more antidepressants and anxiolytics, 18 smoke a greater number of cigarettes, 19 and abuse other types of drugs. 4 Despite the benefits of social support, women who experience IPV often resort to strategies such as resistance or appeasement to cope with the abuse.20,21 This behavior may be influenced by emotions such as shame or guilt, which can be decisive in whether they seek help.
One of the more likely pathways leading to the harmful health effects of IPV is chronic stress to which victims are subjected. A common misconception is that exposure to stress ceases once women exit an abusive relationship. In reality, this stress often persists long after the relationship ends and may even worsen. Women may continue to face intimidation or violence from their former abusers, experience stress from single parenting, or endure the pressure of testifying in legal proceedings.22,23 It is also important to consider the intensity and duration of the violence, the context in which it occurs, and whether the woman has suffered abuse earlier in life (e.g., child abuse) or is facing other stressors. 23
Epigenetics refers to changes in gene activity and expression as well as stable, long-term alterations in the DNA that are not necessarily heritable. Epigenetic changes underlie biological embedding, where life experiences, especially early experiences, can alter biological processes with long-term effects on health and behavior. 24 It has been described that stressors can interact with an individual’s genome, producing epigenetic changes in DNA. 25 Among the types of epigenetic modifications is the DNA methylation. During this process, methyl groups are added to the DNA sequence, which affects transcriptional activity, repressing transcription by binding transcription factors or promoting the binding of other transcriptional repressors.26,27
These epigenetic changes can have unfavorable repercussions on the health of women victims of IPV and even on their offspring. In recent years, massive sequencing techniques have been developed and optimized at great speed, allowing the analysis of a series of genes of interest (gene panels) or the complete genome of a person, with great coverage and depth and reading, allowing the detection of variants found at very low frequencies. Massive sequencing techniques applied to the analysis of the variants in women subjected to gender-based violence can offer a reliable result of the impact and severity of this sustained stress over time, as well as a great speed in its analysis. These studies are crucial for comprehending the profound impact on women victims of IPV, including the significant consequences it has on their health and on their offspring. This understanding is vital for implementing legal and health measures, and for exploring the potential reversibility of these epigenetic marks once IPV ceases, and with the application of psychotherapy to help overcome post-traumatic stress. Because of that, the objectives of this study are to conduct a systematic review of the literature to identify the presence of epigenetic marks associated with IPV.
