Abstract
Background:
Pancreatic neuropathy occurs during the development of pancreatic ductal adenocarcinoma (PDAC), with changes correlating to pancreatic neuropathic pain and increased expression of nociceptive genes in sensory ganglia. Emerging evidence suggests that sphingosine-1-phosphate receptor 1 (S1PR1) plays critical roles in the onset and maintenance of pain. However, whether S1PR1 in sensory ganglia contributes to PDAC-associated neuropathic pain remains unclear.
Methods:
We collected histopathological sections and pain-related data from patients who underwent surgical resection and were pathologically confirmed as having PDAC. S1PR1 levels in intrapancreatic nerves were measured using immunohistochemistry. A mouse model of PDAC-associated pain was established in C57BL/6J mice via orthotopic transplantation of MT5 cells. Pain behaviors were evaluated through abdominal mechanical hyperalgesia, hunch score, and open-field tests. The changes and subcellular localization of S1PR1 in dorsal root ganglia (DRGs) were observed. Subsequently, the S1PR1 antagonists W146 and FTY720 were administered to investigate the underlying molecular mechanisms. We further assessed the analgesic efficacy and its impact on tumor progression of the S1PR1 antagonist FTY720.
Results:
S1PR1 levels in nerves from PDAC patients experiencing cancer-associated pain were significantly higher compared to those without such pain. In the DRGs of a PDAC mouse model, S1PR1 expression was upregulated and colocalized with neurons and satellite glial cells. Intrathecal injection of S1PR1 antagonists W146 and FTY720 effectively alleviated PDAC-induced neuropathic pain hypersensitivity and suppressed the upregulation of transient receptor potential vanilloid 1 (TRPV1) and calcitonin gene-related peptide (CGRP). Additionally, FTY720 alleviated pancreatic cancer-related neuropathic pain and demonstrated partial anti-tumor effects.
Conclusions:
Our findings indicate that S1PR1 in DRGs plays a pivotal role in PDAC-associated neuropathic pain. Inhibition of S1PR1 signaling may alleviate PDAC-related neuropathic pain, and targeting S1PR1 represents a promising strategy for adjuvant management of pancreatic cancer-related pain.
Introduction
Pancreatic ductal adenocarcinoma (PDAC) is the most common pathological phenotype of pancreatic cancer, and characterized by intrapancreatic neuropathy and associated pain.1,2 A longitudinal analysis conducted by Westermann et al. 3 over a 2.5-year period revealed that 93% of surveyed patients reported pain related to pancreatic cancer, with 83% of these individuals experiencing moderate to severe levels of pain intensity. This cancer-associated pain significantly correlates with diminished quality of life, impaired functional capacity, and reduced survival rates among PDAC patients. Current therapeutic approaches are inadequate for achieving sufficient analgesia, leaving a substantial proportion of patients vulnerable to persistent cancer-related pain and its comorbidities. 4 The underlying mechanisms contributing to PDAC-associated pain remain incompletely elucidated, thereby impeding the development of fully effective treatment strategies for pain relief.
The pathophysiology of pain in pancreatic cancer is complex and multifactorial. Current research indicates that pancreatic neuropathy plays an important role in pancreatic cancer pain.1,5,6 Evidence suggests that neuronal damage within the peripheral nervous system occurs during the progression of PDAC. 7 In the tumor microenvironment, aggressive tumor cells invade surrounding nerves, leading to nerve injury and neuroinflammation – a phenomenon referred to as perineural invasion (PNI) or neural invasion (NI).8,9 PNI is observed in approximately 80%–100% of patients and has emerged as one of the most distinctive characteristics of PDAC. 2 Recent studies have established a close relationship between PNI and pain experienced by patients with pancreatic cancer.5,6 The primary afferent neurons located in the dorsal root ganglion are responsible for receiving nociceptive signals and transmitting them to the spinal cord, thereby initiating the pain conduction pathway. 10 Certain mediators within the DRG or injured nerves are recognized to be associated with pain-related behaviors, including transient receptor potential cation channel vanilloid 1 (TRPV1), and calcitonin gene-related peptide (CGRP).11,12 These mediators play a critical role in the onset and progression of pain resulting from nerve injury, facilitating abnormal spontaneous activity in damaged nerve fibers and hypersensitivity among DRG neurons.13,14 Satellite glial cells (SGCs), which uniquely surround sensory neuron cell bodies within sensory ganglia, exhibit significant activation following nerve injury or inflammatory stimuli. 15 Through retrograde labeling of pancreatic nerves and immunostaining, it has been possible to localize the pancreatic afferents in the mouse T9-T12 DRGs. 16 Zhu et al. 17 previously demonstrated that hyperexcitability of DRG neurons contributes to both the onset and persistence of pain related to pancreatic cancer. Furthermore, Stopczynski et al. 7 using a genetically engineered mouse model for spontaneous PDAC, found that peripheral neuronal damage manifests early during disease progression; these changes correlate with reductions in exploratory behavior linked to pain and increased expression levels of nociceptive genes within sensory ganglia. Notably, expressions of nociception-related genes such as TRPV1 and CGRP are elevated in PDAC-afflicted DRGs, indicating an occurrence of peripheral afferent sensitization alongside neurogenic inflammation. 7
