Abstract
Introduction
Postoperative pain is an adaptive response against tissue injury. In most surgical cases, however, the pain intensity is very strong 1 and has a negative impact on patient outcomes. 2 During surgery, strong noxious sensations occur in association with the action potential resulting from mechanical damage to the tissue. 3 This is followed by acute pain hypersensitivity immediately after the surgery. Primary afferent neurons increase their functional activity during surgery, thus influencing postoperative pain hypersensitivity. 4 Importantly, functional activation of the nociceptive neurons in the dorsal root ganglion (DRG) during the immediate postoperative period contributes to the development of chronic pain hypersensitivity. 5 However, the precise mechanisms responsible for activation of the DRG neurons during and immediately after surgery are unknown.
Interleukin-1 beta (IL-1β) is the central mediator of innate immunity and inflammation. 6 It is produced as an inactive precursor (pro–IL-1β) that requires processing at specific sites for its activation and secretion. 7 In the presence of harmful signals, such as damage-associated molecular patterns (DAMPs), the Nucleotide oligomerization domain (NOD)-like receptor (NLR), forms inflammasomes with caspase-1. The inflammasomes activate caspase-1, and the activated caspase-1 cleaves pro-IL-1β, leading to generation of its mature form, cleaved IL-1β (cIL-1β). 8 IL-1β plays a role as a pronociceptive molecule in peripheral tissue. 9 Activation of caspase-1 and subsequent IL-1β processing in the peripheral tissue produces pain hypersensitivity after tissue inflammation and tissue injury.10,11
In addition to its peripheral function, IL-1β is also involved in crucial signal transduction in the nervous system. Approximately 80% of the DRG neurons express pro IL-1β, further emphasizing its role in primary afferent neurons. 12 We previously discovered that both the NLRP2 inflammasome and cIL-1β exist in DRG neurons and contribute to the generation of inflammatory pain hypersensitivity. 13 However, although cIL-1β was also found to be expressed in DRG neurons in the non-inflamed condition, its function in basal nociceptive transduction is not well understood.
In the present study, we hypothesized that tissue injury triggers activation of IL-1β processing in the DRG and contributes to behavioral pain hypersensitivity. To prove this hypothesis, we investigated the expression of cIL-1β in the DRG of mice that received a plantar incision. We also performed behavioral experiments to analyze the effect of IL-1β cleavage inhibition on the development of pain hypersensitivity after the plantar incision.
Materials and methods
Animals
The experimental protocols were approved by the Animal Care Committee of Kyoto Prefectural University of Medicine. Adult male C57BL/6 mice weighing 20–25 g (Shimizu Laboratory Supplies Co, Ltd, Kyoto, Japan) were used in the study protocols, which were performed in accordance with the guidelines of the National Institute of Health and International Association for the Study of Pain (Washington, DC). Mice were housed in groups of six per cage and maintained at a 12-h light/dark cycle with food and water available ad libitum. All the surgeries and injections were performed under isoflurane inhalation anesthesia (3% v/v). At the end of the experiments, all the mice were euthanized painlessly with an overdose of pentobarbital.
Mouse plantar incision model
A mouse plantar incision model was adopted as the animal model of postoperative pain. 14 Following anesthetization with isoflurane inhalation (3% v/v), the plantar surface of the left hind paws of the mice was disinfected with 70% alcohol-impregnated cotton, and a 5 mm longitudinal incision was made with a No. 11 scalpel from 2 mm proximal to the heel to the toes. Next, the plantar muscle was lifted with a curved forceps and crushed with a scalpel and forceps. The skin was then single sutured with 5–0 nylon.
Experiment 1: Expression of cleaved IL-1β in the DRGs of plantar incision model mice
To observe changes in cleaved IL-1β (cIL-1β) expression over time, mice were randomly assigned to naive (n = 6) and at each time point after the plantar incision (5 min, 1 h, 1 day) groups (n = 6 in each group). Following treatment with a plantar incision, as described above, the mice were euthanized by intraperitoneal administration of an overdose of pentobarbital at the time points determined by their grouping, and the left L4 DRG was harvested. Naive animals did not receive any surgical and/or drug treatment. Immunofluorescence was performed to detect the expression levels of cIL-1β in DRG neurons, and to compare the cIL-1β positivity rate in neurons in each group.
Experiment 2: Analgesic effect of a caspase-1 inhibitor and IL-1 receptor antagonist
The analgesic effect of caspase-1 inhibitor on pain following the plantar incision was evaluated by behavioral assessment in response to mechanical and thermal stimulation. Mice were randomly assigned to saline and caspase-1 inhibitor (Z-YVAD-FMK, BioVision, Milpitas, CA) groups (n = 6 per group). The mice were habituated for 2 days and pre-plantar incision thresholds to mechanical and thermal stimulation were measured the day before the plantar incision. Mice were anesthetized with isoflurane (3% v/v), and a caspase-1 inhibitor (0.2 mM/5 μL, diluted in saline) or an equal volume of saline was administered intrathecally between the 5th and 6th lumbar vertebrae using a Hamilton syringe with a 26G needle (Hamilton Company, Reno, NV). 15 Once awake and confirmed no limb paralysis, the mice were re-anesthetized with isoflurane inhalation (3% v/v) 20 min later, and a plantar incision was performed. Spinal cord injury was confirmed by the limb paralysis. Behavioral tests using mechanical and thermal stimulation were performed at each of the following time points after the plantar incision: at 1 h, 3 h, 6 h, and on days 1, 3, 5, and 7.
