Abstract
Introduction
Knee osteoarthritis (KOA) is highly prevalent among older adults 1 and is one of the primary causes of pain and disability worldwide. 2 According to the data from the China Health and Retirement Longitudinal Study, the prevalence of symptomatic KOA is 8% with increased risk as one age until a plateau around age 70. 3
As a multifactorial disease, KOA can cause symptoms including joint pain, stiffness, loss of motion, fatigue, poor sleep, and loss of function as well as radiographic changes including joint space narrowing and osteophyte formation. 4 The pathogenesis of KOA is complex, involving mechanical overload, 5 inflammatory, 6 metabolic alterations, 7 and cellular senescence, 8 which together result in structural deterioration and failure of synovial joints. 6 There was a major opinion that the inflammation reaction plays a critical role in the development of KOA, 9 and joint damage will lead to sterile inflammation. Clinical investigations now show that synovial inflammation is associated with pain sensitization, and similar to KOA, is a common and important feature of hand osteoarthritis, 10 and macrophages are key players in KOA pathology and their activation state has been studied extensively. 11 Targeting macrophages in inflammation is relevant since macrophages are the main producers of a range of pro-inflammatory cytokines. 12 High levels of inflammatory cytokines, such as IL-1β, TNF-α, and proteolytic enzymes, enhance osteoclast differentiation and stimulate various cells in the synovium, such as synovial fibroblasts and leukocytes. 13
NLRP3 is a multiprotein oligomer composed of caspase-1, an apoptosis-related spot-like protein containing CARD (ASC) and nod-like receptor protein 3 (NLRP3). Caspase-1 is recruited to an inflammasome by proximity-induced autocatalytic activation. Activated caspase-1 cleaves the cytokines pro-interleukin-1β (pro-IL-1β) and pro-IL-18 into their mature and biologically active forms. 14 Pyroptosis is an inflammatory form of regulated cell death that relies on cytosolic inflammasome activation, 15 which is dependent on inflammasome-mediated caspase-1 activation and results in the formation of plasma membrane pores due to Gasdermin D (GSDMD) insertion, leading to the release of intracellular proteins, ion decompensation, water influx and cell swelling. 16 Pan et al. found that NLRP1/NLRP3 inflammasome-mediated and caspase-1-dependent FLS pyroptosis increased HMGB1 secretion in KOA. 17 Anders thinks that macrophage pyroptosis in the kidney can lead to renal inflammation and fibrosis. 18 In wang’s report, synovial tissue showed significant NLRP3-mediated cellular pyroptosis in KOA. 14 Hence, we wanted to further investigate the mechanism of pyroptosis in knee osteoarthritis.
Acupuncture is a form of traditional Chinese medicine, which involves the action of inserting needles into specific points, 19 called “acupoints.” 20 Acupuncture intervention is frequently used to control pathologic symptoms in several visceral organs, and a growing number of studies using experimental animal models suggest that acupuncture stimulation may be involved in inducing anti-inflammatory responses. 21 Electroacupuncture (EA), which is widely used in clinical practice, refers to the pulse current input to acupoints based on needle acupuncture. EA is a non-surgical method widely used in the therapy of KOA, and different acupuncture methods have been clinically proven to be effective. 22 As a traditional means of treating pain, its functions such as pain relief, sedation, promotion of “qi” and blood circulation, and adjustment of muscle tension have been proved by a large number of clinical practices to improve joint pain, swelling, deformity, and activity of KOA. It has a remarkable curative effect on clinical symptoms and is widely used in the treatment of KOA.23,24
In summary, KOA is a specific inflammatory disease with pyroptosis characterized by inflammasome activation, which may be regulated by multiple inflammatory signals. We hypothesize that EA can relieve the inflammatory response in the joint, thereby alleviating pyroptosis and thus treating KOA.
