Abstract
Background:
Postpartum depression among mothers living with HIV is a significant public health problem due to its effects on engagement in care, HIV disease progression, and an increased risk of mother-to-child transmission of HIV.
Objective:
The objective of this study was to determine the prevalence and factors associated with postpartum depression among mothers living with HIV.
Design:
The study employed a cross-sectional quantitative research design.
Methods:
In this cross-sectional survey, we consecutively recruited 290 participants among mothers attending postnatal, immunization, and family planning clinics at an urban clinic in Uganda. Using an interviewer-administered questionnaire, we collected data on socio-demographics, obstetric, and HIV-related characteristics. Postpartum depression was assessed using the Patient Health Questionnaire version 9. We classified participants with Patient Health Questionnaire version 9 scores of ⩾10 as having postpartum depression. We conducted logistic regression to examine the association between postpartum depression and independent variables.
Results:
The prevalence of postpartum depression was 15.9%. After controlling for other variables, participants who reported poor male partner support were more likely to experience postpartum depression compared to those who had good partner support (adjusted odds ratio = 4.52, confidence interval = 2.31–8.84, p value < 0.001).
Conclusion:
Mothers living with HIV should be routinely assessed for the presence of depression and male partner support. Health care providers of HIV-infected women should design strategies to promote male partner support for better maternal, infant, and HIV treatment outcomes.
Background
The postpartum period is often a period of joy for both the mother and the entire family. However, this period is also recognized as a period of vulnerability in the life of the woman with several potential complications including postpartum depression (PPD).1,2 PPD is a mood disorder that occurs after birth and is characterized by symptoms such as mood changes, fatigue, inability to sleep, lack of energy, low self-esteem, tearfulness, loss of appetite, feelings of inadequacy, irritability, loss of interest and enjoyment, and reduced energy leading to diminished activity.2,3 The coexistence of HIV and PPD harms adherence to antiretroviral therapy (ART) and other outcomes such as premature progression of HIV infection.4–6
Globally, the prevalence of PPD in the general population of women is estimated at 13%; however, the estimated prevalence for developing countries is 19.8%. 2 A review reported the prevalence of PPD to be 10%–20%. 7
In terms of PPD in women living with HIV in developing countries, studies have reported varying prevalence, with the highest being 74.1% in Thailand and the lowest of 11% in Malawi.6,8 A review of PPD in women living with HIV in Africa was 22.5%. 4 Several studies have also reported variable prevalence of PPD among women living with HIV in Sub-Saharan Africa (SSA); 25.2% in Kenya, 35.6% in South Africa, and 26% in Zimbabwe.9–11 Many of these studies measured PPD at 6 weeks post-delivery whiles other studies screened for PPD between 1 week and 18 months after delivery. The measurement tools used for the prevalence reported were the epidemiological studies depression scale, Edinburgh postpartum depression scale, and Diagnostic and Statistical Manual of Mental Disorders (5th ed.; DSM-5).
The most frequently reported factors associated with PPD are low income level, low educational status, poor partner or male involvement, lack of social support, stigma, and discrimination.10,12 Poor access and adherence to ART for both mother and infant were found to be a factor contributing to PPD in mothers, but the evidence is limited on the subject.13,14
In Uganda, the prevalence of depressive symptoms in the general population of postpartum women and women living with HIV is 43% and 39%, respectively.5,15 However, little is known about the prevalence and factors associated with PPD among mothers living with HIV in Uganda. This study aimed to assess the prevalence and factors associated with PPD among mothers living with HIV at an urban clinic in Uganda.
