Abstract
Purpose:
Migraine is a neurological disorder, affecting approximately 1.16 billion individuals globally. Zinc finger HIT-type containing 1 (Znhit1), a chromatin remodeler, has exhibited a neuroprotective role. This study aims to investigate the role of Znhit1 in migraine.
Methods:
A migraine mouse model was established by repeated intraperitoneal injection of nitroglycerin (NTG). The Znhit1 expression in trigeminal nucleus caudalis (TNC) was detected using reverse transcription quantitative polymerase chain reaction and western blot assays. Behavioral phenotype caused by central sensitization was assessed by Von Frey monofilaments and hot plate assays. Inflammatory response was evaluated through enzyme-linked immunosorbent assay and western blot analysis. Gene set enrichment analysis, western blot, cell counting kit-8 (CCK-8), and flow cytometry were utilized to explore potential molecular mechanisms in vitro.
Results:
Repeated injection of NTG reduced Znhit1 expression in the TNC. Overexpression of Znhit1 alleviated hyperalgesia, upregulated 5-hydroxytryptamine (5-HT) level, and inhibited FBJ osteosarcoma oncogene (c-Fos) and calcitonin gene-related peptide (CGRP) expression. Moreover, overexpression of Znhit1 downregulated the expression of inflammatory markers [interleukin (IL)-6, IL-1β, tumor necrosis factor (TNF)-α, cyclooxygenase-2 (COX-2), and inducible nitric oxide synthase (iNOS)] and inhibited the activation of NOD-like receptor protein 3 (NLRP3) inflammasome. CCK-8 assay found that cell viability was enhanced in lipopolysaccharide (LPS)-induced BV2 cells treated with Znhit1 overexpression, while silencing of Znhit1 resulted in decreased cell viability. In LPS-induced BV2 cells, silencing of Znhit1 enhanced inflammatory response, promoted cell apoptosis, and activated NLRP3 inflammasome, whereas MCC950, a specific inhibitor of NLRP3, reversed these effects induced by silencing of Znhit1.
Conclusion:
Our results demonstrated that Znhit1 ameliorated hyperalgesia and inhibited inflammatory response by suppressing the activation of NLRP3 inflammasome.
Highlights
Repeated injection of NTG reduces Znhit1 expression in TNC.
Znhit1 improves hyperalgesia and inhibits inflammatory response in migraine.
The neuroprotective role of Znhit1 is mediated by suppressing NLRP3 inflammasome.
Introduction
Migraine is a chronic neurological disorder characterized by recurrent attacks of moderate to severe headaches, often accompanied by emesis, photophobia, nausea, and phonophobia.1,2 Based on years lived with disability, migraine is recognized as the second most common disabling disease across all ages. 3 In 2021, migraine was the third leading contributor to disability-adjusted lifer-years (DALYs) among neurological disorders worldwide, affecting approximately 1.16 billion individuals. 4 Notably, migraine incidence has been consistently higher in females than in males from 1990 to 2021. 5 Recent studies have emphasized the key role of central sensitization in the pathophysiology of migraine.6,7 Specifically, inflammation factors and neuropeptides can activate microglia in trigeminal nucleus caudalis (TNC), leading to enhanced central sensitization, and activated microglia and neurons further exacerbate this process.8–10
Zinc finger HIT-type containing 1 (Znhit1), a subunit of the SNF2-related CREBBP activator protein (SRCAP) chromosome remodeling complex, participates in the deposition of H2A.Z histone variant to regulate development and homeostasis.11–13 Emerging evidence highlights the protective role of Znhit1 in Parkinson’s disease (PD). 14 Znhit1 expression in PD patients was decreased, and its overexpression promoted neuronal survival and growth.14,15 However, the function of Znhit1 in migraine remains unexplored.
The NOD-like receptors (NLRs) are cytosolic sensors responsible for immune response and have been reported in neurological disorders, including PD, Alzheimer’s disease (AD), and depressive disorder. 16 Overactivation of NOD-like receptor protein 3 (NLRP3) inflammasome, a member of the NLRs family, has been reported in microglia and associated with migraine.17,18 Currently, the inhibition of NLRP3 inflammasome is considered a promising approach for migraine treatment.19,20 For example, in nitroglycerin (NTG)-induced migraine rats, administration of Xiongshao Zhitong granules alleviated migraine symptoms by regulating the TRPV1-mediated NLRP3 inflammatory pathway. 21 Therefore, further research on the mechanisms regulating the NLRP3 inflammasome in migraine is warranted.