Methods
Literature search
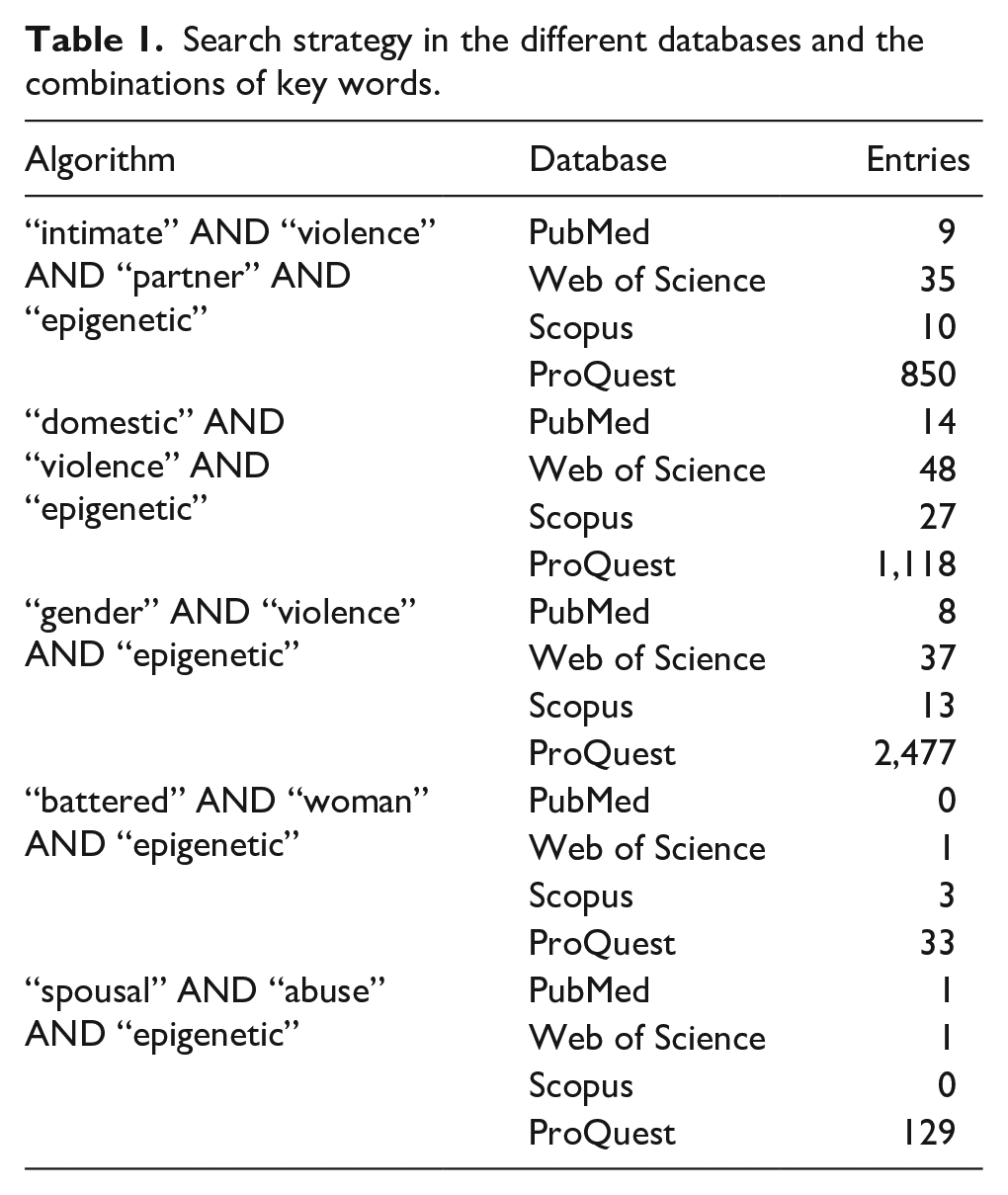
A systematic review of the PubMed, Scopus, Web of Science, and ProQuest databases was carried out, according to Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines. The methodology used to track the articles and the inclusion criteria are detailed below. The search strategy followed was combinations of the keywords “intimate violence partner” OR “domestic violence” OR “gender-based violence” AND “epigenetic” specified in Table 1. In the ProQuest database, newspapers, reports and working documents were discard.
Search strategy in the different databases and the combinations of key words.
Inclusion criteria
The systematic review covered all scientific articles published in journals up to June 15, 2024. Considering the recent development of this research domain, we did not insert time limits. The inclusion criteria for this review were: (i) records published with an experimental approach in scientific journals; (ii) the studies were not carried out on animals; (iii) the experiments were aimed at female survivors of gender-based violence; and (iv) the records provide information on epigenetic changes associated with this gender-based violence. Gender-based violence was defined as any physical, sexual, or psychological abuse directed against a woman by her current or former partner. Neither restrictions were imposed on the language of publication or on the study design, nor on the geographical location, with the aim of including as many relevant studies as possible.
Exclusion criteria
The exclusion criteria for this systematic review were: (i) reviews, systematic reviews, letters, or bioinformatical analysis; (ii) studies were carried out in children; (iii) to study other pathologies associated with gender violence; (iv) studies focused on genetics but not on epigenetics resulting from gender violence; and (v) studies focused exclusively on analyzing the environment or social cause of this gender violence.
Data extraction
For inclusion of the articles in the review, the key characteristics of the study were identified based on the title, abstract, and topic discussed. The included articles were reviewed by two independent reviewers (CGM and CH). When a conflict was identified, the articles were reviewed again, and the discrepancy was resolved following the key points established for the systematic review. Also, book chapters and conference paper from ProQuest and SCOPUS were reviewed as gray literature, to avoid as much as possible search bias. All the references that passed the filter were imported into Mendeley and then transferred to a Microsoft Excel document, where the following data is compiled: authors, year of publication, journal, study population, study sample, and epigenetic alteration.
