Abstract
Despite the known benefits of breastfeeding for both infant and mother, clinical support for problems such as benign inflammation of the lactating breast remain a research frontier. Breast pain associated with inflammation is a common reason for premature weaning. Multiple diagnoses are used for benign inflammatory conditions of the lactating breast which lack agreed or evidence-based aetiology, definitions, and treatment. This article is the second in a three-part series. This second review analyses the heterogeneous research literature concerning benign lactation-related breast inflammation from the perspectives of the mechanobiological model and complexity science, to re-think classification, prevention, and management of lactation-related breast inflammation. Benign lactation-related breast inflammation is a spectrum condition, either localized or generalized. Acute benign lactation-related breast inflammation includes engorgement and the commonly used but poorly defined diagnoses of blocked ducts, phlegmon, mammary candidiasis, subacute mastitis, and mastitis. End-stage (non-malignant) lactation-related breast inflammation presents as the active inflammations of abscess, fistula, and septicaemia, and the inactive condition of a galactocoele. The first preventive or management principle of breast inflammation is avoidance of excessively high intra-alveolar and intra-ductal pressures, which prevents strain and rupture of a critical mass of lactocyte tight junctions. This is achieved by frequent and flexible milk removal. The second preventive or management principle is elimination of the mechanical forces which result in high intra-alveolar pressures. This requires elimination of conflicting vectors of force upon the nipple and breast tissue during milk removal; avoidance of focussed external pressure applied to the breast, including avoidance of lump massage or vibration; and avoidance of other prolonged external pressures upon the breast. Three other key preventive or management principles are discussed. Conservative management is expected to be effective for most, once recommendations to massage or vibrate out lumps, which worsen micro-vascular trauma and inflammation, are ceased.
Keywords
Introduction
Breast pain is one of the most common reasons women give for premature weaning.1,2 Despite the known benefits of breastfeeding for both infant and mother, clinical interventions for problems such as breast inflammation and pain remain a research frontier.3,4
Multiple diagnoses are popularly used for benign lactation-related breast inflammation (BLBI), including blocked ducts, phlegmon, mammary candidiasis, subacute mastitis, subclinical mastitis, mastitis, and white spots. Yet these diagnoses lack agreed or evidence-based aetiological models, definitions, and treatment. The first article in this three-part series on lactation-related breast inflammation synthesizes the latest evidence concerning the functional anatomy and physiology of the lactating breast, the mechanobiology of lactation, the mammary gland immune system including the human milk microbiome and somatic cells, and interactions between these to propose a mechanobiological model of BLBI. 5
Despite European and other dissent over the past two decades, English-language clinical protocols are built on a pathogenic microbiota model of BLBI, which is shown in the first article of this series to be inconsistent with emerging research. The pathogenic model has resulted in overtreatment with antibiotics and antifungal medications, even as the World Health Organization urgently calls for antimicrobial stewardship. 6 Overuse of medical, surgical, and pharmaceutical interventions is an increasingly serious international problem in healthcare more broadly.7,8 Both patients and clinicians typically overestimate the benefits of medical interventions and underestimate potential harms.9–11 It is therefore not surprising, given the relative lack of research into clinical breastfeeding support, that over-medicalization and overtreatment are significant problems in the care of breastfeeding women and their babies, including when BLBI emerges.12–18
This second article in the three-part series analyses the heterogeneous research literature concerning BLBI from the perspectives of the mechanobiological model and complexity science, to re-think classification, prevention, and management of lactation-related breast inflammation. Ethical approval has not been required since this is a theoretical investigation. The third article addresses aetiology, classification, prevention, and management of lactation-related inflammation of the nipple–areolar complex. 19
Postpartum breast cancer has a higher risk of metastatic spread relative to other forms of breast cancer. Axillary and supraclavicular lymph node examination is required when lactating women present with a breast lump. A persistent lactation-related breast lump requires ultrasound imaging and further investigation as indicated, to exclude malignant inflammatory masses or benign non-lactation-related masses. The period of time a new lump in a lactating breast can be observed before investigation depends on clinical context but threshold for imaging should remain low. 20 Please see the Academy of Medicine Clinical Protocol #30: Breast Masses, Breast Complaints, and Diagnostic Breast Imaging in the Lactating Woman for clinical guidelines on identification, differential diagnoses, and management of conditions in the lactating breast that are not BLBI. 20
The complex systems approach to lactation-related breast inflammation detailed in this three-part series forms part of the breastfeeding domain of the programmes known as Neuroprotective Developmental Care (NDC or ‘the Possums programs’), developed and delivered in Australia since 2011. NDC synthesizes the evidence concerning early-life care across the domains of breastfeeding, cry-fuss problems, infant sleep, and parental mood by applying the theoretical frames of evolutionary biology and complexity science, translating this evidence into clinical practice.12,17,21–34 From an evolutionary perspective, breastfeeding is foundational to, and interacts with, each other domain.
A complex systems approach to classification of acute and end-stage non-malignant lactation-related breast inflammation
The interaction between networks of inflammatory signals within the lactating mammary gland immune system, triggered by high intraluminal pressures, determines the severity of breast inflammation, its duration, and resolution. 35 The popular diagnoses of blocked or plugged ducts, phlegmon, subacute mastitis or mammary dysbiosis, and mastitis are poorly defined and have overlapping presentations. This article proposes a classification system using two main categories.
The first, BLBI, is described clinically by relevant presenting signs and symptoms (Table 1). Engorgement, a diagnosis with clearer and agreed definition, is conceptualized as a bilateral and generalized presentation of BLBI. From a complexity science perspective, BLBI emerges out of multiple interacting systems, and is located along a spectrum of severity, spread, and systemic involvement.36,37 When BLBI becomes more severe, whether localized or generalized, systemic responses of fever, myalgia, and rigour are triggered.
Clinical signs and symptoms used to classify acute and end-stage non-malignant lactation-related breast inflammation.

The second category is end-stage non-malignant inflammation of the lactating breast, with sub-categories of active or inactive. Three well-defined diagnoses fall within the category of active, end-stage, non-malignant inflammation of the lactating breast: abscess, fistula, and septicaemia. Although non-malignant, these conditions are also not benign. An abscess is described by selecting the relevant presenting signs and symptoms of lactation-related breast inflammation as detailed in Table 1.
The diagnosis of galactocoele, also well-defined, sits within the category of inactive, end-stage, non-malignant or benign inflammation of the lactating breast. A galactocoele is also described by the relevant presentation of signs and symptoms as detailed in Table 1.
Clinical principles of prevention and management of BLBI
Emerging evidence supports the hypothesis that avoidance of or elimination of very high intra-alveolar and intra-ductal pressures is the fundamental strategy for the prevention of or downregulation of lactation-related breast inflammation, as detailed in the first article of this series. 5 Implementation of the following two key clinical strategies for stabilization of intraluminal pressure is expected to optimize the resilience of the interconnected and interacting intramammary immune systems.
Frequent flexible removal of milk (Table 2).
Elimination of mechanical forces that cause high intraluminal pressures:
a. Avoidance of conflicting vectors of force upon the nipple and breast tissue during milk removal, which compress lactiferous ducts (and also cause nipple pain, inflammation, and damage);5,12,19,27,28 b. Avoidance of micro-vascular trauma in breast stroma, for example, by lump massage, which increases stromal tension, worsening compression of lactiferous ducts;
5
c. Avoidance of prolonged external pressure on the breast, for example, tight bra or breast shells, which may compress lactiferous ducts.
