Abstract
Objective:
Utidelone (UTD1), a recently approved epothilones analog in China for metastatic breast cancer, is endorsed in combination with capecitabine for metastatic breast cancer patients who have encountered first-line therapy failures. Despite its clinically verified therapeutic efficacy, it is concurrently associated with peripheral neuropathic pain, particularly affecting extremities. However, the etiology of UTD1-induced peripheral neuropathic pain remains unclear.
Methods:
The present investigation built a mouse pain model induced by UTD1, resulting in marked mechanical and cold allodynia.
Results:
Examination of the dorsal root ganglia unveiled a notable upregulation of TRPA1, accompanied by noteworthy alterations in oxidative stress-related markers, including ATF4, SOD2, CAT, and Cyt-C. The TRPA1 antagonist HC-030031, resulted in the alleviation of mechanical and cold allodynia in the UTD1-induced pain model, as well as two antioxidants, including Mito-tempo and edaravone.
Interpretation:
The present study will provide new strategies for pain relieving induced by UTD1.
Introduction
According to recent data from the World Health Organization, breast cancer has surpassed lung cancer as the most prevalent cancer globally, with 2.26 million new cases compared to 2.2 million. 1 Despite advancements in treating advanced breast cancer, the prognosis for metastatic breast cancer remains bleak, with a 5-year survival rate of merely 27%. 2 The advent of targeted therapies and immunotherapies has gained prominence in light of the elucidation of breast cancer’s molecular subtypes and the development of specific drugs. Nevertheless, chemotherapy continues to be fundamental in treating metastatic breast cancer. Epothilones, a class of natural microtubule inhibitors, share a mechanism of action with paclitaxel but possess a distinct molecular structure, enabling them to circumvent taxol resistance and enhance patient outcomes. 3 Utidelone (UTD1), an innovative microtubule stabilizing agent and an epothilone B analog, received approval in China in 2021 for use with capecitabine in metastatic breast cancer patients who have not responded to first-line treatments. UTD1 has demonstrated significant antitumor efficacy in breast cancer and is currently undergoing phase III clinical trials.4–6 However, its use is associated with various side effects, notably peripheral neuropathic pain, characterized primarily by heightened sensitivity to mechanical and cold stimuli in the hands and feet. 7 Often, it is the adverse effects, not tumor progression, that necessitate treatment modifications or hospitalizations. Thus, developing clinical strategies to address these side effects is imperative. Yet, the underlying cause of UTD1-induced peripheral neuropathic pain remains elusive.
We have examined the currently understood cellular patho–physiological mechanisms underlying chemotherapy-induced peripheral neuropathic pain (CIPNP) in the peripheral sensory neurons of the dorsal root ganglia (DRG) and the spinal cord. Members of the transient receptor potential (TRP) channel family serve as sensors for temperature and noxious stimuli, playing a crucial role in the genesis of pathological pain.
TRP channels are a vital group of non-selective cation channels situated on cell membranes, each with distinct gating properties. Specific channels, including TRPV1/2/3/4, TRPM2/4/5/8, TRPA1, and TRPC5, are activated by various thermal stimuli and are classified as thermoTRPs. 8 ThermoTRPs participate in the organism’s response to thermal and mechanical stimuli, such as temperature changes and pressure. 9 Furthermore, under specific pathological conditions, thermoTRPs act as nociceptors, contributing to the generation and transmission of pain, thereby facilitating the development of neuropathic pain. 10
TRP ankyrin 1 (TRPA1) is a calcium-permeable cation channel co-expressed with TRP vanilloid 1 (TRPV1) in a specific subset of DRG neurons, playing a crucial role in detecting and transducing noxious stimuli.11,12 Activation of TRPA1 by harmful cold temperatures has been established. 13 Given that TRPA1 responds to oxidative stress 14 and low temperatures, 13 we proposed that it mediates the mechanical and cold hypersensitivity induced by UTD1. In our research, we examined the impact of UTD1 treatment on TRPA1 expression in the DRG and assessed whether a TRPA1 antagonist could mitigate UTD1-induced peripheral thermal neuropathic pain, utilizing a mouse model of UTD1-induced painful peripheral neuropathy.
Our biochemical and pharmacological experiments indicate that TRPA1 plays a significant role in UTD1-induced mechanical and cold allodynia, thereby clarifying the mechanisms of pain-sensory neuropathy linked to UTD1 use. These findings have the potential to inform the prevention and treatment of UTD1-induced peripheral neuropathy pain.
Materials and methods
Experimental animals
Adult ICR mice, weighing between 25 and 30 g, were sourced from Hangzhou Ziyuan Experimental Animal Technology Co., Ltd. (Hangzhou, China). They were accommodated in groups of 4–5 per cage under automatic environmental control, with the temperature maintained at 22 ± 2°C and humidity between 60%–80%. The animals were subjected to a 12-h light-dark cycle (lights on from 06:00 to 18:00) and had ad libitum access to food and water. All mice underwent a quarantine period of 5–7 days upon arrival. Each mouse was utilized in a single experiment only. Each set of experiments consisted essentially of half male and half female mice. The animal study protocol received approval from the Ethics Committee of Shidong Hospital, Affiliated to University of Shanghai for Science and Technology.