Statistical analysis
In this systematic review, a meta-analysis could not be conducted due to the limited number of studies available on this topic. Additionally, the measurement of IPV varied significantly, as did the methods used to assess the stress resulting from this violence and other mental health outcomes, leading to low concordance between the studies. The initial goal was to compile all epigenetic modifications in women exposed to IPV and assess the heterogeneity of these changes across studies using the I2 statistical method, to subsequently identify potential sources of variability in the epigenetic modifications. Given these limitations, we did not attempt to calculate measures of association. Instead, study-specific variables are presented in Table 2 and discussed in “Results” and “Discussion” sections of the manuscript.
Final articles that passed all the filters and their main characteristics: measurement of gender-based violence, sample, sequencing method, and epigenetic change detected.

CAS: Composite Abuse Scale; CTS-2: Conflict Tactics Scale-2; HADS: Hospital Anxiety and Depression Scale; IPV: intimate partner violence; PTSD: post-traumatic stress disorder; PCL-S: post-traumatic symptoms–short version; PCL-C: post-traumatic stress disorder checklist civilian; BPSAQ: Brief Physical and Sexual Abuse Questionnaire; TLEQ: Traumatic Life Events Questionnaire; SD: standard deviation.
The quality and susceptibility to bias of all studies included in the review were evaluated using the 27-item PRISMA 2020 checklist.
Results
Literature search
The PubMed search identified a total of 32 articles, another 122 were found on Web of Science, Scopus returned 53 results, and 4,607 were found using ProQuest. The total number of articles identified was 4,814. Of these, 4,695 articles were duplicated or irrelevant according to the selection criteria. The number of articles evaluated in the full-text selection stage was 119. The greatest cause of exclusion was “seeing epigenetic changes in the offspring” (32.74%), followed by “reviews” (30.1%), “social causes or other pathologies” (22.12%) and, last, non-specific causes (15.04%). Finally, a total of six articles met all the inclusion requirements and were thoroughly reviewed (Table 2). The most important aspects of the following are highlighted below, describing the methodology used, as well as the epigenetic changes resulting from IPV stress (Figure 1).

Literature review workflow following PRISMA guidelines.
Measurement of gender-based violence and mental disorders
Measuring gender-based violence and mental health outcomes poses the challenge of accurately capturing the extent and impact of violence, as well as reliably assessing associated mental health outcomes, given the complexity and variability of victims’ experiences. Additionally, the studies included in the review employed different instruments and scales to measure both parameters.
Regarding the measurement of violence, most studies measure IPV in mothers28–31 or women. 32 Only the study by Serpeloni et al. 33 measures violence in the form of domestic and community violence in triads of grandmothers/mothers/children. All articles except Radtke et al. 31 analyze the mental health of the women included in the study. The most common mental health outcome measured is PTSD,28,30,32,33 followed by anxiety and depression.28,29,33
As for the methodologies used to measure violence, Moser et al. 28 measured the severity of physical partner violence to the mother and herself in the context of adult romantic relationships using the Conflict Tactics Scale-2 (CTS-2), short version. 34 Wadji et al. 29 also employed the CTS-2 to assess IPV in mothers. Cordero et al. 30 assessed IPV through clinical interviews, and Radtke et al. 31 focused on the impact of IPV using the Composite Abuse Scale (CAS) 35 conducted by experienced clinical psychologists. Radtke et al., 29 CAS test was conducted three times separately, focusing on the periods before, during, and after the pregnancy with the particular child whose blood was analyzed in this study. For the periods before and after pregnancy, participants were asked to report acts of domestic violence whenever these happened before or after pregnancy with the relevant child. On the other hand, Piccinini et al. 32 analyzed the violence exerted against women through questionnaires developed by them and complemented with gynecological-forensic examination in cases where had been sexual abuse. Finally, Serpeloni et al. 33 analyzed domestic and community violence in triads of grandmothers/mothers/children through the survey “Things I Have Seen and Heard.” 36
