Abstract
Damage-associated molecular patterns (DAMPs), including mitochondria-derived molecules, are known to trigger immune responses and produce nociceptor sensitization during tissue inflammation. This animal study investigated whether mitochondrial debris promotes inflammatory hyperalgesia through activation of the stimulator of interferon genes (STING) signaling pathway in dorsal root ganglion (DRG) neurons. The results showed that local administration of mitochondrial debris into the hind paws of rats induced significant mechanical hyperalgesia and increased STING expression in DRG neurons. Pretreatment with H-151, a selective STING inhibitor, attenuated both debris-induced hyperalgesia and neuronal STING upregulation. STING expression in DRG neurons was similarly upregulated in a model of tissue inflammation induced by Complete Freund’s Adjuvant (CFA), and administration of H-151 significantly alleviated the inflammatory hyperalgesia and increase in STING expression. These findings suggest that mitochondrial debris released during tissue inflammation activates the STING pathway in primary afferent neurons. Effective suppression of hyperalgesia by pharmacological inhibition of STING in both debris-induced and CFA-induced models in this study highlights the pronociceptive role of STING activation in peripheral sensory neurons. In conclusion, mitochondrial debris-induced STING activation in DRG neurons plays a critical role in the development of inflammatory hyperalgesia, and targeting this pathway might represent a novel therapeutic strategy for inflammatory pain.
Introduction
Despite remarkable progress in pain management, severe pain is common in patients with acute pain. 1 Effective pain management is crucial for optimal recovery and preventing adverse events. 2 A large part of acute pain is nociceptive, in which tissue injury or inflammation sensitizes nociceptors in primary afferent neurons. 3 Sensitization of primary afferent neurons during tissue injury, referred to as inflammatory hyperalgesia, leads to exacerbation of acute pain 4 and the development of persistent pain. 5 However, the molecular mechanisms underlying the nociceptor activation are not yet fully understood.
Bioactive molecules packed in the cell body are released into the extracellular space during tissue injury. These molecules, which are collectively referred to as damage-associated molecular patterns (DAMPs), induce systemic inflammatory responses 6 and/or local immune activation. At the same time, DAMPs exert diverse effects on the excitation of primary afferents and produce inflammatory hyperalgesia. 7 Mitochondria are intracellular organelles that play a crucial role in the production of ATP and in maintaining calcium homeostasis, ensuring normal cell function. N-formyl peptides or mitochondrial DNA, packed inside the mitochondria, can potentially serve as DAMPs. 8 For instance, in myocardial infarction, mitochondrial DAMPs are released from injured cells and are involved in the development of reperfusion injury. 9 However, the precise role of mitochondrial DAMPs in the development of inflammatory hyperalgesia is still unknown.
Mitochondrial DNA is one of the major mitochondrial DAMPs. 10 Mitochondrial DNA activates the cyclic-GMP-AMP synthase (cGAS)/stimulator of the interferon (IFN) genes (STING) signaling pathway, 11 and activation of STING contributes to the development of neuroinflammation and neurodegenerative disease progression. 12 In the present study, we hypothesized that mitochondrial DAMPs are released during tissue inflammation and activate the cGAS-STING signal, which in turn sensitizes primary afferent neurons and produces inflammatory hyperalgesia. To test this hypothesis, we investigated whether: (1) injection of mitochondrial debris leads to behavioral hyperalgesia and induction of STING in the DRG; (2) STING inhibition prevents debris-induced hyperalgesia; and (3) STING is activated and STING inhibitor prevents hyperalgesia in an animal model of inflammatory hyperalgesia.
Methods
Animals and treatments
All experimental procedures in this study were approved by the Animal Care Committee of Kyoto Prefectural University of Medicine and were conducted in accordance with the guidelines of the National Institutes of Health and the International Association for the Study of Pain. Male Sprague-Dawley rats (200–250 g; Shimizu Laboratory Supplies Co. Ltd., Kyoto, Japan) were housed in groups of three per cage under a 12-h light/dark cycle. Surgeries and drug administrations were performed under anesthesia with 2% v/v isoflurane. The sample sizes for all experimental protocols were determined according to the results of our previous research. 5
Experiment 1: Behavioral analysis following local administration of mitochondrial debris
Rats were randomly assigned to naïve, saline and debris groups (n = 6 for each group). Liver tissue was obtained from unrelated rats, and 180–200 mg of the collected tissue was used for mitochondrial debris extraction using the Mitochondria Isolation Kit for Tissue (Thermo Fisher Scientific, Waltham, MA, USA), following the method described by Tsuji et al. 13 Approximately 500 μg of mitochondrial debris was extracted under sterile conditions at 4°C and stored at −80°C.
In the debris group, mitochondrial debris was suspended in 100 μL of saline and injected subcutaneously into the left hind paw using a 29G insulin syringe. In the saline group, 100 μL of saline alone was injected using the same procedure. Behavioral assessments for mechanical and thermal pain sensitivity were conducted as described later, from baseline (0 h) through 7 days post-injection.