Materials and methods
Agents
NLRP3 Rabbit pAb (A5652, A14223), ASC Rabbit pAb (A16672), Caspase-1 Rabbit pAb (A0964), IL-1β Rabbit pAb (A11369, A11370), IL-18 Rabbit pAb (A1115), MMP13 Rabbit pAb (A11755), CD68 Rabbit pAb (A15037), TNF-α Rabbit pAb (A11534), GSDMD Rabbit pAb (A18281), GSDMD Full length+N-terminus Rabbit pAb (A20197), β-actin Rabbit mAb (AC026), HRP’s anti-rabbit IgG antibody (AS014), all antibodies were purchased from Abclonal, Wuhan, China. Sodium iodoacetate (S817623, Shanghai Macklin Biochemical Co., Ltd), Microliter Syringes (50 μL, Shanghai High Pigeon Industry and Trade Co., Ltd, Shanghai, China), Disposable acupuncture needles (HUAN QIU, Suzhou Acupuncture and Moxibustion Appliance Co., Ltd, Suzhou, China), Huatuo brand SDZ-II electronic acupuncture apparatus (Suzhou Medical Equipment Co., Ltd., Suzhou, China), Electric Electric Von Frey (IITC Life Sciences, CA, USA).
KOA model
Forty-five male SD rats weighing 250–300 g were maintained on a 12-h light/dark cycle with free access to food and water. The rats were randomly divided into 3 groups: the control group (n = 15), the KOA group (n = 15), the KOA+EA group (n = 15). The control group was injected with 50 μL of sterile 0.9% saline, and the KOA model was injected with classical monosodium iodoacetate (MIA). 25 In brief, the rats in each group were anesthetized with chloral hydrate (40 mg/kg, intraperitoneal injection), and 50 μL of sodium iodoacetate solution was injected into the right knee joint cavity of the rats with a micro syringe (3 mg dissolved in 50 μL of sterile 0.9% normal saline), the same volume of solvent was injected in the same position in the control group. Subsequently, the rats were placed in a continual drawer-like joint movement after stretching and bending the injected hind limb 10 times. The rats were kept on a heating pad to maintain their temperature at 37°C. The experimental protocol was reviewed and approved by the Animal Research and Care Committee of Tongji Medical College, Huazhong University of Science and Technology (SCXK (XIANG)2019–0014).
Experimental design
In our experiment, behavioral tests were performed 1 day before MIA injection (baseline), 1 day before treatment after the successful model, and 12 h after each course of EA treatment. After two courses of EA treatment and behavioral testing, rats were sacrificed for histological evaluation of joint tissue. The experimental design is shown in Figure 1. Timeline of experiments. KOA was induced in rats by intra-articular injection MIA (3 mg/50 μL) through their right knee, while the rats in the Control group were injected with saline at the same position. Rats in experimental groups underwent the following treatments: EA was performed on the fifth day after modeling and lasted for 2 weeks. Behavioral tests were measured once a week until the treatment was completed. After the last treatment, a part of the rats in each group was randomly selected to be sacrificed, and the right knee joint was taken for pathological observation.
EA operation
Rats were placed in a gentle rat immobilization device. Each EA course was performed for 20 minutes, once a day for 6 days a week. The treatment group received EA after successfully establishing the KOA model, while there was no operation in the control group. The acupoints were located according to the “Map of Acupuncture and Moxibustion Acupuncture Points for Laboratory Animals”. The points are the “Dubi” acupoint (ST35, located in the depression of the lateral patellar ligament of the knee), the “Neixiyan” acupoint (EX-LE4, located in the depression of the medial patellar ligament of the knee), the “Yinlingquan” acupoint (SP9, located at the lower edge of the medial tibial condyle), and the “Yanglingquan” acupoint (GB34, located below the anterior fibular head of the knee). We used 0.25 mm × 13 mm disposable acupuncture needles (HUAN QIU, Suzhou Acupuncture and Moxibustion Appliance Co., Ltd, Suzhou, China), to directly pierce 5 mm deep, all acupoints were connected to electrodes at the root of the needle shank, and one group of Dubi and Neixiyan (“Dubi” is connected to the positive electrode, “Neixiyan” is connected to the negative electrode), another group of Yinlingquan and Yanglingquan (“Yanglingquan” is connected to the positive electrode, and “Yinlingquan” is connected to the negative electrode). The acupoints were connected to the Huatuo brand SDZ-II electronic acupuncture apparatus (Suzhou Medical Equipment Co., Ltd., Suzhou, China). A sparse wave was continuously stimulated for 20 min with a frequency of 2 Hz and an intensity of 1 mA, to the extent that the local muscles of the rats began to shake slightly and the rats did not struggle.
