Abstract
When conventional drug-based Western medicine proves ineffective, complementary and alternative medicine (CAM), including herbal medicine and acupuncture, often gains prominence. Phytochemicals, plant-derived compounds synthesized for self-protection against environmental stressors like ultraviolet radiation and insects, have also shown numerous physiological benefits in humans. Among these, flavonoid compounds – abundant in fruits and vegetables – have garnered significant research interest due to their reported biological activities, such as antioxidant, anti-inflammatory, and anticancer effects. Recent in vitro studies have provided substantial evidence that flavonoids modulate the activity of various ion channels. These channels are crucial for the generation and conduction of activation and action potentials in excitable cells, including those involved in pain transmission. These findings strongly suggest that flavonoids could serve as novel therapeutic agents for pain relief, potentially replacing existing local anesthetics. This paper discusses recent in vivo neurophysiological findings which reveal the potential of flavonoids to substitute existing local anesthetics in nociceptive and inflammatory pain. We explore the possibility of developing new drugs with fewer side effects by focusing on the common chemical structure of flavonoids, addressing the shortcomings of current anesthetics, and outlining future prospects.
Keywords
Introduction
For some patients suffering from chronic pain, conventional Western medical treatments, including drug therapy, offer limited relief. This has led to a growing interest in and use of complementary and alternative medicine (CAM), which shows promise in addressing such persistent conditions.1–3 Traditionally, Western medicine has characterized CAM as a system lacking rigorous scientific validation and widespread clinical adoption. It’s often primarily associated with practices like herbal medicine and acupuncture. However, a growing body of evidence from various studies suggests that natural compounds frequently utilized in CAM possess a range of beneficial biological actions relevant to human health. These include, but are not limited to, anti-oxidative, anti-inflammatory, and cardioprotective effects, as well as significant antinociception (pain relief).4–8
It’s generally known that existing “local anesthetics” used for treatments like surgery in medical settings, and “anti-inflammatory drugs” used to relieve inflammatory pain, both produce “side effects” in addition to their primary pharmacological action of pain relief. In current dental practice, local anesthetics are often co-administered with vasoconstrictors because local anesthetics alone provide insufficient anesthetic efficacy. Local anesthetics function as inhibitors of voltage-gated Na+ channels in excitable cells, while vasoconstrictors act as adrenergic receptor agonists. This combination is known to induce cardiovascular side effects such as arrhythmias, elevated blood pressure, and increased heart rate, posing risks for elderly patients and those with underlying conditions like hypertension or diabetes. 9 Furthermore, when performing dental procedures that require local anesthesia, such as tooth extraction, pulpectomy, or implant surgery, the efficacy of local anesthetics can be diminished if the target site is already accompanied by inflammatory pain. This reduced efficacy is due to peripheral sensitization of trigeminal nerve endings and tissue acidosis. 10
Phytochemicals, which are plant-derived chemical substances, are produced by plants to protect themselves from threats like ultraviolet radiation and insects. However, these compounds also possess numerous physiological effects that are beneficial to human health.5,6 Polyphenols comprise a large variety of molecules including some with a unique phenol ring (phenolic acids, phenolic alcohols) or typically more than one. According to both the number of phenol rings and the structural elements binding these rings to one another, polyphenols can be divided into the following groups: flavonoids, phenolic acids, phenolic alcohols, stilbenes, and lignans.11,12 Flavonoids, a natural polyphenol present in various fruits and vegetables, are an important therapeutic and chemopreventive agent used in the treatment of various illnesses. Flavonoids form the largest group of polyphenols. Categories of flavonoids include flavones (e.g. aptigenin), flavonols (e.g. quercetin), flavones (e.g. naringenin), isoflavones (e.g. genistein), Flavanols (e.g. catechin), and anthocyanin (e.g. cyanidin). 13
Although previous review articles have summarized animal studies suggesting the potential of several natural compounds, including flavonoids,10,11,13,14 there is a scarcity of reviews detailing the neurophysiological mechanisms through which flavonoids modulate the excitability of nociceptive neurons within the pain pathway during nociceptive and pathological states, employing in vivo electrophysiological methods. As shown in Table 1, recent in vitro studies demonstrate that flavonoids can modulate neuronal excitability in the nervous system. Their mechanisms involve effects on nociceptive sensory transmission (via mechanoreceptors and voltage-gated ion channels), across various tissues. These findings indicate the potential of flavonoids as CAM candidates, especially as therapeutic agents for nociceptive and pathological pain.15–23
Effect of flavonoids on the ionic channel activities (in vitro).