Mental health outcomes were also measured differently in each study, with PTSD being the common factor in most studies. Serpeloni et al. 33 assessed the severity of PTSD symptoms using the University of California, Los Angeles PTSD Index to Diagnostic and Statistical Manual of Mental Disorders (DSM)-IV 37 in mothers and grandmothers. Depression symptom severity were evaluated with the Patient Health Questionnaire (PHQ-9) 38 and anxiety symptom severity with the Generalized Anxiety Disorder (GAD-7). 39 Moser et al. 28 quantified stress resulting from gender-based violence using various psychometric assessments: The Clinician Administered PTSD Scale 40 to assess lifetime PTSD and the Checklist of post-traumatic symptoms–short version (PCL-S) 41 for current PTSD symptoms. Physical and sexual abuse during the mothers’ childhood through the Brief Physical and Sexual Abuse Questionnaire (BPSAQ) 42 and supplemented by the Traumatic Life Events Questionnaire (TLEQ). 43 Anxiety was measured with the anxiety subscale of the revised version of the Symptom Checklist 90, 44 and maternal depressive symptoms were assessed via the Beck Depression Inventory-II. 45 Cordero et al. 30 evaluated lifetime maternal IPV-PTSD with the CAPS (Clinician Administered Post-traumatic stress Disorder Scale) questionnaire, physical and sexual abuse during the mothers’ childhood BPSAQ 42 and TLEQ. 43 Wadji et al. 29 employed the Hospital Anxiety and Depression Scale 46 to consider mothers’ anxiety and depression symptoms and cortisol saliva level to measure the stress. On the other hand, Piccinini et al. 32 examined the prevalence of PTSD based on the symptoms collected in the DSM-V and the Post-Traumatic Stress Disorder Checklist Civilian (PCL-C) scale.
Sample used and sequencing method
With regard to the biological material used, the studies by Serpeloni et al., 33 Wadji et al., 29 and from Moser et al. 28 perform DNA extraction from a saliva sample; Radke et al., 31 Cordero et al., 30 and Piccinini et al. 32 take maternal whole blood.
While in Serpeloni’s et al. 33 study, DNA methylation profiling is performed using the Infinium Human Methylation 450 BeadChip kit from the Queen Mary University of London Genome Centre according to standard protocols, the studies by Moser et al. 28 and Cordero et al. 30 performed a first PCR for the amplification of the saliva DNA sample with the HotStarTaq Master Mix kit (Qiagen, San Diego, CA, USA) to later measure the degree of methylation of the CpG islands with the Pyro Q-CpG software (Biotage AB, Uppsala, Sweden). Wadji et al. 29 also employed saliva to isolate the genomic DNA and performed bisulfite conversion using EpiTect Fast 96 DNA Bisulfite Kit from Qiagen. Then, they generated amplicons from the 1F genomic region of interest: NR3C1 gene.
Radke et al. 31 also previously amplified the DNA from the blood sample with FastStart Taq DNA Polymerase (Roche, Mannheim, Germany); the products were purified and cloned. Positive clones were identified and sequenced and analyzed using Sequencher (version 4.2.2.; Gene Codes Corporation, Ann Arbor, MI, USA). Piccinini et al. 32 made a panel of interesting genes for post-traumatic stress and analyzed the methylation in the CpG sites by means of PCR.
Epigenetic changes associated with stress resulting from IPV
Serpeloni et al. 33 noted that exposure to domestic violence is associated with decreased subunit methylation of CPLX, the chaperone of caseinolytic mitochondrial matrix peptidase, as well as of BDNF, the maternal brain-derived neurotrophic factor, but not in the grandmother. Furthermore, there was no correlation between DNA methylation and the mental health variables analyzed (PTSD and depression) in any of the three generations. Moser et al. 28 saw a correlation between the degree of methylation of exon 4 of the promoter region of the BDNF gene and maternal anxiety, correlating the severity of anxiety with the degree of methylation. Maternal exposure to domestic violence as a child was also significantly correlated with the degree of BDNF methylation. In contrast, they saw no correlation between PTSD severity with BDNF methylation. In relation to IPV, Piccinini et al. 32 observed methylation in the BDNF promoter in women who were exposed to IPV compared to those who did not.