5
In addition, applying the mechanobiological model, another three principles are expected to prevent or treat BLBI.
3. Wean or downregulate milk production gradually.
4. Avoid increasing milk production beyond an infant’s physiological need.
5. Gentle manual movement of the breasts.
The Neuroprotective Developmental Care (NDC) model of frequent and flexible breastfeeding. 31

Principle 1: frequent flexible milk removal
The most fundamental step in either prevention or the clinical management of BLBI across the spectrum of presentations is to support mammary immune resilience by repetitive contraction of alveoli and dilation of lactiferous ducts, combined with vacuum milk removal (Table 2). Hand expression of milk also results in contraction of alveoli and dilation of lactiferous ducts.
Building on the mechanobiological aetiological model of BLBI, this article hypothesizes that frequent irregular alveoli contractions and ductal dilations concomitant with milk removal by vacuum counter the compressive effects of high stromal tension, and downregulate the inflammatory cascade caused by the mechanical effects of ductal occlusion and milk backpressure. Frequent contraction of the alveoli and ductal dilations create asynchronous and highly irregular waves or vibrations of pressure gradients within the stroma, acting as a stromal pump that promotes venous and lymphatic flow. 5
A woman with BLBI is advised that milk needs to be removed from the affected breast very frequently and flexibly. NDC principles of frequent and flexible breastfeeding are detailed in Table 2. 31
Principle 2: elimination of mechanical forces that cause high intraluminal pressures
Eliminate conflicting intra-oral vectors of force during milk removal, which cause lactiferous duct compression
The gestalt biomechanical model of infant sucking proposes that multiple factors relating to the fit between the baby’s and mother’s anatomies may create intra-oral vectors of force that conflict with the direction of vacuum generated by mandibular depression, resulting in ‘breast tissue drag’, which is associated with
Nipple discomfort, pain, or damage;
Unsettled infant behaviour at the breast;
The tongue is a muscular hydrostat, which changes shape without changing volume. 45 In the gestalt model, the tongue is conceptualized as a supple, adaptive organ that dynamically responds to and moulds around available intra-oral nipple and breast tissue, rather than as a forcible driver of nipple compression and nipple shape. Elimination of conflicting vectors of force intra-orally (that is, elimination of ‘breast tissue drag’) allows peak vacuum to achieve optimal intra-oral breast tissue volume.12,27,28
With maximum intra-oral breast tissue volume, the mechanical force of the vacuum defuses over the largest possible epithelial area of the nipple–areolar complex and breast skin. The gestalt biomechanical model of breastfeeding proposes that bending and stretching mechanical loads compress lactiferous ducts in the glandular tissue, which are predominantly located directly behind the nipple, and which have been demonstrated in ultrasound studies to compress under even very light touch.46,47 Two-thirds of alveolar glandular tissue lies within a 3 cm radius of the nipple. Combined with frequent and flexible breastfeeds, optimal intra-oral breast tissue volume and elimination of breast tissue drag result in optimal milk transfer, infant satiety, and weight gain.12,27,28,48–52
Because clinical breastfeeding support remains a research frontier, interventions women typically receive for fit and hold (also known as latch and positioning) are based upon experience or opinion.3,53–56 For example, the popularly used cross-cradle hold with the other hand shaping the breast has been shown to increase the risk of nipple pain. 57 When women are taught ‘laid-back breastfeeding’ or ‘biological nurturing’ in hospital immediately after the birth (also known as skin-to-skin or the physiologic initiation of breastfeeding), the incidence of nipple pain and damage decreases modestly.58,59 A 2021 Chinese randomized controlled trial (RCT) of 504 pairs demonstrated that implementing baby-led self-attachment from birth results in a 12% increase in exclusive breastfeeding at day 3, and an 8% and 5% decrease in the number who reported nipple pain at 3 days and 3 months postpartum, respectively. 60 However, baby-led approaches have not been demonstrated effective as clinical interventions for breastfeeding problems. A 2013 Swedish RCT of 103 mothers with infants up to 16 weeks of age with severe latch-on difficulties found that a baby-led or skin-to-skin intervention did not increase the likelihood that the infant would latch on. 61 Remarkably, breast tissue drag and positional instability remain omitted variables in almost all clinical breastfeeding research.12,62
The gestalt method integrates laid-back breastfeeding or biological nurturing with the gestalt biomechanical model of infant suck derived from ultrasound, vacuum, and MRI studies.12,28 In a small case series, the gestalt method changed intra-oral nipple position, increased intra-oral nipple and breast tissue dimensions, and decreased nipple slide in mother–baby pairs who had already received comprehensive lactation support. 28 These preliminary results support the hypothesis that the gestalt method increases intra-oral breast tissue volume by eliminating conflicting vectors of force, and that standard fit and hold interventions may not optimize intra-oral breast tissue volume.12,27,28
Avoid focussed deep pressure on the breast
Lactating women with new breast lumps are commonly advised to engage in lump massage, to massage out the hypothesized blockage in the duct caused by sticky milk, a biofilm, or lactolith. However, the first article in this series demonstrates that there is no evidence to support the hypothesis that lumps are caused by blocked or plugged ducts in this way, and proposes that lump massage causes micro-vascular trauma and haemorrhage in the tense, highly vascular stroma of an inflamed lactating breast. Increased stromal swelling places further pressure on ducts, worsening intraluminal backpressure and inflammation. For this reason, lump massage and vibration risk worsened inflammation, tissue necrosis, and abscess. 5
Avoid other prolonged external pressures on the breast
From the perspective of the mechanobiological model, external applications of pressure on the lactating breast increase the risk of breast inflammation due to mechanical compression of ducts, which increases upstream intra-ductal and intra-alveolar backpressure over time. 5 For this reason, the following should be avoided:
Therapeutic breast massage or manual lymphatic drainage (Appendix 1).
Squeezing, shaping, or compressing the breast during breastfeeds.
Finger on breast to prevent breast occluding infant’s nostrils.
Restrictive or ill-fitting bra or garment.
Sleeping with pressure on the breast.
Bruising of the breast, for example, from an infant’s kick.
Use of mechanical pump in a way that places asymmetric pressure or drag on breast tissue.
Positioning infant with chin or nose pointing towards area of inflammation (which misunderstands biomechanics of milk transfer and risks worsened breast tissue drag).
Principle 3: wean or downregulate milk production gradually
Sudden cessation of breastfeeding or milk removal may result in widespread alveolar rupture across the mammary glands, precipitating an exaggerated and whole-of-breast inflammatory response and the emergence of localized clinical inflammation.63,64 Mammary glands from mice which underwent abrupt involution exhibited higher levels of mammary gland inflammation and cell proliferation, resulting in denser stroma and altered collagen composition. 65 A woman wishing to downregulate her supply or wean her infant needs to do this gradually to avoid BLBI, calibrating her breasts’ response.
Principle 4: avoid increasing milk production beyond an infant’s physiological need
When a lactating woman removes her milk mechanically, that is, by pumping, it is important she pumps physiologically. A cohort study of 346 breastfeeding women by Cullinane et al. 66 in 2015 showed that breastfeeding women who pumped a few times a day were at increased risk of mastitis. (Reasons for pumping were not investigated). 66 From the mechanobiological perspective, milk production which exceeds the infant’s needs increases the risk of excessively high intraluminal pressures and BLBI.
