Abstract
Background:
Acupuncture effectively reduces chronic neck pain and plasma Substance P (SP) levels, but upstream molecular mechanisms remain unknown.
Objectives:
We aimed to identify circulating microRNAs (miRNAs) associated with acupuncture-induced SP reduction and explore potential neuroplasticity mechanisms.
Methods:
We performed longitudinal plasma miRNA profiling (Affymetrix miRNA 4.0 Array, ~4600 miRNAs) in chronic neck pain patients: Acupuncture group (n = 3; 0, 4, 8 weeks) and Control group (n = 3; 0, 4 weeks). Linear Mixed-Effects Models (LMMs) tested associations between each miRNA and SP dynamics (miRNA × Time × Group interaction). Statistical significance was validated using permutation testing (2000 iterations).
Results:
Screening identified 53 miRNAs significantly associated with SP, validated by permutation testing (p < 0.001). Fourteen high-confidence miRNAs showed significant three-way interactions, indicating treatment-specific SP relationships. The most significant was miR-1302-6 (p = 7.65 × 10−6), followed by miR-181b-2. These miRNAs displayed diverse temporal patterns: some (miR-196b, miR-6788) increased during treatment, while others (let-7d, miR-1302-6) decreased parallel to SP. Functional enrichment revealed striking convergence on neuroplasticity pathways: axon guidance (p = 2.61 × 10−6), MAPK signaling (p = 4.18 × 10−5), neuron projection development (p = 7.52 × 10−10), and synaptic structures (p = 9.52 × 10−12).
Conclusions:
This exploratory study provides first molecular evidence for an acupuncture-miRNA-SP axis in chronic pain. The enrichment of neuroplasticity pathways suggests acupuncture may induce structural remodeling of nociceptive circuits rather than simply suppressing inflammation, offering novel mechanistic insights and potential biomarkers for personalized acupuncture therapy. The trial was registered with the Korean Clinical Trial Registry (KCT0005363).
Introduction
Chronic neck pain affects millions globally, imposing substantial healthcare burdens with limited effective treatments. Among non-pharmacological interventions, acupuncture has demonstrated clinical efficacy, yet its molecular mechanisms remain poorly understood, hindering optimization and personalization of therapy.
Our previous work established that acupuncture treatment in chronic neck pain patients leads to parallel reductions in clinical pain scores and plasma Substance P (SP), a key neuropeptide in nociceptive signaling and neurogenic inflammation. 1 However, the upstream molecular events mediating this SP reduction remain unexplored.
MicroRNAs (miRNAs), small non-coding RNAs regulating post-transcriptional gene expression, have emerged as critical modulators of pain pathophysiology.2,3 Circulating miRNAs are particularly attractive as they can cross the blood-brain barrier and reflect central nervous system activity, with evidence demonstrating correlations between circulating and brain tissue miRNA levels in chronic pain states. Their stability in circulation makes them attractive biomarker candidates, while their ability to regulate multiple targets simultaneously positions them as potential therapeutic mediators. Recent evidence indicates miRNAs regulate neuronal excitability, synaptic plasticity, and inflammatory responses in various pain models.4,5
Despite growing interest in miRNA-pain relationships, no studies have examined whether circulating miRNAs mediate acupuncture’s analgesic effects. This knowledge gap represents a critical barrier to understanding acupuncture at the molecular level.
We hypothesized that specific circulating miRNAs, modulated by acupuncture, would show temporal associations with SP reduction. Using longitudinal profiling and sophisticated statistical modeling, we aimed to: (1) identify miRNAs whose dynamics correlate with acupuncture-induced SP changes, and (2) elucidate potential biological pathways through which these miRNAs might mediate analgesia.
This exploratory study employed Linear Mixed-Effects Models (LMMs) to maximize statistical power from limited samples while accounting for individual heterogeneity – a novel approach in acupuncture-miRNA research. By focusing on treatment-specific temporal patterns rather than simple pre-post comparisons, we sought to identify miRNAs most likely involved in acupuncture’s therapeutic mechanism.