Radtke et al.’s 31 and Wadji et al.’s 29 studies detected methylation in the glucocorticoid receptor promoter NR3C1, the first study in both mothers and sons, and the second only in the mothers’ cohort. Also, Radtke et al. 31 reported that in children the presence of methylation was significantly associated with the mother’s exposure to violence from her partner during pregnancy. In contrast, there was no association in offspring’s methylation status when the violence was experienced before or after pregnancy. Cordero et al. 30 also analyzed the methylation status of the NR3C1 gene in mothers and children. Mothers who experienced violence and post-traumatic stress were significantly correlated with NR3C1 methylation, unlike those who did not. The same effect was seen in the children of mothers who had IPV-PTSD. Finally, Piccinini et al. 32 found a correlation in the methylation of the IGF2 promoter in women exposed to IPV with respect to those who did not suffer from it.
Discussion
The works included in this systematic review are carried out in different areas of the world (Brazil, Switzerland, Germany, and Italy) and present diverse social contexts, with one upper-middle-income country (UMIC) (Brazil) and three high-income countries (HIC) (Switzerland, Germany, and Italy). Among them, the article by Wadji et al., 29 conducted in Switzerland, studies IPV in mothers from Cameroon, a lower-middle income country (LMIC) and the study by Radke et al., 31 conducted in Germany, includes women from different countries, including HICs (Czechoslovakia, Germany, Hungary, Poland, Russia, and Taiwan), UMICs (Iraq, Serbia and Montenegro, and Kosovo) and one LMIC (Iran) 47 .
All studies have in common the analysis of the impact of gender-based violence on women. Gender-based violence is presented throughout the studies in different ways (such as domestic violence, community violence, IPV. . .), having in common that there is a continuous violence over time and not just a series of repeated attacks more or less frequently, which generates a final situation of chronic stress on the women.
It is likely that the results differ between studies conducted in countries with very high levels of domestic and community violence and those with more moderate, government-regulated domestic/community violence. Examples of the first scenario are the study by Wadji et al. 29 on women living in Cameroon, a country where 43.2% of all women have experienced domestic violence, or the study by Serpeloni et al., 33 in Brazil, where 43% of women aged 16 or older have experienced physical, psychological, and/or sexual violence by an intimate partner, 48 and which has one of the highest femicide rates in Latin America (1.7 cases per 100,000 women). 49 This contrasts with studies conducted in countries like Switzerland, where 21%–29% of violence against women occurs in the domestic sphere, 50 where it is suspected that the levels of stress resulting from different forms of gender-based violence are relatively lower.
Similarly, it would also be interesting to analyze the resilience of women to gender-based violence in each country based on their political, economic, historical, and religious contexts. A recent study developed in the European Union showed that countries higher on human development index show a tendency to have a greater proportion of resilient women than the average country, 51 suggesting that a combination of individual and contextual policy interventions helps women cope. This also hints that in countries where there is more community violence and less intervention policies, women suffer greater stress related to different forms of gender-based violence.
All the articles included in the final review evaluate gender-based violence in a quantitative way. This quantification is important, since the stress to which these women are subjected not only is associated with epigenetic marks that have an impact on their health but also parameters such as intensity and frequency are of great influence. The study by Moser et al. 28 and Wadji et al. 29 uses the CTS-2 to measure gender-based violence, with Moser’s research focusing on women in Switzerland and Wadji’s on women in Cameroon. The CTS-2’s advantages for measuring IPV include its assessment of both partners’ behaviors and its comprehensive evaluation of five relationship dimensions: psychological, physical, and sexual violence, potential consequences of conflicts, and conflict resolution strategies. Radtke et al., 31 employed the CAS, a validated tool that measures the degree of IPV experienced by an individual across four dimensions: severe combined abuse, physical abuse, emotional abuse, and harassment. By capturing both the frequency and severity of abuse, the CAS provides quantitative data and qualitative insights into the nature of the violence experienced, aiding in understanding the intensity and impact of IPV. Contrary, Serpeloni et al., 33 measure exposure to community and domestic violence, using the survey “Things I Have Seen and Heard.” Although typically used for children, the survey was modified for an adult version in this study. The survey encompasses a wide range of violent and traumatic experiences, including witnessing violence, direct victimization, and hearing about violence, but making it less specific to IPV than other forms of measurement.