Behavioral tests
Using an electric Von Frey (IITC Life Sciences, CA, USA), the severity of joint pain was quantitatively assessed by paw withdrawal threshold. In short, the specific method is as follows: prepare a 25 cm × 10 cm × 10 cm rat cage, place the rats individually in a suspended metal cage in order, fully expose the bottom of the cage, and start the test after the rats are quiet and adapted. After installing and debugging the pain measuring instrument, the probe is perpendicular to the area between the soles of the rat’s feet and slowly upwards to ensure that the display is visible. Record the number while the rat obviously shrinks its feet under the exclusion of walking and jumping. Each rat needs to be tested more than 3 times, the mean value was taken as the final plantar mechanical pain threshold of the rat and statistical analysis was performed.
Measurement of knee joint swelling
After each rat was calm for half an hour after the paw withdrawal threshold was measured, the transverse diameter of the right knee was measured with a sliding caliper, and the horizontal distance between the left and right highest points of the knee joint flexed at 90° was measured and recorded.
Histopathological analysis
Semi-quantitative scoring system recommended from the OARSI.

Notes: Lorenz J, Grässel S. Experimental osteoarthritis models in mice. Methods Mol Biol. 2014;1194:401–19. doi: 10.1007/978-1-4939-1215-5_23. PMID: 25,064,117.
Grading criteria of Mankin score.

Notes: Adapted from Sahin K, Kucuk O, Orhan C, et al. Niacinamide and undenatured type II collagen modulates the inflammatory response in rats with monoiodoacetate-induced osteoarthritis. Sci Rep. 2021;11 (1):14,724.
Grading criteria of synovitis score.

Notes: Gerwin N, Bendele AM, Glasson S, et al. The OARSI histopathology initiative - recommendations for histological assessments of osteoarthritis in the rat. Osteoarthritis Cartilage. 2010 Oct;18 Suppl 3:S24-34. doi: 10.1016/j.joca.2010.05.030. PMID: 20,864,021.
Immunohistochemistry
During immunohistochemistry, a specimen of knee joint tissue was taken out, dewaxed with xylene, and rehydrated with graded alcohol. The deparaffinized and rehydrated sections were washed twice with pure water and then incubated with pepsin antigen retrieval solution at 37° for 30 min. The sections were circled around the tissue with a histochemical pen, and the endogenous peroxidase-blocking solution was added dropwise to incubate at room temperature for 30 min. After blocking for 1 hour, the sections were respectively treated with antibody NLRP3 (1:200), antibody Caspase-1 (1:200), antibody ASC(1:200), antibody IL-1β(1:200), antibody IL-18 (1:200), antibody CD68 (1:200), antibody TNF-α(1:200) and antibody MMP13(1:200), incubated overnight at 4°C. Secondary antibodies were added for 2 hours at room temperature and detected with DAB. Finally, dark brown cells were considered to be positive. Photomicrographs were taken with a microscope. Micrographs of cartilage tissue samples were obtained at 400X magnification.
Western blot
Removal of cartilage tissue from a −80°C refrigerator and the total protein was extracted with cell lysate. After grinding and centrifugation, the tissue supernatant was obtained. Protein quantification was performed using the Bradford method. 30 micrograms of protein samples were added to sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred to a polyvinylidene fluoride membrane. Primary antibodies were incubated overnight at 4°C. Antibody dilution methods are as follows: antibody NLRP3 (1:500), antibody ASC(1:500), antibody Caspase-1 (1:500), antibody IL-1β(1:500), antibody GSDMD (1:1000), antibody GSDMD+N-terminus (1:1000), antibody MMP13(1:1000) and then incubated with HRP’s anti-rabbit IgG antibody (1:5000) for 2 hours at 37°C. The target protein on the polyvinylidene furoyl membrane was visualized using the ECL kit and captured using the BIO-RAD bioimaging system.
