Abstract
Background:
Cancer is the leading cause of death in people living with HIV. In the United States, nearly 1 in 4 people living with HIV are women, more than half of whom rely on Medicaid for healthcare coverage.
Objective:
The objective of this study is to evaluate the cancer burden of women living with HIV on Medicaid.
Design:
We conducted a cross-sectional study of women 18–64 years of age enrolled in Medicaid during 2012, using data from Medicaid Analytic eXtract files.
Methods:
Using International Classification of Diseases, Ninth Revision, Clinical Modification diagnosis codes, we identified women living with HIV (n = 72,508) and women without HIV (n = 17,353,963), flagging the presence of 15 types of cancer and differentiating between AIDS-defining cancers and non-AIDS-defining cancers. We obtained adjusted prevalence ratios and 95% confidence intervals for each cancer and for all cancers combined, using multivariable log-binomial models, and additionally stratifying by age and race/ethnicity.
Results:
The highest adjusted prevalence ratios were observed for Kaposi’s sarcoma (81.79 (95% confidence interval: 57.11–117.22)) and non-Hodgkin’s lymphoma (27.69 (21.67–35.39)). The adjusted prevalence ratios for anal and cervical cancer, both of which were human papillomavirus-associated cancers, were 19.31 (17.33–21.51) and 4.20 (3.90–4.52), respectively. Among women living with HIV, the adjusted prevalence ratio for all cancer types combined was about two-fold higher (1.99 (1.86–2.14)) in women 45–64 years of age than in women 18–44 years of age. For non-AIDS-defining cancers but not for AIDS-defining cancers, the adjusted prevalence ratios were higher in older than in younger women. There was no significant difference in the adjusted prevalence ratios for all cancer types combined in the race/ethnicity-stratified analyses of the women living with HIV cohort. However, in cancer type–specific sub-analyses, differences in adjusted prevalence ratios between Hispanic versus non-Hispanic women were observed. For example, the adjusted prevalence ratio for Hispanic women for non-Hodgkin’s lymphoma was 2.00 (1.30–3.07) and 0.73 (0.58–0.92), respectively, for breast cancer.
Conclusion:
Compared to their counterparts without HIV, women living with HIV on Medicaid have excess prevalence of cervical and anal cancers, both of which are human papillomavirus related, as well as Kaposi’s sarcoma and lymphoma. Older age is also associated with increased burden of non-AIDS-defining cancers in women living with HIV. Our findings emphasize the need for not only cancer screening among women living with HIV but also for efforts to increase human papillomavirus vaccination among all eligible individuals.
Keywords
Introduction
Currently, there are nearly 1.2 million people living with HIV (PLWH) in the United States. Among PLWH, approximately 25% are women, of whom, 75% are from racial or ethnic minorities. 1 In addition, transgender women, those with substance use disorders, and those with lower socioeconomic status are disproportionately represented among women living with HIV (WLWH). 2 With the early initiation of highly active antiretroviral therapy (HAART), life expectancy of WLWH has improved significantly. 3 Nevertheless, it remains shorter among WLWH than in the general population. 4
Cancer in WLWH is a major cause of morbidity and mortality. In the pre-HAART era, AIDS-defining cancers (ADCs), including Kaposi’s sarcoma (KS), non-Hodgkin’s lymphoma (NHL), and cervical cancer, accounted for a large portion of cancer-related deaths. Because of the immunocompromised state caused by HIV infection, cancers associated with viral infections are commonly seen in people with HIV. While KS is the most common cancer in PLWH, 5 the prevalence of KS in non-HIV-infected population is low. As a result, the incidence of KS in the United States has remained stable in recent decades. KS mainly results from the human herpes virus-8 (HHV8, also known as KHSV).5,6 NHL is distinguished from Hodgkin’s lymphoma (HL) based on Reed–Sternberg cells in the lymph fluid. NHL and a proportion of HL are linked to Epstein–Barr virus (EBV) infection. 7 Both HHV8 and EBV belong to the same gamma-herpesvirus family. Cervical cancer is the most common cancer in WLWH, and cervical cancer is caused by human papillomavirus (HPV). 8 Despite the availability of HPV vaccination and screening, cervical cancer is the leading cause of cancer-related deaths in this population.9,10 In the post-HAART era, improved healthcare has dramatically reduced the incidence of ADCs in WLWH,11,12 and non-AIDS-defining cancers (NADCs) now account for a significantly increased fraction of the overall cancer burden in WLWH. 13