Cordero et al. 30 and Piccinini et al. 32 both used clinical interviews to select mothers suffering from IPV for their study, but they did not provide details on the interviews and selection process. This lack of detail complicates the evaluation of the adequacy of these techniques for measuring IPV in women and the analysis of the association between IPV and stress.
Regarding the measurement of PTSD, the CAPS method is employed by several studies in this review28,30 and is considered the gold standard for PTSD diagnosis. CAPS covers all PTSD symptoms in detail, allowing for a comprehensive assessment of the frequency and intensity of each symptom, leading to a nuanced understanding of the individual’s experience. Both studies also used the BPSAQ and TLEQ, established tools with validated reliability and validity in assessing IPV and trauma, ensuring that the data collected is credible and useful for research or clinical purposes. Piccinini et al. 32 used the PCL-C to assess the presence and severity of PTSD symptoms. The PCL-C has demonstrated good reliability and validity in various populations, making it a trusted tool for assessing PTSD symptoms, although it provides less in-depth analysis than CAPS.
Less specific techniques were employed by Serpeloni et al., 33 who did not analyze stress per se but used a 17-item trauma-related event checklist, a tool used to identify exposure to various traumatic events. Wadji et al. 29 measured salivary cortisol levels in women exposed to IPV, providing a physiological measure of stress. Lastly, Radtke et al. 31 did not account for any stress measure or mental health outcome, which makes it difficult to understand the overall epigenetic changes produced and the specific changes in the body due to IPV.
Additionally, half of the studies included in this review28,33,29 considered the anxiety and depression symptoms of women exposed to IPV, which can help in understanding the women’s situation and the severity of their symptoms.
The moment of life in which the woman suffers this violence is also relevant, especially in matters of inheritability of the epigenetic changes associated with this chronic stress, as we see in the article by Radke et al., 31 where the presence of N3RC1 methylation in the children was significantly associated with the mother’s exposure to violence during pregnancy, but said change was not significant when the mother experienced the violence before or after the pregnancy. This is because pregnancy is a time window in which babies are genetically more susceptible to changes, a phenomenon that various researchers have called the “programming effects” of prenatal stress, having serious repercussions on temperament and neuroendocrine regulation of newborn,52,53 on cortisol metabolism, which is reflected in the long run in worse management of stressful events. 54 From the Wadji et al.’s 29 report, we cannot conclude anything about the timing of pregnancy, as the children witnessed IPV after birth. It is striking that methylation occurred in the mothers and not in the offspring, indicating that stress derived from IPV may have a more pronounced impact on mothers.
An article on the genocide that occurred with the Tutsi demonstrated changes in the methylation status of the promoter regions of NR3C1 and NR3C2, the children of women pregnant during the genocide, but this was not the case with women who became pregnant later. In addition, these children had lower cortisol and mineralocorticoid receptor levels, modifications that are suggested as the biological basis for the transmission of Hypothalamic–pituitary–adrenal (HPA) axis abnormalities. 55 This does not mean that for the offspring to inherit the epigenetic alterations associated with violence, it is strictly necessary for this to occur during pregnancy, but it does mean that it is consistently more likely, since otherwise, said epigenetic alterations would have to occur in the female gonads.
The epigenetic changes observed in women as a result of gender-based violence in this systematic review were methylations in the BDNF, CPLX, NR3C1, and IFG2 genes. The first of these, the BDNF gene, is a neurotrophin associated with nerve growth factor and acts as such. This gene has been shown to play an important role in synaptic plasticity 56 and is involved in the pathophysiology of depression, suicidal behaviors,56–60 and in other psychiatric disorders such as bipolar disorder. 61 Specifically, the V66M BDNF allele is strongly associated with bipolar disorder62,63 and anxiety-related disorders such as PTSD.62,64
There are also studies showing the correlation of BDNF with addiction-related behaviors, such as increased intake and development of tolerance to drugs and increased craving after withdrawal.65,66 Recently, it has been seen that this same Val66Met allele of the BDNF gene acts as a moderator of the effect of prenatal exposure to IPV on the regulation of infant temperament and cortisol. Children with this methylated allele have poorer temper regulation and poorer mobilization of the cortisol response. 52
In contrast, Serpeloni et al. 33 did not find that BDNF methylation was related to either PTSD or depression in any of the three generations covered by the study. In contrast, Moser et al. 28 observed a significant correlation between the severity of maternal anxiety with the degree of BDNF methylation, as well as between exposure to domestic violence when the mother was a girl and the degree of methylation, this event not occurring with the severity of the PTSD.