Methods
This was a cross-sectional study that consecutively recruited 290 postnatal mothers living with HIV from a clinic that offers postnatal care and family planning to mothers and immunization to children. The sample size was determined using Leslie Kish’s formula and the prevalence of PPD from a study conducted in Kenya.9,16 Eligible participants were mothers living with HIV between 4 weeks and 1 year after delivery. The period of 4 weeks was selected to exclude mothers who might be having postpartum blues and physiological problems. The study included the range of 1-year to capture mothers who will have a late onset of depression after delivery. Mothers aged 15 years and above were included in the study. However, consent from legal representatives of mothers aged 15–17 was not obtained because the mothers certified the condition for providing independent informed consent (emancipated minors according to Clinical Research Regulation for Uganda). 17 Mothers and children who were very ill were excluded. Before participation, mothers living with HIV were identified by the midwives working at the clinic after receiving care and approached for consent. Written informed consent was provided in English and Luganda. Mothers who accepted to participate were taken through the consenting process and recruited into the study. Data on socio-demographic, obstetric, community, and HIV-related characteristics were collected using interviewer-administered questionnaires. Participants were taken to a separate room for the interview that was conducted by trained research assistants who were nurse counsellors. The interviews were conducted in either English or Luganda.
Sample size determination
The number of study participants was obtained using Leslie Kish’s formula. 16 The calculation is as shown below
N = sample size.
Z α = 1.96 the standard normal value corresponding to 95% confidence level.
p = 25.2% of the prevalence of PPD was obtained from a study done in Kenya. 9
d = total width of confidence interval (CI) with a set precision of 5%. d = 0.05 × 0.05 = 0.0025.
A total of 290 participants were included in the study.
Measurement
PPD was assessed using the Patient Health Questionnaire version 9 (PHQ-9). The PHQ-9 is a diagnostic instrument developed and validated by Kroenke et al. 18 PHQ-9 is a recommended tool for screening and diagnosis of depression among people living with HIV (PLHIV) in Uganda. 19 The tool has also been used in several studies to assess depression among PLHIV.20–22 This is a 9-item scale, and the scores for each of the items are rated as (0 = not at all, 1 = several days, 2 = more than half the days, or 3 = nearly every day). The final assessment of depression using PHQ-9 is completed by summing the item scores for each participant; scores range from 0 to 27 in total. Scores are interpreted as “no depression symptoms” if the total score is less than 5, “minimal depression symptoms” for total score between 5 and 9, and as “major depressive disorder (MDD)” for total scores ⩾10. This depression assessment tool has been widely used across different settings.8,23,24 PHQ-9 has also been translated into several languages. 25 More recent PHQ-9 validation studies reported acceptable diagnostic properties for detecting MDDs at cut-off scores between 8 and 11. 25 The PHQ-9 was translated to Luganda and validated in Uganda and found to be a reliable diagnostic indicator of PPD. 26 It is a recommended tool for assessing and managing depression in primary health care in Uganda. 19 According to the guidelines for diagnosing depression in Uganda, a total score of 5–9 is experiencing depressive symptoms, 10–14 mild depression, 15–19 moderate depression, and ⩾20 severe major depression. 19 A cut-off of ⩾10 as a measure of MDD was applied in this study based on similar evidence from a systematic review and recommendation by Ministry of Health, Uganda, DSM-5, and PHQ-9 diagnostic criteria.19,27,28 The PHQ-9 translated version in Uganda was used with Cronbach’s alpha 0.68. 26 PHQ-9 and other aspects of the questionnaire were read to all participants, and responses were recorded on the questionnaire by trained research assistants who were nurse counsellors.
In addition, we asked participants questions to elicit socio-demographic characteristics: age, marital status, level of education, employment status, number of children, and information about the partners. Community-, health-, and HIV-related characteristics included timing of HIV diagnosis, duration since HIV diagnosis, knowledge of partner’s HIV status, attendance of clinic visits, family support (this excluded information on the partner), partner support, adherence to antiretroviral medication, HIV status disclosure, and recent bad events (death, job loss, and debts). Obstetric and infant factors included parity, preparation for delivery, labor stress, mode of delivery, infant health, and breastfeeding. The structured questionnaire was also translated to Luganda by experienced translators to ensure consistency. The data were collected within a period of 4 weeks from March 2018 to April 2018.
Data analysis
Data were analyzed using Statistical Package for Social Sciences (SPSS) version 21. Descriptive statistical analysis was applied in analyzing the socio-demographic, obstetric health, and HIV-related characteristics.
For logistic regression analysis, mothers with PHQ-9 summated scores of ⩾10 were considered to have PPD. Independent variables found to be significantly associated with PPD with p values of less than 0.05 at bivariate were entered into a regression model for multivariable analysis. The strength of the association between PPD and the independent variables was assessed using odds ratios (ORs) and 95% CIs.