Reagents
The TRPA1 antagonist HC-030031 was acquired from Macklin (Shanghai, China). Mito-tempo was sourced from MedChem Express (New Jersey, USA), and edaravone injection was obtained from Simcere (Nanjing, China). The TRPA1 antibody was procured from Alomone (Jerusalem, Israel), while ATF4, SOD2, CAT, Cyt-C, and NF-H antibodies were purchased from proteintech (Wuhan, China). The CGRP antibody was sourced from Abcam (Cambridge, UK), and the GAPDH antibody was acquired from Affinity (OH, USA).
UTD1-induced neuropathic pain models and drugs administration
Following acclimatization and baseline measurements of pain sensitivity, mice were allocated randomly to various treatment groups. In the initial experiment series, mice received intraperitoneal (i.p.) UTD1 injections at doses of 1, 2, and 4 mg/kg within each experimental group, or a control solution (0.9% saline) for 4 days. The phase aimed to identify the effective UTD1 dose for reliably reproducing the pain model and exploring the potential molecular mechanisms of chemotherapy-induced pain. In the subsequent experiment series, a UTD1 dose of 2 mg/kg was selected. Notably, no weight loss occurred post-UTD1 treatment across the groups. In the final experiment series, mice were divided into four groups: three received an i.p. pre-treatment with UTD1 (2 mg/kg) for 4 days. Subsequently, one group received an intragastric (i.g.) dose of HC-030031 (300 mg/kg) on the fifth and twelfth days. Given HC-030031’s solubility challenges, it was initially dissolved in 10% DMSO and then adjusted to 30 mg/mL with 90% corn oil, with the volume set to 10 mL/kg for i.g. administration. The second group received an i.p. Mito-tempo dose (1 mg/kg) starting on the eighth day post-UTD1 administration, continuing daily for 10 days. The third group underwent i.p. administration of edaravone injection (10 mg/kg) immediately on the first day after UTD1 administration (2 mg/kg daily for 4 days), following a protocol akin to clinical patient treatment, with once-daily administration for 14 days. The fourth group received an i.p. administration of the vehicle (0.9% normal saline) as a control.
Behavioral tests
Behavioral assessments were conducted between 9 a.m. and 4 p.m in a quiet, temperature-controlled environment by an evaluator unaware of the mice’s genotypes and treatments.
Mechanical allodynia
Mechanical allodynia was assessed using the “up-and-down” method. 15 The mechanical nociceptive threshold was determined before (baseline) and after drug administration. Mice were placed in plexiglass chambers (10 × 10 × 7 cm) atop an elevated wire mesh platform and allowed to acclimate for 25–30 min. Von Frey fibers (0.008–1.4 g) were applied to the midplantar area, exerting enough pressure to bend the filament for 5 s. Responses such as paw lifting or licking were noted, and the filament weight causing the behavior was recorded. In the absence of a response after 5 s, a heavier filament was used. Each force level was tested 10 times at 5-min intervals, with a response considered positive if observed in at least five instances. The minimum filament weight at which 50% of paw withdrawals occurred was identified as the mechanical pain threshold.
Heat allodynia
Thermal hyperalgesia was evaluated using the tail-flick test. The mouse’s tail was submerged 3 cm from the tip in water maintained at 48°C. Tail-flick latency time was recorded from the onset of heat exposure to tail withdrawal. To prevent tail scalding, the maximum duration for the tail-flick test was set at 15 s. The test was conducted three times per mouse, with a 15-min interval between each session, and the average of these times was used as the latency time.
Cold behavioral test
Cold hyperalgesia was assessed using the acetone stimulation test. Mice were placed in plexiglass chambers (10 × 10 × 7 cm) on an elevated wire mesh platform and acclimated for 25–30 min. A drop (0.05 mL) of acetone was applied to the center of the plantar skin, and the response was observed within 30 s post-application. Responses were categorized into four grades: 0, no response; 1, quick withdrawal, tap, or stomp; 2, prolonged withdrawal or repeated flicking of the paw; 3, repeated flicking of the paw with ventral paw licking. Acetone was applied thrice per paw with at least a 15-min interval between application, and the average score was calculated.
Tissue isolation and extraction
Following intraperitoneal (i.p.) injection of UTD1 to establish the neuropathic pain model, mice were euthanized via cervical dislocation at various time points. The hind paw skin was promptly excised and flash-frozen in liquid nitrogen. Subsequently, after making an incision in the dorsal fur, the sciatic nerves were isolated and immediately frozen in liquid nitrogen. The muscles adjacent to the spine were dissected to expose the lumbar vertebrae, which were then longitudinally sectioned to extract the L2–L5 dorsal root ganglia (small, millet-sized, and translucent) using tweezers. A portion of these tissues was instantly frozen in liquid nitrogen for later quantitative PCR (qPCR) and Western Blot analyses. The remaining samples were fixed in 4% paraformaldehyde and incubated overnight at 4°C, preparing them for subsequent immunofluorescence studies.