The improvements in viral suppression and survival afforded by HAARTs will likely continue to alter the types of cancers that most impact WLWH. It is projected that while the burden of ADCs will likely further decrease, NADCs, which are mainly composed of HL and cancers of the anus, mouth, throat, liver, lung, and breast, will increase slightly. 14 The current consensus is that the HIV infection itself does not have any direct carcinogenic effect. 15 However, with the aging of the population and increased longevity associated with HAART, WLWH are at increased risk of cancer, due to compromised immune function and co-infection by oncogenic viruses such as HPV, hepatitis B (HBV) or C virus (HCV).16,17 Furthermore, the prevalence of smoking and alcohol drinking, which are independent risk factors for lung, head/neck, liver, and other types of cancer, is much higher in WLWH than in non-infected women, contributing to the increased mortality and morbidity in WLWH. 18 In addition, substance use and high-risk sexual behavior increases exposure to HIV and other oncogenic viruses.6,19,20
Despite the substantial morbidity and mortality caused by cancer in PLWH, the cancer burden in WLWH specifically has not been evaluated comprehensively, given that men comprise the majority of PLWH and studies have traditionally focused on this population. This is an important knowledge gap that needs to be addressed since interventions to reduce cancer burden in WLWH may differ from those of men. Medicaid data provide a unique opportunity to better understand cancer burden in WLWH since 54% of WLWH were enrolled in Medicaid as of 2018. 21 In addition, given that Medicaid is a safety-net program for women with low incomes, Medicaid data allow us to capture those with complex medical needs and heightened vulnerability for poor outcomes. Thus, in this study using Medicaid data from all 50 states and the District of Columbia, we investigated the prevalence of 15 common types of cancer in WLWH compared to women without HIV on Medicaid.
Methods
Medicaid database and study population
This is a cross-sectional study using data from the Medicaid Analytic eXtract (MAX) database, which includes the following files: (1) The Personal Summary (PS) file, which we used to retrieve individuals’ demographics and months of enrollment in Medicaid during the study year and (2) claims files, including Inpatient (IP) and Other Therapy (OT) for care received in IP hospital and institutional and non-institutional outpatient care settings, respectively. We used the claims files to identify the relevant diagnosis codes for HIV and 15 types of cancer (Supplemental Table 1). 22
The study population included 17,426,471 individuals 18–64 years of age and were noted to be female in the administrative records. We excluded Medicaid/Medicare dually eligible individuals and those with recipient indicator “2” (recipient only had premium payment claims (i.e. no healthcare services claims)) or “9” (recipient was not enrolled in State Child Insurance Health Program (S-CHIP) or Medicaid for any months in the study period), consistent with our previous study. 22 We included enrollment and claims data for all other Medicaid beneficiaries, including for non-users (people who were enrolled in Medicaid during 2012 but did not have any claims).
Key variables of interest
Main independent variable HIV status
We identified HIV status based on relevant International Classification of Diseases, Ninth Revision (ICD-9) diagnosis codes included in the Agency for Healthcare Research and Quality (AHRQ) Clinical Classification Software (CCS). 23 To identify WLWH, we required one or more occurrences in the IP file or two more separate occurrences in the OT file, at least 30 days apart. Women were classified as HIV negative in the absence of claims carrying HIV diagnosis.
Outcome ascertainment—cancer
We defined each of the 15 types of cancers as binary variables in women based on relevant ICD-9 diagnosis codes included in AHRQ CCS. 23 These included cancer of the breast, cervix, head/neck, esophagus, stomach, colon, rectum, anus, liver and intrahepatic bile duct, pancreas, bronchus/lung, and other respiratory and intrathoracic organs, as well as HL and NHL, KS, and leukemia. Similar to HIV status, we required at least one occurrence in the IP file or two more separate occurrences in the OT file, at least 30 days apart, to ascertain their cancer diagnosis. ADCs included KS, NHL, and cervical cancer. NADCs included cancer of head/neck, esophagus, stomach, colon, rectum, anus, liver, pancreas, bronchus/lung, other respiratory and intrathoracic organs, breast, HL, and leukemia.