Principle 5: gentle manual movement of breasts
NDC applies an evolutionary lens to propose that in Homo sapiens’ environment of evolutionary adaptedness, the lactating mammary gland was subject to constant and irregular movement, occurring in the context of frequent and flexible breastfeeds. Breast position and conformation shifted in relation to gravity repeatedly throughout the day, including with tissue vibration in response to rapid fall or movements of the breast in space. Bras and more sedentary lifestyles limit breast movement and tissue vibration and may create areas of the breast, depending on breast shape and bra fit, which are exposed to minimal or no movement in response to gravity.
The breast massage popularly referred to as ‘breast gymnastics’ requires lifting and gently moving the breasts in various directions, often with circular movements using the whole of the hand or palm laid gently over the breast. This form of breast massage may have a role, either preventatively in very early lactation or in the context of BLBI. 67
There is no reason to routinize this kind of breast care or to outsource it to a masseuse, since the woman herself can gently move her own breasts as often as she wishes, which may stimulate milk ejections, being careful not to cause discomfort or pain, which may flag the risk of microvascular trauma. Upper limb stretching movements that engage the pectoral muscles may similarly help move the breast tissue, and also do not require burdensome exercise prescriptions or outsourcing.
Commonly recommended strategies for management of BLBI, which have been demonstrated as ineffective, or which lack scientific rationale, are detailed in Table 3. Appendix 1 discusses why therapeutic breast massage of lactation or manual lymphatic drainage are not appropriate treatments for BLBI.
Strategies for management of benign lactation-related breast inflammation, which have been demonstrated as ineffective or lack scientific rationale.
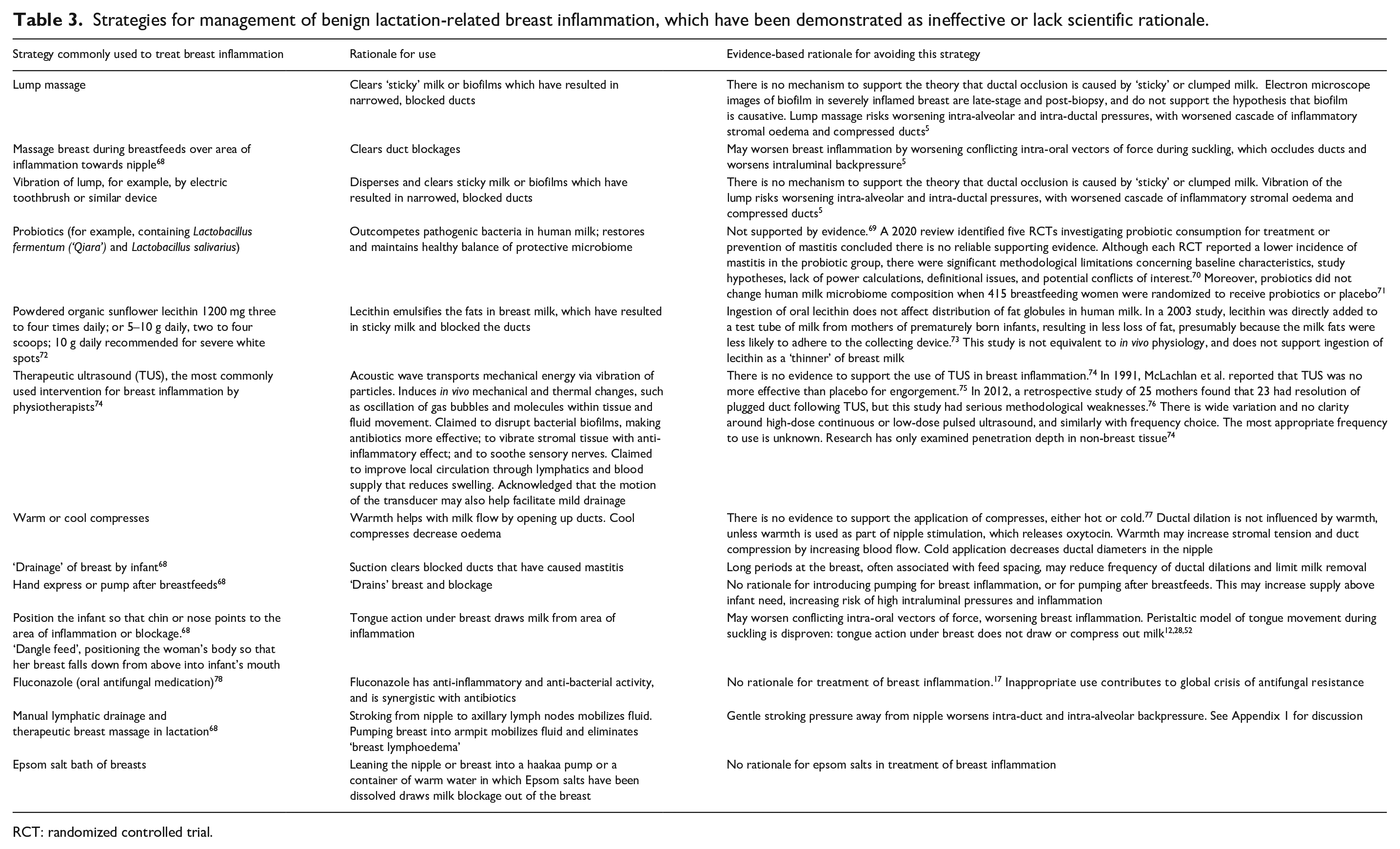
RCT: randomized controlled trial.
BLBI: engorgement
What is engorgement?
Engorgement is a bilateral, generalized BLBI, which like all breast inflammation occurs along a spectrum of severity and is most accurately described by the relevant selection of presenting signs and symptoms shaded in Table 4. It is estimated that between one- and two-thirds of new mothers experience engorgement. 79
Spectrum of signs and symptoms of benign lactation-related breast inflammation commonly referred to as engorgement (shaded).

In the first 5 days post-birth, secretory activation of lactation may be associated with painful, hot, distended breasts. This is often referred to as ‘the milk coming in’. Symptoms occur after (not concomitant with) measurable indicators of the onset of transitional milk secretion, such as increased lactose and decreased sodium content.80,81 Post-birth engorgement has been theorized as overshoot or temporary excess in initial milk synthesis, since Homo sapiens have the potential to produce milk multiple factors above the milk needs of a singleton infant. 82 Although a physiological engorgement is sometimes differentiated from BLBI engorgement, they are both on the spectrum of BLBI. If an infant is offered frequent flexible feeds from birth, with generous skin-to-skin opportunities, and if underlying clinical problems like conflicting intra-oral vectors of force or restrictive feeding practices are resolved, the rate of postpartum milk synthesis quickly upregulates or downregulates to meet the infant’s needs. 5 In the context of underlying clinical breastfeeding problems or restrictive feeding practices, engorgement may develop into intense, localized areas of inflammation (commonly referred to to produce milk as mastitis), poor supply, and premature introduction of formula. 83
Although engorgement is commonly referred to as ‘oedema’, this article argues this is likely inaccurate. Applying the mechanobiological model and a complex systems perspective, uncomfortable or painful, hot, bilateral distension of the breasts is due to:
High intraluminal pressures of milk, which trigger an inflammatory cascade.
Hyperaemia (capillary dilation).
Leakage of proteins from the widened junctions of the venules, which osmotically increase interstitial fluid and associated stromal tension.