Experiment 2: Evaluation of the effect of mitochondrial debris on STING expression in the DRG
Immunofluorescence staining was performed to analyze STING expression in the DRG. Rats were randomly assigned to naïve, saline, and debris groups (n = 6 for each group). The debris group received a subcutaneous injection of mitochondrial debris (dissolved in 100 μL of saline) into the left hind paw using a 29G insulin syringe, and the saline group received an equivalent injection of 100 μL of saline, as in Experiment 1. At 4 hs post-injection, the rats were sacrificed, and the L5 DRG was collected from each rat under terminal anesthesia with isoflurane, and then processed for immunofluorescence staining.
Experiment 3: Evaluation of the effect of a STING inhibitor on debris-induced hyperalgesia
Rats were randomly assigned to naïve, vehicle and H-151 (STING inhibitor) groups (n = 6 for each group). The H-151 group received an intraperitoneal injection of H-151 (30 mg/kg dissolved in 400 μL of 10% dimethyl sulfoxide (DMSO) + 90% corn oil; Selleck Chemicals, Houston, TX, USA) using a 25G needle,14,15 and the vehicle group received an equivalent injection of the vehicle solution. Thirty minutes after the administration of H-151 or vehicle, mitochondrial debris was injected into the left hind paw, as described in Experiment 1. Thereafter, behavioral assessments for mechanical and thermal pain sensitivity were conducted before (0 h) and 4 h after debris injection, as described later.
The percent maximum possible effect (%MPE) was calculated using the formula:
Experiment 4: Evaluation of the effect of H-151 on debris-induced STING expression in the DRG
Immunofluorescence staining and Western blotting were performed to analyze STING expression in the DRG following H-151 and mitochondrial debris injection. For this, rats were randomly assigned to naïve, vehicle and H-151 groups. The H-151 group received an intraperitoneal injection of H-151 (30 mg/kg dissolved in 400 μL of 10% DMSO + 90% corn oil) using a 25G needle, and the vehicle group received an equivalent injection of the vehicle solution.
Thirty minutes after the administration of H-151 or vehicle, mitochondrial debris was injected into the left hind paw. At 4 h post-injection, the rats were sacrificed and L5 DRG was collected from each rat and processed for immunofluorescence staining (n = 6 for each group) and Western blotting (n = 6 for each group).
Experiment 5: Evaluation of the effect of complete Freund’s adjuvant (CFA) on STING expression in the DRG
Tissue inflammation was induced by the intraplantar injection of CFA. For this, rats were randomly assigned to naïve, CFA, and CFA with H-151 groups (n = 6 per group). Rats in the CFA group received a subcutaneous injection of CFA (50 μL; InvivoGen, San Diego, CA, USA) into the left hind paw using a 25G needle. Rats in the CFA with H-151 group received an intraperitoneal injection of H-151 (30 mg/kg, dissolved in 400 μL of 10% DMSO + 90% corn oil) using a 25G needle, followed immediately by injection of CFA (50 μL) into the left hind paw using a 25G needle. At 2 days post-injection, the rats were sacrificed, and L5 DRG was collected from each group and processed for immunofluorescence staining to analyze STING expression in the DRG.
Experiment 6: Evaluation of the effect of the STING inhibitor on CFA-induced inflammatory hyperalgesia
Rats were randomly assigned to vehicle or H-151 groups (n = 6 per group). The H-151 group received an intraperitoneal injection of H-151 (30 mg/kg, dissolved in 400 μL of 10% DMSO + 90% corn oil) using a 25G needle, followed immediately by injection of CFA (50 μL) into the left hind paw using a 25G needle. The vehicle group received the same volume of vehicle intraperitoneally, followed by CFA injection in the same manner.
Behavioral assessments for mechanical and thermal pain sensitivity were conducted at baseline (0 h), and at 4 h, 1 day and 2 days post-CFA injection.
Behavioral assessment
All behavioral experiments were conducted by an experimenter (AH) who was blind to the experimental conditions. Mechanical pain sensitivity was assessed by measuring the withdrawal threshold in response to von Frey filament stimulation (Muromachi Kikai, Tokyo, Japan). Rats were placed in a transparent plastic chamber with a metal wire grid floor and allowed to acclimate to the experimental environment. The von Frey filament was applied to the left hind paw, and the lowest force that elicited a clear withdrawal response at least twice in ten applications was recorded as the threshold. To prevent tissue damage, the maximum stimulus intensity was set at 60 g. Withdrawal thresholds were logarithmically transformed to a linear scale for statistical analysis. 17
Thermal pain sensitivity was assessed by measuring the latency of the withdrawal response to radiant heat stimulation (Plantar Test Apparatus; IITC Life Science Inc., Woodland Hills, CA, USA). For this, rats were placed in transparent plastic cages with a glass floor and allowed to acclimate before testing. A radiant heat source was applied to the left hind paw, with a cut-off latency of 20 s to prevent burns. Heat stimulation was applied three times at 5-min intervals, and the mean withdrawal latency of the three trials was calculated.