Covariates
Independent variables included age categories (18–24, 25–34, 35–44, 45–54, and 55–54 years) and race/ethnicity (non-Hispanic White; non-Hispanic Black; Hispanic of any race, including Hispanic or Latino and one or more races; Asian; American Indian or Alaskan Native; Native Hawaiian or Other Pacific Islander; more than one race; and unknown or missing). Due to the small numbers in the latter race/ethnicity categories, we grouped individuals in the All Other category when examining cancer prevalence. In addition, our models accounted for the US Census divisions (New England, Mid-Atlantic, East North Central, West North Central, South Atlantic, East South Central, West South Central, Mountain, and Pacific) based on the states from which the Medicaid data originated. 24 We also identified and adjusted for co-infections by HBV or HCV, and/or by cervical HPV or any type of HPV (see the relevant diagnosis codes in Supplemental Table 1). Since the length of enrollment in Medicaid may be associated with greater odds to capture HIV and cancer diagnoses, our multivariable models also accounted for the total number of months of enrollment in Medicaid during 2012. In our models, we did not include the behavioral factors that are known to be high risk for certain types of cancers, given the potential under-reporting of these factors in administrative data (e.g. we did not identify patients with smoking behavior by ICD-9 codes). 25
Statistical analysis
We estimated prevalence ratios for each cancer type by HIV status using log-binomial models, 26 in which cancer (specific cancer type or cancer types combined) was the dependent variable, and HIV status was the main independent variable. Covariates included age group, race/ethnicity, US Census divisions, enrollment months, and co-infection by HBV/HCV (in models for liver cancer) or HPV (in models for head/neck, rectal, and anal cancers). For all adjusted prevalence ratios (APRs), the reference category was women without HIV. In the analyses that examined all cancer types combined, we accounted for co-infections by including a binary variable indicating the presence of HBV, HCV, and/or HPV. We did not adjust for co-infections in models for NHL and HL, Kaposi sarcoma, and breast cancer.
We conducted the stratified analyses to examine cancer prevalence by age and race/ethnicity within the WLWH population. We used similar regression models to estimate cancer prevalence in age and race/ethnicity strata with approximately equal proportion in each subgroup. We divided the WLWH into two age groups (18–44 and 45–64 years) based on the age distribution of the cancers with the highest prevalence in WLWH—cervical, breast, and anal cancer. For the APRs of the older age group (45–64), the reference category is younger age group (18–44). For the APRs of a given race/ethnicity stratum, the reference category was composed of women in all other race/ethnicity categories (e.g. APRs for non-Hispanic Black women are presented in comparison to all other women in this study population). To address small numbers in our stratified analyses, we focused on the most common cancer types (breast, anal, rectal, and lymphoma), and all other NADCs in one category. In the stratified race/ethnicity analyses, we presented our data for non-Hispanic Whites, non-Hispanic Blacks, Hispanics, and All Others.
We used SAS software, version 9.4 for UNIX (SAS Institute, Inc., Cary, NC, USA) for data processing and analysis, R version 4.0.0, R Studio 1.3.1093, and the ggplot2 package to generate the forest plots for APRs. 27
Results
We identified 72,508 WLWH and 17,353,963 women without HIV. Table 1 shows the characteristics of the population by demographics, co-infections, and type of cancer. Compared to women without HIV, a higher percentage of WLWH were in the 45–64 age group (46.54% vs 17.48%). Similarly, there was a greater representation of non-Hispanic Black women in WLWH among their non-HIV counterparts (58.55% vs 21.18%).
Distribution of the study population by HIV status, demographics, and cancer type.