Statistical analysis
The statistical analysis was performed using the SPSS 20.0 software (SPSS Inc., Chicago, IL, USA). Data are presented as the mean ± standard deviation. Group comparisons were assessed with one-way ANOVA or two-way ANOVA with Bonferroni’s post hoc test for the comparison of multiple columns. A p value <.05 (two-tailed) was considered statistically significant.
Results
EA reduces mechanical nociceptive pain and joint swelling in KOA rats
First, we observed the knee joints directly, as shown in Figure 2(A), the knee joints showed no redness and swelling, normal flexion and extension functions, transparent light white articular cartilage, and neat joint edges with no deformation in the control group. In contrast, in the KOA group, there was obvious redness and swelling of the knee joint, swelling and deformation, and the synovial tissue was congested and thickened. But the EA treatment can effectively relieve the deformation of the joint and maintain the normal flexion and extension of the joint. Next, we tested the injurious behavior induced by intra-articular injection of MIA into the right knee joint of rats. Before the start of EA treatment, MIA injection significantly reduced the ipsilateral paw withdrawal threshold (PWT) compared with the control group, and EA treatment can significantly elevate the paw withdrawal threshold caused by MIA (Figure 2(B)). Besides, we found that the swollen diameter of the joints in the KOA group could reach nearly 10 mm compared to the control group by measuring the joint diameter of the rats, while the treatment with EA significantly alleviated the swelling of the joints of the rats in the KOA group, almost reaching the normal level (Figure 2(C)). The above results show that EA treatment can relieve pain and reduce joint swelling. EA reduces mechanical nociceptive pain and joint swelling in KOA rats. (A) Effect of EA on the macroscopic view of articular cartilage damage surface in each group. Effect of EA on alleviating mechanical allodynia and improving the joint swelling degree in KOA rats. (B) PWT was tested by Electric Von Frey before modeling and measured every week. (C) Degree of joint swelling. Two-way ANOVA was used in different periods. Data were presented as mean ±standard deviation, n = 10 in each group. KOA group vs Control group (***p < .001, ****p < .0001), KOA+EA group vs KOA group (###p < .001, ####p < .0001).
EA treatment alleviates the joint pathological changes caused by KOA
Pathological changes are one of the typical features of KOA.
26
Next, we observed knee joints with Safranin O/Fast Green and H&E staining. As shown in Figure 3(A), the control group showed no histopathological changes after staining with Safranin O/Fast Green and showed intact articular cartilage tissue structure, sharp coloration of the whole layer, and normal chondrocyte morphology. In the KOA group, the cartilage tissue structure was significantly damaged at multiple levels, coloring was significantly lost, inflammatory cells eroded the cartilage, and chondrocytes were severely damaged. After EA treatment, the degree of cartilage tissue structure destruction was significantly improved compared with the KOA group, the structure was intact, and only the cartilage surface coloring was lost. The OARSI results were consistent with the above results (Figure 3(E)). Then we observed that the cartilage tangential layer was continuous and intact in the control group, with uniform distribution of cytoplasmic and matrix components, while in the KOA group the cartilage tangential layer fibers were thinned, the degree of cartilage destruction was severe, and the tidal line was completely lost by H&E staining (Figure 3(B)). In contrast, after EA treatment, the cartilage surface was continuous and more regular, the chondrocytes were more neatly arranged, and the radiolucent and calcified layers were clearly structured. It’s the same in synovial tissue (Figure 3(C)), the synovial cell layer was thicker, and inflammatory cell infiltration and angiogenesis were significantly increased in the KOA group compared with the control group, while in the EA group we could see less inflammatory cell proliferation and reduced angiogenesis. The results of the Mankin score (Figure 3(F)) and synovitis score (Figure 3(G)) were consistent with the above results. Histological analysis of knee articular cartilage post injury with EA treatment. Effect of EA on protecting cartilage, synovium, and IFP damage in KOA rats. (A) tibial cartilage stained with Safranin O/Fast Green of articular cartilage damage surface in each group, scale bar 100 μm. (B) H&E staining (original magnification), synovial hyperplasia, inflammatory cell infiltration, cartilage surface damage. (C) H&E staining was applied to assess histopathological changes in the synovial membrane. The pathological score of synovitis was used to quantitatively assess the severity of synovitis. Synovial inflammation, scale bar 50 μm. (D) Fibrosis of IFP, scale bar 50 μm. (E) OARSI scores in each group. (F) Mankin scores. (G) Synovitis score. The performance images were selected from the inner side of the tibial plateau. Data was presented as the mean ± standard deviation, n = 5 in each group; KOA group vs Control group (*p < .05, ***p < .001), KOA+EA group vs KOA group (#p < .05, ##p < .01).