Ethical review
Approval to conduct the study was sought from the School of Health Science Research and Ethics Committee (SHSREC REF-2017-055) and permission to carry out the study at Mulago National Referral Hospital was sought from the Research and Ethics Committee (MHREC 1307). All participants were provided written informed consent.
Results
Socio-demographic, obstetric, and HIV-related characteristics of the participants
A total number of 290 participants were recruited for the study. The mean age of the participants was 27.06 (SD: ±5.15) with a range of 15–47 years, the majority (64.5%) of the participants were married and 2.4% were widowed. Almost half (44.2%) of the participants had no or low education and slightly more than half (57.6%) of the participants were not employed. More than half 178 (61.4%) of the mothers were diagnosed HIV positive for the first time during the last pregnancy. The duration since HIV diagnosis ranged from 0.25 to 21 years with a mean and median of 2.2 (SD: ±2.8) and 1 year, respectively. Participants had been on ART for an average of 1.9 (SD: ±2.1) years and missed less than one pill in the previous month (mean: 0.68 and SD: ±2.6), with a range of 0–21 missed pills. More than three-quarters (78%) of the participants rated their adherence to ART as excellent; the rest of the mothers rated their adherence between poor and fair. Almost all (95.5%) of the mothers were multiparous. Less than one-half (124, 42.7%) rated themselves as being prepared for delivery for their previous pregnancy. Some mothers (15.9%) reported having had difficulties in breastfeeding their infants. Participants had disclosed their HIV status to an average of two persons with a mean of 2.6 (SD ±3.1) and a range of 0–12. The details of other socio-demographic and obstetric characteristics are presented in Table 1.
Socio-demographic and obstetric characteristics of participants.
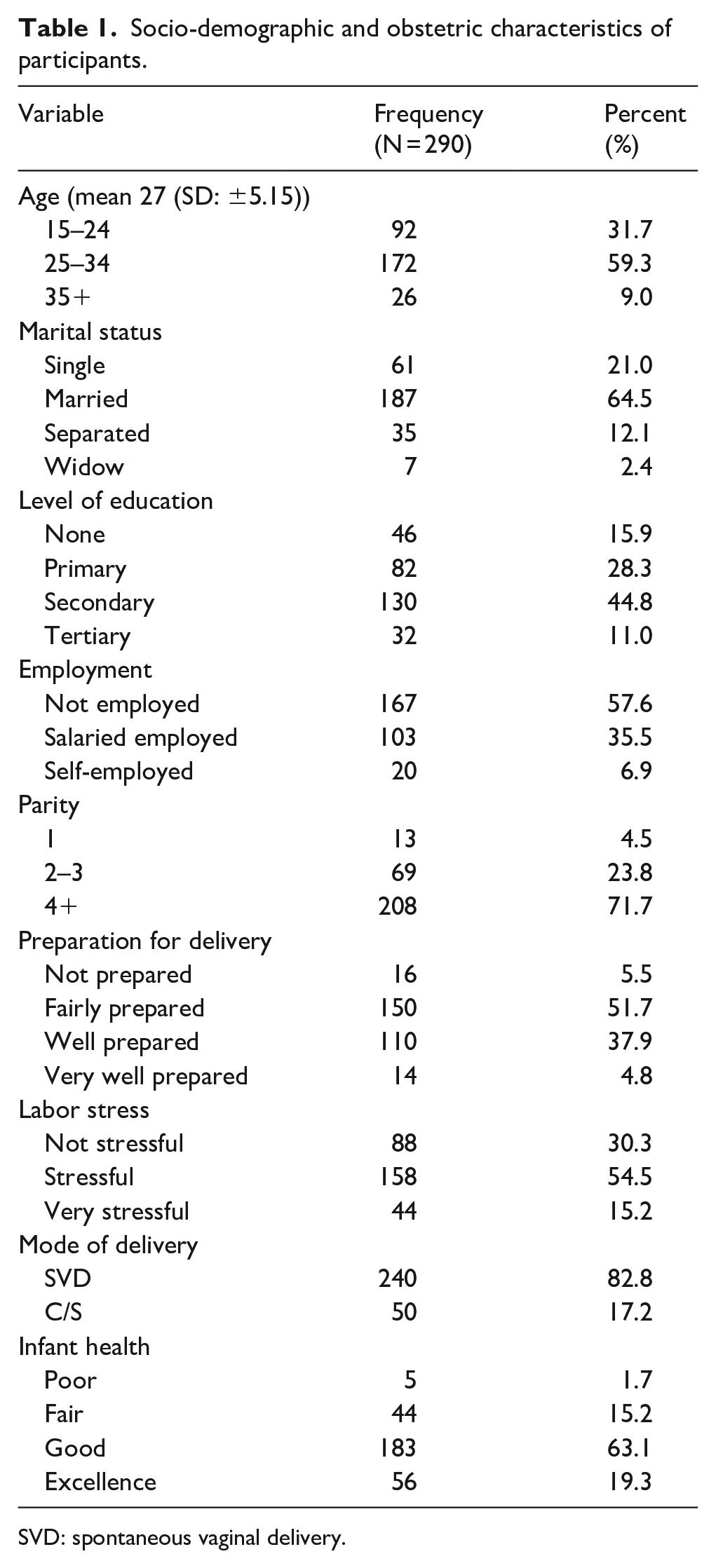
SVD: spontaneous vaginal delivery.
Community- and health facility–related characteristics
One-third (34%) of the participants travelled a distance of >5 km to access health services. About 1 in 10 (10.7%) of the mothers reported having had a recent bad event such as death, loss of job, or debt. More than 1 in 4 of the 86 (29.7%) mothers reported receiving poor social support from their partners, and the rest of the mothers reported receiving fair, good, or excellent support from partners. Other community- and health facility–related characteristics are reported in Table 2.