RNA isolation and quantitative real-time polymerase chain reaction
Total RNA was extracted using Trizol Reagent (Invitrogen, USA) following the manufacturer’s protocol. One microgram of total RNA was reverse-transcribed to cDNA using the Revert Aid First Strand cDNA synthesis Kit (Thermo Fisher Scientific, USA). Quantitative real-time polymerase chain reaction (qPCR) was conducted with SYBR Green PCR Master Mix (Bimake, USA) on a real-time PCR Detection System (ABI 7500, Life Technology, USA), adhering to the manufacturer’s guidelines. The cycling conditions were set as a 10-min initial denaturation at 95℃, followed by 40 cycles of 95℃ for 15 s (denaturation) and 60℃ for 1 min (annealing and extension). The mRNA expression of the target genes was normalized to GAPDH levels. Primer dimerization or nonspecific amplification was assessed through melt curve analysis. Gene expression differences were quantified using the 2−∆∆CT method, normalized against GAPDH expression. Primer sequences were as follows: GAPDH: 5’-GAAGGTCGGTGTGAACGGAT-3’ (Forward), 5’-AATCTCCACTTTGCCACTGC-3’ (Reverse); TRPA1:5’-TTCTTCGTGTGAAGTGCTGAAT-3’(Forward), 5’-TGGCCTTGTG CTCAGTCAAC-3’ (Reverse); SOD2: 5’-AGACCTGCCTTACGACTATGG-3’ (Forward), 5’-CTCGGTGGCGTTGAGATTGTT-3’ (Reverse); Cyt-C: 5’-AAGACTGGACCAAACCT CCA-3’ (Forward), 5’-TCCAT-CAGGGTATCCTCTCC-3’ (Reverse); CAT: 5’-GGAGGCGG GAACCCAATAG-3’ (Forward), 5’-GTGTGCCATCTCGTC-AGTGAA-3’ (Reverse); ATF4: 5’-CTCTTGACCACGTT-GGATGAC-3’ (Forward), 5’-CAACTTCACTGCCTAGCTC-TAA A-3’ (Reverse).
Protein extraction and Western blot analysis
Proteins from hind paw skin and DRGs (L2–L5) were analyzed using Western blotting. Tissue samples were homogenized in Radio-immunoprecipitation assay (RIPA) lysis buffer (Beyotime Biotechnology, China) with added protease and phosphatase inhibitors (Thermo Fisher Scientific, USA). Protein concentrations were determined using the bicinchoninic acid (BCA) assay (Beyotime Biotechnology, China). Proteins were separated using SDS-PAGE (Epizyme, China) and transferred onto polyvinylidene fluoride (PVDF) membranes (Millipore, Germany). Membranes were blocked with 5% skim milk at room temperature for 1 h, then incubated with primary antibodies (TRPA1, SOD2, Cyt-C, CAT, ATF4, GAPDH) overnight at 4°C. Afterward, membranes were incubated with secondary antibodies at room temperature for 1 h. Protein bands were visualized using ECL Detection Reagent (Epizyme, China) and analyzed with Image J software. The GAPDH bands were used as control for normalization.
Immunofluorescence
Paraffin-embedded sections (15 µm) of DRGs (L2–L5) were incubated overnight at 4°C with primary antibodies: rabbit anti-TRPA1 and mouse anti-CGRP, NF-H in 5% bovine serum albumin-PBST. Following this, sections were treated with fluorescent secondary antibodies and images were captured using a Leica fluorescence microscope.
Statistical analysis
Statistical evaluations were conducted using GraphPad Prism 9. Results were presented as mean ± SEM. The Student’s t-test was applied for two-group comparisons. For multiple groups, after assessing variance homogeneity, one-way ANOVA was utilized. Non-parametric tests were applied to data not adhering to a normal distribution. p values <0.05 were deemed statistically significant.
Results
Establishment of UTD1-induced allodynia model in mice
The initial set of experiments was designed to establish the dosage-dependent effects of UTD1 on inducing peripheral neuropathic pain in mice. Following intraperitoneal (i.p.) administration of UTD1 at doses of 1, 2, and 4 mg/kg, the mice maintained normal health, evidenced by stable body weight and no significant changes in the appearance of hind paw skin. The mechanical pain threshold was assessed using the Von Frey test, which showed a significant reduction in the hind paw’s mechanical threshold in all groups on the third day after administration, with the effect lasting beyond 5 weeks (Supplemental Figure 1). Interestingly, there were no significant differences in the changes in mechanical threshold between the doses (Supplemental Figure 1). In the subsequent experiment, which aimed to consistently replicate the UTD1-induced pain model and investigate the potential molecular mechanisms of chemotherapy-induced pain, a dose of 2 mg/kg UTD1 was selected. The results demonstrated a marked decrease in the mechanical allodynia threshold starting on the fifth day after administration, continuing for more than 5 weeks (Figure 1(b); p < 0.01). The cold allodynia threshold also showed a significant reduction starting on the twelfth day post-administration, lasting for over 2 weeks (Figure 1(c); p < 0.05). However, the heat allodynia threshold did not show a significant change compared to the control group (Figure 1(d); p > 0.05). These results suggest that UTD1-induced peripheral neuropathic pain in mice is characterized predominantly by mechanical and cold allodynia, with no significant effect on heat allodynia, aligning with observations in patient cohorts.