This study investigated the roles of Znhit1 in migraine through mouse model induced by NTG and cell model induced by lipopolysaccharide (LPS). Our findings may advance our understanding of migraine pathology.
Materials and methods
Bioinformatics analysis
The transcriptomic dataset (GSE110194) of migraine mice induced by NTG was downloaded from the Gene Expression Omnibus (GEO) database. Differentially expressed genes (DEGs) were identified using the DESeq2 package in R, and Znhit1 expression was compared between the control and NTG groups. Gene set enrichment analysis (GSEA) was used to analyze the pathways related to Znhit1.
Animal
Specific pathogen-free (SPF) C57BL/6 female mice (~8 weeks old, 18–22 g) purchased from Beijing Vital River Laboratory Animal Technology Co., Ltd (China) were bred under a 12-h light-dark cycle (20–25℃, 55%–60% humidity), with sufficient food and water. All animal procedures were approved by the Ethics Committee of Yantaishan hospital (approval number: 2024-169) and complied with the guide for the Care and Use of Laboratory Animals.
Construction of migraine model
The migraine model was established using intraperitoneal (i.p.) injection of NTG (Beijing Yimin, China). Before each application, 1 mg/mL NTG working solution was prepared by diluting the fresh NTG stock solution (5 mg/mL) with 0.9% saline and stored away from light. Following 3 days of acclimation, mice in the control group were administered with 0.9% saline (1 mL/100 g) through i.p. injection, while mice in the NTG group received i.p. injection of NTG (10 mg/kg) at 1, 3, 5, 7, and 9 days. After the last NTG injection for 2 h, mice were intraperitoneally injected with 1% pentobarbital sodium for euthanasia, then TNC tissues were collected at each time point.
Stereotaxic virus injection
Mice were anesthetized by inhaling isoflurane (5% induction, 1.5% maintenance) and fixed on a stereotaxic apparatus (RWD Life Science, Guangdong, China). According to the stereotaxic atlas, the TNC region was located, and a hole was drilled using a skull borer. Then, microinjection was carried out with a 10 μL NanoFil syringe (World Precision Instruments, USA) at a controlled speed of 100 nL/min, and 1 μL adeno-associated virus (AAV)-Znhit1 or negative control (empty vector) was injected into TNC, followed by leaving needle for 10 min. After withdrawing the needle with a slow velocity, the skin was closed with 4-0 lines, and mice were maintained for 2 weeks for further analysis.
Animal treatment
A total of 40 mice were randomized into 4 groups (each group = 10): control, NTG-induced migraine (NTG), NTG + vector (Vector), and NTG + Znhit1 (Znhit1) groups. Mice in NTG, Vector, and Znhit1 groups were administered with NTG (10 mg/kg) through i.p. injection at 15, 17, 19, 21, and 23 days. The control group was received the same volume of saline at the same point.
Behavioral test
Before official testing, mice were underwent adaptation training for 3 days, at least 30 min. On days 15, 17, 19, 21, and 23, the mechanical nociceptive and thermal thresholds were measured before NTG or saline injection (basal response) and after NTG or saline injection for 2 h (post-treatment response). All experiments were performed by the same researchers blinded to the groups. For the mechanical nociceptive threshold, Von Frey monofilaments were used to prick the hind paw for 3 s. The positive responses were manifested as shaking or rapid withdrawal in mice, and the 50% mechanical pain threshold was evaluated using the up-down method.
For the thermal threshold, the hot plate analgesia meter was utilized to detect the thermal threshold of the hind paw. After 30 min of acclimatization, the hot plate was heated to 50 ± 5℃ to reach the test temperature, and then mice were placed onto the hot plate. The positive responses were lick, jump, or stamp.
Cell culture and treatment
Mouse microglia BV2 cells (Procell, China) were cultured in dulbecco’s modified eagle medium (DMEM) containing 10% fetal bovine serum and 1% penicillin-streptomycin solution at 37℃ with 5% CO2. The pcDNA3.1-Znhit1, two siRNAs, or corresponding negative control were transfected into BV2 cells with lipofectamine 3000. After transfection for 48 h, BV2 cells were induced by 1 μg/mL LPS (Beyotime, China) for 24 h,22–24 followed by further analysis. In si-Znhit1 + MCC950 + LPS group, after transfection with si-Znhit1 for 48 h, BV2 cells were treated with MCC950 for 5 h and then stimulated with LPS for 24 h. The siRNAs sequencing was as follows: si1-Znhit1: 5′-GTAGGCTCTTCTGTCAACTGT-3′, si2-Znhit1: 5′-GTCTGAAGTGGACCGTGTAAA-3′, and si-NC: 5′-GGTGACGAACTAGGTGTACTA-3′.