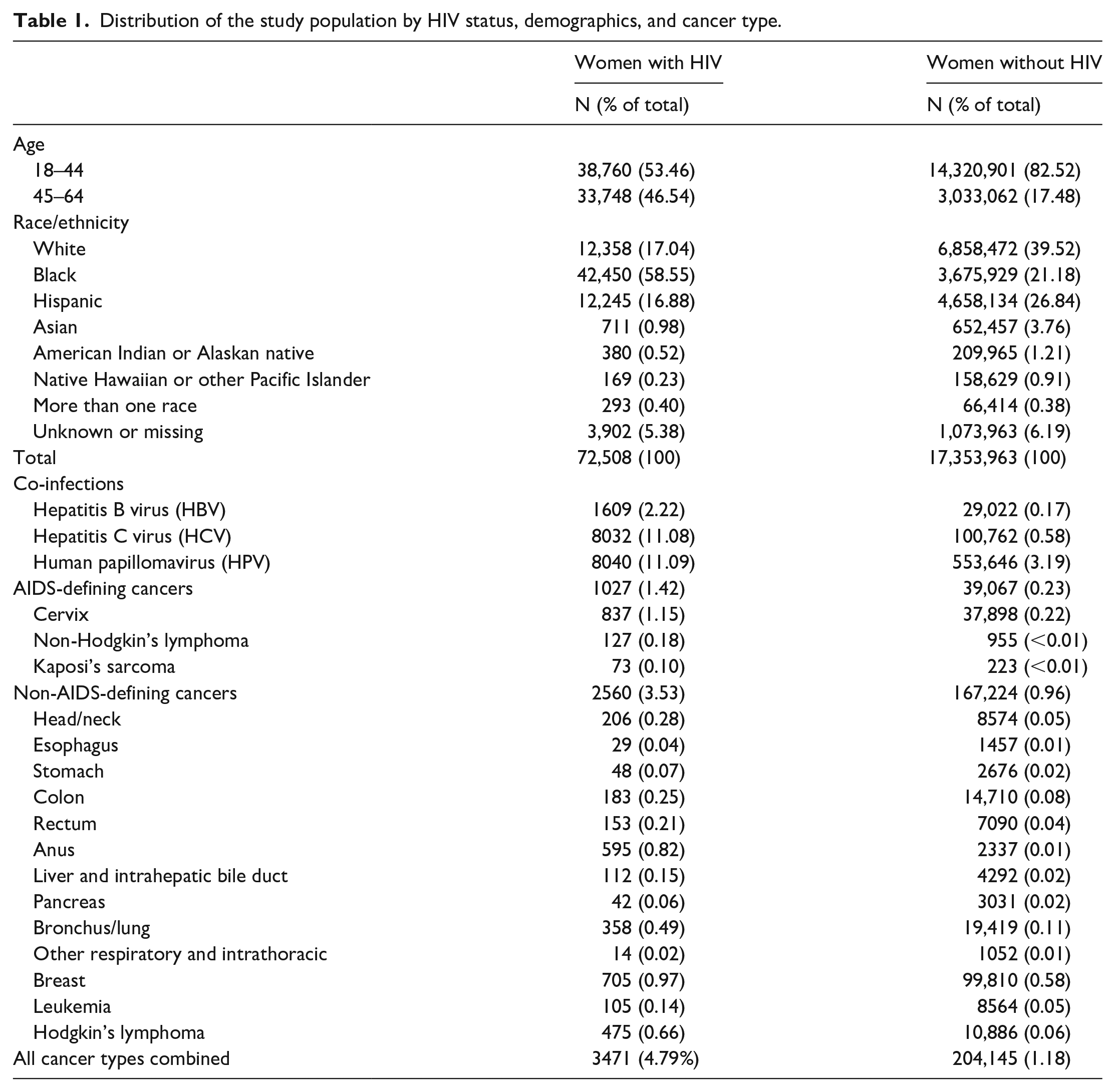
Cancers were more common among WLWH than among women without HIV (4.79% vs 1.18%). Among WLWH with ADCs (1027 cases), cervical cancer was the most prevalent cancer type and composed 81% of ADC cases, while NHL and KS composed 12% and 7% of ADC cases, respectively. Breast, anal, and lung cancers were the most common NADCs, accounting for about 28%, 23%, and 14% of total NADCs (2560 cases), respectively. In women without HIV, breast cancer, cervical cancer, and lung cancers were the most prevalent cancer types, accounting for 49%, 19%, and 10% of total cancers, respectively.