To confirm that the caspase-1 inhibitor suppresses cIL-1β expression in DRG neurons, immunofluorescence analysis was performed on DRG tissue sections 5 min after the incision. Mice were randomly assigned to naive, saline, and caspase-1 inhibitor groups (n = 6 per group). As described above, the mice were intrathecally administered saline or caspase-1 inhibitor and the plantar incision was performed. Naive animals did not receive any surgical and/or drug treatment. Five minutes later, the mice were deeply anesthetized with an overdose of pentobarbital, and left L4 DRGs were harvested and processed for immunofluorescence analysis. The positivity rate of cIL-1β in neurons was compared among the three groups.
The analgesic effect of IL-1 receptor antagonist (RA) on pain following the plantar incision was evaluated by behavioral assessment in response to mechanical and thermal stimulation. Mice were randomly assigned to saline and IL-1 RA (500 ng/5 μL, diluted in saline, Recombinant Human IL-1ra/IL-1F3, #280-RA/CF, R&D Systems, Inc. Minneapolis, MN 55413) groups (n = 6 per group). Intrathecal drug injection and behavioral analysis were conducted as above.
Experiment 3: IL-1β-induced hyperalgesia
To evaluate the contribution of IL-1β to the development of hyperalgesia, IL-1β was administered intrathecally and behavioral assessment in response to mechanical and thermal stimulation was performed. Mice were randomly assigned to saline and IL-1β groups (n = 6 in each group). The mice were anesthetized with isoflurane (3% v/v) and IL-1β (recombinant mouse IL-1β, #I5271 Sigma-Aldrich, St Louis, MO, 5 ng/5 μL, diluted in saline) or an equal volume of saline was administered intrathecally using a Hamilton syringe with a 26G needle in a manner similar to Experiment 2. Upon awakening, behavioral analysis was performed after confirmation of the absence of spinal cord injury (at 1 h, 3 h, 6 h, and on days 1, 3, 5, and 7).
Experiment 4: Caspase-1 expression in mouse DRG
Double staining of DRG tissue sections for evaluation of caspase-1 mRNA expression by in situ hybridization and cIL-1β by immunofluorescence was performed to determine the contribution of caspase-1 to cIL-1β expression. Mice were randomly assigned to naive, saline and caspase-1 inhibitor (Z-YVAD-FMK, BioVision, Milpitas, CA) groups (n = 6 per group). Treatment of the mice and harvesting of DRGs was performed as described in Experiment 2. The rate of cIL-1β positivity in caspase-1 mRNA-positive neurons was compared among the three groups.
Experiment 5: Distribution of cIL-1β and IL-1R1 in the DRG
Double staining of cIL-1β with IL-1R1 mRNA, an IL-1β receptor, was conducted in the L4 DRG of mice 5 min after the plantar incision to assess the relative association between cIL-1β producing neurons and their receptors (n = 6).
Experiment 6: Effect of regional analgesia on cIL-1β expression
Regional analgesia was performed prior to plantar incision in IL-1β cleavage in the DRG. Mice were randomly assigned to naive, saline, or regional analgesia groups (n = 6 per group). In the regional analgesia group, 0.5% levobupivacaine was injected into the plantar (10 μL), medial (5 μL), and lateral (5 μL) aspects of the ankles of the mice using a Hamilton syringe with a 30G needle (Hamilton, Reno, NV). The saline group received the same dose of saline in the same manner as the levobupivacaine was administered in the regional analgesia group. The plantar incision was performed 20 min later under isoflurane anesthesia (3%v/v). Five minutes later, the mice were deeply anesthetized with an overdose of pentobarbital, and the left L4 DRGs were harvested and processed for immunofluorescence assessment. The positivity rate of cIL-1β in the neurons was compared among the three groups.
Experiment 7: Contribution of caspase-1 to neuronal activation
Phosphorylated ERK (pERK) in the dorsal horn of the spinal cord was evaluated to demonstrate the contribution of IL-1β cleavage in the DRG to activation of the nociceptive sensory system. Mice were randomly assigned to naive, saline, or caspase-1 inhibitor groups (n = 5 in each group). As described in experiment 2, the mice in the saline and caspase-1 inhibitor groups were respectively given saline or the caspase-1 inhibitor intrathecally, and a plantar incision was performed. Five minutes after the plantar incision, mice were deeply anaesthetized with an overdose of pentobarbital, and their L4-5 spinal cords were harvested and processed for immunofluorescence.
Behavioral assessment
The pain threshold to mechanical stimulation was measured as the response to von Frey filament stimulation (Muromachi Kikai, Tokyo, Japan). Mice were individually housed and well habituated in cages with clear acrylic partitions and raised wire mesh floors. The plantar surface of the incised side of a stationary mouse was stimulated 10 times with a filament, and its response was evaluated. A positive response was defined as two or more escape responses, such as lifting the foot or licking the plantar surface. The lightest load resulting in a positive response was determined as the pain threshold. To avoid tissue damage, the upper load limit was set at 2 g. The pain threshold to thermal stimulation was measured as the latency period against radiant heat stimulation (plantar test, Ugo Basile SRL, Gemonio, Italy). Mice were individually partitioned on a glass plate with acrylic plates and well habituated in cages with clear acrylic partitions and glass floors. Radiant heat stimuli were applied to the plantar surface of the foot through the glass floor and the latency to demonstration of escape responses, such as vigorous shaking of the foot or licking of the plantar surface, was measured. The intensity of the radiant heat stimulus was set so that the latency under untreated conditions was approximately 10 s (IR = 35). To avoid burns, an exposure time of 20 s was used as the cut-off time for stimulation.