Immunohistochemistry
Rats were transcardially perfused with 300 mL of 0.9% NaCl, followed by 350 mL of 10% neutralized formalin (Wako Pure Chemical Industries, Ltd., Osaka, Japan) under terminal anesthesia with isoflurane. The harvested L5 DRG tissues were cryoprotected in 20% sucrose (0.1 M phosphate buffer) at 4°C for 24 h, then frozen and stored at −80°C. Thin sections (10-µm thick) were prepared using a cryostat (CM1850, Leica Microsystems, Wetzlar, Germany). The sections were blocked with Blocking One P (Nacalai Tesque Inc., Kyoto, Japan) for 1 h at room temperature and incubated at 4°C for 3 days with a rabbit anti-STING antibody (1:1000, Proteintech Group Inc., Rosemont, IL, USA) in 0.1% Tween 20/0.1 M Tris-buffered saline (TBS, pH 7.4) containing 1% blocking reagent (EMD Millipore, Billerica, MA, USA).
After washing with 0.1 M phosphate-buffered saline (PBS), the sections were incubated overnight at 4°C with a Cy3-conjugated anti-rabbit secondary antibody (1:200, Merck Millipore) in 0.1 M TBS.
To demonstrate the distribution of STING expression, double-staining immunofluorescence was performed. Sections were incubated with anti-STING antibody (1:2000) and anti-calcitonin gene-related peptide (CGRP, 1:250, Frontier Institute) at 4°C for 3 days, followed by overnight incubation with Cy3-conjugated anti-rabbit secondary antibody (1:200, Merck Millipore) and Alexa Fluor-conjugated anti-guinea pig secondary antibodies (1:500, Merck Millipore) in 0.1 M TBS at 4°C.
Separately, sections were incubated with anti-STING antibody (1:2000) and anti-isolectin B4 (IB4, 1:200, Vector Laboratories) at 4°C for 3 days, followed by overnight incubation with Cy3-conjugated anti-rabbit secondary antibody (1:200, Merck Millipore) and fluorescein isothiocyanate-conjugated isolectin B4 (1:250, Abcam) in 0.1 M TBS at 4°C.
The stained sections were then washed and visualized using a fluorescence microscope equipped with a digital camera system (Nikon, Tokyo, Japan).
Cell counting
Immunohistochemistry images were analyzed using ImageJ software (NIH, Bethesda, MD, USA) on a Macintosh computer. To assess STING expression in DRG neurons, STING- or DAPI-positive neurons were identified based on nuclear size (>50 µm2). Preliminary experiments confirmed that using this criterion effectively distinguished neuronal STING/DAPI signals while excluding glial signals.
For each rat, the number of STING- or DAPI-positive neurons was counted in five sections spaced at least 100 µm apart, and the percentage of positive neurons was calculated.
Image analysis
Mean STING signal intensity and the cross-sectional area of neurons with visible nuclei were measured using ImageJ software (NIH). Based on previously established methods, regions of interest (ROIs) were manually traced for all neurons in the tissue. To facilitate visualization, the brightness of the STING-stained images was adjusted to enhance neuronal cell profiles. The ROIs were then applied to the original STING-stained images to measure signal intensity.
Relative signal intensity (RSI) was calculated by subtracting the background signal intensity from the mean neuronal signal intensity. Neurons with RSI > 50 were classified as STING-positive. Five sections, spaced at least 100 µm apart, were analyzed per rat, and the mean percentage of STING-positive neurons was calculated.
Western blotting
L5 DRG tissues were homogenized in a buffer containing 20 mM Tris-HCl (pH 8.0), 137 mM NaCl, 2 mM EDTA, 10% glycerol, 1% Triton X-100, 1 µM PMSF, 10 µg/mL leupeptin, and 10 µg/mL pepstatin A, supplemented with a protease inhibitor (Thermo Fisher Scientific). Protein concentrations of the homogenates were determined using the Bradford reagent (Bio-Rad, Hercules, CA, USA).
Fifty micrograms of cell lysates were separated via 8% SDS-PAGE and transferred onto nitrocellulose membranes. The membranes were incubated overnight at 4°C with rabbit anti-STING (1:1000, Proteintech Group Inc.) or rabbit anti-GAPDH (1:20,000, Cell Signaling Technology, Danvers, MA, USA) antibodies. They were then incubated with horseradish peroxidase (HRP)-conjugated secondary antibodies (1:5000 Thermo Fisher Scientific). Detection was performed using the Enhanced Chemiluminescence Select Western Blotting Detection Kit (GE Healthcare, Chicago, IL, USA) and Hyperfilm (GE Healthcare). The intensity of the selected bands was captured and analyzed using ImageJ software (NIH) on a Macintosh computer.