Methods
Study design and participants
This study represents a secondary exploratory analysis utilizing plasma samples from our previous clinical trial 1 (KCT0005363). From the original cohort, we selected six middle-aged women with chronic neck pain: an Acupuncture group (n = 3) and a waitlist Control group (n = 3).
The trial protocol consisted of a 4-week waitlist period (weeks 0–4, no intervention) followed by a 4-week acupuncture treatment period (weeks 4–8, twice weekly, eight sessions total). The Acupuncture group completed the full protocol with assessments at week 0 (baseline), week 4 (pre-treatment), and week 8 (post-treatment), yielding nine samples. The Control group completed only the waitlist period and was assessed at weeks 0 and 4, yielding 6 samples (15 total samples).
This parallel-group design enabled LMM analysis with three-way interactions (miRNA × Time × Group) to isolate treatment-specific temporal dynamics from natural variation during the waitlist period. The study was approved by the institutional review board of Kyonggi University (KGU-20171222-HR-026) with written informed consent.
Acupuncture intervention
Licensed practitioners administered standardized acupuncture to 2 midline proximal points (GV14, GV16) and 10 bilateral points (BL10, GB20, GB12, GB21, TE3, TE17, ST10, SI3, SI14, SI15). Disposable sterile stainless steel needles (0.25 × 30 mm, Dongbang Acupuncture Inc., South Korea) were inserted 10–20 mm depth using guide tubes. All bilateral points were needled on both sides. After insertion, needles were manually rotated 2–3 times bidirectionally to elicit de-qi sensation, then retained for 15 min without further manipulation. Each session was administered twice weekly for 4 weeks.
Sample collection and analysis
Whole blood was collected in EDTA-anticoagulated tubes (BD Vacutainer, BD Biosciences, Franklin Lakes, NJ, USA). Plasma was isolated by centrifugation at 3000 RPM for 15 min at 4°C and stored at −80°C until analysis. SP was quantified using a commercial ELISA kit (KGE007, R&D Systems, Minneapolis, MN, USA) according to the manufacturer’s protocol. Total RNA was extracted using the mirVana miRNA Isolation Kit (Ambion, Austin, TX, USA). miRNA profiling was performed using the Affymetrix GeneChip™ miRNA 4.0 Array (Affymetrix, Santa Clara, CA, USA), which assays 4603 human miRNAs. Expression values were RMA-normalized and log2-transformed using Affymetrix Expression Console software (Affymetrix, Santa Clara, CA, USA).
Statistical analysis
All analyses were performed using R version 4.5.1 (R Foundation for Statistical Computing, Vienna, Austria). We fitted separate LMMs for each miRNA using lme4 package (version 1.1-37): ‘SP ~ miRNA_expression * Time * Group + (Time | Subject_ID)‘
This approach leveraged full longitudinal data while accounting for repeated measures correlation. The three-way interaction term (miRNA × Time × Group) specifically identified treatment-dependent relationships. p-values were adjusted using Benjamini-Hochberg False Discovery Rate (FDR) correction.
Permutation testing
To validate overall significance despite multiple testing, we performed 2000 permutations, randomly shuffling temporal SP measurements within subjects while preserving individual distributions. This generated a null distribution against which observed results were compared.
Functional analysis
Target genes for significant miRNAs were predicted using multiMiR. KEGG pathway and Gene Ontology enrichment analyses were performed using clusterProfiler with adjusted p < 0.05 significance threshold.
Sample size justification
As an exploratory, hypothesis-generating study, sample size was determined by high-quality sample availability from the parent trial. The LMM approach maximized statistical efficiency from limited samples, while permutation testing provided rigorous validation.
Results
Participant characteristics and plasma Substance P
Six participants were included in this miRNA profiling study. Baseline characteristics and individual plasma SP levels at all timepoints are presented in Supplemental Table S1. Consistent with our previous report, 1 plasma SP decreased during the acupuncture treatment period (weeks 4–8) while remaining stable in the Control group during the waitlist period (weeks 0–4), providing the phenotypic foundation for identifying associated miRNA changes.