Lymphatic capillary dilation triggered by increased interstitial fluid. 5
Ultrasound analysis shows that main lactiferous ducts are immediately behind the nipple. Peri-areolar engorgement is likely to be as much or mostly due to severely engorged superficial milk ducts and hyperaemia, rather than interstitial fluid. 49
A 2015 observational study of 20 postpartum women found that those who received intrapartum intravenous fluids had more breast swelling and tenderness in the first 10 days. 84 Intravenous fluids are likely to cause increased interstitial fluid of the breasts only if the woman has received a significant dose, which also results in lower limb oedema.
After the first week post-birth, engorgement is typically due to compromised milk removal and restricted breastfeeding practices. It may be associated with damaged nipples, which are a marker of conflicting intra-oral vectors of force during breastfeeding. Engorgement risks low supply (due to alveolar rupture and involution) and premature introduction of formula or weaning. 83 This is because the mechanical load of high intra-alveolar and intra-ductal pressures and microscopic areas of alveoli rupture trigger the wound-healing environment of inflammation, microscopic involution, and downregulation of milk synthesis. These mechanisms are discussed in detail in the first article in this series. 5
How is engorgement treated?
Zakarija-Grkovic and Stewart’s 2020 Cochrane review shows that commonly recommended interventions for engorgement, including cabbage leaves, compresses, acupuncture, Gua Sha, therapeutic ultrasound, enzyme therapy, and massage are not demonstrated to be efficacious. 83
From the perspective of the mechanobiological model, the previously detailed Principles 1–5 of management of BLBI, in particular, frequent flexible access to the breast from birth (Table 2) and elimination of conflicting intra-oral vectors of force when the infant is suckling, are essential for the prevention or management of engorgement.
Anecdotally, reverse pressure softening of the areola may be helpful if an infant has difficulty coming on due to engorgement. 85 Reverse pressure softening is application of a gentle but firm pressure for 20 s or so on the areola with fingertips, which disperses milk. 85 Manual expression is not as effective as vacuum for milk removal but has a place in the management of BLBI for some women. Manual expression risks backpressure and micro-vascular trauma and should be used carefully, without causing pain. Gentle manual expression applies sensory stimulation to the breast and nipple, using positive pressure to press residual milk in the ducts towards the nipple. This triggers the alveolar contractions and ductal dilations of milk ejection. 5 Safe manual expression technique is described in a freely available short video https://possumsonline.com/video/handexpression.
BLBI: ‘blocked ducts’ or ‘plugged ducts’
What are blocked or plugged ducts?
Healthy lactating breasts are often lumpy, but these lumps decrease or disappear after breastfeeds. A blocked or plugged duct has been described as a palpable lump, which develops gradually, sometimes associated with localized tenderness or pain in the absence of erythema or systemic symptoms, and which may persist for a week or two. There is no prevalence data.
As discussed in detail in the first article of this series, there is no evidence to support the hypotheses that lactiferous ducts are blocked by sticky milk due to increased fatty acids or fat globules or by biofilm, causing obstruction and inflammation. Electron microscope images of biofilm in severe breast inflammation are late-stage and post-biopsy, and do not support the hypothesis that biofilm is causative. 5 This article proposes that a new non-erythematous lump in a lactating breast is most accurately referred to as BLBI, described by relevant signs and symptoms shaded in Table 5.
Spectrum of signs and symptoms of benign lactation-related breast inflammation commonly referred to as blocked or plugged ducts (shaded).

How is BLBI (commonly referred to as blocked or plugged ducts) treated?
From the perspective of the mechanobiological model, the previously detailed principles of management of BLBI, in particular, frequent flexible milk removal (Table 2) and elimination of conflicting intra-oral vectors of force when the infant is suckling, should be applied as treatment for the signs and symptoms shaded in Table 5. Lump massage or vibration risks worsened inflammation and abscess and should not be applied.
BLBI: ‘phlegmon’
What is a phlegmon?
A phlegmon has been described as a poorly defined indurated lump in the breast of a lactating woman, though the term lacks clear definition and is usually a diagnosis made by imaging investigation. The term is extrapolated from the surgical literature, where inflammatory masses referred to as phlegmons may surround a hollow organ, for example, in appendicitis and diverticulitis. On ultrasound imaging, a phlegmon may appear as an irregular and heterogeneous inflammatory mass, often containing some visible fluid in the tissues. There is no prevalence data. 20
This article proposes that there is no role for the term phlegmon, but that lactation-related breast lumps which have been determined to be benign are best described clinically according to presenting signs and symptoms on the spectrum of inflammation, shaded in Table 6.
Spectrum of signs and symptoms of benign lactation-related breast inflammation commonly referred to as phlegmon (shaded).

How is BLBI (commonly referred to as phlegmon) treated?
From the perspective of the mechanobiological model, the previously detailed principles of management of BLBI, in particular, frequent flexible milk removal (Table 2) and elimination of conflicting intra-oral vectors of force when the infant is suckling, should be applied for the signs and symptoms shaded in Table 6. Lump massage or vibration risks worsened inflammation and abscess and should not be applied.
BLBI: ‘subacute mastitis’ or ‘mammary dysbiosis’
What is subacute mastitis or mammary dysbiosis?
There is no agreement on the definition of subacute mastitis, also referred to as subclinical mastitis and mammary dysbiosis, and no prevalence data for these diagnoses.
In 2018 Kaski and Kvist
86
observed, As there is no clear scientific consensus on the definition of human lactational mastitis it would seem incautious to introduce the term ‘subacute mastitis’: there is certainly no clear definition of what this condition might entail in humans. Also, it would be difficult to prescribe treatment for a condition that has not yet been scientifically described and classified. . . . Treatment of a ‘subacute’ condition should not be recommended as it may be of little value to the individual and might be of great detriment to the global community.
Proponents of the diagnosis of subacute mastitis or mammary dysbiosis apply the pathogenic microbiota model of breast inflammation to hypothesize that bacterial overgrowth or mammary dysbiosis creates sticky milk or biofilm, which causes plugging of ducts, indurated painful areas, decreased milk synthesis, pain with latch, and increased risk of mastitis.87–89 Subacute mastitis or mammary dysbiosis has also been characterized as an inflammatory condition, which does not present with systemic illness or localized breast erythema. 90 Signs and symptoms attributed to subacute mastitis or mammary dysbiosis are detailed in Table 7. Because of hypothesized bacterial biofilm formation, 2–6 weeks of treatment with antibiotics is typically recommended (cephalosporin, amoxicillin/clavulanate, or dicloxacillin, or erythromycin).68,72,78,88,91 Some clinicians also hypothesize that women with symptoms of mammary dysbiosis are more likely to develop nipple blebs or white spots, proposing that white spots are distal extensions of biofilm.72,87,88 The third article of this series also contests this pathogenic bacterial or biofilm hypothesis concerning aetiology of white spots. 19
Spectrum of signs and symptoms of benign lactation-related breast inflammation commonly referred to as subacute mastitis or mammary dysbiosis (shaded).