In Figure 1, we present the APRs obtained from the log-binomial models for each cancer type. Among ADCs, the highest APR was for KS (81.79 (57.11–117.22)), followed by NHL (27.69 (21.67–35.39)) and cervical cancer (4.20 (3.90–4.52)). Among NADCs, the highest APRs were observed for anal cancer (19.31 (17.33–21.51)) and HL (4.92 (4.47–5.41)). The APRs of cancer of head/neck, rectal, bronchus/lung, esophageal, stomach, pancreas, colon, and leukemia were significantly higher in WLWH compared to women without HIV. For breast cancer, however, we observed a lower prevalence (0.70 (0.65–0.76)) in WLWH, compared to women without HIV. For liver cancer, the APR was 33% lower (0.75 (0.61–0.92)) after adjusting for HBV/HCV co-infection in the multivariable models.

Adjusted prevalence ratios (APRs) and 95% confidence interval for various types of cancer stratified by ADC/NADC.
Table 2 presents the APRs for cancers among WLWH in the older age group (45–64 years) compared to the younger age group (18–44 years), based on the number of cancer cases in the two age groups listed in Supplemental Table 2. Our results showed that the overall APR for all cancer types combined was about two-fold higher (1.99 (1.86–2.14)) in older WLWH compared to their younger counterparts. Among WLWH with ADC, there was no statistical difference in the APRs of each type of cancer in the older WLWH compared to those of younger WLWH. For NADCs, the APRs for cancer of the anus, rectum, breast, HL, and other NADCs combined (including cancer of head/neck, esophagus, stomach, colon, liver, pancreas, lung, other respiratory and intrathoracic organs, and leukemia) were significantly higher in older than in younger WLWH.
ARP and 95% CIs for select cancers by age group from WLWH.

For the APRs of older age group (45–64 years), the reference category is younger age group (18–44 years). APRs for which the 95% CI crosses 1.0 are not statistically significant at p < 0.05. Models are adjusted for race/ethnicity, US Census divisions, and months of enrollment in Medicaid during 2012. The model for cervical cancer is adjusted for cervical HPV, and models for rectal and anal cancers adjusted for any type of HPV. Models for All Other Cancers and All Cancers Combined are adjusted for the presence of any co-infection (HBV/HCV and/or HPV). Models for non-Hodgkin’s and Hodgkin’s lymphoma and breast cancer are not adjusted for co-infections. APR: adjusted prevalence ratio; CI: confidence interval; WLWH: women living with HIV; HPV: human papillomavirus; HBV: hepatitis B virus; HCV: hepatitis C virus.
Table 3 presents the age-adjusted APRs by race/ethnicity in WLWH based on the numbers of cancer cases in the four race/ethnicity groups listed in Supplemental Table 3. Across four race/ethnicity groups, the APR for all cancer types combined did not differ significantly. For cancer-specific APRs, we noted some variation in the APRs across race/ethnicity categories. Among ADCs, the highest APR for NHL was observed in Hispanic women (2.00 (1.30–3.07)). For NADCs, lower APRs for breast cancer (0.73 (0.58–0.92)), and other NADC cancer types combined (0.76 (0.63–0.93)) were observed among Hispanic women. Other types of cancers were not different across the four race/ethnicity groups.
APRs and 95% CIs for select cancers by race/ethnicity from WLWH.

For the APRs of each race/ethnicity strata, the reference category is the all the other women except the race/ethnicity strata, the reference category is all the other women except the race/ethnicity examined (i.e. non-Hispanic White vs all other race/ethnicities (reference), non-Hispanic Black vs all other race/ethnicities (reference), Hispanic vs all other race/ethnicities (reference), all other race vs non-Hispanic White/non-Hispanic Black/Hispanic (reference)). All models are adjusted for age, US Census divisions, and months of enrollment in Medicaid during 2012. Models for cervical cancer are adjusted for cervical HPV, and models for rectal and anal cancers are adjusted for any type of HPV. Models for All Other Cancers and All Cancers Combined are adjusted for the presence of any co-infection (HBV/HCV and/or HPV). Models for non-Hodgkin’s and Hodgkin’s lymphoma and breast cancer are not adjusted for co-infections. APRs: adjusted prevalence ratios; CI: confidence interval; WLWH: women living with HIV; HPV: human papillomavirus; HBV: hepatitis B virus; HCV: hepatitis C virus.