Identification of SP-associated miRNAs and treatment-specific miRNA dynamics
LMM screening of 4603 miRNAs identified 53 with significant associations to plasma SP dynamics (FDR < 0.05). Permutation testing strongly validated this discovery (p < 0.001), confirming these associations exceeded chance expectations from multiple testing.
Fourteen miRNAs exhibited significant three-way interactions (miRNA × Time × Group), indicating treatment-specific temporal relationships with SP (Table 1). The most significant was miR-1302-6 (p = 7.65 × 10−6, FDR = 0.0195), followed by miR-181b-2 (p = 1.27 × 10−5, FDR = 0.0195).
Candidate miRNAs with significant three-way interaction effects.
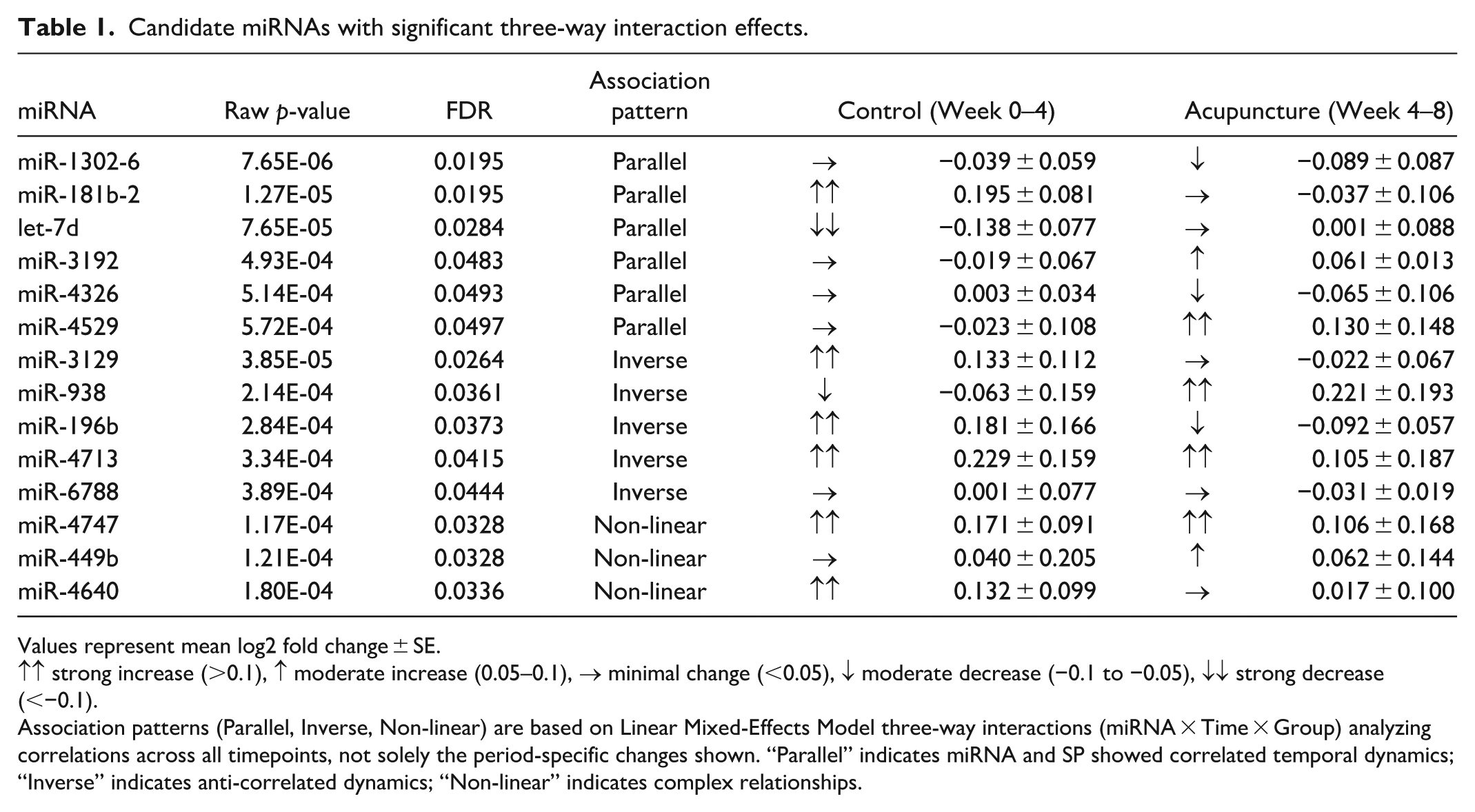
Values represent mean log2 fold change ± SE.