EA can relieve the inflammatory response in KOA
The inflammatory response is one of the important features in the pathogenesis of KOA and plays a crucial role in the development of KOA.
27
To confirm the EA treatment could alleviate the inflammatory response in KOA, we performed Immunohistochemistry staining of the knee joints. As shown in Figure 4, the IHC analysis showed that in the KOA group, CD68 staining was significantly deepened both in cartilage and synovium, and a significant increase in CD68-positive cells, indicating infiltration of macrophages that may cause an inflammatory response. We detected the pro-inflammatory cytokines (TNF-α) and cartilage degradation biomarkers (MMP-13), Compared with the Control group and the KOA group result showed that both TNF-α and MMP13 were elevated in cartilage and synovium. Similarly, EA inhibited the TNF-α and MMP13 expression. The above results suggest that EA can alleviate the inflammatory response in KOA. Effect of EA on the expression of CD68, TNF-α, and MMP13 in each group of cartilage and synovium. (A–C). Immunohistochemistry staining of CD68, TNF-α, and MMP13. Scale bar = 50 μm, n = 5 in each group. The performance images were selected from the inner side of the tibial plateau. (D–I). Immunohistochemical quantification of CD68, TNF-α, and MMP13 in three groups. All data are presented as the mean ± SD. KOA group vs Control group (**p < .01, ***p < .001, ****p < .0001), KOA+EA group vs KOA group (#p < .05, ##p < .01, ###p < .001, ####p < .0001).
EA treatment can alleviate pyroptosis caused by an inflammatory response
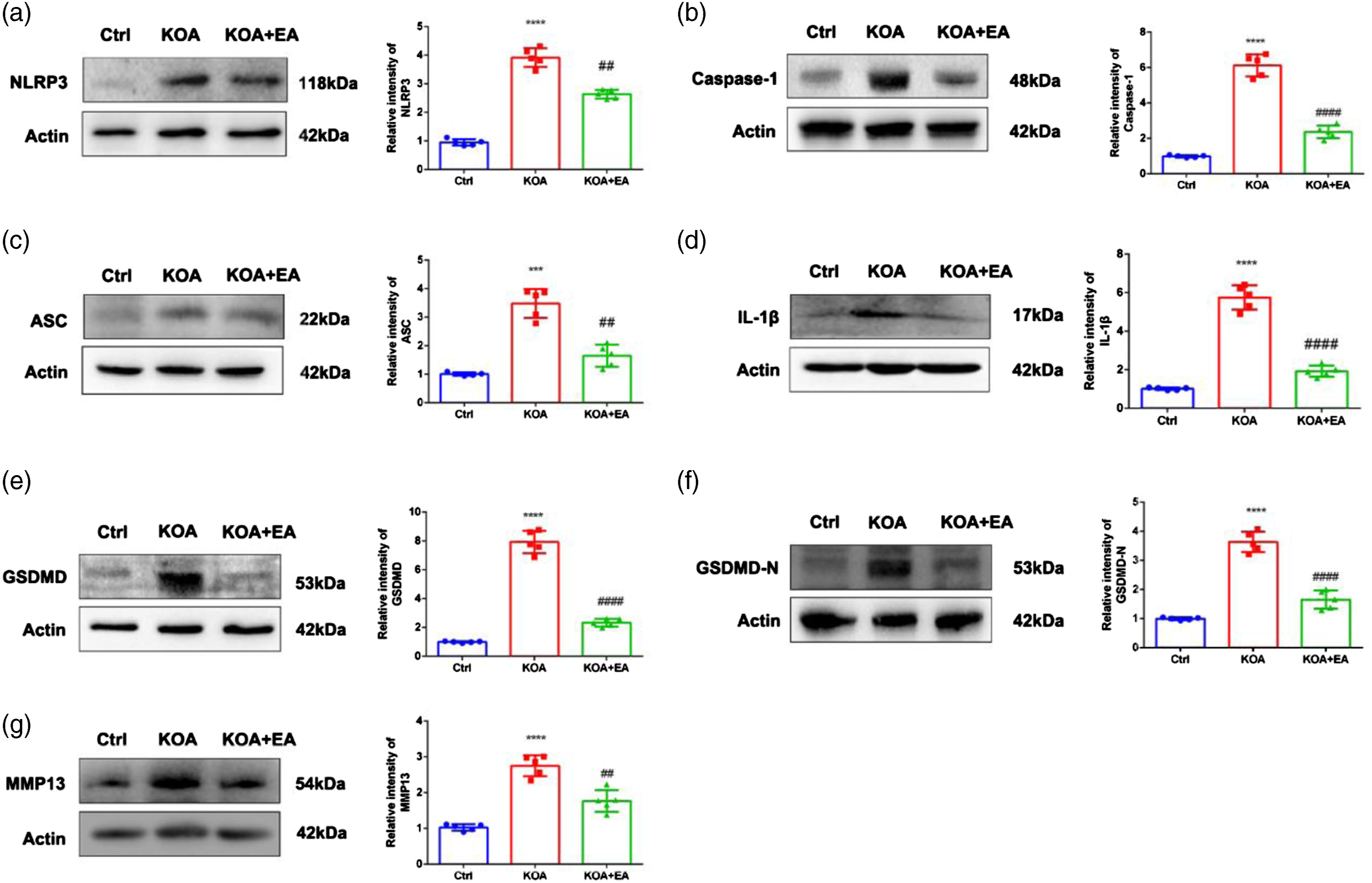
In the previous results, we identified many inflammatory factors that accumulate at the bone joint in the pathogenesis of KOA. Since there is increasing evidence that NLRP3 inflammasome is involved in a large number of inflammatory diseases,28,29 we speculated whether the inflammatory factors that accumulate in the bone tissue can cause pyroptosis and whether EA can alleviate pyroptosis. We can still visualize that in the Immunohistochemistry results (Figure 5), the NLRP3 inflammasome and related proteins (ASC, Caspase-1, IL-1β, and IL-18) in the KOA group showed significantly higher expression of protein-positive cells and heavier staining in the same field of view compared to