Discussion
Using national Medicaid data, we documented a high burden of ADCs and several types of NADCs in WLWH than in women without HIV on Medicaid. Our findings showed large differences in prevalence across cancer sites and age groups. Compared to women without HIV on Medicaid, the prevalence of cervical cancer was about 4.2-fold higher, and the prevalence of anal cancer was about 19.3-fold higher among WLWH. Given that additional risk factors (e.g. smoking and non-HPV co-infections) increase cancer risk in PLWH, 28 our findings highlight the importance of cancer risk evaluation and screening. Preventive measures should address multiple risk factors,29,30 and promote HPV vaccination in all eligible individuals. As noted in previous reports,31–33 these highly prevalent cancer types should be targeted for cancer screening in WLWH. Related to HPV infection, both cervical and anal cancers are preceded by high-grade squamous intraepithelial lesions (HSILs), and precancerous cells are even more common on anal pap test than cervical pap test for WLWH. 31 Screening of cervical HSIL has been shown to be critical in preventing cervical cancer for WLWH, but the evidence-based guidelines for the screening anal HSIL in WLWH is still lacking. Recently, the multi-site ANal Cancer HSIL Outcomes Research (ANCHOR) study suggested that the treatment of anal HSIL reduced the risk of anal cancer by more than half, 34 highlighting that the screening and timely treatment of HSIL could prevent anal cancer in WLWH.
The highest APR observed in our study was for KS, which was 82 times higher in women with HIV than women without HIV, a notable but unsurprising finding. KS rarely develops in healthy persons carrying HHV8, the virus responsible for this type of cancer, and less than 5% of the general US population is infected by HHV8. 5 In contrast, the risk of HIV-associated KS is high, even among patients who receive effective HAART, and have normal CD4+ counts. 5
To account for the increased risk of cancer due to advanced age, we conducted age-specific analyses. The crude numbers from the younger and older age strata showed a higher prevalence of NADCs in the older age group than in the younger age group (Supplemental Table 2). Thus, the age-stratified analysis of APR is necessary to show site-specific cancer prevalence in WLWH population (Table 2). Indeed, the result of age-specific analyses showed the prevalence for NADC at specific sites was higher in older than in younger WLWH, demonstrating that the NADC prevalence increased with age in women with HIV infection across multiple cancer sites. Interestingly, our study showed that the adjusted prevalence of three types of ADCs, including NHL, cervical cancer, and Kaposi’s lymphoma were similar for young and old age group, possibly suggesting that the co-infection of HIV and HPV/EBV virus is an even more potent risk factor for the prevalence of ADCs than age.
WLWH are at an increased risk for contracting a range of co-infections by oncogenic viruses, particularly various types of HPV.8,35,36 Our data show that HPV-associated cancers (primary sites such as cervical, anal, rectal, and head/neck) contribute the most to the cancer burden in WLWH enrolled in Medicaid. Invasive cervical cancer is the most common cause of cancer-associated death in WLWH, and often occurs at a relatively young age and presents at a more advanced stage than women without HIV. 37 Therefore, screening for cervical cancer at the appropriate age is highly recommended for this group of women. 38 Given the time window from HIV/HPV infection to progress to the precancerous lesion and malignancy formation, early HPV vaccination would be crucial to prevent anogenital cancer. However, studies have shown that most HIV-infected women have not received timely vaccination to prevent initial HPV infection. 39 Thus, ensuring completion of the HPV vaccine series in teens and eligible adults is paramount. In addition, both HBV and HCV can cause hepatocellular carcinoma. In the United States, about 6%–10% of PLWH have co-infection of HBV, 40 and approximately 6%–16% of them have co-infection with HCV. 41 Furthermore, other non-infection-related risk factors, such as smoking, alcohol abuse, and drug use, could contribute to the increased risk of cancer in WLWH.