↑↑ strong increase (>0.1), ↑ moderate increase (0.05–0.1), → minimal change (<0.05), ↓ moderate decrease (−0.1 to −0.05), ↓↓ strong decrease (<−0.1).
Association patterns (Parallel, Inverse, Non-linear) are based on Linear Mixed-Effects Model three-way interactions (miRNA × Time × Group) analyzing correlations across all timepoints, not solely the period-specific changes shown. “Parallel” indicates miRNA and SP showed correlated temporal dynamics; “Inverse” indicates anti-correlated dynamics; “Non-linear” indicates complex relationships.
The 14 candidate miRNAs showed 3 distinct patterns:
- Inverse pattern: miR-196b, miR-6788, miR-3129, miR-4713, and miR-938 increased during treatment while SP decreased
- Parallel pattern: let-7d, miR-1302-6, miR-181b-2, miR-4326, miR-4529, and miR-3192 decreased alongside SP
- Non-linear pattern: miR-4747, miR-449b, and miR-4640 showed complex temporal dynamics that did not follow consistent parallel or inverse patterns
Neuroplasticity pathways dominate functional enrichment
Bioinformatic analysis revealed striking convergence on neuroplasticity-related pathways (Figure 1). KEGG pathway analysis showed highest enrichment for Axon guidance (adj. p = 2.61 × 10−6), Hippo signaling (adj. p = 1.37 × 10−6), and MAPK signaling (adj. p = 4.18 × 10−5).
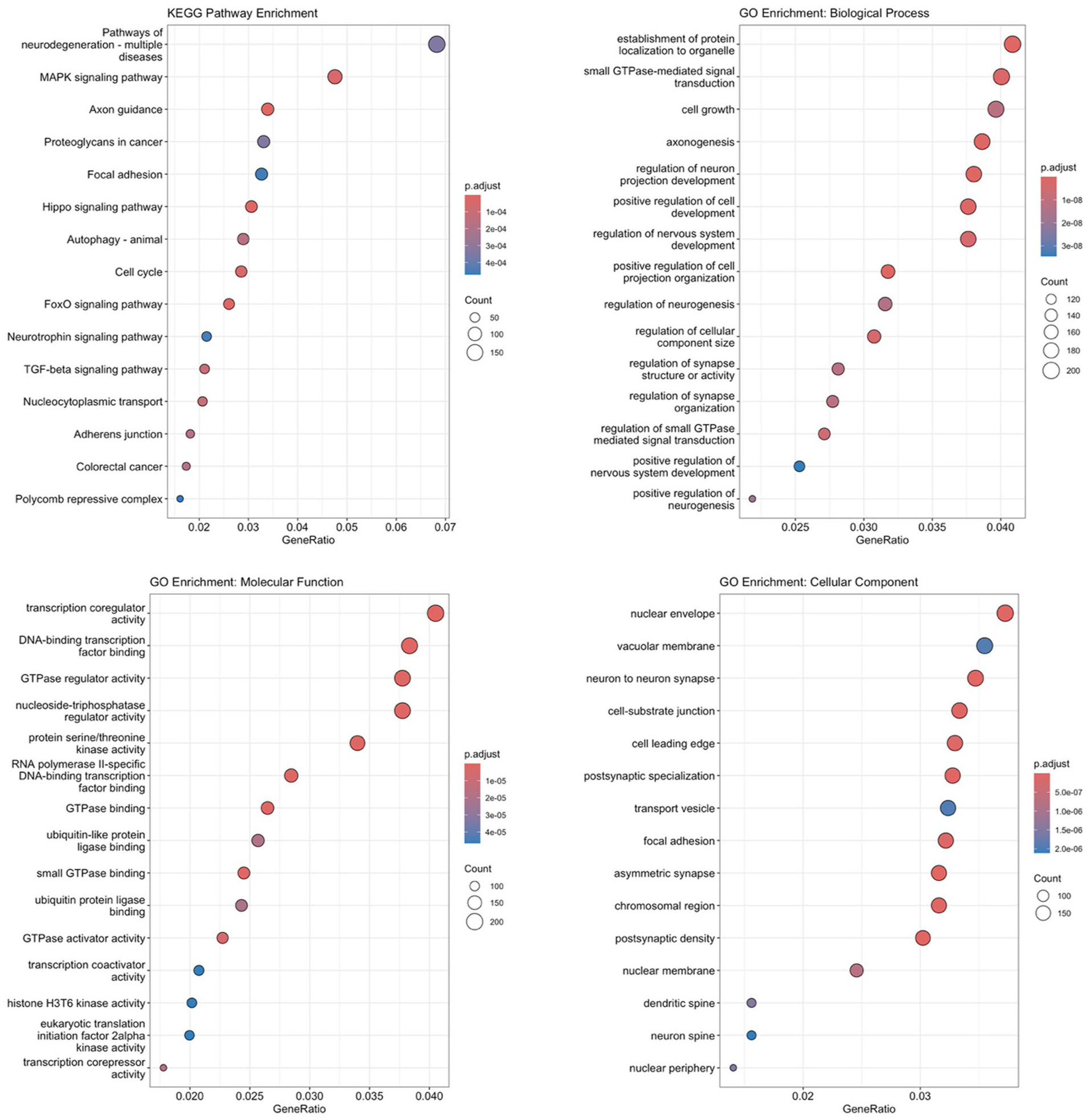