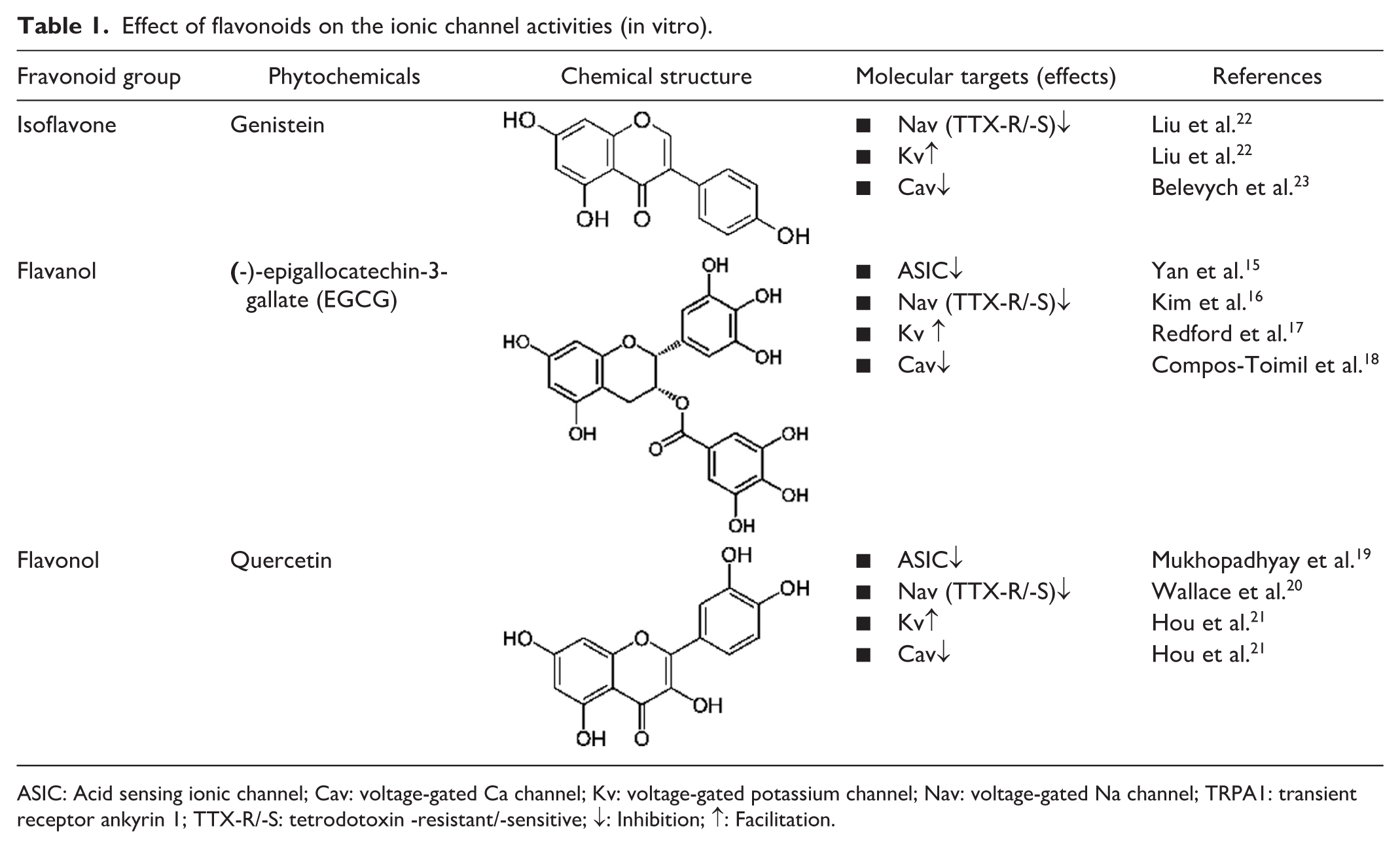
ASIC: Acid sensing ionic channel; Cav: voltage-gated Ca channel; Kv: voltage-gated potassium channel; Nav: voltage-gated Na channel; TRPA1: transient receptor ankyrin 1; TTX-R/-S: tetrodotoxin -resistant/-sensitive; ↓: Inhibition; ↑: Facilitation.
We recently conducted animal studies in our laboratory using a neurophysiological approach to investigate the pain-relieving effects of flavonoids present in foods. Our published research has shown the following effects of natural compounds: (i) local anesthetic effect on nociceptive pain; (ii) local anesthetic effect on acute inflammatory pain relief. In this review, we aim to discuss the contribution of Flavonoids to the relief of nociceptive and/or pathological pain and their potential clinical application, and specific chemical structure is important role for analgesia informed by our recent published in vivo studies.24–29 (Table 2)
Effect of flavonoids on the nociceptive neuronal activity responding mechanical stimulation under non-inflamed and inflamed conditions (in vivo). (a) Nociceptive pain.

(b) Inflammatory pain.

NNS: Non-noxious stimulation; NS: Noxious stimulation.