Also, maternal depression and anxiety during pregnancy can induce methylation changes in the insulin growth factor (IGF) system.67,68 The IGF system includes IGF1 and IGF2, as well as several other genes related to IGF-binding proteins. Although each of them is expressed in various parts of the body, IGF1 and IGF2 are also synthesized by the placenta, where they are involved in the regulation of fetal, placental, and fetal function. 69 Human studies have found associations between birth weight and differential methylation in both IGF1 and IGF2.70,71 In addition, the relationship between changes in insulin-like growth factors with eating disorders has been described, such as bulimia or anorexia nervosa, 72 alcohol-related disorders, 73 PTSD, 74 and schizophrenia, in the latter case with IGF2 hypomethylation.75,76
Other essential components are the other changes produced on the HPA axis and the NR3C1 gene, one of its main regulators. Its methylation is involved in the development of mental disorders such as depression, anxiety,77,78 and schizophrenia. 79 NR3C1 is found to be hypermethylated in adolescents who have experienced bullying or depression, 80 as well as in adults who have experienced other traumatic events such as emotional, physical, or sexual abuse or early parental death in childhood. 81 Maternal IPV is associated with methylation of the glucocorticoid receptor NR3C1 gene, specifically implicated in newborn weight through cortisol-related pathways, as well as other HPA axis genes such as CRH, CRHBP, and FKBP5.82,83
There appears to be some ambiguity in the results obtained; and in some cases, they appear to be contradictory. This may be due to the difficulty of establishing a clinical diagnosis of PSTD and mental health outcomes, as well as the difficulty involved in its measurement, limitations that also occur in IPV. On the contrary, it does not seem that the type of biological sample studied influences the results, since the study by Radtke et al., 31 who used total blood sample, and the others by Cordero et al. 30 and Wadji et al., 29 who used saliva sample, both show methylation at the glucocorticoid receptor. What this systematic review does show is that both mental health outcomes and PTSD and, more specifically, IPV-PTSD associated with epigenetic changes both in women and in their offspring.
Limitations
This review has the intrinsic limitations of a systematic review, such as variability in the study sample, the analysis technique, and the cohort itself. In addition, it presents the difficulty of enormous variability and subjectivity when it comes to the measurement of gender-based violence and post-traumatic stress, due to the lack of consensus regarding a universal technique to measure its intensity.
In addition to the different resilience capacities that several women may present in the same situation of violence.
Conclusions
Building on the contribution and quality of previous research, this review concludes that IPV leaves epigenetic marks in women. Furthermore, there is evidence that these marks can be inherited for up to two generations. The genes most affected by this violence are primarily related to stress, including NR3C1, IGF1, and IGF2, and are involved in brain pathways associated with neurotrophins like BDNF, which is linked to nerve growth. However, larger cohort studies and longitudinal follow-ups are necessary to validate these findings and to determine whether these epigenetic marks are reversible over time.
Supplemental Material
sj-docx-1-whe-10.1177_17455057241290335 – Supplemental material for Epigenetic changes produced in women victims of intimate partner violence: A systematic review
Supplemental material, sj-docx-1-whe-10.1177_17455057241290335 for Epigenetic changes produced in women victims of intimate partner violence: A systematic review by Coral González-Martínez, Christian Haarkötter, Elena Carnero-Montoro, Jose A Lorente and Miguel Lorente in Women's Health
Footnotes
Acknowledgements
This work was supported by the University of Granada (Department of Legal Medicine, Toxicology and Physical Anthropology) and by GENyO, Centre for Genomics and Oncological Research: Pfizer-University of Granada.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