Reverse transcription quantitative polymerase chain reaction (RT-qPCR)
The total RNA was extracted from TNC using Trizol reagent (Invitrogen, USA), and the QuantiTect RT-PCR Kit (Qiagen, USA) was utilized to quantify Znhit1 expression. Through 2−ΔΔCT method, mRNA expression levels were calculated. The primer sequences for 5′-3′ were: Znhit1: GATCGGAGGCAGAAGTTACGG (forward primer) and GTCCTGGGAGCGAACTTG (reverse primer); glyceraldehyde 3-phosphate dehydrogenase (GAPDH): TCAGGAGAGTGTTTCCTCGTC (forward primer) and ATGAAGGGGTCGTTGATGGC (reverse primer).
Western blot
BV2 cells or TNC tissues were lysed on ice using RIPA lysis buffer containing 1% protease and phosphatase inhibitor (Sangon Biotech, China). Then, the lysis buffer was transferred to a centrifuge tube and centrifuged for 20 min at 12,000 rpm and 4℃, followed by the collection of the supernatant. The BCA Protein Assay Kit (Beyotime) was used to measure protein concentration. After protein denaturation, samples (30 μg) were loaded into sodium dodecyl sulfate polyacrylamide gel electrophoresis to separate protein and transferred into the polyvinylidene fluoride membrane. The membrane was blocked using tris-buffered saline with Tween-20 with 5% skimmed milk powder for 40 min in a shaker (Eppendorf, Germany) and then incubated with following antibodies: anti-Znhit1 (1:2000, 16595-1-AP, Proteintech, China), anti-GAPDH (1:20000, 10494-1-AP, Proteintech), anti-FBJ osteosarcoma oncogene (c-Fos, 1:40000, 66590-1-Ig, Proteintech), anti-calcitonin gene-related peptide (CGRP, 1:1000, 14959, Cell Signaling Technology, USA), anti-interleukin (IL-6, 1:1000, ab290735, Abcam, UK), anti-IL-1β (1:3000, 26048-1-AP, Proteintech), anti-tumor necrosis factor (TNF)-α (1:1000, 17590-1-AP, Proteintech), anti-cyclooxygenase-2 (COX-2, 1:1500, 27308-1-AP, Proteintech), anti-inducible nitric oxide synthase (iNOS, 1:1000, ab178945, Abcam), anti-NLRP3 (1:6000, 30109-1-AP, Proteintech), anti-cleaved caspase-1 (1:1000, 89332, Cell Signaling Technology), and anti-apoptosis-associated speck-like protein (ASC, 1:2000, 83858-3-RR, Proteintech) overnight at 4℃. After washing three times, the membranes were incubated with secondary antibodies (1:5000, ab205719; 1:50000, ab205718; Abcam) at room temperature (RT) for 1 h. The enhanced chemiluminescence (ECL) work buffer (Vazyme, China) was added to the membrane for 2 min at RT to detect bands and then analyzed using Image J software.
Enzyme-linked immunosorbent assay (ELISA)
The TNC supernatant was collected after lysing using RIPA buffer with 1% protease and phosphatase inhibitors. Then, the Mouse IL-6 ELISA Kit (RAB0309, Sigma, USA) and the Mouse IL-1β ELISA Kit (RAB0275, Sigma) were utilized to detect the IL-6 and IL-1β levels according to their respective protocols. Mouse Tumor necrosis factor α, TNF-α ELISA KIT (CSB-E04741m, Cusabio, China) was used to measure TNF-α level.
Mouse whole blood in tubes with an anticoagulant was centrifuged at 1000 rpm for 15 min at 4℃. The plasma was used to analyze 5-hydroxytryptamine (5-HT) and CGRP levels using ST/5-HT ELISA Kit (D751013, Sangon) and Mouse Calcitonin Gene Related Peptide (CGRP) ELISA kit (CSB-EQ027706MO, Cusabio).
Measurement of nitric oxide (NO) in serum
Mouse whole blood in the tube without anticoagulant was coagulated at RT, followed by centrifugation at 1000 rpm for 20 min at 4℃. The NO Content Assay Kit (D799309, Sangon) was used to measure the NO level. The absorbance value at 550 nm was determined.
