Abstract
EA effectively treats gastrointestinal diseases, pain symptoms, and emotional disorder. Furthermore, vHPC and mPFC are two of the crucial nuclei involved in controlling chronic pain and anxiety-like behaviors. In the present study, it is investigated whether EA may reduce visceral pain and anxiety associated with inflammatory bowel disease (IBD) by inhibition of vHPC-to-mPFC pathway. We found that EA alleviated visceral hyperalgesia and anxiety in TNBS-treated IBD mice. EA decreased the numbers of c-Fos and neurogranin (labeled glutamatergic neurons) co-labeled neurons in both vHPC and mPFC. EA suppressed the activation of vHPC and mPFC pyramidal neurons associated with anxiety-like behaviors and EA suppressed the activation of vHPC neuronal response to von Frey filament. In addition, chemogenetic inhibition of the vHPC-to-mPFC pathway alleviated mechanical allodynia, visceral hyperalgesia and anxiety in IBD mice. However, chemogenetic activation of vHPC-to-mPFC pathway antagonized the effect of EA on anxiety and visceral hyperalgesia, but not on mechanical allodynia in IBD mice. In conclusion, our findings revealed that vHPC-to-mPFC pathway is involved in the inhibitory effect of EA on anxiety and pain sensitivity in IBD mice. EA may exert anti-anxiety effect via inhibition of vHPC-to-mPFC pathway. Thus, our study provides new information about the cellular circuits mechanisms of the therapeutic effect of EA on the comorbidity of visceral pain and anxiety induced by IBD.
Introduction
Inflammatory bowel disease (IBD), including Crohn’s disease (CD) and ulcerative colitis (UC), has been drawing growing attention recently.1–3 In addition to gastrointestinal symptoms, IBD patients usually accompany with multiple emotion disorders such as anxiety and depression,4,5 which in turn often exaggerate gastrointestinal symptoms of IBD patients. 6 Electroacupuncture (EA) consists of placing thin needles into the skin at specific locations (acupoints) with electrical stimulation 7 and has been proven to be effective in treating gastrointestinal diseases, pain symptoms as well as emotional disorders.8–11 Although the therapeutic effect of EA on comorbidity of visceral pain and anxiety in IBD patients has been well recognized clinically, 12 its underlying mechanism remains unknown.
The ventral hippocampus (vHPC) and media prefrontal cortex (mPFC) are two of the crucial nuclei involved in controlling anxiety-like behaviors.13–16 In fact, the vHPC is not only anatomically connected but is also functionally associated with the mPFC.17–20 For example, bilateral inhibition of glutamatergic neurons from the vHPC to mPFC reduces anxiety, 21 whereas chemogenetic activation of mPFC-projecting vHPC glutamatergic neurons increases anxiety. 22 Thus, the vHPC-mPFC pathway is likely the principal component of the anxiety circuit.21,22 However, it remains unknown that the vHPC-to-mPFC pathway participates in the anxiolytic effect of EA on IBD mice.
Neurons in the vHPC that project to the PFC were associated with chronic pain. It has been reported that large decreases in hippocampal connectivity with the mPFC are found in patients with back pain 23 and weakening of information flowing in the fronto-hippocampal circuit results from SNI model of neuropathy. 24 However, it needs be investigated that the vHPC-to-mPFC pathway participates in the analgesia effect of EA on IBD mice.
Therefore, in this study, we investigated whether EA alleviated the comorbidity of visceral pain and anxiety in 2,4,6-trinitrobenzene sulfonic acid (TNBS)-induced IBD mice. We then examined the responses of pyramidal neurons of vHPC and mPFC to EA stimulation using in vivo recording of neuronal calcium activity in IBD mice. Furthermore, we determined whether chemogenetic manipulating of the vHPC-to-mPFC pathway altered the effect of EA on visceral pain and anxiety in IBD mice. Our findings provide new evidence that EA inhibits the comorbidity of visceral pain and anxiety associated with IBD mice by inhibiting pathway from vHPC to mPFC.
Materials and method
Animals
Adult male C57 BL/6 mice (8 week old; 20–25 g) were housed in the environment of 23°C ± 2°C and a 12 h light/dark cycle, and was free access to food and water. All animal procedures followed the Institutional Animal Care and Use Committee at Huazhong University of Science and Technology and conformed to the ethical guidelines of the International Association for the Study of Pain (IASP).
Viruses constructs and surgery
Adeno-associated viruses (AAV/2-9) were designed to achieve CRE-DIO system-mediated chemogenetic manipulation strategy 25 : Retro-AAV-Camkii-CRE-WPRE-pA combined with rAAV-hysn -DIO-hM3D(Gq)-mCherry-WPRE-pA were designed to excite glutamatergic neurons inputting from vHPC to mPFC. Retro-AAV-Camkii-CRE-WPRE-pA combined with rAAV-hSyn-DIO-hM4D(Gi)-mCherry-WPRE-pA were designed to inhibit glutamatergic neurons inputting from vHPC to mPFC. AAV2/1-Camkii-GCaMP6s 26 was designed to label glutaminergic neurons expressed GFP in the vHPC and mPFC. All viruses used in this research were purchased from the Wuhan BrainVTA scientific and technical corporation.