Immunofluorescence and in situ hybridization
Tissue sections were prepared as follows. Mice were deeply anesthetized with an overdose of pentobarbital, perfused with 0.9% NaCl from the cardiac apex, and perfusion fixed in 10% neutral buffered formalin (NBF; Fujifilm Wako Pure Chemicals, Osaka, Japan). The left L4 DRGs or L4-L5 spinal cord segments were immediately harvested, fixed by overnight immersion in 10% NBF at 4°C, and then immersed in 0.1 M phosphate buffer containing 20% sucrose overnight at 4°C for cryoprotection. The DRG or spinal cord was embedded in OCT compound (Tissue Tek, Sakura Finetek, Tokyo, Japan), cut into 10 µm and 16 µm frozen sections for the DRG and spinal cord, respectively, using a cryostat (Leica Biosystems, Wetzlar, Germany) and mounted on amino-silane-coated glass slides (Matsunami Glass Ind., Ltd, Osaka, Japan).
The immunofluorescence procedure was performed as follows. Prior to primary antibody incubation, sections were immersed in Blocking One P (Nacalai Tesque Inc., Kyoto, Japan) for 30 min at room temperature to avoid nonspecific reactions. The sections were then incubated with rabbit anti-cleaved IL-1β monoclonal antibody (1:50; #63124 Cell Signaling Technology, Danvers, MA), and rabbit anti-phospho-p44/42 MAPK (Erk1/2) antibody (1:1000; #9101 Cell Signaling Technology). Primary antibodies were diluted in 0.1% Tween-20 in 0.1 M Tris-buffered saline (TBS, pH 7.4) containing 1% blocking reagent, and incubated for 3 days at 4°C. The sections were then washed in 0.1 M phosphate-buffered saline and incubated with donkey anti-rabbit IgG (H + L) Cy3 conjugated antibody (1:500 #AP182 C; Merck Millipore, Burlington, MA), donkey anti-rabbit IgG (H + L) FITC conjugated antibody (1:100 #AP182 F; Merck Millipore) overnight at 4°C. Secondary antibodies were diluted in 0.1% Tween-20 in 0.1 M Tris-buffered saline (TBS, pH 7.4) containing 1% blocking reagent. Sections were then washed in 0.1 M phosphate-buffered saline and cover-slipped with DAPI-containing anti-fading agent (Nacalai Tesque). Specificity of anti-cleaved IL-1β monoclonal antibody verification analysis by the absorption testing (Supplemental material).
The distribution of caspase-1 mRNA and IL-1R1 mRNA was detected using the RNAscope® system (Advanced Cell Diagnostics, Newark, CA), an in situ hybridization kit. The procedures were performed according to the manufacturer’s protocol for the Multiple Fluorescent Kit v2, using Mm-Casp1 (#404551; Advanced Cell Diagnostics) and Mm-Il1r1 (#413211; Advanced Cell Diagnostics) probes. For double immunofluorescence staining, in situ hybridization was performed first, followed by immunofluorescence, as described above, starting just before application of the anti-fading agent. After staining, the tissue images were observed under a fluorescence microscope (ECLIPSE E600; Nikon, Tokyo, Japan), and the images captured with a digital camera (DS-Ri1; Nikon) were assessed using image integration software (NIS-Elements BR; Nikon).
Image analysis
Digital immunohistochemistry images were captured and analyzed using Image-J (NIH, Bethesda, MD) on a Macintosh computer system (Apple, Inc., Cupertino, CA). 5 The mean signal intensity and cross-sectional area of the neurons with visible nuclei was calculated. The signal intensity of each neuron was subtracted by the background signal intensity to give the signal intensity (SI). Scatter plots were generated using prism eight software (GraphPad Software, San Diego, CA) based on the neuronal cross-sectional area and SI. Based on scatter plots, cIL-1β positivity thresholds were defined as SI >5 (rabbit anti-cIL-1β monoclonal antibody #63124), caspase-1 mRNA positivity threshold as SI >3, and IL-1R1 mRNA positivity threshold as SI >7. The digital profile count obtained by this procedure correlated well with the results of manual counting performed by the investigator who was blinded to the study procedure. For DRG sections, two sections per mouse at least 100 µm apart from each other were randomly selected, and a total of approximately 200 nucleated neurons were freehand circled and designated as regions of interest. For spinal cord sections, five sections of the L4-5 lumbar spinal cord per mouse, at least 160 µm apart from each other, were randomly selected. pERK expression was measured by visually counting pERK-positive neurons in the L4-5 spinal cord dorsal horn.
Statistical analysis
Statistical analyses were performed using GraphPad Prism eight software (GraphPad Software, La Jolla, CA). Each dataset was presented as the mean ± standard error of the mean (SEM). Behavioral scores were subjected to repeated measures two-way analysis of variance (ANOVA) with Geisser Greenhouse correction; Bonferroni’s multiple comparison test compared two groups at each time point, and Bonferroni’s multiple comparison test compared the pre- and post-treatment time points for each group. The results of immunofluorescence and in situ hybridization were subjected to one-way ANOVA with Tukey’s multiple comparison test. For all statistical analyses, the significance level α was set at 0.05 and a p value of <0.05 was considered statistically significant.
Results
Experiment 1: Expression levels of cIL-1β in DRGs in the mouse plantar incision model
cIL-1β was expressed in the cell bodies and processes of neurons in the DRG. Scatter plots of signal intensity versus cross-sectional area revealed that cIL-1b was highly expressed in relatively small neurons (Figure 1(a)). Comparison of the positivity rate of cIL-1β in the naive group versus the plantar incision revealed that cIL-1b expression was increased and peaked at 5 min and continued for 1 h after plantar incision. The positivity rate of cIL-1β after plantar incision was 28.9% in the naive group, 41.3% in the 5 min group, 36.4% in the 1 h group, and 30.5% in the 1 day group. (Figure 1(b)). Expression of cIL-1β in the DRG. (a) The left panels demonstrate cIL-1β expression (red signal). cIL-1β was detected in the neurons of the DRG. Expression of cIL-1β increased after the plantar incision. Scale bar = 50 μm. The right panels demonstrate scatter plots showing the relationship between neuronal cross-sectional area and signal intensity (SI). The cIL-1β-positive threshold was defined as SI >5. (b) Percentage of cIL-1β-expressing neurons in relation to the total profile. Neurons positive for cIL-1β increased significantly 5 min after the plantar incision. The data in the graph are presented as the mean ± SEM (n = 6 in each group). Data were analyzed by one-way ANOVA with Tukey’s multiple comparison test (*p < .05, ***p < .001, vs naive).