Immunofluorescence (IF) staining
Mouse brain tissues were fixed in 4% paraformaldehyde overnight, followed by dehydration in 20% sucrose solution for 24 h and 30% sucrose solution for 24 h. The brain tissues was embedded in optimal cutting temperature (OCT, SAKURA, USA) and sliced into 10 μm thickness. After washing and blocking, the TNC slices were incubated with anti-c-Fos (1:1000, ab208942, Abcam) and anti-CGRP (1:100, ab81887, Abcam) at 4℃ overnight. Next, 200 μL secondary antibody solution (1:1000, ab150113 and ab150115, Abcam) was added to incubate slices for 1 h at RT, avoiding light. Subsequently, slices were stained with 4′,6-diamidino-2-phenylindole (DAPI, Beyotime) for 5 min at RT and observed under a fluorescence microscopy (Leica). At last, ImageJ software was utilized for measuring the mean fluorescence intensity (MFI).
Cell counting kit-8 (CCK-8) assay
The BV2 cell viability was assessed using Cell Counting Kit-8 kit (Solarbio, China). In brief, 10 μL CCK-8 solution was added to incubate the treated BV2 cells for 2 h. The absorbance values at 450 nm were determined.
Flow cytometry
The treated BV2 cells were digested using trypsin and harvested. Cells were washed in pre-cold PBS and then resuspended in Annexin V-FITC binding buffer (Beyotime). Next, 5 μL Annexin V-FITC and 10 μL propidium iodide (PI) staining solution were added to cell suspension for 20 min in the dark. Finally, the apoptotic cells were examined using a flow cytometer (BD Biosciences, USA). Cells were divided into Q1 quadrant (necrotic cells), Q2 quadrant (late apoptosis), Q3 quadrant (early apoptosis), and Q4 quadrant (living cells).
Statistical analysis
All experimental results were shown as the mean ± standard deviation (SD) and analyzed in GraphPad Prism 9.0. Before statistical comparison analysis, Shapiro-Wilk test was utilized for testing data normality, Brown-Forsythe test was utilized for testing variance homogeneity. Differences between the two groups were compared using Student’s t-test. One-way analysis of variance (ANOVA) or two-way ANOVA followed by Tukey’s post hoc test was utilized to analyze differences in multiple groups. p < 0.05 was considered statistically significant.
Results
Repeated injection of NTG reduces Znhit1 expression in the TNC tissue
In the GSE110194 dataset, Znhit1 expression was downregulated in the TNC of NTG-induced mice (Figure 1(a)). Then, the migraine model was constructed by repeated injection of NTG to further confirm the expression of Znhit1 (Figure 1(b)). Compared with control group, Znhit1 expression at mRNA and protein levels in NTG groups was gradually decreased with increasing time (Figure 1(c) and (d)).

Repeated injection of NTG reduced Znhit1 expression in the TNC tissue. (a) The Znhit1 expression level in the GSE110194 dataset (n = 10), Student’s t-test was utilized for statistical analysis. (b) The schematic diagram illustrates the establishment of the migraine model by injection (i.p.) of NTG (10 mg/kg) at 1, 3, 5, 7, and 9 days. (c–d) The mRNA and protein expression of Znhit1 in TNC was detected using RT-qPCR (c) and western blot (d) (n = 5), one-way analysis of variance followed by Tukey’s post hoc test was utilized to perform statistical analysis.
Znhit1 improves hyperalgesia in NTG-induced mice
To explore the role of Znhit1 in vivo, we overexpressed Znhit1 by AAV intracranial injection before injection of NTG, and TNCs were collected on day 24 (Figure 2(a)). Compared to the control group, Znhit1 expression was significantly decreased in the NTG group, while Znhit1 expression in the Znhit1 group was markedly increased compared with vector group (Figure 2(b)). The mechanical and paw withdrawal latency thresholds were assessed before NTG injection (basal response) and after NTG injection for 2 h (post-treatment response) at 15, 17, 19, 21, and 23 days. For the basal response, there were no differences at the first time point among the four groups; except for first time point, the mechanical and paw withdrawal latency thresholds in the NTG group were notably decreased compared to the control group (Figure 2(c) and (d)). Compared to the vector group, Znhit1 overexpression notably increased mechanical and paw withdrawal latency thresholds in both basal and post-treatment responses (Figure 2(c) and (d)), suggesting that Znhit1 might ameliorate NTG-induced hyperalgesia.