Clinical characteristics of nociceptive pain and pathological pain
Patients with congenital insensitivity to pain 30 are constantly exposed to life-threatening disabilities such as joint deformities due to fractures, or tissue necrosis and sepsis caused by burns. This highlights that pain functions as a crucial “biological warning signal” that protects the body from tissue damage resulting from noxious stimuli, emphasizing its essential role in our survival. 31 Physiological pain, also known as nociceptive pain, generally serves as this warning signal.
However, pathological pain refers to a state where this “biological warning signal” function is lost. It is characterized by changes in neurons within pain transmission pathways and persistent activation of signaling, leading to chronic pain. This chronic pain significantly reduces the quality of life and often persists even after the tissue injury has healed. 31 Pathological pain that no longer functions as a warning signal includes inflammatory pain and neuropathic pain. Hyperalgesia and allodynia are frequently reported symptoms in pathological pain.32,33 Hyperalgesia involves an increased sensitivity to painful stimuli, while allodynia refers to pain elicited by stimuli that are normally non-painful. 34 These pathological pain conditions are hypothesized to originate from plastic changes in sensory neurons within the somatosensory pathway, often stemming from peripheral tissue inflammation or injury. 35 Generally, peripheral sensitization, including hyperexcitability of peripheral nerve endings due to peripheral inflammation or injury, and chemical-mediated communication between neurons and between neurons and glial cells in the sensory ganglia, is thought to induce central sensitization, which includes hyperalgesia.33,36–38
A comprehensive overview of trigeminal pain transmission pathways
This review focuses on the potential contributions of flavonoids to the alleviation of nociceptive and pathological pain. Therefore, we will introduce the general characteristics of trigeminal pain pathways and nociceptive neurons. The trigeminal nociceptive sensory pathway is divided into lateral and medial pathways. The lateral pain system is responsible for transmitting sensory input regarding the discriminative features of pain, particularly its location and intensity. In contrast, the medial pain system mediates the emotional and affective components of pain experience. The trigeminal system is characterized by unique anatomical organization and physiological mechanisms specialized for processing nociceptive input and non-nociceptive sensory information from the orofacial region. Specifically, the trigeminally innervated areas (oral mucosa, tooth pulp, and temporomandibular joint) receive innervation from small-diameter myelinated Aδ fibers and unmyelinated C fibers, which play crucial roles in the transmission and perception of orofacial nociception. 33
Nociceptive sensory input originating from areas innervated by trigeminal ganglion (TG) neurons is transmitted from trigeminal afferent fibers to second-order neurons located in the trigeminal spinal trigeminal nucleus (SpV) in the brainstem and the upper cervical spinal cord (C1-C2).33,34 The SpV, a pivotal relay point in the processing and transmission of orofacial sensory information, exhibits a functional organization consisting of three distinct subnuclei: the oralis, interpolaris, and caudalis.33,34 The SpV caudalis (SpVc), along with the C1-C2 dorsal horn, serves as a primary relay site for trigeminal nociceptive signals originating from inflammation and tissue injury.33,34 