Community-, health facility–, and HIV-related characteristics of study participants.

ART: antiretroviral therapy.
PPD
Of the 290 mothers living with HIV who were interviewed, 46 (15.9%) had MDD, 90 (31%) had minimal symptoms of depression, and 154 (53.1%) had no symptoms of PPD. Although the prevalence was lower than the global estimate, postpartum mothers living with HIV who contributed to the 15.9% prevalence had major PPD that warranted prompt treatment. For logistic regression analysis, the 46 mothers who had major postpartum disorder were considered to have PPD, and the rest of the mothers (244) were considered to have no PPD. Following bivariate analysis, single mothers were more likely to have PPD (OR = 1.69, 95% CI: 1.48–5.36) compared to those who had partners. Sixty-six (76.7%) of the mothers had poor partner support, whereas 37 (18.1%) had good partner support. Single mothers were 13 times more likely to have poor partner support compared to mothers who were married (OR = 13.37, 95% CI: 7.06–25.32). Mothers who rated the health of their infants as poor had more than twice the odds of having PPD compared to those who rated the health of their children as good (OR = 2.25, 95% CI: 1.08–4.69). In addition, mothers who were not prepared for delivery were more likely to have PPD (OR = 2.40, 95% CI: 1.19–4.80) compared to those who were prepared for delivery. Knowledge of the HIV status of the partner was associated with reduced odds of having PPD (OR = 0.47, 95% CI: 0.24–0.90). Participants who had experienced a recent major bad event like death, loss of job, or debt were more than twice likely to have PPD (OR = 2.46, 95% CI: 1.05–5.74). Furthermore, mothers who rated support from partners as poor were five times more likely to have PPD compared to those who received good partner support (OR = 5, 95% CI: 2.56–9.66). Refer to Table 3 for detailed bivariate analysis results. After controlling for socio-demographic, HIV-related, and obstetric variables, mothers who reported partner support as poor were more than four times more likely to have PPD compared to those who reported having fair or good partner support (adjusted odds ratio (AOR) = 4.52, 95% CI: 2.31–8.84).
Factors associated with postpartum depression at bivariate analysis.