After adjusting for HBV and HCV infection, our findings suggest that HIV infection was not associated with higher risk of liver cancer among WLWH compared with women without HIV. This may be explained by the sustained viral suppression afforded by HAART since liver cancer risk has been directly associated with HIV-related immunodeficiency level. Furthermore, some HAART regimens can also be used to manage HBV co-infection concurrently, so people on HAART may have a lower liver cancer risk than the general population with un-diagnosed HBV infection. 42
One of the major strengths is that our study included a large fraction of all WLWH. As previously mentioned, 54% of WLWH were enrolled in Medicaid in 2018, 21 our study represented the WLWH population in the United States at large. Another strength of this study is our in-depth stratified analysis of prevalence by age and race/ethnicity group. In particular, while the overall prevalence of all cancer types was similar across race/ethnicity groups, the prevalence varied by cancer sites across race/ethnicity group (Table 3 and Supplemental Table 3).
Our study has several limitations. First, given our use of data from the Medicaid program, demographic variables (age, race/ethnicity, and sex) are as documented in the administrative records. This limits our ability to examine certain subpopulations, such as transgender individuals, for whom documented sex may be incorrect or incomplete. Second, due to the nature of administrative data, we did not have any reliable measures on behavioral risk factors such as, smoking, drug abuse, or sexual behaviors. Third, this study does not account for the co-infection of all oncogenic viruses. While we accounted for HPV and HBV/HCV co-infection, we could not account for EBV and other oncogenic infection or opportunistic infections, given the limited sensitivity of administrative ICD-9 codes to identify these conditions. Fourth, we note that these results reflect data from 2012. Since then, Medicaid enrollment fluctuated considerably, first by increasing due to its expansion in 2014, then declining in 2017–2019, 43 and increasing again during the pandemic. 44 Given that the pool of Medicaid is dynamic, getting a fully representative snapshot of the Medicaid population is difficult. Nonetheless, the associations identified in this study are likely to be consistent over time. Fifth, our findings from this study should apply to WLWH on Medicaid and may not be generalizable to the WLWH population overall, especially given that many individuals join the Medicaid program upon being diagnosed with cancer, or after having depleted their resources. Finally, the sample in this study includes those women diagnosed with HIV but does not include those who have HIV but were not yet diagnosed. Therefore, it will be necessary to continuously monitor cancer burden in Medicaid-insured WLWH. 45
Conclusion
This study highlights the high prevalence of ADCs and certain NADCs in WLWH, compared with women without HIV on Medicaid. Our findings highlight the importance of proactive measures to promote cancer screening and more widespread HPV vaccination to reduce the burden in this population.
Supplemental Material
sj-docx-1-whe-10.1177_17455057231170061 – Supplemental material for Cancer burden in women with HIV on Medicaid: A nationwide analysis
Supplemental material, sj-docx-1-whe-10.1177_17455057231170061 for Cancer burden in women with HIV on Medicaid: A nationwide analysis by Guangjin Zhou, Siran M Koroukian, Suparna M Navale, Nicholas K Schiltz, Uriel Kim, Johnie Rose, Gregory S Cooper, Scott E Moore, Laura J Mintz, Ann K Avery, Sudipto Mukherjee and Sarah C Markt in Women's Health
Supplemental Material
sj-docx-2-whe-10.1177_17455057231170061 – Supplemental material for Cancer burden in women with HIV on Medicaid: A nationwide analysis
Supplemental material, sj-docx-2-whe-10.1177_17455057231170061 for Cancer burden in women with HIV on Medicaid: A nationwide analysis by Guangjin Zhou, Siran M Koroukian, Suparna M Navale, Nicholas K Schiltz, Uriel Kim, Johnie Rose, Gregory S Cooper, Scott E Moore, Laura J Mintz, Ann K Avery, Sudipto Mukherjee and Sarah C Markt in Women's Health
Supplemental Material
sj-docx-3-whe-10.1177_17455057231170061 – Supplemental material for Cancer burden in women with HIV on Medicaid: A nationwide analysis
Supplemental material, sj-docx-3-whe-10.1177_17455057231170061 for Cancer burden in women with HIV on Medicaid: A nationwide analysis by Guangjin Zhou, Siran M Koroukian, Suparna M Navale, Nicholas K Schiltz, Uriel Kim, Johnie Rose, Gregory S Cooper, Scott E Moore, Laura J Mintz, Ann K Avery, Sudipto Mukherjee and Sarah C Markt in Women's Health
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