Subsequently, projection neurons located within the SpVc and the C1-C2 segments of the spinal cord extend axonal projections to thalamic nuclei, specifically the ventral posteromedial nucleus (VPM) and medial thalamic nuclei, as well as the parabrachial nucleus (PBN).39–42 Nociceptive neurons that receive nociceptive sensory input from the orofacial region are somatotopically organized within the VPM thalamic nucleus. However, this precise spatial arrangement is not observed in the medial thalamic nuclei or the PBN. Specifically, noxious stimuli originating from intraoral structures are relayed to the somatosensory cortex via the medial division of the VPM, whereas nociceptive signals from the face and head are transmitted via the lateral division of the VPM. 42
Mechanisms of pain transmission: The role of electrical and chemical signals
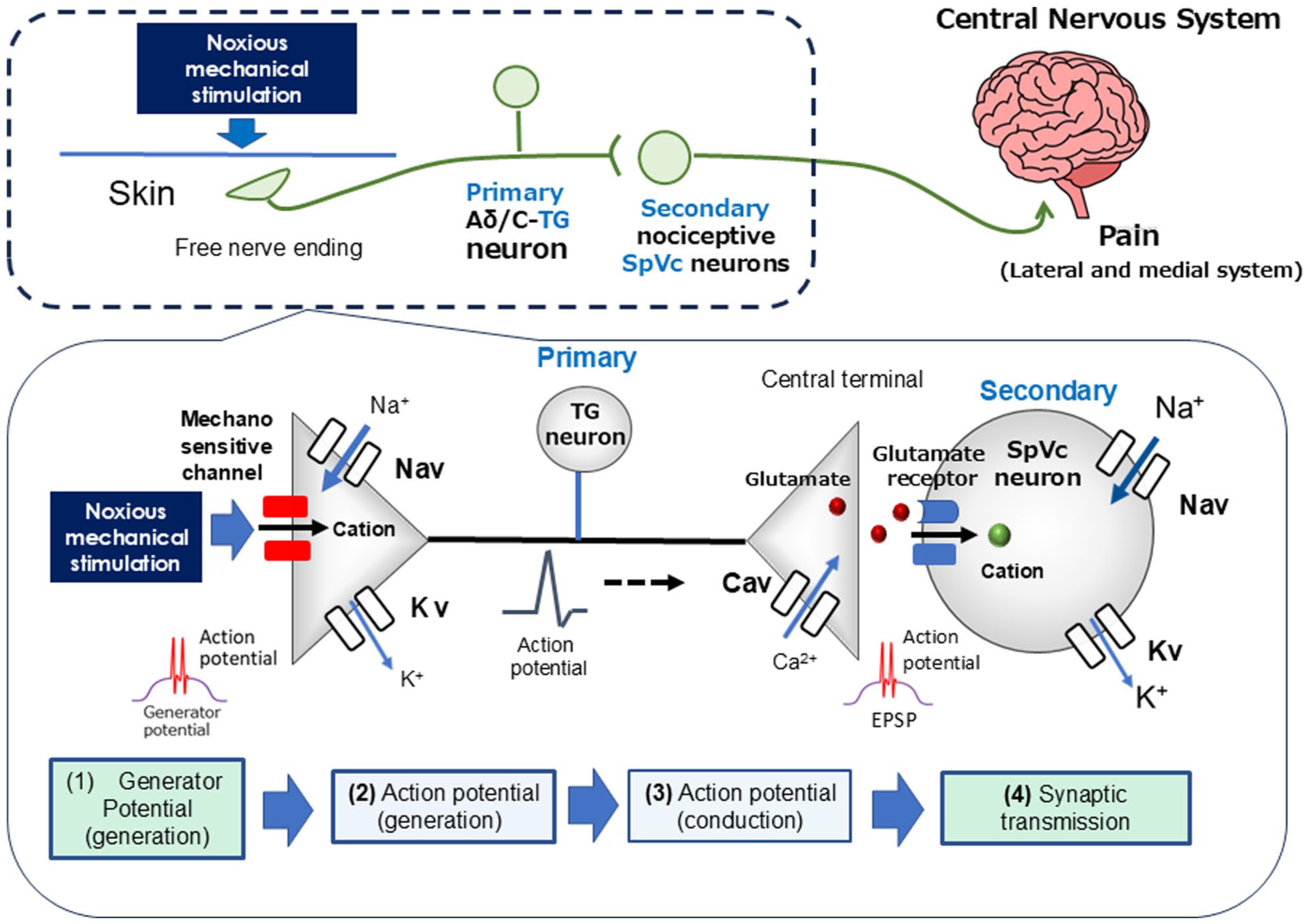
Primary afferent nerve fibers include both myelinated Aδ fibers (thin, slow-conducting) and unmyelinated C fibers, both of which are essential for pain transmission, including those from TG neurons.33,34 Aδ fibers mediate sharp, well-localized “fast” pain, while C fibers transmit dull, poorly localized “slow” pain.33,34 TG neurons are characterized by a pseudounipolar morphology, possessing a central axon that forms chemical synaptic connections with second-order neurons. Conversely, their peripheral axons form nociceptive free nerve endings.33,34 Nociceptors function as energy transducers, converting external noxious stimuli (thermal, cold, mechanical, or chemical energy) into electrical signals. 32 As illustrated in Figure 1, the processing of sensory information in primary afferent fibers can be broadly categorized into four stages: (i) Transduction, where peripheral terminals convert external stimuli; (ii) Generation and initiation of action potentials; (iii) Conduction of action potentials along the neuron; and (iv) Transmission, where central terminals form the presynaptic component of the first synapse in the central nervous system (CNS) sensory pathway.33,43

Trigeminal nociceptive sensory transduction involves a precise signaling cascade. When noxious mechanical stimulation is applied to the tissue, it activates a generator potential in the peripheral terminal of trigeminal ganglion (TG) neurons. This depolarization subsequently opens Nav and Kv channels, thereby generating action potentials. These action potentials are conducted through primary afferent TG neurons to the central terminal in nociceptive neurons within the spinal trigeminal subnucleus caudalis (SpVc). Here, presynaptic Cav channels open, leading to the release of neurotransmitters into the synaptic cleft. These neurotransmitters then bind to postsynaptic ionotropic glutamate receptors, activating excitatory postsynaptic potentials (EPSPs). If the EPSP amplitude surpasses the action potential threshold, a barrage of action potentials is conducted to higher centers in the pain pathway, resulting in pain perception.
When noxious mechanical stimuli are applied to the peripheral receptive field of the skin, candidate nociceptive mechanosensitive ion channels expressed in free nerve endings, such as TRPA1 (transient receptor potential ankyrin 1) and ASICs (acid-sensing ion channels), are activated. This activation increases the cation permeability of the cell membrane, resulting in an inward current and the generation of a depolarizing generator potential.44–47 The non-propagating generator potential, acting as an analog signal with an amplitude proportional to stimulus intensity, can initiate a digital action potential governed by the all-or-none law.