UTD1-induced pain-like behaviors in mice. (a) Mice received a single intraperitoneal (i.p.) dose of UTD1 (2 mg/kg) to assess behavioral changes. A marked decrease in the mechanical allodynia threshold starting on the fifth day after administration, continuing for more than 5 weeks. UTD1 triggered time-dependent mechanical (b) and cold (c) allodynia, but not heat (d) allodynia.
TRPA1 expression increases in DRGs after UTD1 treatment
Considering the established role of TRP channel family members in conditions associated with neuropathic pain, we conducted detailed analyses. DRGs (L2–L5) and hind paw skin samples were collected and processed at specific time points—before administration and at 3, 7, 14, and 28 days post-UTD1 administration. We evaluated mRNA expression changes of crucial TRP channel family members in both DRGs and skin using qPCR and Western blot techniques. The results showed a significant elevation in TRPA1 mRNA expression in DRGs by the 7th day post-UTD1 treatment (Figure 2(a); p < 0.001), whereas TRPV1 and TRPV4 mRNA levels did not change markedly (Figure 2(b); p > 0.05; Figure 2(c); p > 0.05). Consistent with qPCR results, Western blot analysis revealed a significant rise in TRPA1 protein levels in DRGs on the 7th day post-UTD1 treatment (Figure 2(d); p < 0.001). These findings indicate TRPA1’s potential role in UTD1-induced neuropathic pain. In contrast, skin samples displayed a significant reduction in TRPA1 mRNA and protein levels on day 7 post-UTD1 treatment relative to controls (Supplemental Figure 2), suggesting a possible compensatory mechanism in the skin to counteract neuropathic pain induced by UTD1, which represents a protective bodily response, although the specifics remain unclear.

The mRNA levels of ion channels were quantified using qPCR, while their protein levels were assessed by Western blot. A significant elevation in TRPA1 mRNA expression in DRGs by the 7th day post-UTD1 treatment (a), whereas TRPV1 and TRPV4 mRNA levels did not change markedly (b, c). Consistent with qPCR results, Western blot analysis revealed a significant rise in TRPA1 protein levels in DRGs on the 7th day post-UTD1 treatment (d).
UTD1 induces oxidative stress in DRGs and skins
To thoroughly evaluate the changes in oxidative stress-related biomarkers in the DRG and hind paw skin after UTD1 administration, PCR and Western blot analyses were performed. The results demonstrated a marked upregulation of catalase (CAT), superoxide dismutase 2 (SOD2), cytochrome C (Cyt-C), and activating transcription factor 4 (ATF4) mRNA in DRGs on the 7th day post-UTD1 treatment compared to controls (Figure 3(a); p < 0.0001; Figure 3(b); p < 0.001; Figure 3(c); p < 0.01; Figure 3(d); p < 0.01). Western blot findings corroborated the PCR data, as shown in Figure 3(e)–(h). This increase suggests a compensatory boost in antioxidant defenses within the DRG following UTD1 administration, indicative of oxidative stress in this pain model. In the hind paw skin, there was a significant decrease in CAT, SOD2, and ATF4 mRNA levels on day 7 post-UTD1 treatment compared to controls, while Cyt-C mRNA levels remained relatively unchanged (Supplemental Figure 3). In contrast, Cyt-C protein levels were elevated, consistent with the PCR observations (Supplemental Figure 3). With the exception of Cyt-C mRNA in the skin, the other measured parameters exhibited notable fluctuations, with the most significant changes occurring on the 7th day and returning to baseline by the 28th day. This pattern suggests a reduction in oxidative and antioxidant markers in the skin, pointing to impaired oxidative and antioxidant mechanisms and triggering sustained oxidative stress in the skin of mice.

A marked upregulation of CAT, SOD2, Cyt-C, and ATF4 mRNA in DRGs on the 7th day post-UTD1 treatment compared to controls (a–d). Western blot findings corroborated the PCR data, as shown in (e)–(h). Data are presented as mean ± SEM. *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001 compared to control mice (n = 4–6 mice).
Cellular distribution of TRPA1 in DRG neurons after UTD1 treatment
Seven days after UTD1 administration, DRGs were extracted from the lumbar segment (L2–L5) of mice. Immunofluorescence was employed to assess TRPA1’s distribution and expression in DRG neurons. This analysis indicated a significant upregulation of TRPA1 expression in DRG neurons on day 7 post-UTD1 treatment (Figure 4(A) and (B); p < 0.05). For precise localization of TRPA1 in DRG neurons, co-staining with TRPA1 and neuronal markers—NF200 (large-diameter neurons), and CGRP (peptidergic neurons)—was performed on DRG sections. These double immunofluorescence studies showed TRPA1 presence in both large and small-diameter neurons (Figure 4(C)).