the control group. Compared with the KOA group, the EA group showed reduced expression of NLRP3 and related protein-positive cells, lighter staining, and occasionally lighter staining of positive cells. The IHC analysis showed that the positive cells in the EA group was reduced (Figure 5(F)-O). Therefore, we extracted cartilage tissues from rats and examined the expression of related proteins in cartilage tissues by western blot. In Figure 6, We found that the relative expression of NLRP3 and ASC proteins was upregulated significantly higher in cartilage tissues of the KOA group, and EA treatment can alleviate the expression of NLRP3 and ASC (Figures 6(A) and (C)). In addition, the relative expressions of Caspase-1 and IL-1β proteins were significantly higher in cartilage tissues of the KOA group compared with the control group, while there was a significant downregulation in the EA group (Figures 6(B) and (D)). And the expression levels of GSDMD and its N-terminal protein showed the same trend (Figures 6(E) and (F)). We also found that the MMP13 was abundantly expressed in cartilage tissue, and its expression could be significantly down-regulated by EA treatment. The above results suggest that EA treatment can inhibit pyroptosis caused by NLRP3 inflammasome response, thereby protecting cartilage tissue and treating KOA and the mechanism may be related to the inhibition of NLRP3 inflammasome-mediated pyroptosis. Immunohistochemistry staining. Effects of EA on expressions of NLRP3, Caspase-1, ASC, IL-1β, and IL-18 of cartilage and synovium in each group. (A–E). Immunohistochemistry staining of NLRP3, Caspase-1, ASC, IL-1β, IL-18. Scale bar = 50 μm, n = 5 in each group. The performance images were selected from the inner side of the tibial plateau. (F–O). Immunohistochemical quantification of NLRP3, Caspase-1, ASC, IL-1β, and IL-18 in three groups. All data are presented as the mean ± SD. KOA group vs Control group (**p < .01, ***p < .001, ****p < .0001), KOA+EA group vs KOA group (#p < .05, ##p < .01). Western blot for protein expression and representative results of NLRP3, caspase-1, ASC, IL-1β, GSDMD, GSDMD-N terminus, and MMP13 in cartilage tissues of each group (n = 5). KOA group vs Control group (***p < .001, ****p < .0001), KOA+EA group vs KOA group (##p < .01, ####p < .0001).