The nociceptor of a primary sensory neuron is known as a “trigger potential” because it initiates action potentials. When the membrane potential of a free nerve ending reaches the threshold for action potential generation in response to a noxious stimulus, an action potential is triggered. This rapid electrical phenomenon consists of a depolarization phase mediated by the opening of voltage-gated sodium (Nav) channels and subsequent influx of Na+ ions, and a repolarization phase driven by the opening of voltage-gated potassium (Kv) channels and subsequent efflux of K+ ions.33,43 Nociceptive neurons express both tetrodotoxin-sensitive (TTX-S) and tetrodotoxin-resistant (TTX-R) Nav channels. While Aδ neurons express both TTX-S and TTX-R Nav channels, C neurons primarily express TTX-R Nav channels. 48
Noxious stimuli applied to the receptive field induce generator potentials with amplitudes proportional to the stimulus intensity, which then modulate the firing frequency of action potentials.33,43 Action potentials initiated at the peripheral free nerve endings are actively propagated along the axon to the central terminals through the sequential opening and closing of Nav and Kv channels. Depolarization of the central nerve terminal upon arrival of the action potential activates voltage-gated calcium (Cav) channels, leading to an influx of calcium ions (Ca2+). The increase in intracellular Ca2+ levels at the presynaptic terminal triggers the release of excitatory neurotransmitters, including glutamate, into the synaptic cleft. These neurotransmitters bind to ionotropic glutamate receptors on second-order sensory neurons, causing cation influx and subsequent neuronal excitation. The cation influx resulting from glutamate receptor activation generates an excitatory postsynaptic potential (EPSP). The depolarization caused by an EPSP, if it reaches the firing threshold, can initiate an action potential in the postsynaptic neuron. The amount of neurotransmitter released is believed to determine the amplitude of the EPSP. Subsequently, the firing frequency of action potentials, which correlates with the EPSP amplitude, is processed by higher central nervous system centers as encoding pain intensity.33,43
Flavonoids: In vitro modulation of excitable tissues
Table 1 summarizes the in vitro effects of several flavonoids (Genistein, (-)-epigallocatechin-3-gallate, Quercetin) on ion channel activity. Genistein, a naturally occurring isoflavone found in soybeans, is a polyphenol with various biological functions, including cardioprotective, neuroprotective, and anticancer properties. 49 Genistein’s modulatory role on voltage-gated ion channels, specifically Nav, Kv, and Cav channels, has been previously reported.22,23,50 For instance, Liu et al. 22 showed that genistein inhibits action potential firing in capsaicin-sensitive nociceptive TG neurons by reducing both TTX-R and TTX-S Nav channel currents in vitro. Notably, TTX-R Nav currents are selectively expressed in small- and medium-diameter sensory ganglion neurons, such as nociceptive neurons.33,48
Catechins, a type of flavonoid found in green tea, are naturally occurring polyphenols. (-)-epigallocatechin-3-gallate (EGCG) is the primary active ingredient, comprising over 10% of the dry weight and 65% of the total catechin content of green tea extracts. 51 EGCG is known for its beneficial biological properties, including antioxidant, anticancer, and anti-inflammatory effects. 51 EGCG influences neuronal excitability by modulating various ion channels, such as Nav, Kv, Cav channels, and acid-sensing ion channel 3 (ASIC3).15–18 Kim et al. 18 previously showed that EGCG dose-dependently inhibits both TTX-R and TTX-S Nav channels in dorsal root ganglion (DRG) neurons in vitro. TTX-R Nav channels are selectively expressed in small- and medium-diameter sensory ganglion neurons, such as nociceptive neurons.33,48
The flavonoid quercetin is a phytochemical known for its diverse biological functions, including antioxidant, anti-inflammatory, and cardioprotective properties.52–54 It has a documented modulatory role on Nav, Kv, and Cav channels in cardiac muscle.20,21 For instance, in an in vitro rat coronary arterial smooth muscle preparation, quercetin induces vasorelaxation by enhancing Kv channels and depressing Cav channels. 21 Interestingly, a study by Wallace et al. 20 using a whole-cell patch clamp technique in rat cardiac myocytes showed that red grape polyphenols, specifically quercetin, catechin, and resveratrol, all inhibited Nav currents. The half-maximal inhibitory concentrations (IC50) followed the order: quercetin > catechin > resveratrol. Furthermore, quercetin has been shown to inhibit ASIC. 19 Previous research suggests that ASICs may function as mammalian cutaneous mechanoreceptors in the nerve terminals of TG neurons.44,46,47
Flavonoids: In vivo modulation of nociceptive neurons
Nociceptive pain
Table 2 summarizes the in vivo effects of flavonoids (Genistein, EGCG, Quercetin) on nociceptive neuronal activity in response to mechanical stimulation under nociceptive conditions. Building on in vitro studies that showed genistein reversibly suppresses both TTX-R and TTX-S Na (Nav) channel currents in acutely isolated TG neurons, 22 Yamaguchi et al. 24 investigated its in vivo effects. Their study examined whether local administration of genistein could attenuate the excitability of rat spinal trigeminal nucleus caudalis (SpVc) wide-dynamic-range (WDR) neurons in response to mechanical stimulation. 24 The key findings were: (i) The mean firing rate of SpVc WDR neurons was dose-dependently and reversibly reduced by local injection of genistein (0.1–10 mM) in response to both non-noxious and noxious mechanical stimuli. (ii) The magnitude of inhibition from 10 mM genistein was comparable to that of 1% lidocaine (37 mM). (iii) A half-dose of lidocaine could substitute for a half-dose of genistein, suggesting a similar mechanism of action. These results suggest that local injection of genistein into the peripheral receptive field suppresses the excitability of SpVc neurons, likely by inhibiting Nav channels in the nociceptive nerve terminals of the TG. Consequently, genistein shows promise as a potential local anesthetic for trigeminal nociceptive pain, possibly with fewer side effects, making it a candidate for CAM.