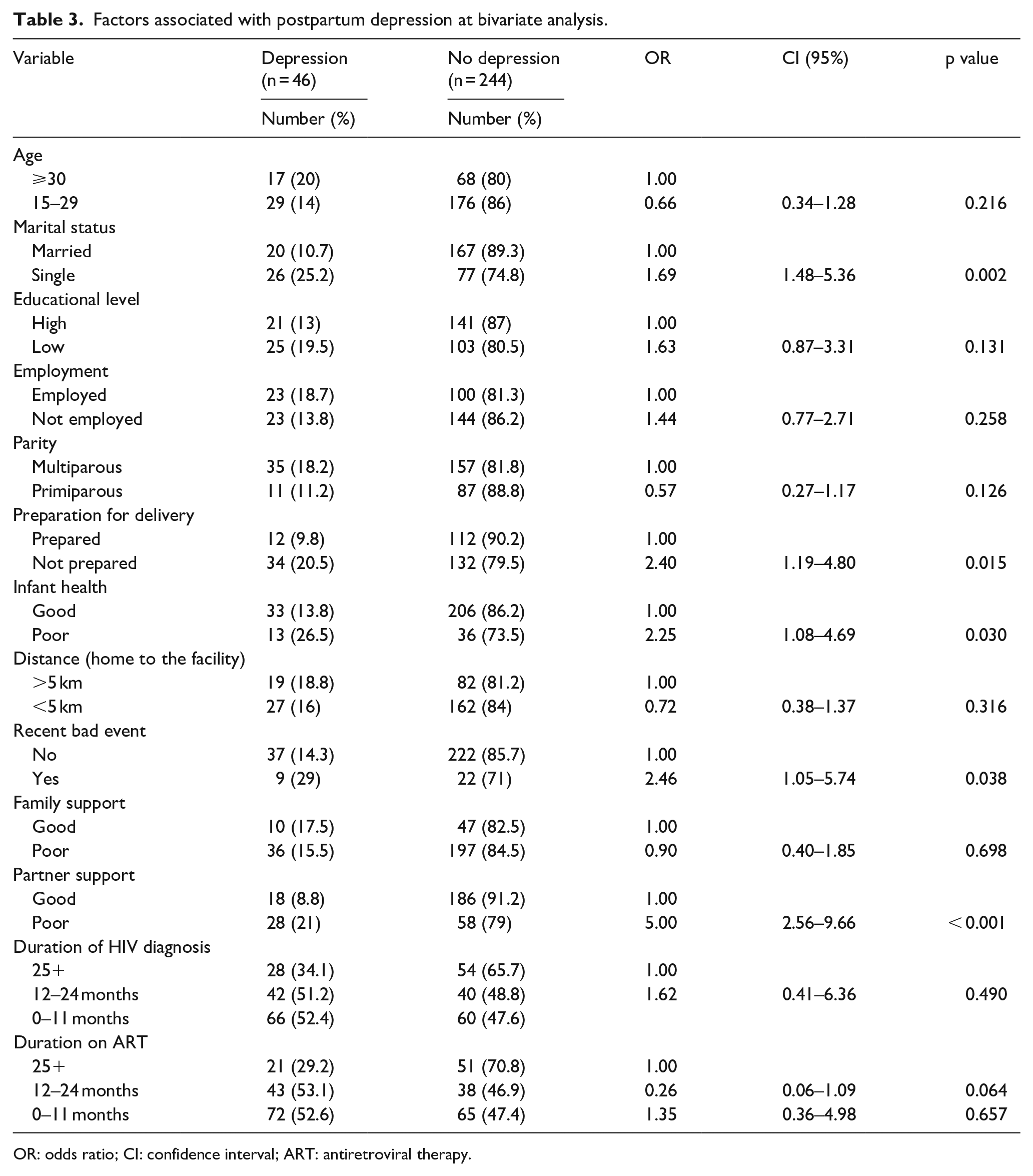
OR: odds ratio; CI: confidence interval; ART: antiretroviral therapy.
Discussion
PPD is a major health issue for mothers as well as their families. The general objective of this study was to determine the prevalence and factors associated with PPD among mothers living with HIV. The prevalence of major depression was 15.9%, while 31% had minor depressive symptoms. The factor that was associated with PPD was poor partner support.