Discussion
The purpose of this study was to reveal the potential therapeutic effect of EA on KOA and its relationship with NLRP3 inflammasome. In the present study, EA reduced mechanical allodynia and the degree of joint swelling in KOA rats, suggesting that EA has an analgesic effect and improves functional mobility impairment. Moreover, the results of the staining of the histologic section further confirmed the conclusion that EA reduced not only cartilage damage but also synovial inflammation and IFP fibrosis. Besides, we found that EA could inhibit the activation of the NLRP3 inflammasome, which was mainly manifested by downregulating caspase-1, ASC, and NLRP3, thus reducing the inflammatory state in KOA. EA also inhibited the expression of pro-inflammatory factors IL-18 and IL-1β, which are downstream substances released after NLRP3 inflammasome activation. These data are consistent with our hypothesis that EA can reduce pyroptosis by modulating NLRP3 inflammasome, and therefore, we suggest that EA has a protective effect on cartilage tissue in KOA.
In this study, we used the Dubi acupoint, the Neixiyan acupoint, and the Yinlingquan and Yanglingquan acupoints, which are two groups of “Complementary Acupoints” in clinical practice. 30 Those pairs of acupoints locate respectively at yin meridians and yang meridians which are opposite locations. The “Complementary Acupoints” reflect the acupuncture theory of “conducting yang from yin, and conducting yin from yang.” 31 The Neixiyan acupoint is the opposite of the Dubi acupoint, in the medial recess of the patellar ligament, and is used for treating knee pain and swelling. Wu’s group found EA (Dubi acupoint and Neixiyan acupoint) delays cartilage degeneration by Modulating Nuclear Factor-κB Signaling Pathway. 32 Yanglingquan is an acupoint of the Gallbladder meridian. It is a healing acupoint and a tendon-meeting acupoint of the eight meeting acupoints. Yinlingquan is the entry acupoint of the Spleen meridian, regulating water and fluid, and promoting water-dampness, treating abdominal distension, edema, and knee pain. Yanglingquan is located on the lateral side of the knee joint and is Yang; Yinlingquan is located on the medial side of the knee joint and is Yin. The two acupoints, one inside and one outside, one water and one earth, are mutually restraining, mutually promoting, and mutually transforming, together playing the role of clearing heat and relieving dampness, soothing tendons, activating channels, and relieving swelling and pain. Finally, we found that stimulating these acupoints by EA can significantly relieve KOA injury, reduce deformation of the knee joint, and achieve the purpose of treating KOA.