Before normal operation, tribromoethanol (100 mg/kg, i.p.) was applied for anesthesia and stereotaxic apparatus (RWD Instruments, China) fixed mice brain. An incision with a depth of 1.5 cm along the midline of the skull was made and the periosteum was removed on the surface of the skull. Virus were injected based on the coordinates of vHPC (2.95 mm from the bregma, 2.75 mm lateral from the midline, and 3.75 mm ventral to the skull) 27 and mPFC (2.95 mm from the bregma, 0.25 mm lateral from the midline, and 1.5 mm ventral to the skull). 28 Desired virus vectors (200 nL) were injected into the vHPC and mPFC at a rate of 50 nl per 60 s. 29 It was excluded in the statistics when the virus infection area exceeds the vHPC and mPFC area.
Chemogenetic manipulation
Three weeks after virus injections, mice were intraperitoneally injected with clozapine N-oxide (CNO, Sigma) 60 min before the behavioral assessment. All baselines for visceral and mechanical sensitivity plus anxiety-like behavior test were recorded after the virus injections and before the CNO administration. 1 mg/kg CNO was administered for both hM3Dq activation and hM4Di inhibition. 29 In the chemogenetic reversing experiment of EA treatment, CNO was applied just before EA treatment 30 min.
IBD model
According to related Dunford et al., 30 this model got prepared. Tribromoethanol (100 mg/kg, i.p.) was used for anesthesia. A PVC-Fr4 catheter (Φ 2.7 mm, YN Medical Instrument, Yangzhou, China) was lubricated by corn oil, which was inserted into the anus to the colon at a depth of approximately 4 cm. The other end of PVC-Fr4 catheter was connected with a 1 ml syringe for drug injection. The TNBS intra-rectal (IR) solution comprised 50 μl of 5% w/v TNBS (Sigma-Aldrich, St. Louis, MO, USA solution with 50 μl absolute ethanol, which was poured into the colon of anesthetized mice. Mice of the vehicle group got an injection solution, comprised with 50 μl distilled water and 50 μl absolute ethanol. After drug injection, keeping upside down for at least 5 min for prohibition from IR solution leakage.
EA treatment
Specialized fabric equipment restrained mice. The bilateral Dachangshu points (BL25) were stabbed obliquely downward with a depth of 2.5 mm into the skin. The tail of the acupuncture needle was connected to an electro-acupuncture (EA) instrument (Huatuo brand, Suzhou) and the EA parameters were 1 mA current, 15 Hz frequency, intermittent wave, and 30 min treatment time. In the sham EA group, the acupuncture needles adhered to points but did not penetrate the skin. Half an hour after EA treatment, the related behavior tests were performed.
Behavioral tests
Measurement of mechanical allodynia
Mice were first habituated to the test environment for 30 min. Mice were placed in a transparent plexiglass box on an elevated mesh floor with a metal mesh pad (5 mm × 5 mm mesh area) at the bottom. The mechanical thresholds were tested for 3 days before TNBS injection and the average value of the 3-day tests was calculated as the baseline threshold. After TNBS injection, the mechanical thresholds were tested once a day for five consecutive days. Then a series of calibrated Von Frey filaments (Wood Dale, USA) were applied erectly to the midplantar surface of the left hind paw to bend the filament for 6 s. The mechanical threshold of mice was measured by using the “up and down” method 31 and the test of withdrawal threshold was repeated two times in each mouse, and the mean value was calculated. Paw withdrawal or licking feet was considered as a pain-like response.
Measurement of visceral hyperalgesia
The colorectal distension (CRD) method was applied to observe the abdominal withdrawal reflex score (AWRs) of mice to evaluate the degree of visceral hyperalgesia. 32 Biefly, mice were lightly anesthetized with Halothane, and a lubricated flexible latex balloon (3 cm) was inserted intra-anally (after the distal part of the rectum was gently cleared by massage) into the descending colon at 0.5 cm proximal to the anus and secured by taping the catheter of the balloon to the base of the tail. After recovery from anesthesia, animals equipped with the balloon were placed in a Plexiglas cylinder for 30 min.
The CRD test was performed in a step-by-step compression mode (20/40/60/80 mmHg). Each pressure value was measured twice, each test lasting 30 s, with an interval of 4 min, AWRs was recorded and averaged. The scoring criteria refer to Al-Chaer’s method 33 : no behavioral response to CRD was rated as 0 point, short pauses in head or body movements during stimulation was rated as 1 point; abdominal muscle contraction during stimulation was rated as 2 point; abdominal lifting was rated as 3 point; body arch, pelvic cavity or scrotum lifting was rated as 4 point.