Functional enrichment analysis reveals neuroplasticity as the dominant mechanism. Target genes of the 14 candidate miRNAs were analyzed for (a) Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment and (b–d) Gene Ontology (GO) enrichment across Biological Process (BP), Molecular Function (MF), and Cellular Component (CC) domains.
Gene Ontology analysis reinforced this neuroplasticity theme. Biological Process terms were dominated by “regulation of neuron projection development” (adj. p = 7.52 × 10−10) and “axonogenesis” (adj. p = 8.26 × 10−10). Cellular Component analysis precisely localized activity to “neuron to neuron synapse” (adj. p = 9.52 × 10−12) and “postsynaptic specialization” (adj. p = 7.15 × 10−11). Molecular Function analysis revealed enrichment of transcriptional regulatory activities, including transcription coactivator activity (adj. p = 3.2 × 10−4), DNA-binding transcription factor binding (adj. p = 8.7 × 10−4), and GTPase regulator activity (adj. p = 1.2 × 10−3).
Notably, TAC1 (encoding SP) was not among predicted direct targets, suggesting these miRNAs modulate SP through broader upstream regulatory networks rather than direct suppression.
Discussion
This exploratory study provides the first molecular evidence linking circulating miRNAs to acupuncture-induced SP reduction in chronic neck pain. The identification of 14 treatment-specific miRNAs, validated through rigorous statistical approaches, suggests a novel acupuncture-miRNA-SP regulatory axis.