Experiment 2: Analgesic effect of cIL-1β inhibition in the mouse plantar incision model
The saline group showed a significant decrease in pain threshold from 1 h to 5 days after the plantar incision compared to pre-incision. The caspase-1 inhibitor group showed a significantly higher pain threshold for 1 day after the plantar incision, as compared to the saline group (Figure 2(a)). The saline group showed a significant decrease in radiant heat latency for 5 days after the plantar incision, while the caspase-1 inhibitor group showed a longer latency time at 1 h compared to the saline group (Figure 2(b)). Analgesic effect of the caspase-1 inhibitor. Behavioral analysis assessing the analgesic effect of the caspase-1 inhibitor (a) and (b). (a) The plantar incision induced a reduction in mechanical threshold against von Frey stimulation. Intrathecal caspase-1 inhibitor administration significantly inhibited the reduction of the mechanical threshold for 1 day after the treatment. (b) The plantar incision induced a reduction in latency time against thermal stimulation. Intrathecal caspase-1 inhibitor administration significantly inhibited reduction in the thermal pain latency for 1 h after the treatment. Data were analyzed by repeated measures two-way ANOVA, with Bonferroni’s multiple comparison test comparing pre- and post-treatment time points for each group (#p < .05, ##p < .01, ###p < .001), and Bonferroni’s multiple comparison test comparing both groups at each time point (*p < .05, **p < .01). The data in the graphs are presented as the mean ± SEM (n = 6 in each group). i.t.: intrathecal administration. (c) and (d) Effect of the caspase-1 inhibitor on cIL-1β expression in the DRG. (c) The panels on the left demonstrate expression of cIL-1β in the DRG in naive, saline, and caspase-1 inhibitor groups. Neurons positive for cIL-1β are labeled with FITC (Scale bar = 50 μm). The panels on the right demonstrate scatter plots showing the relationship between neuronal cross-sectional area and signal intensity. The threshold for cIL-1β positivity was defined as SI >5. Expression of cIL-1β was suppressed in the CASP1 inhibitor group. (d) Percentage of cIL-1β-expressing neurons per total profile. Compared to the naïve group, neurons expressing cIL-1β significantly increased in the saline group, although this was not observed in the caspase-1 inhibitor group. The data in the graph are presented as the mean ± SEM (n = 6 in each group). Data were analyzed by one-way ANOVA with Tukey’s multiple comparison test (**p < .01).
cIL-1β expression in DRG neurons significantly increased in the saline group 5 min after the incision. However, cIL-1β expression did not increase in the caspase-1 inhibitor group (Figure 2(c)). The cIL-1β positivity rate was significantly higher in the saline group than in the other two groups; there were no significant differences between the naive and caspase-1 inhibitor groups. The positivity rate of cIL-1β was 33.9% in the naive group, 49.9% in the saline group, and 36.9% in the caspase-1 inhibitor group (Figure 2(d)).