Open field test (OFT)
OFT was used to evaluate anxiety emotion and motor activity of mice. Before normal experiment, mice usually adapt for experiment room at least 30 min. The apparatus comprised a polystyrene enclosure (50 * 50 * 50 cm) and video recorder. 34 Mice were placed in the enclosure and had no limit to move freely for 5 min. The centric 25 * 25 cm area was defined as center area. The apparatus need be cleaned with 75% ethanol between each test and LabState (Xinruan Information Technology Co., LTD, Shanghai, China) recorded each experiment track. Total distance moved (cm) and time spent in the center area were analyzed.
Elevated plus-maze (EPM)
EPM was used to evaluate anxiety emotion and motor activity of mice. Before normal experiment, mice usually adapt for experiment room at least 30 min. The apparatus comprised 100 cm open arms and 100 cm close arms. 34 Mice were placed in the enclosure and had no limit to move freely for 5 min. The apparatus need be cleaned with 75% ethanol between each test and LabState (Xinruan Information Technology Co., LTD, Shanghai, China) recorded each experiment track. Total distance moved (cm) and time spent in the open arms were analyzed.
Immunofluorescence
Mice under deep anesthesia were transcardially perfused with 37°C normal saline and then followed by 4% paraformaldehyde in 0.1 M PBS (pH, 7.4; 4°C). The brain were abstracted immediately and post-fixed in the paraformaldehyde. Overnight the tissues were cryoprotected in 30% sucrose in 0.1 M PBS for 24 h at 4°C. The OCT embedded blocks were sectioned for 30-μm thickness.
The sections were washed in 0.01 M PBS and blocked for 2 h with blocking liquid (5% donkey serum and 0.2% tween-20 in 0.01 M PBS) at room temperature. The sections were incubated with the following antibodies: rabbit anti–neurogranin (1:1000, abcam, UK), guinea pig anti-c-fos (1:1000, abcam, UK). Subsequently, the free-floating sections were washed with 0.01 M PBS for 3 times, and incubated with following secondary antibodies (Jackson ImmunoResearch) for 2 h: donkey anti-rabbit IgG conjugated with Dylight 594 (1:500, abcam, UK), donkey anti-guinea pig IgG conjugated with Dylight 488 (1:500, abcam, UK). The sections were cover-slipped with mounting fluid containing DAPI for the nucleus staining. The samples were observed under a fluorescence microscope (Olympus) for the immunofluorescence staining. Images were analyzed by using NIH Image J software (Bethesda, MD, USA). The layouts of images were based on the Photoshop (ADOBE company, USA).
Calcium signal optical fiber recorder
One week before the behavioral tests, mice were prepared for optical fiber or cannula implantation. For vHPC and mPFC local fiber photometry recordings, mice were unilaterally implanted with optical fibers (200 mm O.D., 0.37 NA, Inper, Hangzhou, China) 2 weeks after injecting GCaMP6s (see sup Figure 1). The fiber photometry system (ThinkerTech Nanjing Bioscience Inc.) has been described previously. Briefly, a 488-nm laser beam (OBIS 488LS, Coherent) was reflected off a dichroic mirror (MD498, Thorlabs), focused with a 103 objective lens (0.3 NA, Olympus Inc., Japan), and then coupled to an optical commutator (Doris Lenses) to record calcium signals. The commutator and implanted fiber were connected by a 2-m optical fiber (200 mm O.D., 0.37 NA). The laser power at the tip of the optical fiber was adjusted to 0.01 mW to decrease laser bleaching. The GCaMP6s fluorescence was bandpass filtered (MF525-39, Thorlabs). An amplifier was used to convert the photomultiplier tube current output to a voltage signal, which was further digitized at 100 Hz and recorded by a Power 1401 digitizer and Spike2 software (CED, Cambridge, UK). The heatmap and averaged Ca2+ traces were plotted by a GraphPad Prism ver8.0 (GraphPad Inc, USA).

EA relieved the pain sensitivity and anxiety-like behaviors of IBD mice. (a) Experimental flowchart. (b) Schematic diagram of Dachangshu points (BL25) on the skin surface. (c) Time course of mechanical threshold after EA treatment. (d) Visceral score after EA treatment.(e) Anxiety-related behaviors in mice for anxiety was recorded as time in center zone in the OPF. (f) Anxiety-related behaviors in mice for anxiety-like behaviors was recorded as time in open arms in the EPM. The data are expressed as mean ± SEM (n = 9 mice).
Laser power: The laser power measured at the fiber tip was maintained at 0.02–0.03 mW to ensure signal quality while minimizing photobleaching effects.
Recording duration: The total duration for each fiber photometry recording session was 10 min.
Correction method: A double-exponential function fitting was applied to the raw fluorescence signal (F) to model and subtract the baseline drift caused by photobleaching. The corrected signal (F_corrected) was used for calculating ΔF/F.