A review of the research finds the following:
The diagnosis of subacute mastitis derives from the dairy industry. 86
The condition popularly diagnosed as nipple thrush or mammary candidiasis is not supported by evidence. Candida albicans is commonly present in the healthy human milk microbiome. Antifungals or antibiotics have not been demonstrated to resolve the symptoms of painful nipples and deep stabbing or radiating breast pain between breastfeeds, commonly diagnosed as mammary candidiasis or subacute mastitis. 17
The concept of dysbiosis is increasingly contested by human microbiome researchers, since it inaccurately assumes a comparative state of eubiosis against which to determine dysbiosis.5,92,93
Identification of Staphylococus and Streptococcus in human milk does not imply causation of pathology. 94 The hypothesis that pathogenic biofilms develop in mammary ducts, cause breast inflammation, and resist medication penetration is not supported by evidence. Electron microscopy images of biofilm in severe breast inflammation are late stage and post-biopsy, and do not support this causative hypothesis. 5 Lactating women may occasionally express cords of inspissated milk in the context of BILBI and this may also very occasionally contain mucinous solid. This more solid expressed milk and products would be expected to contain very high leukocyte and epithelial counts, and perhaps secondary biofilm formation. There is nevertheless no reason to believe that intra-ductal biofilm formation is causative.
In a 2021 audit of medical records in a breastfeeding medicine clinic, 8 of 25 patients who were unsuccessfully treated with antifungals prior to presentation were diagnosed with subacute mastitis. Betts et al. 87 observed that for seven of these patients, symptoms resolved within 42 days on a 4–6-week course of a macrolide antibiotic; in the eighth case, with probiotic treatment. Unfortunately, this retrospective review is methodologically flawed and does not support the validity of the diagnosis of subacute mastitis nor the efficacy of the antibiotics. 87
Proponents of the diagnosis of subclinical mastitis argue that it is an asymptomatic inflammatory condition of the lactating mammary gland caused by lactocyte tight junction leakage, linked to early lactation failure and poor infant weight gain and which may progress to clinical mastitis. In 2020, Samuel et al. 90 diagnosed 40% of 305 breastfeeding mothers at day 2 post-birth with subclinical mastitis, decreasing to 10% at day 17, and 5% at day 30. Samuel et al. 90 applied a milk sodium to potassium ratio greater than 0.6 as diagnostic.
The authors found that their diagnosis of subclinical mastitis was associated with lower lactose levels, changes in fatty acid, mineral, and trace element composition, and elevated interleukin and inflammatory proteins. They found more subclinical mastitis in mothers who underwent caesarean, attributing this to delay in breastfeeding leading to breast engorgement and inadequate breast emptying. They found no difference in infant milk intake, number of feeds per day, infant weight gain, and head circumference between those diagnosed with and those without subclinical mastitis. The authors observed that the prevalence of subclinical mastitis varied remarkably across countries, proposing that it was impacted by differences in breastfeeding practices. 90 But in this and earlier related studies, subclinical mastitis remains very poorly defined.
This article argues that Samuel et al. do not, in fact, demonstrate the existence of a clinically relevant diagnosis, subclinical mastitis. Their study does, however, demonstrate that multiple factors related to inflammation are identifiable in human milk, and corroborates the central hypothesis in this three-part series, that subclinical pro-inflammatory feedback loops downregulate milk secretion within the multiple complex systems of the lactating breast, impacted by both biological and behavioural factors. The interindividual variability of pro-inflammatory factors in mothers’ milk reflects the impact of multiple genetic and clinical breastfeeding factors on intra-luminal pressures, including frequency of feeds and conflicting intra-oral vectors of force during suckling. For example, the transition from colostrum to transitional to mature milk occurs at different rates between women depending on myriad factors. There may be an excess of milk secretion relative to the infant’s needs post-birth, which downregulates in response to mechanobiological factors through a pro-inflammatory response, or milk secretion may downregulate due to restrictive feeding practices or compromised poor milk transfer. These same multiple factors will have an impact on infant weight gain and introduction of formula.
How is BLBI (commonly referred to as subacute mastitis or mammary dysbiosis) treated?
This article proposes that there is no role for the diagnoses of subacute mastitis, mammary dysbiosis, or subclinical mastitis. From the perspective of the mechanobiological model, the previously detailed principles of management of BLBI, in particular, frequent flexible milk removal (Table 2) and elimination of conflicting intra-oral vectors of force when the infant is suckling, should be applied for the signs and symptoms shaded in Table 7.
BLBI: ‘mastitis’
What is mastitis?
Mastitis means ‘inflammation of the breast’, but the term has not been applied to all lactation-related breast inflammations, and the definition of mastitis lacks international consensus. 95 Mastitis is most commonly described as an erythematous painful lump in the breast, usually towards the outer part of the breast, with or without systemic symptoms of fever, myalgia, rigours, and fatigue.68,96 The cluster of signs and symptoms commonly used to diagnose a mastitis emerge variably on a continuum from mild to severe (Table 8).36,37 Because of the highly subjective nature of fatigue, this symptom is not used in this article’s classification system.
Spectrum of signs and symptoms of benign lactation-related breast inflammation commonly referred to as mastitis (shaded).
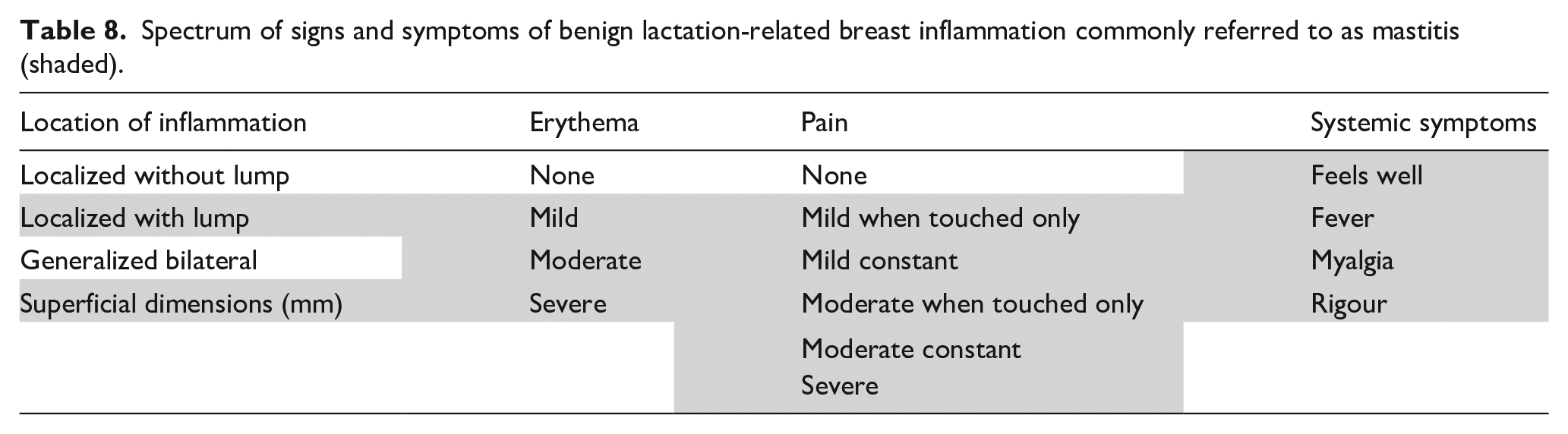
Despite diagnostic uncertainty, the BLBI commonly referred to as mastitis remains a serious lactation-related problem, associated with low milk supply and weaning. 37 This article proposes that BLBI, which encompasses mastitis, is more accurately described by clinical presentation, by selecting the relevant presenting signs and symptoms described in Table 8.