Our most striking finding is the overwhelming enrichment of neuroplasticity pathways among miRNA targets. Rather than primarily targeting inflammatory mediators, these miRNAs appear to orchestrate structural and functional remodeling of neural circuits. This suggests acupuncture’s lasting analgesic effects may stem from inducing adaptive neuroplastic changes rather than transiently suppressing nociceptive signals. The enrichment of axon guidance and synaptic structure components particularly supports this neuroplasticity hypothesis. Previous studies have shown peripheral and central sensitization in chronic pain involves maladaptive synaptic plasticity. 6 Our findings suggest acupuncture might reverse these changes through miRNA-mediated reprogramming of synaptic architecture. The absence of TAC1 as a direct target indicates these miRNAs likely modulate SP through complex upstream networks. The MAPK pathway enrichment provides a plausible mechanistic link, as MAPK signaling integrates diverse stimuli to regulate both neuroplasticity and neuropeptide expression. 7
Our LMM approach represents a methodological advance in acupuncture research, extracting maximum information from limited samples while properly modeling individual heterogeneity. The three-way interaction term specifically isolated treatment-dependent effects, avoiding confounding from non-specific temporal changes. The permutation testing critically validated our multiple testing approach. This empirical validation, rarely employed in biomarker discovery studies, substantially strengthens confidence in our findings despite the small sample size.
These findings suggest potential for miRNA-based biomarkers to predict or monitor acupuncture response. The diverse temporal patterns observed – early responders (miR-196b, miR-6788) versus late responders (miR-1302-6, let-7d) – may reflect different regulatory roles in a mechanistic cascade: early responders potentially initiating therapeutic cascades while late responders maintain analgesic states through sustained neuroplastic remodeling.
Importantly, while acupuncture produces rapid analgesic effects through mechanisms such as endogenous opioid release and segmental inhibition, our neuroplasticity findings suggest miRNA-mediated pathways contribute to the sustained pain relief observed at our 4-week post-treatment follow-up. This temporal framework implies acupuncture might be particularly effective for chronic pain conditions involving maladaptive plasticity, guiding patient selection, and treatment optimization. Future studies should examine both immediate and delayed molecular responses to clarify this relationship.
Several limitations require acknowledgment. The small sample size (n = 3/group) limits statistical power and generalizability. Results should be considered preliminary and hypothesis-generating, requiring validation in larger cohorts. The correlational design cannot establish causality. Whether miRNA changes drive SP reduction or both reflect upstream processes remains undetermined. Functional studies using miRNA mimics/inhibitors are needed to establish causal relationships. Our cohort included only middle-aged women, limiting generalizability. The waitlist control design cannot exclude placebo effects, though the specific molecular changes observed argue against purely psychological mechanisms.
Priority should be given to validation in larger, diverse cohorts with appropriate control interventions. Mechanistic studies should examine whether manipulating identified miRNAs affects SP expression and pain behaviors in preclinical models. Longitudinal studies with more frequent sampling could elucidate temporal cascades and identify optimal biomarker assessment timepoints. Integration with neuroimaging could link molecular changes to neural plasticity measures.
Despite limitations, this study pioneers a novel molecular perspective on acupuncture analgesia. By revealing associations between specific miRNAs, neuroplasticity pathways, and SP reduction, we provide a testable framework for understanding acupuncture’s mechanisms. These findings lay groundwork for developing molecular markers to optimize and personalize acupuncture therapy.
Supplemental Material
sj-docx-1-mpx-10.1177_17448069251401661 – Supplemental material for Circulating microRNAs associated with acupuncture-induced Substance P reduction in chronic neck pain: Evidence for a neuroplasticity mechanism
Supplemental material, sj-docx-1-mpx-10.1177_17448069251401661 for Circulating microRNAs associated with acupuncture-induced Substance P reduction in chronic neck pain: Evidence for a neuroplasticity mechanism by Seung-Nam Kim in Molecular Pain
Footnotes
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea funded by the Korean government (MSIT) (RS-2025-25413539).
Ethical considerations
The study was approved by the institutional review board of Kyonggi University (KGU-20171222-HR-026) with written informed consent.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