Analysis parameters: We employed an automatic detection algorithm based on standard deviation. The specific parameters were: a detection threshold set to more than 3 standard deviations (SD) above the baseline, a 1-s smoothing window, and a 100 Hz sampling rate.
Reported data: In addition to the integrated calcium signal, we have now quantitatively analyzed and report the statistical data for event frequency (events/min) and mean peak amplitude (ΔF/F).
Synchronized video recording: All fiber photometry recordings were synchronized with video tracking to capture the mice’s locomotion trajectories and velocities.
Data exclusion criteria: We established that calcium signal data at any time point where the mouse’s instantaneous velocity exceeded 5 cm/s were considered potentially contaminated by significant motion artifacts. Consequently, these data points were excluded from the analysis of event frequency and peak amplitude and did not contribute to the statistical analysis.
Signal correlation analysis: We calculated the correlation between locomotion velocity and the ΔF/F signal, which confirmed the absence of systematic motion artifact influence on the signal during periods of rest and low-speed movement.
Statistical analysis
The experiments analysis for behavioral tests were blinded to the treatment. All data were presented as mean ± standard errors of means (s.e.m.), unless otherwise specified. Each data set was firstly tested for normal distribution, and those fitted Gaussian distribution were used for parametric analysis. Student t-test (paired or unpaired) was used for comparison between two groups, and one-way analysis of variance was used to analyze the difference among >2 groups, followed by Tukey post hoc comparison. When two independent variables were considered, two-way ANOVA was used. For those data that did not fit the Gaussian distribution, Wilcoxon matched-pairs rank test was used for paired comparison, and Kolmogorov–Smirnov test was employed to compare between two independent samples. A statistical significance was defined as p < 0.05. All statistical analysis and data plotting were performed by GraphPad Prism ver8.0 (GraphPad Inc, USA).
Results
EA alleviated mechanical allodynia, visceral hyperalgesia, and anxiety in TNBS-treated mice
Five days after TNBS injection, from days 3 to 5, TNBS-treated mice also exhibited mechanical allodynia, a referred somatic pain, as evidenced by reduced mechanical threshold of left hindpaw (Figure 1(c), p < 0.05) and visceral hyperalgesia (Figure 1(d), p < 0.05), which was also significantly attenuated by EA treatment. These results support the analgesic effect of EA on mechanical allodynia and visceral hyperalgesia in TNBS-treated mice.
Furthermore, behavioral results showed that, the time in the center zone of OPF and the open arms of EPM of TNBS-treated mice is significantly shortened compared with the vehicle-treated group. EA significantly increased the time spent in the center zone (Figure 1(e) and (f), p < 0.05). It suggested that EA decreased anxiety behaviors induced by TNBS, but sham EA did not relieve pain and anxiety induced by TNBS.
EA inhibited hyperactivity of pyramidal neurons in the vHPC and mPFC in TNBS-treated mice
To investigate the neural pathway mediating the analgesic and anxiolytic effects of EA, we examined the responses of pyramidal neurons in the vHPC and mPFC. The neural activities of vHPC and mPFC are differentially involved in neuropathic pain and anxiety processes.21,22,35,36
Using c-Fos as a biomarker of neuronal activation, our immunohistochemical results showed that the numbers of c-Fos and neurogranin (labeled glutamatergic neurons 37 co-labeled neurons in both vHPC and mPFC were increased in mice treated with TNBS, suggestive of the hyperactivity of these pyramidal neurons in IBD mice (Figures 2(a)–(h), 3(a)–(h), p < 0.05). EA, but not sham EA, reduced the proportion of c-Fos+ pyramidal neurons in the vHPC and mPFC (Figures 2(a)–(h), 3(a)–(h), p < 0.05).

EA inhibited the expression of c-Fos of pyramidal neurons in the vHPC of IBD mice. (a) Immunofluorescence images of c-Fos expressed neurons in the HPC. (b) Immunofluorescence images of Camkii expressed neurons (labeled glutamatergic neuron) in the HPC. (c) Immunofluorescence images of c-Fos expressed neurons co-labeled with Camkii expressed neurons in the HPC. Scale bar for merge images, 50 mm. (d) the number of c-Fos expressed neurons in the mPFC. (e) The number of Camkii expressed neurons in the mPFC. (f) Percentage of c-Fos-expressed neurons co-labeled with Camkii, which is (c-Fos and Camkii co-labeled neurons /total neurogranin neurons) * 100%. (g) magnified cell image from the white outlined box in panel c. (h) Co-localization of c-FOS and Camkii in single cells, quantified by Pearson’s coefficient.