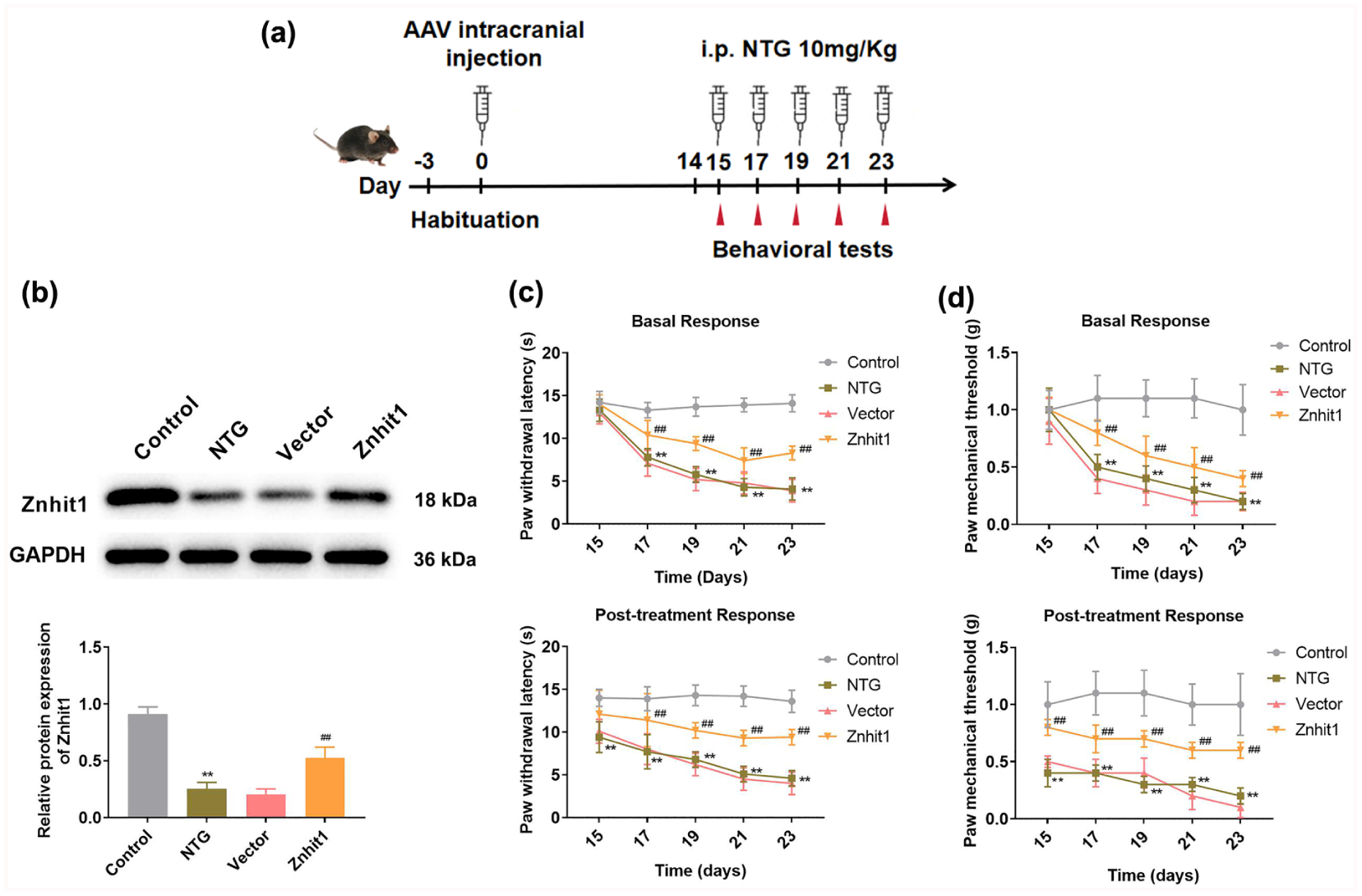
Znhit1 alleviated hyperalgesia in NTG-induced mice. (a) The schematic diagram illustrates the protocol of this study; mice were injected with AAV to overexpress Znhit1 for 14 days. Mice were injected (i.p.) with NTG (10 mg/kg) or saline on 15, 17, 19, 21, and 23 days. (b) The Znhit1 expression in the TNC was detected using western blot (n = 5); One-way analysis of variance followed by Tukey’s post hoc test was utilized to perform statistical analysis. (c–d) Paw withdrawal latency (c) and paw mechanical thresholds (d) of mouse hind paws were evaluated before the injection of NTG or saline and after the injection of NTG or saline for 2 h (n = 10). Two-way analysis of variance followed by Tukey’s post hoc test was utilized to perform statistical analysis.