Effect of UTD1 treatment on TRPA1 protein expression in mouse DRG neurons. (A) Representative photomicrograph of TRPA1 immunofluorescence on day 7 post-treatment with either vehicle or UTD1 (2 mg/kg, i.p.). DRG (L2–L5) sections were immunostained for TRPA1. (B) Quantitative analysis of relative fluorescence intensity indicates an increase in TRPA1 expression in DRG neurons on day 7 following UTD1 administration. Scale bar = 100 µm. *p < 0.05 compared to the vehicle group. n = 5 per group. (C) Distribution of TRPA1 expression across different neuron sizes. TRPA1 was co-stained with NF200 (a–c), CGRP (d–f), with arrows highlighting co-labeled neurons. Scale = 100 μm.
TRPA1 antagonists alleviated mechanical and cold allodynia induced by UTD1
Our study aimed to evaluate the therapeutic potential of TRPA1 antagonists in alleviating UTD1-induced allodynia, encompassing both mechanical and cold modalities, as confirmed in our mouse model. After administering UTD1 (2 mg/kg) daily for 4 days, mice were treated with the TRPA1 selective antagonist HC-030031 (300 mg/kg) via intragastric (i.g.) route on day 5. HC-030031 demonstrated a time-dependent decrease in the mechanical paw withdrawal threshold, fully reversing mechanical hyperalgesia 2 h post administration (Figure 5(a); p < 0.05). Notably, HC-030031’s suppression of both mechanical and cold hypersensitivity followed a similar time course (Figure 5(b); p < 0.01), although this effect waned 6 h post administration, presumably due to drug metabolism. A second i.g. dose of HC-030031 on day 12 post-UTD1 treatment led to a significant increase in mechanical threshold from 1 h to 6 h post-treatment, completely reversing mechanical hyperalgesia 2 h after administration (Figure 5(a); p < 0.01). A comparable significant rise in the cold threshold was observed, fully reversing cold hyperalgesia 2 h after treatment (Figure 5(b); p < 0.01). Since UTD1 had no marked impact on heat allodynia, HC-030031’s effect on heat allodynia was not investigated in this context. Our findings suggest that TRPA1 is implicated in the onset of UTD1-induced mechanical and cold allodynia.
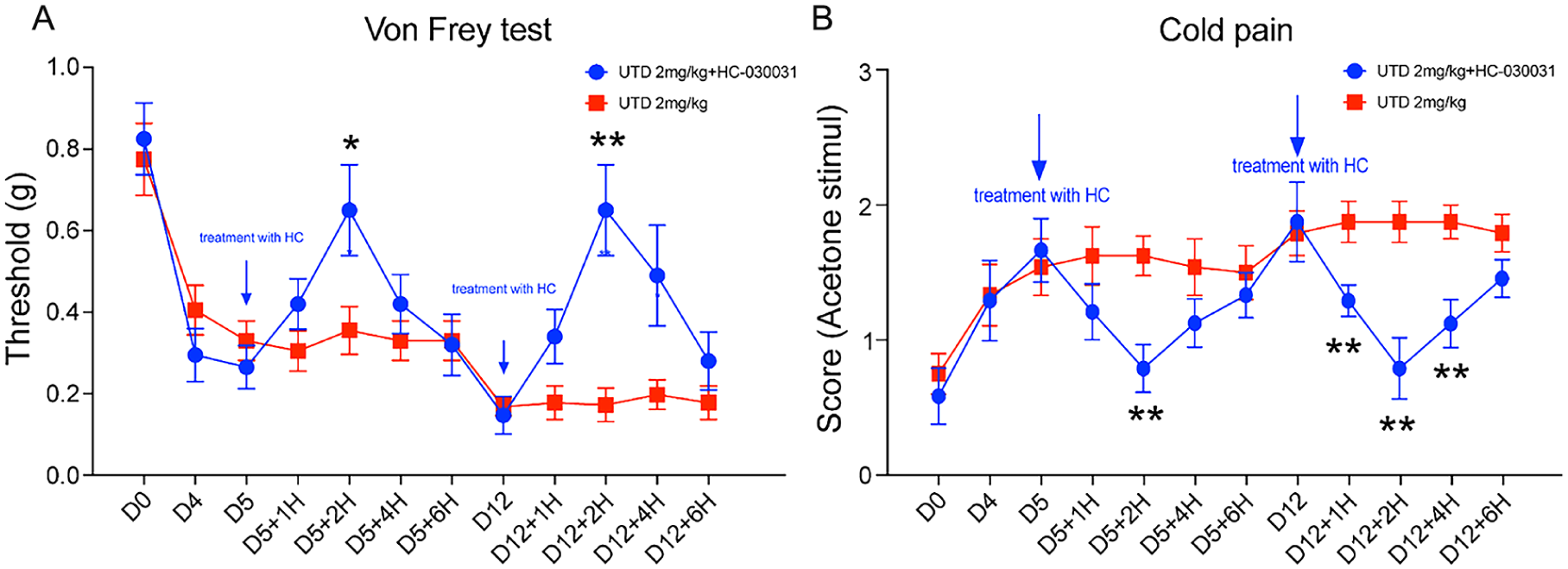
Impact of TRPA1 antagonists on pain-like behaviors after UTD1 treatment. (a) Mechanical allodynia was evaluated using the Von Frey test after intragastric (i.g.) administration of HC-030031 (300 mg/kg). (b) Cold pain response post-HC-030031 treatment was measured using the acetone test.