Building on previous in vitro whole-cell voltage-clamp studies by Kim et al., 16 which demonstrated that EGCG dose-dependently inhibits both TTX-R and TTX-S Nav channel currents in DRG neurons, Uchino et al. 25 investigated the in vivo effects of EGCG. Using extracellular single-unit recordings, 16 they found that local administration of EGCG significantly and reversibly inhibited the mean firing frequency of SpVc WDR neurons. This inhibition was dose-dependent and occurred in response to both non-noxious and noxious mechanical stimuli. The study also revealed that the magnitude of inhibition by EGCG was similar to that of the local anesthetic 1% lidocaine. Furthermore, a half-dose of lidocaine could effectively substitute for a half-dose of EGCG, suggesting a similar mechanism of action. These results, combined with the finding that EGCG application can activate Kv channels to cause vasodilation, 17 suggest that EGCG suppresses the excitability of nociceptive SpVc neurons by inhibiting Nav channels and possibly by opening Kv channels in the TG. Therefore, EGCG holds promise as a potential local anesthetic for trigeminal nociceptive pain, potentially offering a therapeutic option with fewer side effects.
Previous research on the local anesthetic effects of flavonoids, such as genistein and EGCG, on nociception has primarily focused on secondary neurons, leaving the effects on primary afferent activity largely unexamined. This highlights the need for further investigation into whether local administration of these flavonoids can attenuate the excitability of nociceptive primary neurons in response to in vivo mechanical stimulation. Our recent in vivo studies, building on prior in vitro investigations, have addressed this gap. In vitro studies previously demonstrated that the phytochemical quercetin inhibits Nav and Cav channels while activating Kv channels in cardiac muscle tissue.20,23 Consistent with these findings, our work 26 revealed that local administration of quercetin into the peripheral receptive field suppressed the excitability of rat nociceptive primary sensory neurons in the TG. This effect is possibly mediated by the inhibition of Nav channels and the activation of Kv channels at the nociceptive nerve terminals.
Consequently, under non-neuropathic and non-inflammatory conditions, the local anesthetic application of flavonoids may provide a means of alleviating trigeminal nociceptive pain with a reduced incidence of adverse effects, thus contributing to the integration of CAM. A comparative analysis of the inhibitory effects of these phytochemicals and lidocaine on the discharge frequency of nociceptive neurons indicates the following relative local anesthetic efficacy against noxious stimuli: genistein (10 mM) ≈ EGCG (10 mM) ≈ quercetin (10 mM) > 1% lidocaine (37 mM). This suggests that these flavonoids may achieve a similar effect at a concentration roughly one-quarter that of 1% lidocaine. 55 The rationale for this difference in potency likely stems from the fact that these phytochemicals target a wider range of molecules at nerve terminals than lidocaine. This includes inhibiting Nav channels, suppressing mechanosensitive channels, and activating Kv channels. However, further patch-clamp investigations on TG neurons are necessary to fully corroborate this hypothesis. 56 Figure 2 summarizes the possible molecular targets for flavonoids as local anesthetic agents.
Potential molecular targets for flavonoids as local anesthetic agents include mechanosensitive ionic channels, Nav channels, and Kv channels. When locally applied to peripheral tissues, natural compounds (genistein, quercetin, (-)-epigallocatechin-3-gallate) inhibit the generation of generator potentials and/or action potentials in the peripheral terminals of primary afferents following nociceptive stimulation by blocking ASICs, inhibiting Nav channels, T-type Cav and facilitating Kv channels.