There is an association between nipple damage and the kind of breast inflammation commonly referred to as mastitis. A prospective cohort study of 946 breastfeeding women by Foxman et al. 97 found that the presence of nipple cracks and damage was linked with a three- to sixfold increase in the risk of mastitis. In 2007, Kvist et al. 98 investigated 210 cases of lactation-related breast inflammation, finding that 36% of the women also had nipple damage. Although nipple damage was linked with slower resolution of inflammation, it was not linked with increased need for antibiotics. 98 In 2015, Cullinane et al. 66 showed that in 70 breastfeeding women who developed mastitis in the first 8 weeks post-birth, those who reported nipple damage had twice the incidence of mastitis. In 2020, Wilson et al. 99 conducted a systematic review, which investigated the incidence of and risk factors for mastitis, and found that cracked nipples were significantly associated with lactational mastitis in all eight studies analysed. Engorgement, difficulties attaching the baby to the breast, and blocked ducts have also been associated with increased risk of mastitis.77,99 Kvist et al. 98 found that women with breast inflammation who were using nipple shields had less favourable outcomes.
These associations have been explained using the pathogenic model of breast inflammation, in which it is hypothesized that pathogenic bacteria, for example, Staphylococcus and Corynebacterium, enter the milk from nipple cracks to cause breast inflammation.66,99 However, new evidence about the composition of the human milk microbiome, detailed in the first article in this series, demonstrates why it is unlikely that mastitis is caused by retrograde spread of ‘pathogenic’ bacteria from visible nipple damage. Bacteria and fungi identified on the nipple–areolar complex in the presence of nipple pain and damage are also regularly identified in healthy human milk microbiomes.5,94 Moreover, most women with mastitis (64% in the study by Cullinane et al. 66 ) do not have nipple damage. Clinically, only a small proportion of nipple cracks and ulcers show signs of infection, and nipple damage is often not adjacent to the duct openings, but at the junction of the nipple and areola.
From the perspective of the gestalt biomechanical model of breastfeeding, nipple damage, BLBI, and difficulties bringing the infant on to the breast have a shared aetiology. Conflicting intra-oral vectors of force, which exert high mechanical load on breast and nipple tissue, result in nipple pain and damage, difficulty bringing the baby onto the breast, and/or positional infant motor instability.12,27,28 Applying the gestalt model, nipple shield use is often an indicator of underlying and unresolved breast tissue drag and positional stability problems.
Prevalence
Prevalence data for mastitis are based on variable definitions, in the absence of agreement about underlying mechanisms. 96 The systematic review by Wilson et al. 99 of incidence and risk factors for lactational mastitis, which included 26 articles, concluded that lactational mastitis affects about one in four women during the first 6 months postpartum. However, the authors note that the quality of studies is poor.100–103 Mastitis appears to be the most common reason given for weaning in the first 3 weeks post-birth,103,104 and 70% of cases occur in the first 4 to 8 weeks.66,102 Although an episode of mastitis mostly occurs in just one breast, it may occur more than once, and on either side, and some women experience mastitis multiple times with the same child. Women who experienced mastitis with previous children are two to four times more likely to experience mastitis in subsequent lactations. 96 Because the incidence varies widely across locations, Wilson et al. 99 propose that mastitis, regardless of variable definitions, may be mostly preventable.
Prevention
A 2020 Cochrane review by Crepinsek et al.
37
analysed 10 RCTs investigating the prevention of mastitis and concluded: We cannot be sure what the most effective treatments are for preventing mastitis because the certainty of evidence is low due to risk of bias, low numbers of woman participating in the trials, and large differences between the treatments which make it difficult to make meaningful comparisons. Interventions which do not prevent mastitis are detailed in Table 9. Crepinsek et al.
37
found a moderate certainty of evidence that acupoint massage helped prevent mastitis.
Interventions which do not prevent mastitis.

BLBI: benign lactation-related breast inflammation.
From the perspective of the mechanobiological model, the preventive strategies that are likely to have substantial impact, requiring research investment, relate to the mechanical impact of elevated intra-alveolar and intra-ductal pressure, which trigger inflammatory cascades and BLBI. Applying this theoretical frame, prevention focusses on Principles 1–5, detailed above. Frequent flexible milk removal and elimination of mechanical forces which cause high intraluminal pressures are fundamental.
In 2021, a multi-country study by Jiminez et al., 105 funded by Danone Nutricia, randomized 328 women at the 35th week of their pregnancy to either placebo or Ligilactobacillus salivarius PS2 supplementation until 12 weeks postpartum. About 9.7% (29 of 156) women who received the probiotic experienced mastitis, compared to 14% (20 of 144) who received placebo. Both groups exclusively breastfed for a median duration of 77 days. Further investigation in larger trials is required. 105 Prior to this study, a 2020 Cochrane Review by Crepinsek et al. 37 found there was only low-quality evidence to support the effectiveness of probiotics in prevention of mastitis. This was corroborated by the scoping review by Barker et al. 70 of probiotics and mastitis published the same year. Crepinsek et al. 37 note that the findings of the biggest study they could locate of probiotic use for mastitis prevention were withheld by the manufacturer, and surmise that the findings did not advance the manufacturer’s commercial interest. 69 Importantly, probiotics have not been shown to affect the overall composition of the human milk microbiome. 71 At this time, there is little evidence to justify use of probiotics for prevention of mastitis.
How is BLBI commonly referred to as mastitis treated?
Wilston et al.
99
conclude in their 2020 systematic review that: Provision of care to breastfeeding women at risk for or affected by mastitis is currently constrained due to a critical lack of high quality epidemiological evidence about its incidence and risk factors.
From the perspective of the mechanobiological model, Principles 1, 2, and 4 are essential management. Principle 5 may have a role if it is applied gently and does not cause further pain. See the end of this section for a summary of management strategies which derive from a clinical translation of the mechanobiological model and synthesis of relevant research applying a complexity lens.
Cold or hot compresses are not demonstrated to improve outcomes but may be used according to an individual woman’s preference. Cold applications have been shown to narrow lactiferous ducts in the nipple, which may act against the positive effects of ductal dilations.
Due to the highly variable nature of the human milk microbiome between lactating women, the inability of sampling methods to determine a difference between normal and abnormal members of the milk microbiomes, and the increased risk of inappropriate and unnecessary antibiotic prescribing, attempts to isolate causative agents, either by culture or polymerase chain reaction (PCR), from milk or nipple–areolar culture swab, do not contribute to management of breast inflammation. 5 There is no scientific rationale for midstream milk culture and sensitivities from a breastfeeding mother’s milk in the context of breast inflammation, unless abscess is identified. There is no scientific rationale for investigating C-reactive protein or the full blood count, as both white cells and C-reactive protein are markers of inflammation, not necessarily infection.36,106,107 Investigation with ultrasound imaging is vital if signs and symptoms of localized breast inflammation are significant and not resolving, to exclude abscess or other pathology. 68
Lactating women with localized erythematous and painful BLBI may use paracetamol or acetaminophen, or ibuprofen, for comfort. However, patients should be aware that overuse of antipyretics may negatively affect the body’s capacity to downregulate the inflammatory response.5,108,109 The use of warm showers and antipyretics did not improve outcomes in the 2007 Kvist et al. 36 study.