The prevalence of PPD varies considerably among different studies. In the population of mothers living with HIV, evidence across different settings reported both high and low prevalence in the postpartum period due to numerous challenges encountered by mothers in the period after birth. The prevalence in this study is lower than the prevalence reported in studies conducted in Kenya, South Africa, and Zimbabwe.10,14 This low prevalence could be attributed to the use of a diagnostic tool that measured major or severe PPD according to Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM-IV) and only considered mothers with major symptoms lasting for 2 weeks. Other studies which measured major or severe depression using PHQ-9 also reported lower prevalence.29,30 The percentage of postpartum mothers experiencing major or severe PPD could be credited to difficulties encountered by a mother living with HIV such as caring for the infant, following diagnosis, and ART treatment for herself and the baby. Some studies conducted in Kenya reported the prevalence of PPD among women living with HIV to be 48% and 25.2%.9,14 Similar studies conducted in different settings across Africa reported prevalence ranging from 11% to 35.6% in the population of postpartum mothers living with HIV.8,10,11 These studies employed different tools to measure PPD which might have accounted for the difference. The disparity may also be attributed to the differences in population, geographic location, and the definition of PPD.
The low prevalence could be an indicator that mothers in HIV care are adapting well as a result of counselling which is offered as a part of HIV care and support.5,31
On the contrary, the disparity in the prevalence could be a result of the varying degree of support given to mothers during the period postpartum. In the past, elderly and experienced women would take care of women during the postpartum period. However, the practices have changed due to rural–urban migration which poses economic and distance constraints for postpartum mothers.32,33
In this study, mothers living with HIV who rated the support from their partners as poor had higher odds of having an MDD. Mothers with poor partner support were almost five times more likely to be depressed during the postpartum period compared to those who had good partner support (AOR = 4.52, 95% CI: 2.31–8.84, p value < 0.001). In this scenario, the partner is the source of support in various ways such as finances, emotional support, and physical support. The result is consistent with findings from studies conducted in Saudi Arabia and the United States.34,35
Partner support in terms of finances in the home is compromised by polygamous or multiple partnerships leading to poor or inadequate support from the male partner, and this results in stress and possible maternal depression. 5 Making decisions together by partners is a pillar in communication that strengthens the relationship; joint decision-making by couples has been reported to be better than a decision taken alone. 36 This provides emotional support for the mother in the postpartum period and improves communication in the relationship. 37 Partner involvement is a very important factor for women’s health-seeking behavior in the postpartum period; in fact, in many SSA settings, men control household resources and often make critical decisions that affect maternal health including the choice of health services. 38 According to a study conducted in Zambia, mothers who had their partners accompanying them during antenatal care delivered at a health facility and attended their postnatal visits regularly. 39 Partner support during pregnancy and the postpartum period offers the mother a sense of belonging and acceptance by the partner. This improves the mother’s overall emotional and physical well-being, thus improving infant well-being.40,41 Partner support should be encouraged to decrease the occurrence of PPD; therefore, interventions in the pregnancy and postpartum period should target couples to encourage them to be involved in the care of the mother and infant to improve maternal and infant health as well as HIV care outcomes.
Limitation of the study
The study was conducted at the hospital; hence, generalization to the entire population becomes difficult. The cross-sectional nature of the study allowed mothers to be assessed for PPD once; hence, future studies can adopt longitudinal studies to assess PPD at various points in time. The wide time range for assessing PPD after delivery might have affected mothers’ experiences of PPD. The quantitative nature of this study did not allow exploration of partner support; hence, focused discussion can be employed in later studies.
Conclusion
The study showed a prevalence of PPD among HIV-infected mothers of 15.9% which was slightly lower than the global estimate of 19.8% in developing countries. The single associated factor of PPD among HIV-infected mothers was poor partner support. The involvement of partners in the care of HIV-infected postpartum mothers will promote the health of mothers psychologically and emotionally and by default the health of the infants. To achieve optimal maternal and infant HIV outcomes, periodic depression screening during the postpartum period should be encouraged to facilitate early diagnosis and treatment.
Footnotes
Acknowledgements
This work was supported by the NURTURE: Research Training and Mentoring Program for the career development of junior and mid-level faculty at Makerere University College of Health Sciences and the School of Nursing at Johns Hopkins University. The authors wish to acknowledge the contribution of all the study participants who made this work possible.