Our recent findings indicate that local administration of EGCG to peripheral receptive fields diminishes the responsiveness of both primary sensory neurons in the TG and secondary SpVc neurons. 29 A comparison of EGCG’s inhibitory effects on these two neuronal populations reveals that the magnitude of inhibition is greater in secondary neurons than in primary neurons in response to both non-noxious and noxious stimuli. This finding differs from our earlier study. 25 The precise reason for this disparity in the inhibition of discharge frequency is not fully understood, but it is likely linked to the convergence of sensory information. Primary sensory neurons relay information from the periphery to second-order neurons, where signals from multiple primary neurons and interneurons converge. 43 This convergence at the secondary neuron level, which does not occur at the primary level, is hypothesized to be a major contributing factor. The more pronounced suppressive effect of EGCG on second-order neurons is particularly interesting, as it suggests that the flavonoid may be acting on a higher pain transmission center, thereby amplifying the degree of pain relief.
Inflammatory pain
Previous studies suggest that ASICs are candidate mechanoreceptors in mammalian cutaneous nerve terminals of TG neurons. 44 Furthermore, the phytochemical quercetin has been shown to inhibit ASIC currents. 19 Under inflamed conditions, tissue pH can fall below 6.0, activating primary nociceptive afferents via ASICs. 57 This is clinically relevant, as inflammation often diminishes the efficacy of local anesthetics, particularly dental anesthetics. 58 Research by Fu et al. 59 has indicated that peripheral inflammation can upregulate ASICs in TG neurons, suggesting that specific ASIC inhibitors may have a significant analgesic effect on orofacial inflammatory pain. These findings suggest that direct local administration of quercetin could inhibit generator potentials and subsequently suppress action potential firing in nociceptive TG neurons by inhibiting ASIC and Nav channels while opening Kv channels in inflamed tissues. In a recent study, Sashide et al. 27 tested this hypothesis by reporting the suppressive effects of local quercetin injection on the excitability of nociceptive primary sensory neurons in the TG under inflamed conditions. Their key findings were: (i) Quercetin dose-dependently (1–10 mM) and reversibly inhibited the mean firing frequency of TG neurons in response to both non-noxious and noxious mechanical stimulation. (ii) The local anesthetic 1% lidocaine (37 mM) also reversibly inhibited the mean firing frequency of inflamed TG neurons in response to mechanical stimuli. (iii) The mean magnitude of inhibition of TG neuronal discharge frequency with just 1 mM quercetin was significantly greater than with 1% lidocaine.
Figure 3 shows that locally administered quercetin suppresses the excitability of nociceptive primary sensory TG neurons in inflamed tissue. This is likely due to its inhibition of ASIC, Nav, and Cav channels and activation of Kv channels. Consequently, quercetin may be a more effective local analgesic than selective Nav channel blockers, as it targets both generator and action potentials in nociceptive nerve terminals. 27 This highlights quercetin’s potential in CAM. We expect that quercetin will become a viable alternative to conventional local anesthetics, especially for inflammatory conditions, thus broadening the clinical application of CAM. Furthermore, structural analysis of quercetin analogs is anticipated to be key in designing new local anesthetics specifically for inflamed tissues.

The local anesthetic effect of the flavonoids (quercetin and EGCG) is influenced by inflammatory conditions. Inflammation-induced tissue acidity lowers the pH, affecting the ionization equilibrium of lidocaine. The subsequent reduction in the non-ionized form of lidocaine diminishes its binding to Nav channels, thereby decreasing its local anesthetic efficacy in inflamed environments. In contrast, quercetin’s inhibitory action on inflamed tissues may stem from the suppression of action potential firing frequency via the inhibition of nociceptive mechanosensitive channels (ASICs), voltage-gated sodium channels (Nav), T-type calcium channels (Cav), and the activation of potassium channels (Kv). As quercetin and EGCG demonstrates a significantly greater inhibitory potency on discharge frequency compared to lidocaine, it possesses a robust local anesthetic effect on inflamed tissues, suggesting its potential utility within complementary and alternative medicine.