EA inhibited the expression of c-Fos of pyramidal neurons in the mPFC of IBD mice. (a) Immunofluorescence images of c-Fos expressed neurons in the mPFC. (b) Immunofluorescence images of Camkii expressed neurons (labeled glutamatergic neuron) in the mPFC. (c) Immunofluorescence images of c-Fos expressed neurons co-labeled with Camkii expressed neurons in the mPFC. Scale bar for merge images, 50 mm. (d) the number of c-Fos expressed neurons in the mPFC. (e) the number of Camkii expressed neurons in the mPFC. (f) percentage of c-Fos-expressed neurons co-labeled with Camkii, which is (c-Fos and Camkii co-labeled neurons /total neurogranin neurons) * 100%. (g) magnified cell image from the white outlined box in panel c. (h) Co-localization of c-FOS and Camkii in single cells, quantified by Pearson’s coefficient.
Furthermore, to determine the functional significance of pyramidal neurons in the vHPC and mPFC in pain and anxiety-like behaviors, we injected an adeno-associated virus (AAV) vector expressing a Ca2+-sensor protein (GCaMP6s) into the vHPC and mPFC and measured GCaMP6s fluorescence activity of pyramidal neurons. We found that, compared with the vehicle-treated mice, the GCaMP6s fluorescence of pyramidal neurons in the vHPC in TNBS-treated mice was significantly increased when von-Frey filaments were applied on the hindpaw or when mice stayed in the center zone or open arms (Figure 4(a)–(g), p < 0.05). These findings suggest that the pyramidal neurons in the vHPC likely mediate the mechanical allodynia and anxiety-like behaviors in TNBS-treated mice. Importantly, EA treatment reduced the elevated intracellular Ca2+ signaling during each behavior test, suggesting that EA inhibits the hyperactivity of pyramidal neurons in the vHPC (Figure 4(a)–(g), p < 0.05).

EA inhibited the hyperactivity of pyramidal neurons in the vHPC of IBD mice. (a) Heatmap of vHPC CaMKII+ neurons in mice receiving von Frey punctate. (b) Heatmap of vHPC CaMKII+ neurons in mice receiving OPF test. (c) Heatmap of vHPC CaMKII+ neurons in mice receiving EPM test. (d) Average Ca2+ transients of vHPC CaMKII+ neurons in mice receiving von Frey punctate. (e) Average Ca2+ transients of vHPC CaMKII+ neurons in mice receiving OPF test. (f) Average Ca2+ transients of vHPC CaMKII+ neurons in mice receiving EPM test. (g) Total integrated calcium of vHPC CaMKII+ neurons in mice receiving von Frey punctate, OPF test and EPM test. The data are expressed as mean ± SEM (n = 9 mice).
Similarly, the GCaMP6s fluorescence signal of pyramidal neurons in the mPFC in TNBS-treated mice was increased during mechanical stimulation of the hindpaw or when anxiety-like behaviors occurred (Figure 5(a)–(g), p < 0.05). These data support the involvement of mPFC neurons in mediating these behavioral reflexes in TNBS-treated mice. EA suppressed the activation of mPFC pyramidal neurons associated with anxiety-like behaviors but did not alter the neuronal response to von Frey filament stimulation (Figure 5(a)–(g)).