Statistical analysis
Statistical analyses were conducted using GraphPad Prism 9 software (GraphPad Software, La Jolla, CA, USA). Group comparisons were performed using the Brown-Forsythe test, one-way ANOVA with Tukey’s multiple comparison test, Greenhouse-Geisser test, or two-way ANOVA with Tukey’s multiple comparison test. Statistical significance was set at p < 0.05. All data are expressed as the mean ± SEM.
Results
Experiment 1: Development of hyperalgesia following mitochondrial debris administration
Figure 1(a) demonstrates the mechanical threshold against von Frey stimulation. Local administration of mitochondrial debris significantly reduced the pain threshold at 4 h after treatment compared to baseline, the effect continuing for 24 h after the treatment (n = 6, F(8,52) = 3.729, p = 0.002 between treatment and time by repeated two-way ANOVA, #p < 0.05, ##p < 0.01, ###p < 0.001 by Tukey’s multiple comparison test). The debris group showed a clear reduction in pain threshold compared to the naïve and saline groups (n = 6 for each group, F(8,52) = 3.729, p = 0.002 between treatment and time by repeated two-way ANOVA, *p < 0.05, **p < 0.01, ***p < 0.001 vs the naïve and saline groups by Tukey’s multiple comparison test). Pain threshold trends were similar in the naïve and saline groups. In contrast, no significant changes in withdrawal latency against thermal stimulation were observed after debris administration (Figure 1(b)).
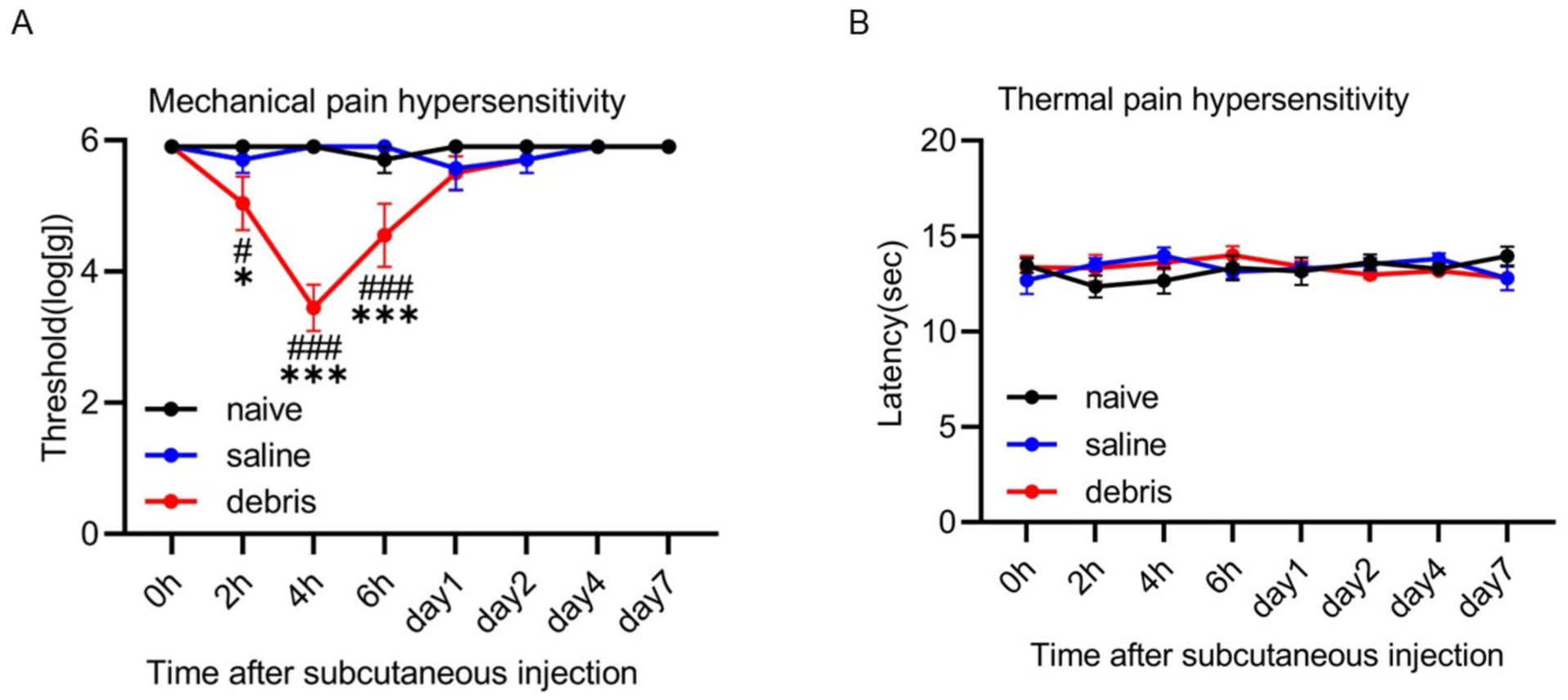
Mitochondrial debris induces hyperalgesia. (a) Mechanical withdrawal threshold against von Frey filament stimulation. In the debris group, local administration of mitochondrial debris significantly reduced the pain threshold at 4 h after treatment compared to baseline, the effect continuing for 6 h after subcutaneous injection of the mitochondrial debris (n = 6, F(14,105) = 9.049, p < 0.001 between treatment and time by repeated two-way ANOVA, #p < 0.05, ###p < 0.001 by Tukey’s multiple comparison test). Compared to the naïve and saline groups, the debris group showed a significant decrease in pain threshold 2, 4, and 6 h after the treatment (n = 6 for each group, F(14,105) = 9.049, p < 0.001 between treatment and time by repeated two-ways ANOVA, *p < 0.05, ***p < 0.001 vs the naïve and saline groups by Tukey’s multiple comparison test). (b) Latency against radiant heat stimulation. No significant changes after subcutaneous injection of mitochondrial debris were observed in the debris group, and there was no clear difference between naïve and debris groups.