This 2007, RCT by Kvist et al. of 210 episodes of breast inflammation in 205 lactating women concluded that daily follow-up and support while a woman awaited her body’s anti-inflammatory response was effective treatment for the great majority of lactating mothers with breast inflammation. This was regardless of how long symptomatic women waited before presenting at the midwifery clinic, which ranged from 1 to 7 days. All patients received usual care, which included unspecified fit and hold adjustments and advice to decrease inter-feed intervals.36,98,106 Kvist reflects that with daily follow-up, women were able to tolerate systemic symptoms while resolution occurred without recourse to antibiotics. Daily follow-up also allowed detection of the 15% of patients who required antibiotics. This article recommends daily follow-up for BLBI, which presents with pain, erythema, or systemic signs and symptoms.
In their 2007 study, Kvist et al. found that 85% of 210 episodes of breast inflammation in 205 breastfeeding women recovered without recourse to antibiotic therapy.36,98 The presence of nipple damage did not increase the need for antibiotics. Women received care when they presented with any or a mix of the following: a tense breast not relieved by breastfeeding, and/or lumps in the breast tissue, breast redness, fever, or pain. The treating midwives used the term breast inflammation with patients, rather than mastitis. The time that elapsed before presenting at the clinic did not affect outcomes. All women were provided with ‘essential care’, which included both advice to decrease feeding intervals and fit and hold support (though the techniques used for this fit and hold support are not described). 36 Women who took longer to recover had significantly higher scores for increased breast tension not relieved by breastfeeding and for erythema. But there was no difference in experience of pain between those who required antibiotics and those who did not. The authors concluded that clinical signs and symptoms of breast inflammation do not help clinicians make a decision about antibiotic use.36,95,98,106
In a 2008 comparison of the milk of 192 women with mastitis or breast inflammation and 466 healthy breast milk donors, Kvist et al. 106 found no correlation between higher bacterial counts and symptoms. There were no differences in bacterial counts between those prescribed and not prescribed antibiotics or those with and without breast abscess. 106 A 2013 Cochrane review by Jahanfar et al. 101 found insufficient evidence to support antibiotics in the treatment of mastitis.
In Australia, 77%–89% of women diagnosed with mastitis are prescribed antibiotics, sometimes on the basis of breast milk culture. This is unsurprising, since antibiotic use in Australia is high relative to Europe and Canada; for example, Australian doctors prescribe more than twice the amount of antibiotics overall compared with their European counterparts. 110 Scandinavians are much less likely to prescribe antibiotics for diagnoses of mastitis or breast inflammation: just 38% in a Finnish study 111 and 15% in the Swedish Kvist et al. trial.36,112 In the United States, 86%–97% of women diagnosed with mastitis are prescribed antibiotics, and rates are similar in New Zealand and Canada.97,98,106,113
Rate of antibiotic use does not have an impact on rates of mastitis recurrence, and the rate of abscess formation remains at approximately 3% of women diagnosed with mastitis, regardless of country or rate of antibiotic prescription for mastitis. 106 The presence of even small quantities of antibiotics in human milk alters the diversity and perhaps the resilience of the human milk and other microbiomes, and infant gut microbiota.36,112 PCR analysis shows that antibiotic administration reduces Lactobacilli and Bifidobacterium in human milk. 114 Kvist has challenged the high levels of antibiotic use in women diagnosed with mastitis in some countries.95,96,112,115,116
Overuse of antibiotics for BLBI occurs in the context of the World Health Organization’s urgent call for responsible antimicrobial stewardship. 6 Antibiotic over-prescribing is described as a tragedy of the commons in which a shared resource is over-exploited by some, to the eventual detriment of all.117,118 As Director General Dr Tedros stated in 2020, ‘Antimicrobial resistance is a slow tsunami that threatens to undo a century of medical progress’. 119 Unnecessary use of antibiotics for BLBI contributes to the increased risk of methicillin-resistant Staphylococcus aureus (S. aureus) breast inflammation in lactating women world-wide.
Antibiotics remain a treatment of last resort when signs and symptoms of BLBI persist without resolution or are particularly severe. If antibiotics are required, clinical protocols have been developed from findings linking S. aureus with mastitis. 5 Flucloxacillin or dicloxacillin 500 mg four times daily is recommended; cephalexin 500 mg four times daily if allergic to penicillin; and clindamycin if the patient is known to have an anaphylactic reaction to penicillin, because of the risk of cross-reactivity between cephalexin and penicillin. If methicillin-resistant S. aureus is suspected or proven, bactrim or clindamycin are prescribed. If breast inflammation does not improve with oral antibiotics, intravenous flucloxacillin, cephazolin, or vancomycin may be necessary. 68
The following is a summary of management strategies for BLBI commonly referred to as mastitis:
Frequent flexible milk removal, including by pumping or hand expression (Table 2).
a. Eliminate conflicting intra-oral vectors of force during milk removal, which cause lactiferous duct compression. b. Avoid focused deep pressures on the breast e.g. lump massage or vibration. c. Avoid other prolonged external pressure on the breast.
Avoid increasing milk supply beyond an infant’s physiological need.
Gentle manual movement of breasts if this does not worsen pain.
Judicious use of anti-pyretics.
Daily follow-up by phone or face to face as indicated.
Antibiotic use if severe and failing to resolve, according to clinical judgement.
Ultrasound, if lump persists.
Active end-stage (non-malignant) lactation-related breast inflammation
Abscess
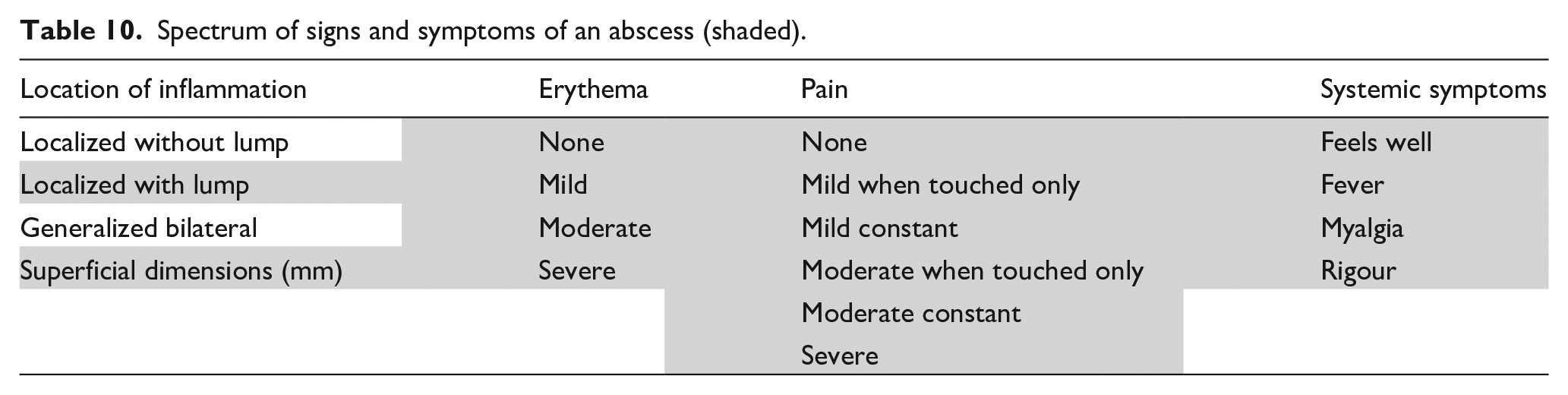
A lactational abscess is a localized collection of milk and purulent fluid, characterized by high white cell counts, mammary epithelial cells, bacteria, and fibrotic septations, and walled off by a stromal inflammatory reaction. The range of clinical signs and symptoms accompanying lactation abscess are detailed in Table 10. Abscess is diagnosed by ultrasound imaging.20,68
Spectrum of signs and symptoms of an abscess (shaded).