Znhit1 enhances 5-HT level and inhibits the expression of c-Fos and CGRP
In migraine mice induced by NTG, NO is considered a trigger, which leads to the activation of c-Fos and CGRP to induce central sensitization.25,26 Additionally, 5-HT dysregulation has been implicated in migraine pathogenesis. 27 Therefore, the levels of NO, c-Fos, CGRP, and 5-HT were determined. In the NTG group, we observed decreased 5-HT level and elevated the expression of CGRP, NO, and c-Fos compared with the control group (Figure 3(a)–(e)). However, compared to the vector group, the 5-HT level was markedly increased, while CGRP and NO levels were notably decreased in the Znhit1 group (Figure 3(a)–(c)). Moreover, c-Fos and CGRP levels were significantly downregulated in TNC tissues in the Znhit1 group compared with the vector group (Figure 3(d) and (e)).

Znhit1 facilitated 5-HT expression and reduced the expression of c-Fos and CGRP in NTG-induced mice. (a–b) ELISA was used to assess 5-HT (a) and CGRP (b) levels in plasma (n = 10). (c) The level of NO in the serum was evaluated using the colorimetric method (n = 10). (d–e) The c-Fos and CGRP levels were detected in the TNC tissues using western blot (d) and IF (e) assays (n = 5). One-way analysis of variance followed by Tukey’s post hoc test was utilized to perform statistical analysis.
Znhit1 alleviates NTG-induced inflammation in TNC
Inflammation activates the trigeminal nervous system and contributes to migraine. 28 The levels of pro-inflammatory factors (IL-6, IL-1β, and TNF-α) and related enzymes (COX-2 and iNOS) were measured. Repeated NTG injection significantly increased the levels of IL-6, IL-1β, and TNF-α, while Znhit1 overexpression remarkably diminished their levels in TNC (Figure 4(a)–(c)). Similar results were also observed in the western blot assay (Figure 4(d)). Moreover, COX-2 and iNOS expression was notably upregulated in the NTG group compared to the control group, whereas their expression was dramatically downregulated after Znhit1 overexpression (Figure 4(d)). Our findings indicated that Znhit1 might relieve NTG-induced inflammation.

Znhit1 inhibited inflammatory response in TNC. (a–c) The levels of IL-6 (a), IL-1β (b), and TNF-α (c) were evaluated in TNC tissues using ELISA. (d) The levels of IL-6, IL-1β, TNF-α, COX-2, and iNOS were determined in TNC tissues by western blot assay. One-way analysis of variance followed by Tukey’s post hoc test was utilized to perform statistical analysis, n = 5.
Znhit1 suppresses NOD-like receptor pathway in vivo
To explore the potential molecular mechanism of Znhit1 involved in migraine, GSEA and western blot were performed. GSEA found that Znhit1 could suppress the NOD-like receptor pathway (Figure 5(a) and (b)). Next, the expression of three proteins in the NOD-like receptor pathway was analyzed in TNC. Western blot revealed that NLRP3, cleaved caspase-1, and ASC expression was remarkably elevated in the NTG group compared to the control group; in contrast, their expression in the Znhit1 group was significantly reduced compared to the vector group (Figure 5(c) and (d)).

Znhit1 suppressed NOD-like receptor pathway in vivo. (a) Based on the GSE110194 dataset, GSEA was utilized for analyzing the potential pathways that might be regulated by Znhit1, including NOD-like receptor signaling pathway (red frame). (b) The NOD-like receptor pathway enrichment plot in the GSE110194 dataset. (c–d) Western blot was used to detect the NLRP3, cleaved caspase-1, and ASC expression in TNC tissues. One-way analysis of variance followed by Tukey’s post hoc test was utilized to perform statistical analysis, n = 5.
Znhit1 inhibits LPS-induced inflammation and NOD-like receptor pathway in vitro
LPS could induce the pro-inflammatory state of BV2 cells. 29 Therefore, the function of Znhit1 in LPS-induced BV2 cells was further investigated. We first successfully overexpressed Znhit1 in BV2 cells (Figure 6(a)). CCK-8 assay showed that cell viability was markedly impaired in BV2 cells treated with LPS, whereas Znhit1 overexpression notably enhanced cell viability (Figure 6(b)). Flow cytometry revealed that the rate of apoptotic cells in the LPS group was significantly increased compared to the control group, while apoptosis rate in the Znhit1 group was remarkably decreased compared to the vector group (Figure 6(c)). Consistent with findings in migraine mice, Znhit1 overexpression dramatically diminished the expression of IL-6, IL-1β, TNF-α, NLRP3, cleaved caspase-1, and ASC in LPS-induced BV2 cells (Figure 6(d) and (e)).