KOA is a complex chronic disease in which the inflammatory responses play an important role in the pathogenesis of KOA, 6 which is a metabolic disease of the knee joint and has a major impact on the maintenance of homeostasis in the internal environment of the knee joint. Among the progression of KOA, IL-1β can induce an inflammatory response causing chondrolysis independently, or it can act together with other inflammatory factors to exert a pro-inflammatory effect and is considered a key factor in the development of KOA. 33 Jia’s group found that IL-1β, MMP3, and MMP13 in the KOA groups were slightly increased, 34 also the IL-1β, at both mRNA and protein levels was upregulated in cultured fibroblast-like synoviocytes. 35 TNF-α is mainly produced by macrophages and chondrocytes, and it has been found that an increase in TNF-α can control the degeneration of articular cartilage and thus the development of KOA. 36 Similarly, in our results, we found the macrophages were increased around cartilage tissue, which can lead to an inflammatory response at the onset. Macrophages appeared to mediate structural progression and pain in KOA, 37 which can secrete large amounts of proinflammatory cytokines and mediators, such as TNF-α, interleukin IL-1, IL-6, IL-12, cyclooxygenase-2 (COX-2), and low levels of IL-10. 38
Activation of the inflammasome is the key to the occurrence of pyroptosis,39,40 and the NLRP3 inflammasome is the core factor of the inflammatory response, which is composed of scaffolding protein NLR, bridging protein ASC, pro-caspase-1 and several other proteins as one of the receptors in the intrinsic immune cells. 41 When the inflammasome is assembled, it can cleave pro-IL-18 and pro-IL-1β and induce the release of mature IL-18 and IL-1β. 42 In addition, caspase-1 can cleave GSDMD and produce GSDMD-N and GSDMD-C. The resulting GSDMD-D is the main domain that induces the production of pyroptosis. It had shown that pyroptosis occurred in RSC-364 cells upon LPS treatment which simulated RA in vitro. 43 Besides, Pan’s group had found that gallic acid suppresses ROS generation, thereby limiting NLRP3 inflammasome activation and pyroptosis dependent on Nrf2 signaling. 44 This is consistent with previous reports demonstrating that RA-associated monocytes can release IL-1β through NLRP3-mediated pyroptosis. 45 Same as the model of RA, in our results, the expression of NLRP3, the core molecule of pyroptosis, and similarly, the expression of caspase-1 and GSDMD-N was abnormally increased. It had been reported that EA could influence the expression of IL-1β and NLRP3 inflammasome in the Hippocampus of Alzheimer’s Disease animal model. 46 Besides, Sun’s group found that EA can improve cognitive function in senescence-accelerated P8 (SAMP8) mice via the NLRP3/Caspase-1 pathway. 47 Above all, the expression of pyroptosis-related signaling molecules was significantly alleviated by EA treatment, indicating that EA treatment can treat KOA by inhibiting NLRP3 inflammasome and reducing pyroptosis.
However, there are still some limitations to our study. First, we failed to detect any differences in inflammatory cytokines in the serum of KOA rats. As it is a localized disease, 48 the detection of effusion-synovitis is of more importance. In addition, the lack of an appropriate cellular model of KOA, as well as EA, limits the further investigation of mechanisms of the protective effect of EA in KOA. The lack of a cellular model is a common shortcoming in the research of EA, and Wu raised that using the serum of EA-treated rats can somehow mimic the effect of EA ex vivo 49 However, due to the striking difference between in vivo and in vitro environments, this approach is far from satisfying to fully reveal the mechanism of EA. Although with such limitations, our work indeed provided evidence about the protective effect of EA in treating KOA.
Conclusions
In conclusion, the present study provided evidence of the role of EA in the treatment of KOA and found that EA can inhibit NLRP3 inflammasome and reduce pyroptosis, which results in the protection of cartilage tissue and the treatment of KOA. It provides reliable evidence for the development of EA in the treatment of KOA and the clinical application of acupuncture.
Footnotes
Author contributions
WZ and LLZ performed the experiments. SY undertook the statistical analysis and BW wrote the manuscript. JChang and JChen designed the study. All authors contributed to the article and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research authorship, and/or publication of this article: This work in its entirety was supported by Inflammation and Immune Mediated Diseases Laboratory of Anhui Province (Hefei, IMMDL202006), Foundation of Anhui Medical University(2020xkj209).
Data availability
The datasets generated and analysed during the current study are available from the corresponding authors on reasonable request.