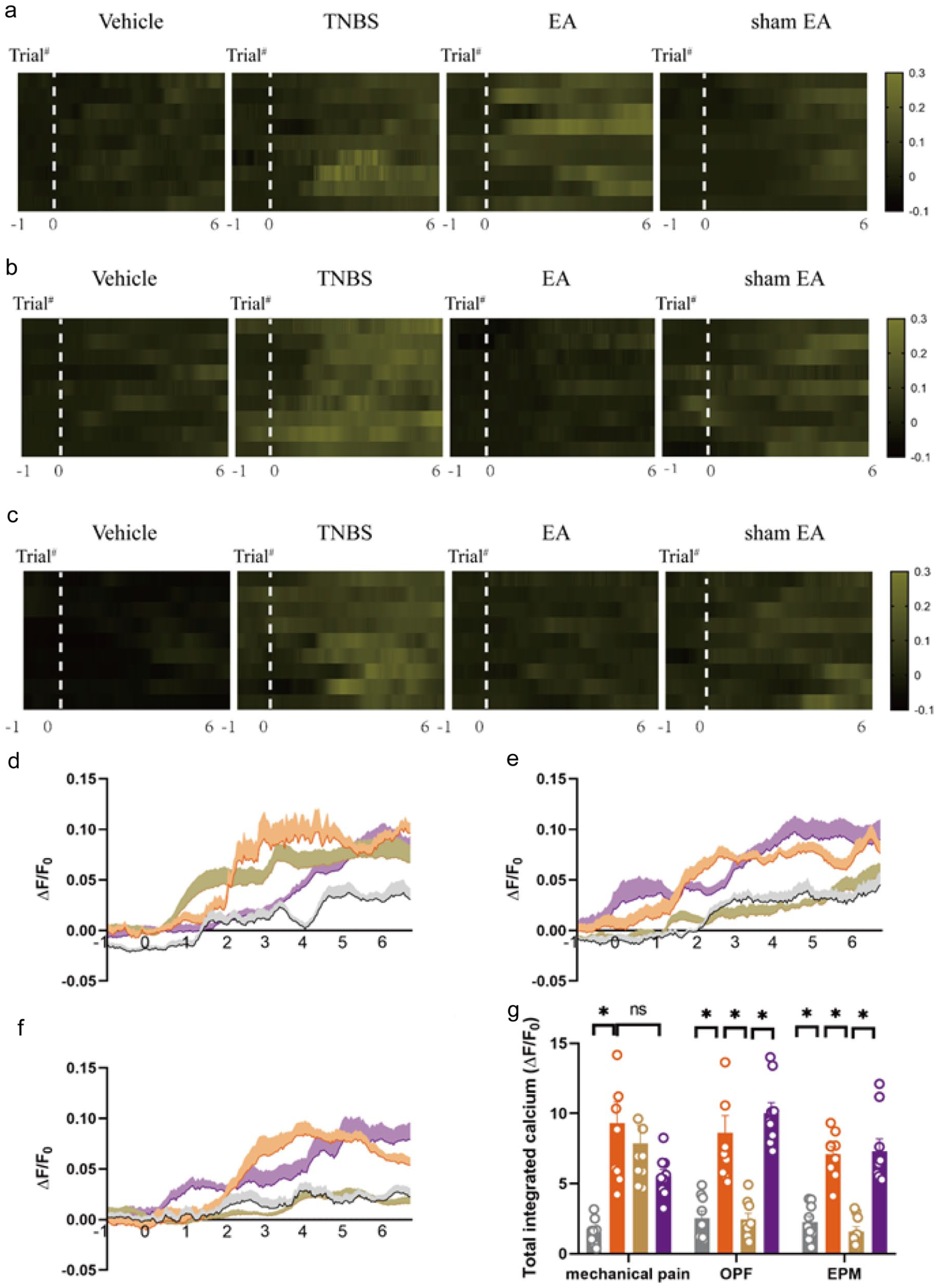
EA inhibited the hyperactivity of pyramidal neurons in the mPFC of IBD mice. (a) Heatmap of mPFC CaMKII+ neurons in mice receiving von Frey punctate. (b) Heatmap of mPFC CaMKII+ neurons in mice receiving OPF test. (c) Heatmap of mPFC CaMKII+ neurons in mice receiving EPM test. (d) Average Ca2+ transients of mPFC CaMKII+ neurons in mice receiving von Frey punctate. (e) Average Ca2+ transients of mPFC CaMKII+ neurons in mice receiving OPF test. (f) Average Ca2+ transients of mPFC CaMKII+ neurons in mice receiving EPM test. (g) total integrated calcium of mPFC CaMKII+ neurons in mice receiving von Frey punctate, OPF test and EPM test. The data are expressed as mean ± SEM (n = 9 mice).
Chemogenetic inhibition of the vHPC-to-mPFC pathway alleviated mechanical allodynia, visceral hyperalgesia and anxiety in TNBS-treated mice
To determine whether the vHPC-to-mPFC pathway participates in anxiety, mechanical allodynia and visceral hyperalgesia in TNBS-treated mice, mPFC-projecting pyramidal neurons in the vHPC were inhibited using a chemogenetic approach. A retrograde adeno-associated virus (AAV) carrying cre under the control of the CamKIIα promoter was injected bilaterally into the mPFC of wild-type mice and an adeno-associated virus carrying hM4Di, an inhibitory designer receptor exclusively activated by designer drugs (DREADD), bilaterally into the vHPC (Figure 6(b)). Four weeks were allowed for viral expression to achieve maximal expression levels in pyramidal neurons from the vHPC to mPFC (Figure 6(b)).

Chemicogenetic inhibition of vHPC-to-mPFC pathway alleviated anxiety, mechanical pain and visceral pain in IBD mice. (a) Experimental flowchart. (b) Schematic diagram of virus injection site. (c) Time course of mechanical threshold. (d) Visceral hyperalgesia was evaluated by CRD. (e) Anxiety-related behaviors in mice for anxiety was recorded as time in centre zone in the OPF. (f) Anxiety-related behaviors in mice for anxiety was recorded as time in open arms in the EPM. The data are expressed as mean ± SEM (n = 8 mice).
We found that the mechanical pain threshold was remarkably increased in the Gi + TNBS + CNO group compared with that in the control virus + TNBS + CNO group and Gi + TNBS + saline group beginning from the 3rd day after TNBS injection (Figure 6(c), p < 0.05). Additionally, The AWRs score of CRD in the Gi + TNBS + CNO group was lower than that in the control virus + TNBS + CNO group and Gi + TNBS + saline group (Figure 6(d), p < 0.05).