A recent study by Utugi et al. 29 investigated whether acute local administration of EGCG could reduce the excitability of nociceptive TG neurons in response to mechanical stimulation under inflammatory conditions. The principal findings were: (i) Day 1 CFA-inflamed rats showed a significantly reduced mechanical escape threshold compared to baseline, demonstrating mechanical hyperalgesia. (ii) The edematous area in the whisker pad significantly increased in CFA-inflamed rats. (iii) CFA-inflamed rats exhibited hyperexcitability, marked by a decreased mechanical threshold and increased spontaneous and mechanically-induced discharges. (iv) EGCG significantly and dose-dependently reduced the mean firing frequency of TG neurons in response to both non-noxious and noxious stimuli. This inhibition was maximal within 45 min, persisted for 60 min, and was reversible. (v) Vehicle Control: The vehicle administration had no significant effect on discharge frequency. These results indicate that localized EGCG administration effectively mitigates the excitability of primary sensory neurons in inflamed tissue (Figure 3). This outcome is further supported by our previous research, which showed that another phytochemical, quercetin, also reversibly and dose-dependently inhibits TG neuronal activity in inflamed tissues. 27
Flavonoids and pain: Functional significance and therapeutic perspectives
The growing interest in CAM underscores the need for effective alternatives when Western medicine-based pharmacotherapy fails. Our recent in vivo neurophysiological studies have shown that natural compounds, flavonoids target multiple molecular pathways to produce local anesthetic and anti-inflammatory pain effects. The absorption and distribution of flavonoids in tissues are not yet fully understood. 60 However, it is speculated that locally administered flavonoids bypass this first-pass metabolism, allowing pharmacological concentrations of the unchanged parent compound to reach the target molecules. Research focused on harnessing the pain-relieving potential of natural compounds, which can circumvent the various side effects of conventional drugs, holds significant importance for the development of highly safe, non-pharmacological treatment modalities. Considering the available evidence regarding the potential toxicity of flavonoids, the limitations of animal studies, and the restricted epidemiological data, it is currently difficult to draw definitive conclusions about the toxicological effects of flavonoids on human health, to the best of our knowledge. 61 Our in vivo research, as detailed in this review, has established that natural compounds demonstrate: (i) local anesthetic properties against nociceptive pain, with an efficacy profile closely matching that of lidocaine, a standard Nav channel antagonist; (ii) a potent local analgesic effect in acute inflammatory pain, exceeding that of conventional Nav channel blockers at the inflammatory site.
Consequently, these findings suggest that flavonoids offer a potential avenue for alleviating both nociceptive and inflammatory pain, highlighting their promising clinical utility. We expect that future research building upon these results will lead to impactful discoveries that significantly advance medical care, particularly in drug discovery for functional foods with analgesic properties, drug-free CAM therapies, and the development of safer analgesics with reduced side effects. Yan et al. 15 conducted whole-cell patch-clamp experiments using various types of flavonoids to determine the chemical structures involved in inhibiting the ASIC3 channel current. They reported that multiple chemical structural sites of EGCG are involved in the inhibition of the ASIC3 channel current. The common chemical structure found in the flavonoids (genistein, catechin, quercetin) listed as polyphenols in Table 1 is speculated to have the potential to modulate voltage-gated ion channels (Nav, Kv, Cav). Thus, exploring this potential moving forward will be critical for advancing local anesthetics beyond current Na channel inhibitors. Finally, given that the current study focused on elucidating the “discriminative aspects” of pain, future investigations should explore the influence of natural compounds on the “emotional aspects” of pain.
Finally, this review highlighted the role of flavonoid compounds (such as isoflavones, quercetin, and catechins) – plant-derived secondary metabolites – as potential alternatives to local anesthetics. We specifically hypothesize that their common diphenylpropane structure modulates voltage-gated and mechanosensitive ion channels on nociceptive primary neurons, thus suppressing neuronal excitability. These fundamental insights are crucial for developing new analgesics, including those that could substitute for local anesthetics in surgery or be formulated as effective topical ointments for relief. The potential contribution to drug-free CAM and the creation of analgesics free of side effects makes this research highly significant in terms of creativity and impact.
Concluding remarks
A variety of flavonoids, including genistein, catechin, quercetin have been previously hypothesized to modulate voltage-gated and mechanosensitive ion channels based on in vitro studies. Building on this knowledge, a neurophysiological analysis was performed in vivo to investigate how the excitability of primary and secondary nociceptive neurons is altered in both non-inflamed and inflamed tissues. The analysis revealed that in non-inflamed tissues, flavonoids inhibited excitability in response to noxious stimuli at concentrations as low as one-fourth that of conventional local anesthetics. Notably, in inflamed tissues where lidocaine was ineffective, both quercetin and catechin successfully suppressed nociceptive excitability, exhibiting a local anesthetic effect equivalent to that observed in healthy tissue. This body of evidence suggests that flavonoids have the potential to act as a substitute for conventional local anesthetics, thus contributing to the field of complementary and alternative medicine. The common chemical structure found in the flavonoids (genistein, catechin, quercetin) listed as polyphenols is speculated to have the potential to modulate voltage-gated ion channels (Nav, Kv, Cav). Thus, exploring this potential moving forward will be critical for advancing local anesthetics beyond current Na channel inhibitors.
Footnotes
Author contributions
US, RT, YS contributed to the acquisition, analysis, interpretation for the work. MT designed the framework of the manuscript and wrote the manuscript. All the authors have read and approved the paper.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a Grant-in-Aid for Scientific Research (C) from the Japanese Society for the Promotion of Science (No.22K10232).