Mitochondria-targeted antioxidant Mito-tempo attenuated mechanical and cold allodynia induced by UTD1
Given the observed changes in oxidative stress markers in DRG and skin of UTD1-treated mice, we explored the potential analgesic effects of Mito-tempo, a mitochondria-targeted antioxidant. Starting on day 8 post-UTD1 (2 mg/kg) administration over 4 days, one mouse group was treated daily with an intraperitoneal (i.p.) injection of Mito-tempo (1 mg/kg) for 10 days. A significant increase in the mechanical nociceptive threshold from the baseline was observed on day 13 (Figure 6(a); p < 0.01), returning to baseline levels by day 26 post-UTD1 administration (Figure 6(a); p < 0.001). Cold allodynia was similarly mitigated in the Mito-tempo-treated group by day 13 post-UTD1 administration (Figure 6(b); p < 0.05), and fully reversed by day 26 (Figure 6(b); p < 0.05). These results suggest that Mito-tempo, a mitochondria-targeted antioxidant, can ameliorate peripheral neuropathic pain in mice. This study further supports the notion that oxidative stress, alongside TRPA1 activation, plays a significant role in the development of mechanical and cold allodynia induced by UTD1.

Impact of mitochondria-targeted antioxidant Mito-tempo on pain-like behaviors after UTD1 treatment. (a) Mechanical allodynia was evaluated using the Von Frey test after intraperitoneal (i.p.) injection of Mito-tempo (1 mg/kg). Mito-tempo treatment commenced on day 8 post-UTD1 administration and continued for 10 days. (b) Cold pain response following Mito-tempo treatment was assessed using the acetone test.
Edaravone injection as a treatment for UTD1-induced peripheral neuropathic pain in mice
Building on our initial investigations into the roles of TRPA1 and oxidative stress in UTD1-induced peripheral neuropathic pain in mice, we assessed the therapeutic efficacy of edaravone injection. This agent, recognized for its antioxidant properties, was administered intraperitoneally (i.p.) at a dosage of 10 mg/kg immediately on the first day following UTD1 administration (2 mg/kg daily for 4 days). Consistent with protocols used in clinical settings, edaravone was administered daily for 14 days. Our results indicated a gradual reduction in mechanical pain in the edaravone-treated group, with significant pain alleviation noted by the 10th day post-treatment (Figure 7(a); p < 0.05) and complete pain resolution by the 26th day. Cold allodynia also began to diminish in the edaravone group by the 13th day post-UTD1 treatment (Figure 7(b); p < 0.05). These findings underscore edaravone’s potential as a therapeutic agent for managing peripheral neuropathic pain induced by UTD1, offering valuable insights for its clinical application in treating such conditions.

The effects of edaravone treatment on pain-like behaviors after UTD1 administration. (a) Mechanical allodynia after intraperitoneal injection of edaravone (10 mg/kg) was assessed by Von Frey test. (b) Cold pain after edaravone administration was assessed by acetone test.
Discussion
UTD1, an innovative microtubule stabilizing agent, is an analog of epothilone B. The discovery in 1995 16 that epothilone B possesses inhibitory effects on tumor cells marked a significant advancement, revealing a mechanism akin to that of paclitaxel—it inhibits microtubule depolymerization, arresting cell division at the metaphase of mitosis, which leads to apoptosis of tumor cells. 17 Subsequent research indicated that epothilone B surpasses paclitaxel in anti-tumor efficacy, capable of circumventing multi-drug resistance (MDR) through various mechanisms. These include the inhibition of P-glycoprotein-mediated mutations in the paclitaxel binding region, overexpression of multi-drug resistance-related protein (MRP-1), and β-III tubulin overexpression in paclitaxel-resistant breast cancer patients.18–21 Additionally, epothilone can target the actin cytoskeleton and its crucial regulator, Rac1GTPase, 22 which are pivotal in multi-drug resistance.23,24 UTD1, derived from epothilone through genetic engineering, has demonstrated substantial antitumor activity in breast cancer, and it is anticipated to show similar efficacy in other tumors. Consequently, addressing the peripheral neuropathic pain associated with UTD1 treatment is becoming a critical clinical necessity.