Antibiotics are required. First-line treatment is dicloxacillin or flucloxacillin 500 mg four times daily orally for 10–14 days. Methicillin-resistant S. aureus may require clindamycin 300 mg orally four times daily for 10–14 days. In the human body, undrained purulent fluid is usually not successfully treated by antibiotics alone. Drainage of symptomatic fluid collection is required to avoid fistula formation or septicemia.68,80
A 2015 Cochrane review by Irusen et al. 120 found that antibiotic treatment did not improve outcomes in breastfeeding women with breast abscess who were also treated with incision and drainage, although all studies were poorly conducted. Low-quality evidence suggests that treatment failure was more common among women treated with needle aspiration compared to those who underwent incision and drainage, but that women treated with needle aspiration were more likely to continue breastfeeding. 120 Breast surgeon Dr Katrina Mitchell proposes from clinical experience that a small ‘penrose’ stent allows for passive decompression of a lactational abscess and can be removed 1–4 days after the procedure, with drainage site closure occurring 7 days post-procedure. This stent is packed with gauze into a bra, allowing women to continue to breastfeed. A video of incision technique of an abscess or galactocoele is available at: https://www.youtube.com/watch?v = JOOKKLgrE28. Fluid from an abscess should be cultured. Ultrasound imaging is repeated after incision and drainage.20,68
Fistula
Fistula is a complication of untreated lactational abscess, and is rare. It is a pathological connection between a lactiferous duct and the skin, which drains milk. There is one published case report of a fistula, which occurred during a second lactation after an abscess ruptured in the first lactation. In this case, resolution occurred when breastfeeding was ceased. 121 A retrospective audit by breast surgeons, published only as an abstract, found that no fistula developed after surgical intervention for breast masses including abscess from 4 pregnant and 43 lactating patients (surgical excision of the mass, percutaneous drain insertion, stab incision and drainage, core-needle biopsy, and punch biopsy). 122 The authors state, drawing on clinical experience, that milk fistula after incision and drainage of a lactational abscess is a very rare occurrence.121,122 They argue that the remote risk of fistula formation is not a reason to delay incision and drainage of an abscess, and that a fistula, if it occurs, typically closes quickly due to the highly vascular wound-healing environment of the lactating breast, with no reason to cease breastfeeding. 80
Septicaemia
Before the advent of antibiotics, puerperal septicaemia from severe inflammation of the lactating breast was a common cause of maternal death in the post-birth period. Intravenous antibiotic treatment prevents this catastrophic outcome.
Inactivated end-stage (non-malignant or benign) lactation-related breast inflammation: galactocoele
A galactocoele is a milk retention cyst. 20 It comprises a dilated terminal lactiferous duct, surrounded by a layer of epithelial cells and myoepithelial cells, and contains either milk or, if chronic, semisolid material. 46 Galactocoeles are asymptomatic, and are not infected (Table 11). There is no prevalence data.
Spectrum of signs and symptoms of a galactocoele (shaded).
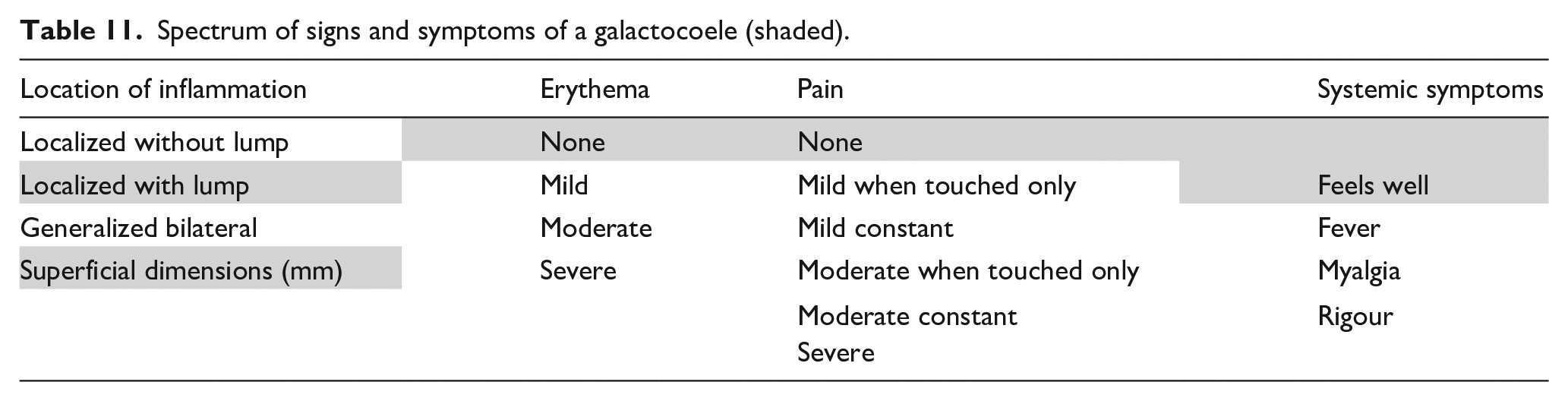
This article hypothesizes that a galactocoele develops in an area subject to very high intra-alveolar pressures, with widespread tight junction rupture between lactocytes and involution of alveoli on a scale which has not been able to repair with tissue remodelling. Clinical symptoms of inflammation either subside or do not emerge, and the collection of fluid is quiescent. Over time, galactocoeles may calcify. Some breast surgeons propose, from clinical experience, that a galactocoele larger than 3 cm requires serial aspiration, or drainage and incision. Drainage is also required if a galactocoele becomes inflamed (or activates), risking an abscess.20,80
Conclusion
The benefits of breastfeeding for infant and maternal well-being have been established. 123 But there remains, relative to most aspects of healthcare, a paucity of methodologically sound research investigating the management of common clinical breastfeeding problems. Clinical breastfeeding support remains a research frontier.
The first article in this three-part series synthesized the research concerning the mechanobiology of the lactating breast, the mammary gland immune system including the human milk microbiome and cell composition, and the functional anatomy of the lactating breast, to propose a mechanobiological model of inflammation of the lactating breast. This second article synthesizes the evidence to propose a new approach to classification, prevention, and management of benign lactation-related breast inflammation and end-stage lactation-related breast inflammation.
This complex systems approach has been developed as the foundational breastfeeding domain of NDC or ‘the Possums programs’. NDC integrates evidence from the perspective of complexity science and evolutionary biology. Given current knowledge about the Developmental Origins of Disease and the burgeoning health system costs of chronic disease, there is urgent need for high-quality evaluation of interventions for BLBI, including evaluation of strategies which aim to eliminate conflicting intra-oral vectors of force during suckling, to optimize the long-term immune-protective benefits an infant receives from his or her mother’s milk.
Footnotes
Appendix 1
Author contribution(s)
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The author is the Medical Director of Possums & Co (www.possumsonline.com), a registered health promotion charity, which educates parents and health professionals with free resources and by the sales of education products developed from the evidence-base of Neuroprotective Developmental Care (NDC or ‘the Possums programs’), including as Milk & Moon www.milkandmoonbabies.com and the NDC Education Hub ![]() . The charity invests all revenue raised back into education and research, which supports the well-being of mothers and babies.
. The charity invests all revenue raised back into education and research, which supports the well-being of mothers and babies.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The charity Possums & Co funded the open access publication fee.