Znhit1 inhibited inflammation and NOD-like receptor pathway in vitro. (a, f) After overexpressing (a) or knocking down (f) Znhit1 in BV2 cells, the Znhit1 expression was determined using the western blot. (b, g) CCK-8 was performed to detect BV2 cell viability. (c, h) Flow cytometry was applied to assess the apoptotic rate in BV2 cells. (d, e, i, j) The expression of IL-6, IL-1β, TNF-α, NLRP3, cleaved caspase-1, and ASC was evaluated using the western blot in BV2 cells. Student’s t-test or one-way analysis of variance followed by Tukey’s post hoc test was utilized to perform statistical analysis, n = 3.
Furthermore, we also knocked down Znhit1 in BV2 cells (Figure 6(f)). The cell viability was significantly decreased in the LPS group, and the inhibition of Znhit1 further downregulated LPS-induced BV2 cell viability (Figure 6(g)). Additionally, the apoptotic rate was increased in the LPS group, and suppression of Znhit1 further promoted cell apoptosis (Figure 6(h)). The expression of IL-6, IL-1β, TNF-α, NLRP3, cleaved caspase-1, and ASC was notably increased in LPS-induced BV2 cells, and silencing of Znhit1 further increased these inflammatory markers in LPS-induced BV2 cells (Figure 6(i) and (j)). Our results indicated that Znhit1 might suppress inflammation and the NOD-like receptor pathway in LPS-induced BV2 cells.
Znhit1 mitigates cell apoptosis and inflammation by inhibiting activation of NLRP3 inflammasome
Based on the inhibitory effects of Znhit1 on inflammation and the NOD-like receptor pathway, the relationship between Znhit1 and NLRP3 was explored in BV2 cells using MCC950, a selective NLRP3 inhibitor. CCK-8 assay showed that silencing of Znhit1 in LPS-induced BV2 cells reduced cell viability, while administration of MCC950 elevated cell viability compared with the si1-Znhit1 + LPS group (Figure 7(a)). Moreover, silencing of Znhit1 promoted IL-6, IL-1β, and TNF-α expression and increased apoptotic rate in LPS-induced BV2 cells; in contrast, these indicators were reversed after treatment with MCC950 (Figure 7(b) and (c)). These findings suggested that Znhit1 might relieve inflammation and cell apoptosis through suppressing NLRP3 inflammasome.

Znhit1 alleviated cell apoptosis and inflammation by inhibiting the activation of NLRP3 inflammasome. (a) BV2 cell viability was detected using CCK-8 assay. (b) IL-6, IL-1β, and TNF-α expression in BV2 cells was assessed using western blot. (c) The flow cytometry was conducted to analyze the apoptotic rate of BV2 cells. One-way analysis of variance followed by Tukey’s post hoc test was utilized to perform statistical analysis, n = 3.
Discussion
Migraine represents a widespread and debilitating neurological condition that greatly impacts the quality of life for patients and imposes a considerable economic burden on society. 30 Current migraine treatment approaches remain inadequate for many patients, with studies showing that over one-third of sufferers fail to respond to triptans. 31 Lately, Znhit1 has been identified as a protective gene in PD. 14 Here, the effects of Znhit1 on NTG-induced mice and LPS-stimulated BV2 cells were investigated, revealing that it may play a neuroprotective role in migraine.
Znhit1 regulates cell apoptosis and participates in cell fate determination and organ development by modulating chromatin structure.11,13,32–34 Interestingly, gene co-expression analysis revealed that Znhit1 was lost in patients with PD, and overexpression of Znhit1 enhanced neuron growth and viability in dopaminergic neurons treated with α-Synuclein. 14 In NTG-induced mice, we found that Znhit1 expression was decreased. Additionally, in vitro, overexpression of Znhit1 enhanced cell viability and repressed cell apoptosis, while silencing of Znhit1 diminished cell viability and promoted cell apoptosis in LPS-induced BV2 cells. These results indicated that Znhit1 might play a protective role in migraine through enhancing microglia viability and inhibiting apoptosis.