Experiment 2: STING expression in DRGs following mitochondrial debris administration
Immunohistochemistry showed that STING expression was detected in only a few neurons in the naive group. In the debris group, however, STING expression was increased in neurons with small cell size (<1000 μm2; Figure 2(a) and (b)), with a significantly higher percentage of STING-positive neurons in the debris group compared to the naïve and saline groups. The percentage of STING-positive neurons was similar between naïve and saline groups (n = 6 for each group, F(2,10) = 7.530, p = 0.01 between treatments, two-way ANOVA, *p < 0.05 by Tukey’s multiple comparison test, Figure 2(c)). At 4 h after debris administration, double-staining immunofluorescence for STING with CGRP or IB4 showed that 16.4% of CGRP-positive neurons were STING-positive, which accounted for 93.5% of all STING-positive neurons. Additionally, 3.2% of IB4-positive neurons were STING-positive, representing 29.9% of STING-positive neurons (Figure 2(d) and (e), Table 1).

STING expression in the DRG following mitochondrial debris administration. (a) Immunofluorescence staining for STING in the DRG at 4 h post-treatment. The debris group showed intense expression of STING in the DRG neurons compared to naïve and saline groups (Scale bar = 50 μm). (b) Scatter plot showing the relationship between STING immunoreactivity signal intensity and neuronal area. There was an increase in the number of small neurons with higher signal intensity in the debris group. (c) The percentage of STING-positive neurons was significantly higher in the debris group compared to both the naïve and saline groups. No significant difference was observed between the naïve and saline groups (n = 6 for each group, F(2,10) = 7.530, p = 0.01 between treatments by two-way ANOVA, *p < 0.05 by Tukey’s multiple comparison test). (d) Double-staining immunofluorescence for STING and CGRP in the DRG at 4 h post-treatment. STING and CGRP immunostaining showed substantial colocalization. The arrows indicate double-labeled neurons. (e) Double-staining immunofluorescence for STING and IB4 in the DRG at 4 h post-treatment. STING and IB4 immunostaining showed only partial colocalization. The arrows indicate double-labeled neurons.
Colocalization of STING with CGRP or IB4.

Experiment 3: Changes in pain threshold and STING expression after intraperitoneal administration of the STING inhibitor followed by injection of mitochondrial debris
Prior intraperitoneal administration of H-151 suppressed the induction of mechanical hyperalgesia induced by debris injection. The debris + vehicle group showed a significantly lower pain threshold at 4 h after the debris injection. On the other hand, the debris + H-151 group showed a significantly higher pain threshold compared to the debris + vehicle group at this time (n = 6 for each group, F(2,15) = 6.553, p = 0.009 between treatments, one-way ANOVA, **p < 0.01, ***p < 0.001 by Tukey’s multiple comparison test, Figure 3(a)).There were no significant changes in withdrawal latency against thermal stimulation in any group after debris and drug administration (Figure 3(b)).