Treatment with IL-1 RA showed similar alleviation of pain hypersensitivity after the plantar incision. The IL-1 RA group are shown to have significantly higher pain threshold for 6 h after the plantar incision when compared to the saline group (Figure 3(a)). The IL-1 RA group showed significantly longer latency time against noxious heat compared to the saline group (Figure 3(b)). (a) The plantar incision induced a reduction in mechanical threshold against von Frey stimulation. Intrathecal IL-1RA administration significantly inhibited the reduction of the mechanical threshold for 6 h after the treatment. (b) The plantar incision induced a reduction in latency time against thermal stimulation. Intrathecal IL-1RA administration significantly inhibited reduction in the thermal pain latency for 6 h after the treatment. Data were analyzed by repeated measures two-way ANOVA, with Bonferroni’s multiple comparison test comparing pre- and post-treatment time points for each group (#p < .05, ##p < .01, ###p < .001), and Bonferroni’s multiple comparison test comparing both groups at each time point (*p < .05, **p < .01, ***p < .001). The data in the graphs are presented as the mean ± SEM (n = 6 in each group). i.t.: intrathecal administration. Intrathecal administration of IL-1β induced reduction of the mechanical threshold against von Frey stimulation (c) and reduction of latency time against thermal stimulation (d). Data were analyzed by repeated measures two-way ANOVA, with Bonferroni’s multiple comparison test comparing pre- and post-treatment time points for each group (#p < .05, ##p < .01) and Bonferroni’s multiple comparison test comparing both groups at each time point (*p < .05, **p < .01). The data in the graphs are presented as the mean ± SEM (n = 6 in each group).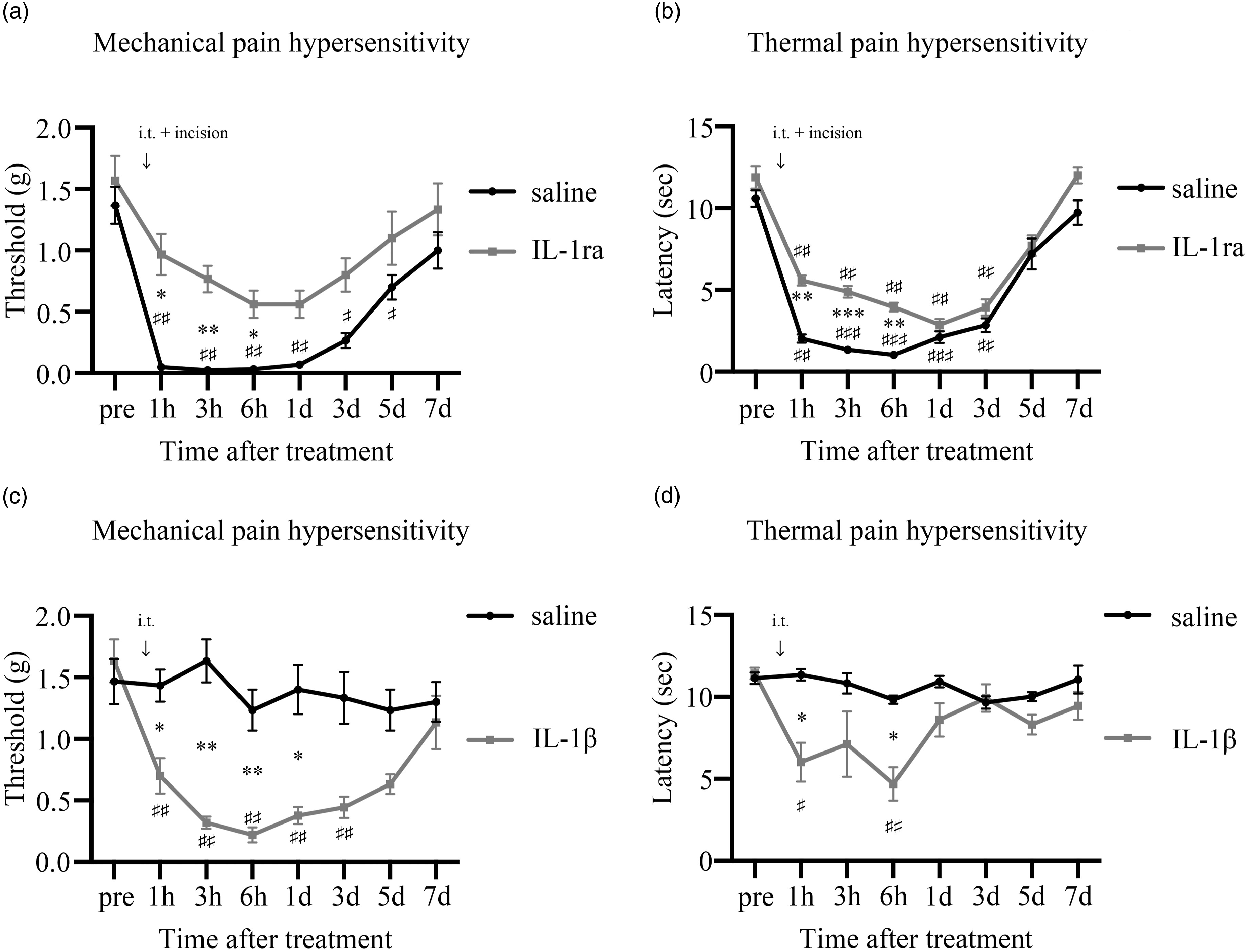
Experiment 3: IL-1β-induced hyperalgesia
The IL-1β group showed a significant decrease in mechanical pain threshold from 1 h to day 3 after administration of IL-1β. On the other hand, the saline group showed no significant decrease in mechanical pain threshold. When comparing the two groups, the IL-1β group showed a significant decrease in mechanical pain threshold from 1 h to day 1 compared to the saline group (Figure 3(c)). The IL-1β group showed a significant decrease in thermal pain threshold at 1 h and 6 h compared to pre-administration. On the other hand, the saline group showed no significant decrease in thermal pain threshold compared to its pre-dose value. When comparing the two groups, the IL-1β group showed a significant decrease in thermal pain threshold at 1 h and 6 h compared to the saline group (Figure 3(d)).
Experiment 4: Caspase-1 expression in mouse DRG