Chemotherapy-induced peripheral neuropathic pain (CIPNP) is a significant adverse effect of cytotoxic chemotherapy. Symptoms can vary from acute pain, such as paclitaxel acute pain syndrome (P-APS), to paresthesia, sensory ataxia, and mechanical and cold allodynia, typically initiating in the fingers and toes and extending proximally in a “glove and stocking” pattern.25,26 CIPNP is commonly induced by the administration of taxanes, platinum compounds, and vinca alkaloids, while it is less frequently observed with ixabepilone, thalidomide, and bortezomib.27–29 The mechanisms underlying UTD1-related peripheral neuropathic pain remain elusive due to the relatively recent introduction of UTD1 to the market.
The DRG is a crucial neural tissue for pain perception, serving as the primary neuron for pain signal afferent in the trunk and limbs, and it is instrumental in processing and conveying afferent signals, including those for pain and inflammatory responses. 30 Presently, research on CIPNP, particularly from platinum-based agents, predominantly concentrates on the neurotoxic effects these drugs exert on DRG neurons. This focus is attributed to the peripheral nervous system’s (PNS) lack of an intricate vascular-nerve barrier, facilitating drug diffusion to the DRG. Platinum-based chemotherapies are known to damage DRG neurons by forming adducts with the nuclear and mitochondrial DNA of sensory neurons within the DRG.29,31
TRPA1, a non-selective cation channel activated by various stimuli, is predominantly expressed in the dorsal root ganglion and trigeminal ganglion. These sensory neurons are pivotal in detecting pain stimuli in targeted tissues, transmitting signals, and eliciting pain sensations.11,32 Recently, TRPA1 has been identified as playing a significant role in tumor resistance, with some strategies targeting TRPA1 to curb tumor growth. 33
In our research, we observed that a single UTD1 dose induced rapid and time-dependent mechanical and cold hypersensitivity responses. TRPA1 was implicated in the development of UTD1-induced mechanical and cold allodynia. Pharmacological intervention using the TRPA1 antagonist HC-030031 indicated TRPA1’s involvement in the mechanical and cold hypersensitivity responses triggered by UTD1 in mice. While HC-030031 effectively reversed pain behaviors, its effect was transient, potentially due to its short half-life.34,35 Therefore, TRPA1 is clearly implicated in UTD1-associated pain neurotoxicity.
Based on PCR and Western blot findings, we postulated that UTD1-induced neuropathic pain might stem from the upregulation of TRPA1 protein expression in DRG neurons. Immunofluorescence analysis confirmed an increase in TRPA1 expression in DRG neurons 7 days post-UTD1 administration, indicating that a substantial number of C-fiber neurons started to express TRPA1 following UTD1 exposure. Pain, as a nociceptive response, primarily involves small to medium-sized neurons. To pinpoint TRPA1’s localization in DRG neurons, we performed double labeling with TRPA1 and markers TRPA1 and NF200 and CGRP. The findings revealed TRPA1 expression in both large and small DRG neurons, suggesting its significant role in pain’s onset and progression. Cata et al. 36 demonstrated heightened excitability in spinal cord dorsal horn neurons in rats experiencing paclitaxel-induced neuropathic pain. We theorize that TRPA1’s upregulated expression may correlate with decreased inactivation and increased reactivation rates of calcium ion channels, enhancing neuronal excitability, potentially leading to ectopic discharge and pain manifestation.
In our study, we observed a significant increase in TRPA1 mRNA and protein levels in mouse DRG homogenates on the 7th day post-UTD1 administration, which then rapidly declined. The temporal pattern of mechanical and cold hypersensitivity, with peak effects noted on days 15 and 20, respectively, seems challenging to correlate directly with these transient TRPA1 expression changes, especially as TRPV1 and TRPV4 levels remained unchanged in our experiments. This suggests that additional factors contribute to UTD1-induced mechanical and cold allodynia.