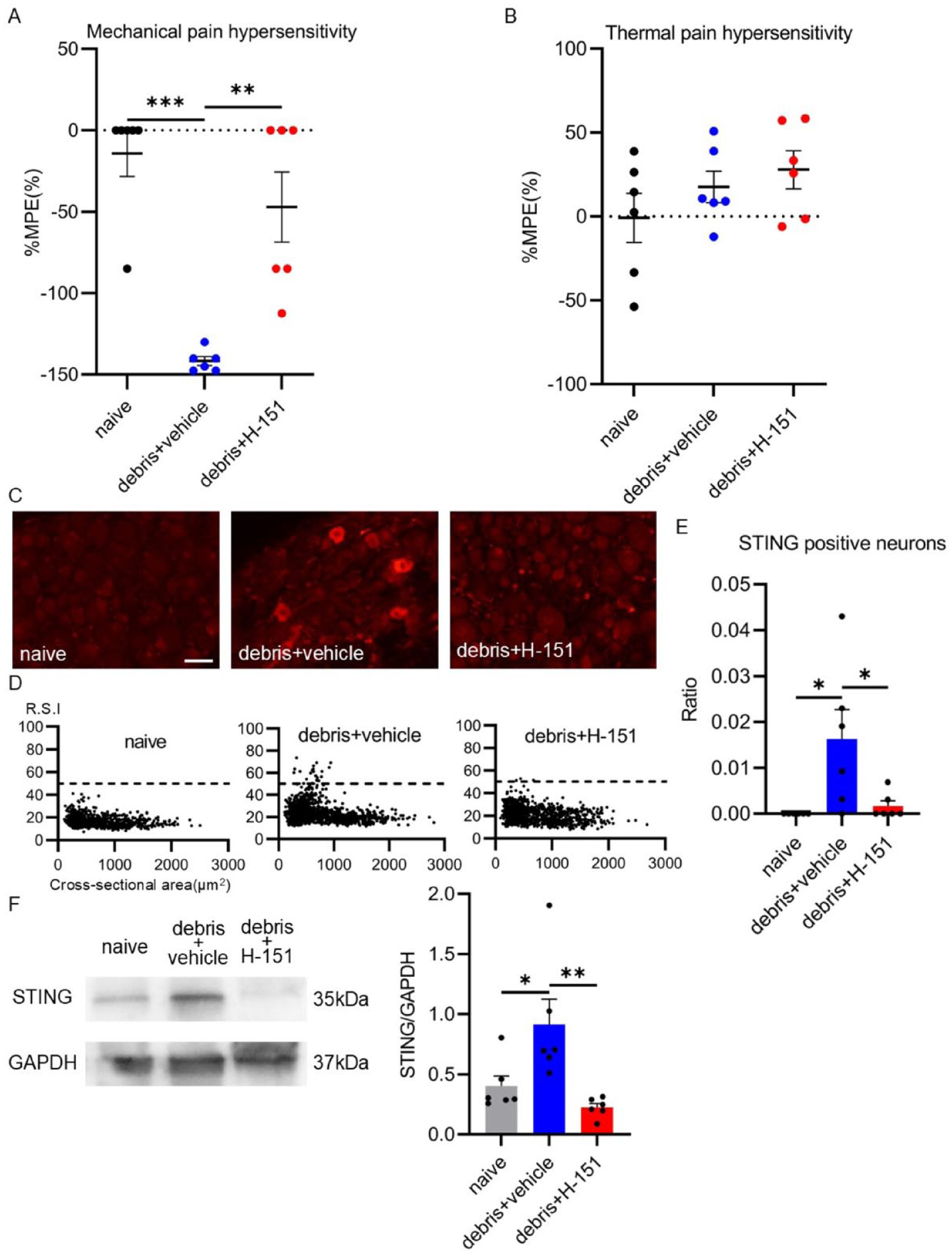
Changes in pain threshold and STING expression after intraperitoneal administration of the STING inhibitor followed by subcutaneous injection of mitochondrial debris. (a) After administration of H-151 or vehicle, mitochondrial debris was locally injected, and the withdrawal threshold was measured 4 h later using the von Frey filament for mechanical stimulation. The debris + vehicle group showed a decrease in pain threshold from baseline. In contrast, the debris + H-151 group showed no significant decrease in the pain threshold from baseline (n = 6 for each group, F(2,15) = 6.553, p = 0.009 between treatments, one-way ANOVA, **p < 0.01, ***p < 0.001 by Tukey’s multiple comparison test). (b) After administration of H-151 or vehicle, mitochondrial debris was locally injected, and the withdrawal threshold to radiant heat stimulation was measured 4 h later. No decrease in threshold from baseline was observed in either the debris + vehicle or debris + H-151 groups. Additionally, there was no significant difference between these two groups and the naïve group. (c) Immunofluorescence staining for STING in the DRG at 4 h post-treatment. The debris + vehicle group showed stronger STING expression in the DRG compared to the naïve and debris + H-151 groups (Scale bar = 50 μm). (d) Scatter plot showing the relationship between STING immunoreactivity signal intensity and neuronal area. Small neurons with higher signal intensity were more abundant in the debris + vehicle group. In contrast, no higher signal intensity neurons were observed in the debris + H-151 group. (e) The percentage of STING-positive neurons was significantly higher in the debris + vehicle group compared to both the naïve and debris + H-151 groups. No STING-positive neurons were observed in the naïve and debris + H-151 groups (n = 6 for each group, F(2,10) = 12.48, p = 0.002 between treatments by two-way ANOVA, **p < 0.01 by Tukey’s multiple comparison test). (f) Western blot analysis of STING expression in the DRG at 4 h post-treatment revealed increased STING expression in the debris + vehicle group, whereas the debris + H-151 group showed a significant reduction in STING expression (n = 6 for each group, F(2,10) = 8.922, p = 0.006 between treatments by two-way ANOVA, *p < 0.05, **p < 0.01 by Tukey’s multiple comparison test).