In situ hybridization showed that caspase-1 mRNA was expressed in the DRG neurons (Figure 4(a)). Double staining experiments showed that average cIL-1β positivity in caspase-1 mRNA-positive neurons was 37.7% in the naive group, 55.2% in the saline group, and 40.4% in the caspase-1 inhibitor group. The saline group had a significantly higher cIL-1β positivity rate than the other two groups; there were no significant differences between the naive and caspase-1 inhibitor groups (Figure 4(b)). The average degree of positivity for caspase-1 mRNA in DRG neurons was 60.9% in the naive group, 61.8% in the saline group, and 63.8% in the caspase-1 inhibitor group, with no significant differences among the three groups (Figure 4(c)). Distribution of caspase-1 mRNA and cIL-1β in the DRG. (a) The red areas represent caspase-1 mRNA-positive neurons and the green areas are cIL-1β-positive neurons. Scale bar = 50 μm. The scatter plots demonstrate the relationship between the signal intensity of caspase-1 mRNA and cIL-1β. The thresholds for positive expression of cIL-1β and caspase-1 mRNA were defined as SI >5 and SI >3, respectively. SI: signal intensity. (b) Percentage of cIL-1β-expressing neurons among caspase-1 mRNA-positive neurons. cIL-1β-expressing neurons significantly increased in the saline group compared to the naive group 5 min after the incision. In the caspase-1 group, the percentage of cIL-1β-expressing neurons did not increase and was significantly lower than in the saline group. The data in the graphs are presented as the mean ± SEM (n = 6 for each group). Data were analyzed by one-way ANOVA with Tukey’s multiple comparison test (***p < .001, vs saline group). (c) Percentage of caspase-1 mRNA-positive neurons. Caspase-1 mRNA expression was similar between groups. The data in the graphs are presented as the mean ± SEM (n = 6 in each group). Data were analyzed by one-way ANOVA with Tukey’s multiple comparison test.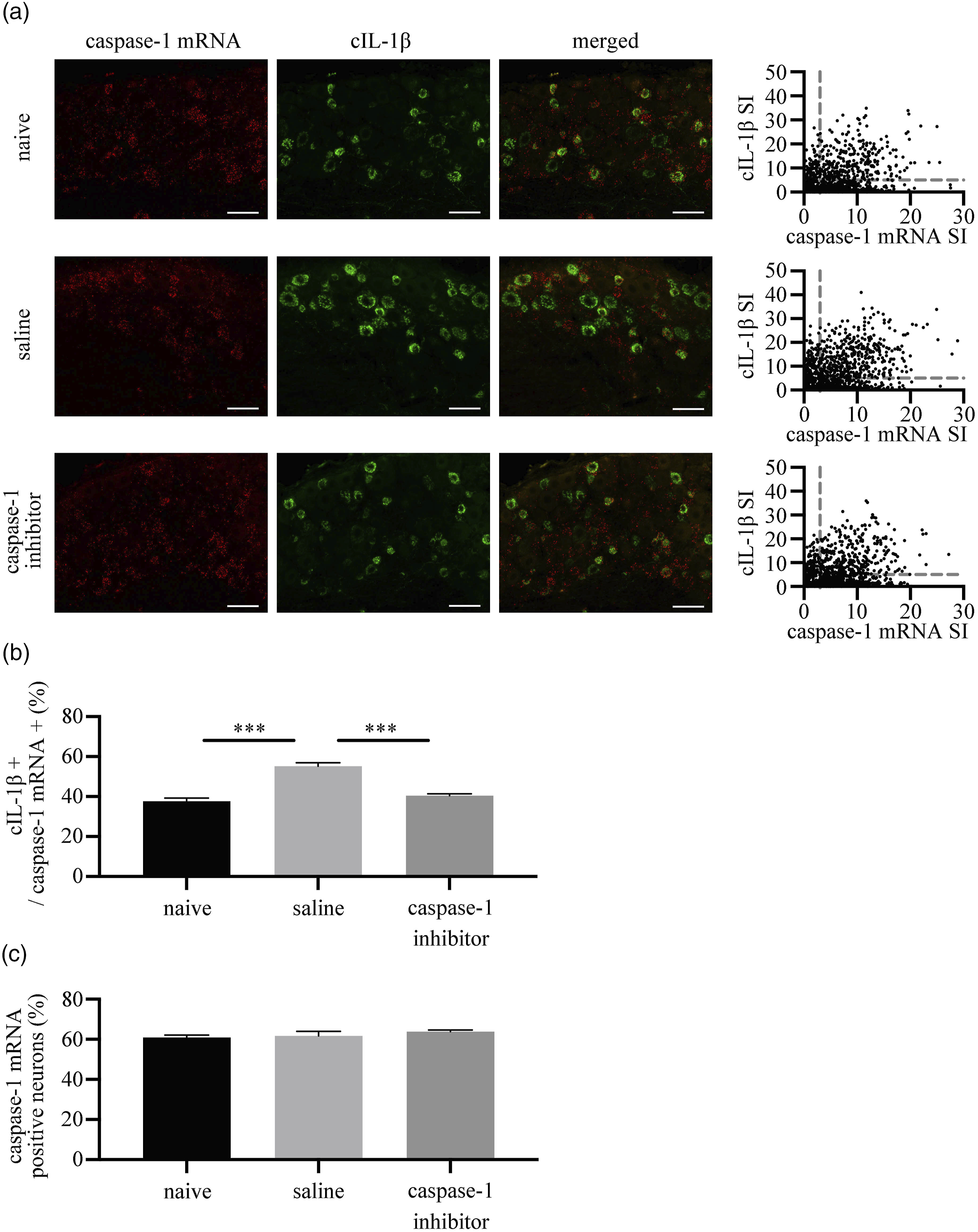
Experiment 5: Distribution of cIL-1β and IL-1R1 in the DRG
In situ hybridization showed that IL-1R1 mRNA was expressed in neurons in the DRG (Figure 5(a). IL-1R1 mRNA was detected in 36% of DRG neurons in the saline group. Double staining showed that co-localization of IL-1R1 mRNA and cIL-1β was seen in 12.2% of the DRG neurons. IL-1R1-positive and IL-1β-negative neurons averaged 23.8%, whereas IL-1R1-negative and IL-1β-positive neurons averaged 30.5% of the DRG neurons (Figure 5(b)). Distribution of cIL-1β- and IL-1R1-positive neurons in the DRG. (a) Double staining for cIL-1β and IL-1R1 mRNA. The cIL-1β-positive neurons are labeled with green and IL-1R1 mRNA-positive neurons with red (Scale bar = 50 μm). (b) Scatter plots generated from signal intensities of cIL-1β mRNA and IL-1R1 mRNA. Thresholds of positivity for cIL-1β mRNA and IL-1R1 mRNA were defined as SI >5 and SI >7, respectively. The distribution of cIL-1β and IL-1R1 is shown in the Venn diagram. SI: signal intensity.