In the OPF and EPM test, compared with the control virus + TNBS + CNO group and Gi + TNBS + saline group, the time staying in center zone and open arms of the Gi + TNBS + CNO group was increased (Figure 6(e) and (f), p < 0.05). Thus, our results suggest that the vHPC-to-mPFC pathway is important for the comorbidity of visceral pain and anxiety in IBD mice.
Chemogenetic activation of vHPC-to-mPFC pathway antagonized the effect of EA on anxiety and visceral hyperalgesia, but not on mechanical allodynia, in TNBS-treated mice
The vHPC connects multiple populations of inhibitory neurons in deep layers of the infralimbic area of mPFC, 38 and neurons in the vHPC that project to the PFC are associated with chronic pain and emotion disorder.21,22,39 We therefore investigated whether the vHPC-to-mPFC pathway participates in the effect of EA on mechanical allodynia, visceral hyperalgesia and anxiety in TNBS-treated mice.
After expressing AAV carrying hM3Dq, the mPFC-projecting pyramidal neurons in vHPC were activated by CNO (Figure 7(b)). Chemogenetic activation of mPFC-projecting pyramidal neurons in the vHPC partially reversed the effect of EA on visceral hypersensitivity in IBD mice, as the AWR score in the Gq + CNO + EA group was higher than in the control virus + CNO + EA group and Gq + saline + EA group (Figure 5(d), p < 0.05). However, there was no difference in the mechanical allodynia among three groups (Figure 5(c)), suggesting that chemogenetic activation of the vHPC-to-mPFC pathway did not antagonize the effect of EA on mechanical allodynia in TNBS-treated mice. One explanation is that the effect of EA on mechanical allodynia is likely mediated via other pain-related pathways, such as the mPFC-PAG 40 and mPFC-amygdala circuits. 41

Chemicogenetic activation of vHPC-to-mPFC pathway antagonized EA effect on anxiety and visceral pain, but not mechanical pain, in IBD mice. (a) Experimental flowchart. (b) Schematic diagram of virus injection site. (c) Time course of mechanical threshold. (d) Visceral hyperalgesia was evaluated by CRD. (e) Anxiety-related behaviors in mice for anxiety was recorded as time in center zone in the OPF. (f) Anxiety-related behaviors in mice for anxiety was recorded as time in open arms in the EPM. The data are expressed as mean ± SEM (n = 8 mice).
In the OPF and EPM test, compared with the control virus + CNO + EA group and Gq + saline + EA group, the time of mice stayed in the center zone and open arms in the Gq + CNO + EA group was decreased (Figure 7(e) and (f), p < 0.05). Thus, stimulation of the vHPC-to-mPFC pathway antagonized the effect of EA on anxiety and visceral pain, but not on mechanical pain, in IBD mice.
Discussion
Patients with IBD normally presented chronic visceral disorders and mood disorders, such as anxiety or depression.5,42,43 Patients with IBD still lacked an effective drug treatment on somatic symptoms and emotion disorders, while EA offered a complementary option. Several reports showed the effect of EA relieved mechanical allodynia and visceral hyperalgesia for analgesia.44–48 In addition, EA had an anxiolytic role in anxiety-like behaviors. 49 This study showed that EA significantly alleviated mechanical pain and visceral pain, as well as anxiety in IBD mice.
In basic research, mPFC processed ascending pain pathway 40 and losses in PFC grey matter were often reversible after successful treatment of chronic pain.50,51 Moreover, mPFC implemented top-down control of anxiety state and low-frequency optogenetic stimulation of mPFC afferents in BLA attenuated anxiety-like behavior. 52 In this study, EA decreased c-FOS expression in neurogranin-positive excitatory neurons induced by TNBS, it suggested that EA suppressed the hyperactivity of mPFC pyramidal neurons. Furthermore, EA inhibited neuronal Ca2+ hyperactivities of mPFC pyramidal neurons induced by anxiety-like behaviors, but not mechanical allodynia, in the IBD mice. Hence, EA may inhibit anxiety-like behavior through inhibition of mPFC pyramidal neurons. However, the analgesic effect of EA may be related to other brain areas, such as vHPC and periaqueductal gray (PAG). 40
The hippocampus, is not only a key region for development of memory and learning, but also related with chronic pain and anxiety.22,53 Several experimental studies have found that direct manipulation of the hippocampus altered nociceptive behavior without being aversive, supporting the hippocampal contribution to pain awareness. 35 Interestingly, the hippocampus is a therapeutic target for anxiety.22,54 In this study, EA reduced c-FOS expression in neurogranin-positive excitatory neurons of vHPC, it suggested that EA suppressed hyperactivity of vHPC pyramidal neurons. Moreover, EA inhibited neuronal Ca2+ hyperactivities of vHPC pyramidal neurons induced by mechanical allodynia and anxiety in IBD mice. In a word, the activity of pyramidal neurons in the vHPC is necessary and sufficient for the coding and processing of pain and anxiety in IBD mice.