Chemotherapeutic agents are known to generate reactive oxygen species, nitrogen species, and lipid peroxides by disrupting normal mitochondrial energy metabolism, leading to oxidative stress. 37 Reactive free radicals and various endogenous peroxidation products can act as agonists for thermoTRPs. Research has established that oxidative stress-generated free radicals in mammalian DRGs can activate TRP channel signaling pathways, including TRPV1 and TRPA1, thereby inducing pain.38,39 This indicates that chemotherapeutic drugs might activate relevant thermoTRPs through oxidative stress pathways, contributing to the development of CIPNP.40,41
Evidence suggests that oxaliplatin and cisplatin induce oxidative stress.42,43 More critically, antioxidants such as acetyl-L-carnitine, alpha-lipoic acid, and vitamin C have been shown to mitigate oxaliplatin-induced hyperalgesia in rats. 44 Various oxidative stress byproducts have been reported to intensify oxaliplatin’s capacity to induce mechanical45,46 and cold hypersensitivity 47 by targeting TRPA1. There is substantial evidence that reactive oxygen species (ROS) and reactive nitrogen species (RNS) cause peroxidation of plasma membrane phospholipids, influencing TRPA1 activity, which in turn leads to pain and neurogenic inflammation.48–50 Additionally, beyond oxaliplatin, numerous studies have demonstrated that antioxidants like N-acetylcysteine can counteract the increase in intracellular ROS levels triggered by paclitaxel, thereby offering protection against paclitaxel-induced sensory neuropathy.51,52
Dysfunction in mitochondrial energy production and tissue acidosis, under hypoxic conditions, can precipitate neuropathic pain. Hypoxia impairs mitochondrial energy output and leads to tissue acidosis, concurrently elevating ROS53,54 production. This condition also diminishes Na+/K+ ATPase activity, reducing membrane potential, which, in turn, exacerbates mitochondrial energy dysfunction, creating a detrimental feedback loop that intensifies oxidative stress. Drawing from prior research, we investigated Mito-Tempo, a mitochondrial-targeted superoxide scavenger, to assess its effects on neuropathic pain. By the 13th day post-UTD1 administration, there was a significant alleviation of mechanical pain in mice treated with Mito-Tempo compared to the control group, with complete pain resolution by day 26. Similarly, cold allodynia in the Mito-Tempo group began to subside on the 13th day post-UTD1 treatment and was fully resolved by day 26, indicating that Mito-Tempo can mitigate peripheral neuropathic pain in mice. Our results suggest that oxidative stress is a key factor in the mechanical and cold allodynia induced by UTD1, with UTD1-induced oxidative stress in the skin and DRG likely contributing to TRPA1 channel activation and subsequent pain response in mice.
Is there an effective treatment for UTD1-induced peripheral neuropathic pain? Research has indicated that glutathione (GSH) can mitigate paclitaxel-induced neuropathic pain in mice. 55 Consequently, we tested edaravone injection, a clinical treatment known to scavenge oxygen free radicals, in our mouse model. We observed that in the edaravone-treated group, mechanical pain gradually decreased by the 10th day post-treatment and was completely resolved by the 26th day. Similarly, cold allodynia began to improve on the 13th day post-UTD1 treatment, suggesting edaravone’s effectiveness in treating UTD1-induced neuropathic pain, which could inform future clinical management.
In summary, our research established that TRPA1 plays a significant role in UTD1-induced mechanical and cold allodynia. The data indicate that inhibiting TRPA1 is effective in reducing allodynia triggered by UTD1. Additionally, our findings demonstrate that UTD1 treatment leads to oxidative stress. Antioxidative interventions were effective in mitigating pain in our mouse model of UTD1-induced pain. Edaravone, in particular, has shown therapeutic potential against peripheral neuropathic pain attributed to UTD1. While it is known that oxidative stress byproducts can activate TRPA1 in the context of various chemotherapies, the specific relationship between UTD1 and this mechanism remains unexplored. Consequently, further research is essential to elucidate both the upstream (oxidative stress) and potential downstream processes that contribute to UTD1-induced and TRPA1-mediated hypersensitivity.
Supplemental Material
sj-docx-1-mpx-10.1177_17448069251377633 – Supplemental material for Utidelone induces mechanical and cold allodynia in mice via TRPA1 activation
Supplemental material, sj-docx-1-mpx-10.1177_17448069251377633 for Utidelone induces mechanical and cold allodynia in mice via TRPA1 activation by Wenwen Gao, Cunjin Su, Liya Dai, Jialong Tao and Yusong Zhang in Molecular Pain
Supplemental Material
sj-tif-2-mpx-10.1177_17448069251377633 – Supplemental material for Utidelone induces mechanical and cold allodynia in mice via TRPA1 activation
Supplemental material, sj-tif-2-mpx-10.1177_17448069251377633 for Utidelone induces mechanical and cold allodynia in mice via TRPA1 activation by Wenwen Gao, Cunjin Su, Liya Dai, Jialong Tao and Yusong Zhang in Molecular Pain
Supplemental Material
sj-tif-3-mpx-10.1177_17448069251377633 – Supplemental material for Utidelone induces mechanical and cold allodynia in mice via TRPA1 activation
Supplemental material, sj-tif-3-mpx-10.1177_17448069251377633 for Utidelone induces mechanical and cold allodynia in mice via TRPA1 activation by Wenwen Gao, Cunjin Su, Liya Dai, Jialong Tao and Yusong Zhang in Molecular Pain
Supplemental Material
sj-tif-4-mpx-10.1177_17448069251377633 – Supplemental material for Utidelone induces mechanical and cold allodynia in mice via TRPA1 activation
Supplemental material, sj-tif-4-mpx-10.1177_17448069251377633 for Utidelone induces mechanical and cold allodynia in mice via TRPA1 activation by Wenwen Gao, Cunjin Su, Liya Dai, Jialong Tao and Yusong Zhang in Molecular Pain
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grants from the international team of gastrointestinal tumor project funding (SZYJTD201804), the project of State key laboratory of radiation medicine and protection (GZK12023017), Interdisciplinary Basic Frontier Innovation Program of Suzhou Medical College of Soochow University(YXY2304035), Bethune Medical Research Fund (2022-YJ-085-J-Z-ZZ-013)
Supplemental material
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