Experiment 6: Effect of regional analgesia on cIL-1β expression
Immunofluorescence showed that, on average, cIL-1β positivity in DRG neurons was seen in 28% of the tissue samples from the naive group, 42.5% of the saline group, and 31.6% of the regional analgesia group. The saline group had a significantly higher cIL-1β positivity rate than the other two groups, with no significant difference between the naive and regional analgesia group (Figure 6). Effect of regional analgesia on injury-induced IL-1β cleavage. (a) The panels on the left show cIL-1β expression in naive, saline, and regional analgesia groups. Scale bar = 50 μm. The panels on the right present scatter plots showing the relationship between neuronal cross-sectional area and signal intensity. cIL-1β expression increased in the saline group, but remained unchanged in the regional analgesia group 5 min after the incision. The threshold for cIL-1β positivity was defined as SI >5. (b) Percentage of cIL-1β-expressing neurons relative to the total profile among the three groups. cIL-1β-expressing neurons significantly increased in the saline group compared to the naive group. cIL-1β expression did not change in the regional analgesia group and was significantly lower than that in the saline group. The data in the graphs are presented as the mean ± SEM (n = 6 in each group). Data were analyzed by one-way ANOVA with Tukey’s multiple comparison test (***p < .001, vs saline group). Expression of phosphorylated ERK (pERK) in the dorsal horn of the spinal cord after the plantar incision. (a) In the saline group, pERK expression increased in the dorsal horn of the spinal cord 5 min after the incision. pERK induction in the caspase-1 group was significantly lower than that in the saline group. (Scale bar = 100 μm). (b) Number of pERK-positive neurons in the dorsal horn of the spinal cord after the plantar incision. The number of pERK-positive neurons increased significantly in the saline group 5 min after the incision. The number of pERK-positive neurons was significantly lower in the caspase-1 group than the saline group. The data in the graph are presented as the mean ± SEM (n = 5 in each group). Data were analyzed by one-way ANOVA with Tukey’s multiple comparison test (**p < .01, ***p < .001, vs saline group).
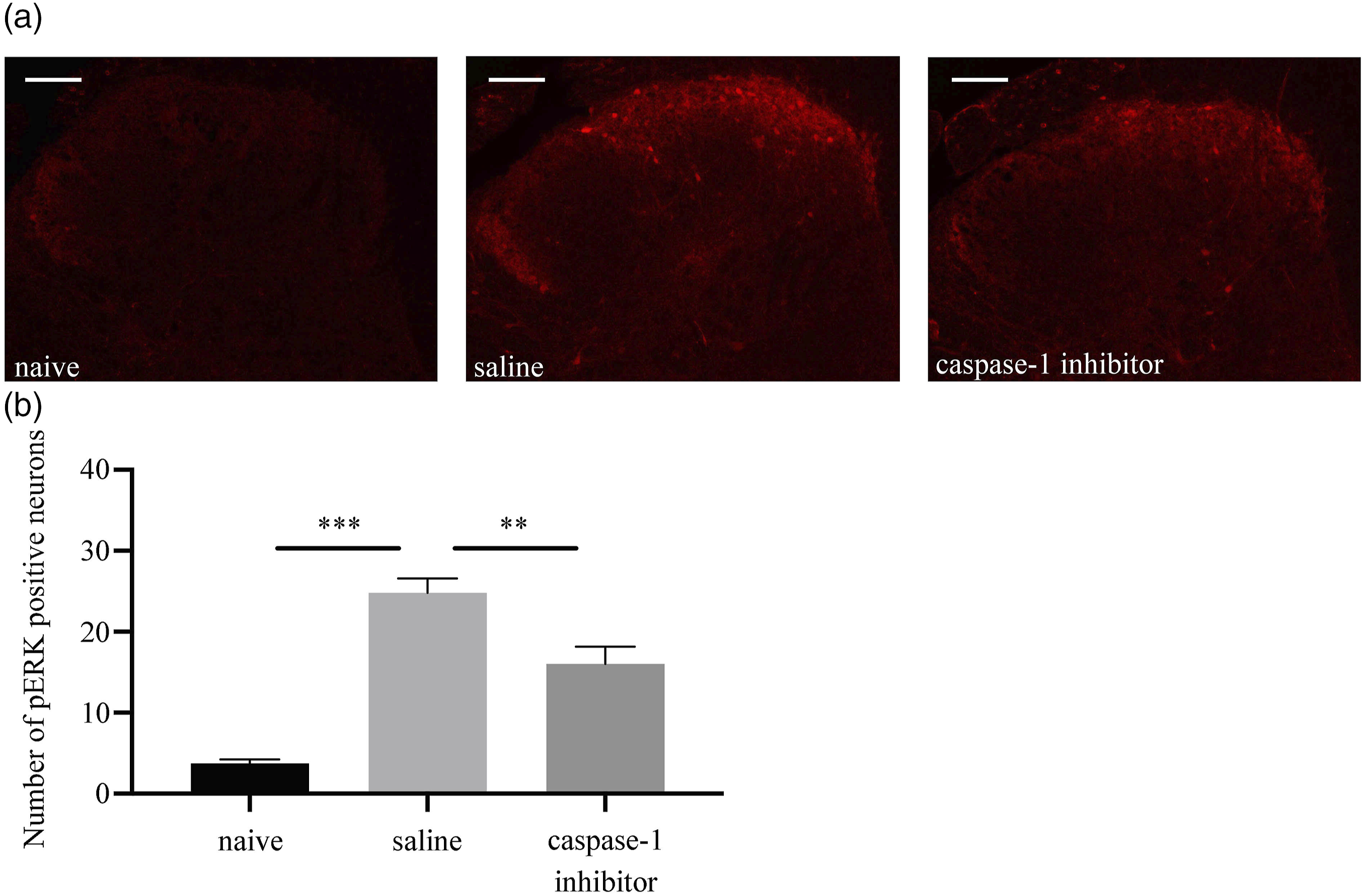
Experiment 7: Role of caspase-1 in neuronal activation
Evaluation of pERK expression in the dorsal horn of the spinal cord after the plantar incision (Figure 7) showed that the number of pERK-expressing neurons significantly increased in the saline group, while induction of pERK was not observed in the caspase-1 inhibitor group. The average number of pERK-expressing neurons was 3.7 in the naive group, 24.8 in the saline group, and 16 in the caspase-1 inhibitor group.