Neurons in the vHPC that project to the mPFC were associated with chronic pain.21,40 In chronic pain clinical practice, large decreases in hippocampal connectivity with the mPFC are found 23 and SNI model of neuropathy reduces the overall amount of information flowing in the fronto-hippocampal circuit. 24 Moreover, the pathway from vHPC to mPFC are thought to serve as principal components of the anxiety. Optogenetic and pharmacological studies have demonstrated that manipulation of the vHPC connections to certain brain regions can regulate anxiety behaviors. In the present study, chemogenetic inhibition of the vHPC-to-mPFC pathway alleviated mechanical allodynia, visceral hyperalgesia and anxiety in IBD mice. In contrast, chemogenetic activation of vHPC-to-mPFC pathway antagonized the effect of EA on anxiety and visceral hyperalgesia, but not mechanical allodynia, in IBD mice. It indicated that the vHPC-to-mPFC pathway may participate in visceral hyperalgesia, mechanical allodynia and accompanying anxiety in IBD mice, and EA may alleviate anxiety and visceral hyperalgesia through the vHPC-to-mPFC pathway. As for the effect of EA on mechanical allodynia, it may correlate with inputs arising from vHPC to other areas, including amygdala. 55
In conclusion, our findings revealed that the vHPC-mPFC pathway plays an important role in visceral pain and anxiety associated with IBD and the effect of EA. Thus, our study provides new information about the cellular and molecular mechanisms of the therapeutic effect of EA on the comorbidity of visceral pain and anxiety in IBD (Figure 8).

Hypothesis diagram of EA alleviating anxiety and visceral pain through inhibiting the vHPC-mPFC pathway. (a) In TNBS-induced IBD mice, hyperactivity of pyramidal neurons was in both vHPC and mPFC receiving each behavior test and vHPC-to-mPFC pathway was overactivated; (b) In TNBS-induced IBD mice with EA treatment, EA reduced hyperactivity of pyramidal neurons in the vHPC during each behavior test and EA suppressed the activation of mPFC pyramidal neurons associated with anxiety-like behaviors but did not alter the neuronal response to von Frey filament stimulation. Further, EA downregulated overactivation of vHPC-to-mPFC pathway. To sum up, the effect of EA on anxiety and visceral pain, but not on mechanical pain, in IBD mice was via inhibition of the vHPC-to-mPFC pathway.
We have discussed the considerations behind our current control group settings and acknowledge that for a more precise dissection of the neural circuitry, future studies could incorporate more complex control groups (e.g., groups receiving viral injections but no optogenetic stimulation/inhibition) to further exclude potential non-specific effects. Building on this, we propose that future research should focus on elucidating the hierarchical and synergistic relationships between the vHPC→mPFC pathway and other known analgesic/anti-anxiety circuits (such as the PAG-amygdala pathways), aiming to construct a more comprehensive neural network model underlying the mechanisms of EA.
Supplemental Material
sj-docx-1-mpx-10.1177_17448069251396875 – Supplemental material for Electroacupuncture reduces visceral pain and anxiety associated with inflammatory bowel disease by inhibition of vHPC-to-mPFC pathway
Supplemental material, sj-docx-1-mpx-10.1177_17448069251396875 for Electroacupuncture reduces visceral pain and anxiety associated with inflammatory bowel disease by inhibition of vHPC-to-mPFC pathway by Xue-Fei Hu, Wen-Qiang Ge, Teng-Fei Hou, Pan-Pan Lv, Fan Wu, Qiang Fu, Qi-Shun Lin, Li-Xue Lin, Hong Zhang, Li-Jun Zhang, Hong-Chun Xiang, Shu-Xiu Zhu and Man Li in Molecular Pain
Footnotes
Author contributions
Man Li and Xue-Fei Hu designed all experiments. Xue-Fei Hu performed the calcium fiber recording studies and analyzed data. Wen-Qiang Ge, Teng-Fei Hou, Hong Zhang, Li-Jun Zhang, and Hong-Chun Xiang assisted in molecular, pathological and behavioral assays and data analysis. Xue-Fei Hu drafted the manuscript, which was revised by Qiang Fu, Fan Wu, Hong-Chun Xiang, Qi-Shun Lin, and Li-Xue Lin. All experiments were supervised by Man Li, Xue-Fei Hu, and Shu-Xiu Zhu. All authors approved the final edited version.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the National Natural Science Foundation of China (No. 82405550, 82174500, 82305049), Key Program of the National Natural Science Foundation of China (No.82130122), the Natural Science Foundation of Hubei province (grant number 2022CFB982 and 2024AFB1052).
Ethics statement
All animal experimental protocols conformed to the Animal Management Rules of the Chinese Ministry of Health, and the study was approved by the Animal Ethics Committee of the Chinese Academy of Medical Sciences.
Data availability statement
The original contributions presented in the study are included in the article, and further inquiries can be directed to the corresponding authors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