NTG, a donor of NO as an endogenous signaling molecule, is widely used in migraine models. 35 NO could cause migraine attacks by promoting vasodilation and activation of trigeminal nerves.36,37 Trigeminovascular system activation represents a hallmark pathophysiological feature of migraine, and its persistent activation contributes to central sensitization, with c-Fos serving as a biomarker of this process.38,39 Hyperalgesia, commonly observed during migraine, is one manifestation of central sensitization. 40 At present, CGRP and 5-HT are considered two primary mediators involved in migraine. 41 During migraine episodes, trigeminal nerve fibers release CGRP, inducing vasodilation and activating nociceptors on the trigeminal nerve. 42 Elevated CGRP level in the TNC have been detected in migraine patients. 43 5-HT functions as a vasoconstrictor, and its reduction can result in intracranial vasodilation, heightened sensitivity of the trigeminovascular pathway, and enhanced trigeminal nociception.44,45 Moreover, based on the abundant expression of 5-HT receptors in the cerebral vasculature, triptans, a class of 5-HT receptor agonists, were developed as a first-line drug for the acute treatment of migraine.41,42 These 5-HT receptor agonists effectively suppress CGRP release and diminish vasodilation to alleviate migraine. 46 In this study, we observed hyperalgesia, elevated CGRP, NO, and c-Fos levels, and a decreased 5-HT level in NTG-induced mice. However, Znhit1 overexpression improved hyperalgesia, reduced the expression of CGRP, NO, and c-Fos, while increased 5-HT level. These findings suggested that Znhit1 regulated the levels of neurotransmitters, thereby playing a neuroprotective role in migraine.
Cortical spreading depolarization (CSD) is the trigger of migraine aura. 47 Under migraine conditions, CSD can activate microglia, which subsequently enhances neuronal stimulation, resulting in elevated inflammatory factors. 48 These inflammatory factors can be responsible for TNC sensitization. 28 IL-6, IL-1β, and TNF-α were the common neurogenic neuroinflammation cytokines. 49 Many studies have reported their increased levels in patients with migraine.50–52 Furthermore, inhibition of iNOS and COX-2-mediated inflammation has been demonstrated to improve migraine.35,53–55 In our study, the expression of IL-6, IL-1β, TNF-α, COX-2, and iNOS was upregulated in NTG-induced mice, whereas overexpression of Znhit1 downregulated these inflammatory markers, suggesting that Znhit1 might relieve inflammation in migraine.
Recently, He et al. 18 reported that elevated NLRP3 expression was detected in microglia in TNC of NTG-induced mice. The NLRP3 inflammasome comprises the sensor protein NLRP3, articulation protein ASC, and effector proteinase caspase-1. 56 Upon activation, caspase-1 cleaves pro-IL-1β or pro-IL-18 to generate activation of inflammatory factors to participate in migraine.57,58 Similarly, in this study, NLRP3, cleaved caspase-1, and ASC expression was increased in NTG-induced mice and LPS-induced BV2 cells. In LPS-induced BV2 cells, silencing of Znhit1 further upregulated the expression of NLRP3, cleaved caspase-1, ASC, IL-6, IL-1β, and TNF-α. In contrast, MCC950 treatment reversed these inflammatory markers’ changes induced by silencing of Znhit1. Moreover, the effects of silencing of Znhit1 on BV2 cell viability and apoptosis were reversed by MCC950 treatment. Our findings suggested that Znhit1 might exert a neuroprotective role in migraine by inhibiting the activation of NLRP3 inflammasome.
However, some limitations exist in the current study. For example, our study mainly relied on a migraine model induced by NTG. Although this is a commonly used model, it may not fully simulate all the complex pathophysiological features of human migraine, especially the spontaneous onset and chronicity processes. Therefore, the generalizability of the research results in other migraine models or clinical situations needs further validation. The neuroinflammation in migraine involves complex interactions among multiple cell types, such as neurons, astrocytes, peripheral immune cells, etc. In vitro experiments only used the BV2 microglial cell line, which may overlook the contributions of other important cells in this process. In addition, the specific molecular mechanism by which Znhit1 regulates the NLRP3 inflammasome (e.g. through direct interactions or by affecting other signaling pathways) has not been fully elucidated. In future studies, we will further explore the underlying mechanisms.
Conclusions
The neuroprotective role of Znhit1 in migraine was confirmed in our study. Znhit1 alleviated NTG-induced central sensitization and inflammation response by inhibiting the activation of NLRP3 inflammasome. Our results may offer a new target to improve migraine.
Footnotes
Author contributions
XW designed the study and performed the research; JHT, YWH, and CBS performed the research and analyzed data; CBS wrote the paper. All authors have read and approved the manuscript. The authors declare that all data were generated in-house and that no paper mill was used.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics approval and Consent to participate
The experimental protocol of our study was performed in accordance with the Guide for the Care and Use of Laboratory Animals and approved by Yantaishan Hospital (approval number: 2024-169).
Availability of data and material
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