Discussion
Tissue injury leads to immediate activation of IL-1β processing within the DRG neurons. In this study, the increase in IL-1β expression was reversed by inhibition of caspase-1, suggesting its involvement in IL-1β processing. Inhibition of IL-1β processing and IL-1 receptor signaling successfully inhibited functional activation of the sensory neurons and behavioral pain hypersensitivity after the tissue injury, suggesting the crucial role of neuronal IL-1β in physiological nociceptive signal transduction.
IL-1β is synthesized and stored in the cell body as an inactive precursor (pro–IL-1β). It requires further processing by cleavage to become active and be secreted. 16 Inflammasomes are the intracellular machinery responsible for the sensing of cell stress and processing of IL-1β cleavage. 17 We previously discovered the presence of NLRP2 inflammasomes in primary afferent neurons. 13 In line with this observation, the present study observed expression of cIL-1β and caspase1 in the DRG neurons. Expression of cIL-1β rapidly increased after the plantar incision, indicating its role in nociceptive signal transduction after tissue injury.
The intrathecal treatment with caspase-1 inhibitor successfully inhibited induction of IL-1β cleavage after the plantar incision. Caspase-1 mRNA was detected in the DRG neurons, and expression of cIL-1β increased in the caspase1 mRNA-positive neuronal population. Therefore, caspase-1 is likely to be involved in the IL-1β cleavage processing observed in the present study. However, since previous studies suggested that there are alternative pathways responsible for IL-1β processing, such as MMP-9 18 or caspase-8, 19 comprehensive analysis investigating the mechanism of IL-1β processing is still required in the future.
Behavior analysis showed that intrathecal treatment of IL-1 RA successfully alleviated pain hypersensitivity after the tissue injury, whereas intrathecal IL-1β injection produced pain hypersensitivity. Therefore, induction of cIL-1β in the DRG has crucial role in the pathophysiology of pain hypersensitivity after the plantar incision. The analgesic efficacy of the IL-1 RA continued longer than cIL-1β expression. Intrathecal IL-1β injection produced pain hypersensitivity for 3 days. Longer duration of pronociceptive effect of IL-1β might responsive for the difference cIL-1β expression and its effect.
Analysis of the ERK phosphorylation experiment was also conducted to investigate the intensity of noxious signals in the spinal cord. Previous study showed that nociceptive specific signals arriving from the peripheral nerve induces phosphorylation of ERK in the spinal cord. 20 In our study, plantar incision induced rapid phosphorylation of ERK, and caspase-1 inhibitor inhibited ERK phosphorylation. Taken together, IL-1β increases nociceptive input to the spinal cord and produces pain hypersensitivity.
IL-1β produces hyperalgesia by activating COX-2 to synthesize prostaglandins after the tissue injury. 10 In addition to this inflammatory action, IL-1β directly activates nociceptive neurons 21 and generates action potentials. 22 The former mechanisms are characterized as production of inflammatory mediators 9 and requires some time for protein synthesis. On the other hand, the latter induces immediate nociceptive response. The involvement of IL-1β in the immediate neuronal activation observed in this study, implies contribution of non-inflammatory function of IL-1β.
Among IL-1 receptor family members, IL-1R1 is the only receptor that can bind to extracellular IL-1β and activate intracellular signal transduction. 23 In the present study, IL-1R1 mRNA signals were detected in the DRG neurons. Interestingly, neurons expressing both cleaved IL-1β and IL-1R1 were at a minority compared to those expressing either IL-1β or IL-1R1. This suggests that IL-1β acts mainly in a paracrine manner, playing a crucial role in nociceptive signal propagation in the DRG. 4
In this study, elevation of cleaved IL-1β occurred as early as 5 min after the surgical intervention. In clinical situations, activation of neuronal IL-1β in the DRG might be completed during the surgical procedure itself, given that most surgical procedures requires more than an hour. In line with our previous observation showing a beneficial effect of regional anesthesia for prevention of nociceptor activation after tissue injury, the results of the present study also suggest the merit of regional analgesia. In cases contraindicated for regional analgesia, novel pharmacological interventions that inhibit IL-1β processing might be promising therapies for the prevention of severe postoperative pain.
In conclusion, the present study demonstrated that IL-1β is rapidly cleaved in DRG neurons after tissue injury. The cleavage process of IL-1β is mediated by caspase-1 in an activity-dependent manner. IL-1β induces functional activation of the sensory nervous system and is involved in physiological nociceptive signal transduction.
This study has several limitations to note. We previously confirmed that intrathecal injection by the protocol in the present study delivers drug to L5 DRG. 13 Indeed, intrathecal caspase-1 inhibitor inhibited cIL-1β induction in the DRG neurons. However, we cannot totally exclude the involvement of spinal cord in the nociceptive effect of IL-1β. In the present study, cleaved IL-1β was detected in cell body of DRG neurons in naive condition. The relationship between cleaved IL-1β and inflammatory reaction should be analyzed in the future study.
Supplemental Material
Supplemental Material - Rapid cleavage of IL-1β in DRG neurons produces tissue injury-induced pain hypersensitivity
Supplemental Material for Rapid cleavage of IL-1β in DRG neurons produces tissue injury-induced pain hypersensitivity by Daisuke Fujita, Yutaka Matsuoka, Shunsuke Yamakita, Yasuhiko Horii, Daiki Ishikawa, Kousuke Kushimoto, Hiroaki Amino and Fumimasa Amaya in Molecular Pain
Footnotes
Author contributions
DF designed the study protocol, conducted experiments, and wrote the article.
YM, SY, DI, KK, HA and YH conducted experiments.
FA designed the study protocol and wrote the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: FA was supported by Grants-in-Aid for Scientific Research from the Japan Society for Promotion of Science (21H03026). SY was supported by Grants-in-Aid for Scientific Research from the Japan Society for Promotion of Science (22K16593). YH was supported by a Grant-in-Aid for Scientific Research from the Japan Society for Promotion of Science (22K16614).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