Immunohistochemistry showed an increase in neurons with strong STING signal intensity in small sized cells (<1000 μm2) in the debris + vehicle group, while the induction of STING expression was suppressed in the debris + H-151 group (Figure 3(c) and (d)). The percentage of STING-positive neurons was significantly higher in the debris + vehicle group compared to the naïve and debris + H-151 groups. STING-positive neurons were not observed in the naïve and debris + H-151 groups (n = 6 for each group, F(2,10) = 12.48, p = 0.002 between treatments, two-way ANOVA, **p < 0.01 by Tukey’s multiple comparison test, Figure 3(e)).
Western blotting demonstrated that STING expression level in the DRG was increased in the debris + vehicle group at 4 h after treatment, and significantly decreased in the debris + H-151 group. There were no significant differences in STING expression levels between the naïve and debris + H-151 groups (n = 6 for each group, F(2,10) = 8.922, p = 0.006 between treatments, two-way ANOVA, *p < 0.05, **p < 0.01 by Tukey’s multiple comparison test, Figure 3(f)).
Experiment 4: STING expression in the DRG following CFA injection
Immunohistochemistry showed that STING expression was detected in only few neurons in the naive group. However, STING expression was increased in neurons with small cell size (<1000 μm2) in the CFA group compared to the naïve group (Figure 4(a) and (b)). Elevated STING expression was significantly inhibited by H-151 treatment. The percentage of STING-positive neurons was significantly higher in the CFA group than in the naïve group or CFA with H-151 group (n = 6 per group, F(2,10) = 8.408, p = 0.007 between treatments, two-way ANOVA, *p < 0.05 by Tukey’s multiple comparison test; Figure 4(c)).

STING expression in the DRG following CFA injection and changes in pain hypersensitivity after intraperitoneal administration of the STING inhibitor. (a) Immunofluorescence staining for STING in the DRG at 2 days post-treatment. STING expression in the DRG was lower in the naïve or CFA + H-151 group compared to the CFA group (Scale bar = 50 μm). (b) Scatter plot showing the relationship between STING immunoreactivity signal intensity and neuronal area. A greater number of small neurons with higher signal intensity were observed in the CFA group. (c) The percentage of STING-positive neurons was significantly higher in the CFA group compared to the naïve or CFA + H-151 group (n = 6 for each group, F(2,10) = 8.408, p = 0.007 between treatments, two-way ANOVA, *p < 0.05 by Tukey’s multiple comparison test). (d) Mechanical withdrawal thresholds measured by von Frey filament stimulation at 4 h, 1 day and 2 days after CFA injection and administration of either H-151 or vehicle. In the CFA + vehicle group, a significant reduction in mechanical threshold against von Frey stimulation was observed 4 h after the CFA injection, which continued for at least 2 days. In contrast, the CFA + H-151 group showed attenuation of the decrease in mechanical pain threshold on days 1 and 2 (n = 6, F(1,10) = 11.41, p = 0.007 between treatments, two-way ANOVA, ##p < 0.01, ###p < 0.001 by Tukey’s multiple comparison test). In the CFA + H-151 group, the reduction in pain threshold observed in the CFA + vehicle group was significantly suppressed both 1 and 2 days after the treatment (n = 6 for each group, F(1,10) = 11.41, p = 0.007 between treatments by two-way ANOVA, *p < 0.05 by Tukey’s multiple comparison test). (E) Thermal withdrawal thresholds measured by radiant heat stimulation at 4 h, 1 day and 2 days after CFA injection and administration of either H-151 or vehicle. Latency against radiant heat stimulation was reduced in the CFA + vehicle group after the CFA treatment. Latency against heat stimulation was also reduced in the CFA + H-151 group 4 h and 1 day after the CFA treatment (n = 6, F(1,10) = 14.1, p = 0.004 between treatments, two-way ANOVA, ##p < 0.01, ###p < 0.001 by Tukey’s multiple comparison test). In the CFA + H-151 group, the reduction in thermal threshold observed in the CFA + vehicle group was significantly suppressed at 2 days after CFA injection (n = 6 for each group, F(1,10) = 14.1, p = 0.004 between treatments by two-way ANOVA, ***p < 0.001 by Tukey’s multiple comparison test).
Experiment 5: Effect of STING inhibition on CFA-induced inflammatory hyperalgesia
Intraperitoneal administration of H-151 prior to subcutaneous CFA injection significantly suppressed the development of mechanical and thermal hyperalgesia. In the CFA + vehicle group, a significant reduction in mechanical threshold against von Frey stimulation was observed 4 h after the CFA injection, continuing for at least 2 days. In contrast, the CFA + H-151 group showed attenuation of the decrease in mechanical pain threshold on days 1 and 2 (n = 6, F(1,10) = 11.41, p = 0.007 between treatments, two-way ANOVA, ##p < 0.01, ###p < 0.001 by Tukey’s multiple comparison test). Mechanical threshold was significantly higher in the CFA + H-151 group compared to the CFA + vehicle group (n = 6 per group, F(1,10) = 11.41, p = 0.007 between treatments, two-way ANOVA, *p < 0.05 by Tukey’s multiple comparison test; Figure 4(d)). Latency against radiant heat stimulation was reduced after CFA treatment in the CFA + vehicle group. Latency against heat stimulation was also reduced in the CFA + H-151 group 4 h and 1 day after the CFA treatment (n = 6, F(1,10) = 14.1, p = 0.004 between treatments, two-way ANOVA, ##p < 0.01, ###p < 0.001 by Tukey’s multiple comparison test). Two days after the treatment, latency against heat stimulation was significantly longer in the CFA + H-151 group compared to the CFA + vehicle group (n = 6 per group, F(1,10) = 14.1, p = 0.004 between treatments, two-way ANOVA; ***p < 0.001 by Tukey’s multiple comparison test; Figure 4(e)).
Discussion
In the present study, we demonstrated that injection of mitochondrial debris induces behavioral hyperalgesia and increases STING expression in neurons of the DRG. STING inhibition successfully alleviated debris-induced hyperalgesia and CFA-induced hyperalgesia, suggesting a critical role for the STING signaling pathway in the development of inflammatory hyperalgesia.
In the present study, local injection of mitochondrial debris was found to induce hyperalgesia at the injection site, suggesting its local effect. Specifically, mechanical, but not thermal, hyperalgesia was observed after the debris treatment. There are, reportedly, subsets of DRG neurons that are responsive to a specific modality of nociception. 18 Differential hyperalgesia, which is limited to mechanical sensation, suggests that mitochondrial debris is likely to stimulate specific subpopulations of DRG neurons that are responsive to the noxious mechanotransduction 19 in a direct manner. Similarly, exposure to mitochondrial debris leads to the production of inflammatory cytokines in neurons in the central nervous system, and is associated with the progression of neuroinflammation in the central nervous system during the progression of Alzheimer’s disease.20,21
Mitochondrial DNA is included in mitochondrial debris and promotes an immune reaction by activating the cGAS-STING signaling pathway. 11 In the present study, only a few DRG neurons in the DRGs of naive rats were found to express STING. Although the percentage of DRG neurons expressing STING was low in this condition, it increased dramatically after the debris treatment. In the DRG, STING induction was detected in both CGRP and IB4-positive populations, with a preference for CGRP-positive neurons. CGRP and IB4-positive neuronal populations constitute C-fiber nociceptors in the primary afferent neurons. 22 This suggests that local injection of mitochondrial debris activates STING signaling in the nociceptors of the primary afferent neurons. Importantly, STING expression was also significantly increased in the DRG neurons in the CFA-induced inflammatory hyperalgesia model. Mitochondrial debris activates immune cells by causing local or systemic inflammation.13,23–25 In addition, our study demonstrated that STING activation is a major molecular event that occurs in primary afferent neurons during the development of inflammatory hyperalgesia.
Previous studies examining the role of STING in nociceptive function yielded conflicting results. STING in DRG neurons plays an antinociceptive role in the resolution of inflammatory pain, 26 and intrathecal injection of a STING agonist was shown to alleviate hyperalgesia in chronic pain models. 27 On the other hand, STING also has a pronociceptive function in dorsal horn and DRG neurons.28,29 Taken together, STING signaling in the DRG neurons might have multifaceted, context, and/or time-dependent roles in pain modulation. Our study demonstrated the crucial pronociceptive role of STING during the acute phase of tissue inflammation. STING expression coincided with the development of hyperalgesia after debris treatment or tissue inflammation. In addition, treatment with STING inhibitors successfully alleviated the hyperalgesia induced by mitochondrial debris injection and tissue inflammation. Therefore, it is very likely that mitochondrial DNA activates STING signaling, which drives sensitization of primary afferent neurons and, contributes, at least in part, to the development of the acute period of inflammatory hyperalgesia.
There are several limitations to this study. First, although we obtained mitochondrial debris under sterile conditions to avoid contamination, we did not sterilize or filter isolated debris due to the technical difficulty. We, therefore, cannot completely exclude endotoxin contamination of the debris. Second, systemic administration of H-151 was seen to produce an antinociceptive effect by inhibiting STING expression in the DRG neurons. However, we cannot completely exclude the possibility that H-151 might have inhibited STING signaling at other sites as well, such as the immune system and/or glial cells.
In conclusion, we identified the pronociceptive effect of mitochondrial debris. STING signaling in DRG neurons is activated by mitochondrial debris, and controls, at least in part, the development of hyperalgesia after tissue inflammation. Inhibition of mitochondrial debris and STING signaling in the DRG neurons might be a promising therapeutic target for treating inflammatory pain.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: SY received Grants-in-Aid for Scientific Research, Japan Society for Promotion of Science (24K12074). YH received Grants-in-Aid for Scientific Research, Japan Society for Promotion of Science (24K19469). FA received Grants-in-Aid for Scientific Research, Japan Society for Promotion of Science (24K02540).
Ethical considerations
All experimental procedures in this study were approved by the Animal Care Committee of Kyoto Prefectural University of Medicine and were conducted in accordance with the guidelines of the National Institutes of Health and the International Association for the Study of Pain.