Sphingosine-1-phosphate (S1P), a metabolite of sphingomyelin, binds to G protein-coupled S1P receptors (S1PRs) and activates numerous intracellular signaling pathways. 18 Preclinical and clinical studies suggest that sphingolipid metabolism alterations contribute to neuropathic pain development. 19 In the peripheral nervous system, S1PR1 expression has been observed in both whole DRG and cultured sensory neurons. 20 Chi and Nicol 21 demonstrated that S1PR1 plays a significant role in directly enhancing the excitability of small-diameter capsaicin-sensitive sensory neurons. Conditional knockout mice lacking S1PR1 specifically in nociceptive DRG neurons exhibited reduced thermal hypersensitivity and hyperalgesia induced by inflammation following S1P exposure. 22 Xie et al. 23 demonstrated that the knockdown of S1PR1 in rat sensory ganglion cells significantly reduces pain-related behaviors induced by local inflammation within the sensory ganglia. These findings suggest that S1PR1 within the DRG may play a crucial role in pain modulation. Furthermore, emerging evidence indicates that blocking S1PR1 at various targets along the pain pathway can alleviate neuropathic pain, inflammatory pain, cancer-induced bone pain, chronic migraine, and opioid-induced hyperalgesia.19,22,24–27 For instance, intrathecal or systemic administration of competitive functional antagonists of S1PR1 attenuated pain behaviors in models of cancer-induced bone pain. 25 Recently, Pan et al. demonstrated that S1PR1 contributes to central sensitization in chronic migraine model; notably, expression levels of S1PR1 increased within the trigeminocervical complex. Inhibition of S1PR1 signaling could mitigate central sensitization. 26 Visceral afferents within the DRG encode and process both the physical and nociceptive aspects of visceral pain. Whether S1PR1 signal contributes to PDAC associated neuropathic pain remains unknown; thus, this study aims to investigate the role of S1PR1 as well as its potential downstream targets in pancreatic cancer-related pain.
This study initially collected histopathological sections and pain-related information from patients who were surgically resected and pathologically confirmed as PDAC, evaluating the relationship between S1PR1 expression in pancreatic nerves and associated pain symptoms. Then we investigated the alterations in S1PR1 expression and localization within the dorsal root ganglia of a mouse model of pancreatic ductal adenocarcinoma. Subsequently, we assessed whether S1PR1 in the DRG is involved in the induction and maintenance of pain associated with perineural invasion (PNI) induced by pancreatic cancer. Finally, we elucidated the mechanisms through which S1PR1 in the DRG contributes to PNI-induced neuropathic hypersensitivity. These findings position S1PR1 as a potential therapeutic target for addressing neuropathic pain associated with PDAC.
Materials and methods
Human PDAC specimens
All human PDAC specimens were obtained from 42 patients who underwent pancreaticoduodenectomy for pancreatic cancer with a histologically confirmed diagnosis of PDAC from January 2020 to December 2023 at the Affiliated Huaian No.1 People’s Hospital of Nanjing Medical University, and this study was approved by the Institutional Review Board of the hospital (ID: KY-2023-141-01). We analyzed pancreatic tumor specimens in patients with a histologically confirmed diagnosis of PDAC, for whom archived paraffin-embedded tissue blocks were in suitable condition and data on pain were available.
Experimental animals
The immunocompetent male and female C57BL/6J mice (7–8 weeks, weighed 20–25 g) were purchased from the Model Animal Research Center of Nanjing University. Mice were housed in pathogen-free conditions at 22–26°C under an automatic 12 h light–dark cycle and provided with food and water as desired. This study was approved by the Animal Care and Use Committee of Nanjing Jinling Hospital, Affiliated Hospital of Medical School, Nanjing University (ID: DZYGKTZY240003), in accordance with the ethical guidelines of the International Pain Research Association and the National Institutes of Health. Mice were given 1 week to adapt to the experimental environment before the experiment began. All efforts were made to reduce the suffering of mice and minimize the number of animals.
Cell culture
The MT5 cell line from mouse PDAC was obtained by dissociating tumor cells from KPC genetically engineered mice, in which pancreas specific expression of oncogenic Kras and mutant p53 drives development of PDAC. The development and culture method of murine MT5 cell lines was previously described. 28 MT5 KPC-luciferin cell was transfected with enhanced firefly luciferase. MT5-luc cells were maintained in DMEM medium (Invitrogen, Waltham, MA, USA) and cultured at 37°C in a 5% CO2 atmosphere in medium containing 10% fetal bovine serum (Biological Industries, Cromwell, CT, USA), penicillin (100 U/ml), 0.2% puromycin (2 μg/ml).
Mouse model of pancreatic ductal adenocarcinoma associated pain
The mouse pancreatic orthotopic injection model was performed as described previously with a few modifications.29,30 Mice were anesthetized by inhalation with isoflurane, a very small abdominal incision was made, and the pancreas was exteriorized. MT5-luc cells (2 × 104) suspended in 50 μl mixed medium (Matrigel: PBS = 1:1) were inoculated into the pancreas of mice with a sterile insulin needle to build an orthotopic tumor model. The pancreas was then returned to the peritoneal cavity and the abdominal cavity was closed. The same surgical procedure was used for sham operation animals except that mixed medium was injected instead of the MT5-luc cells. Animals were allowed a 7-day period for surgical recovery and tumor manifestation. Seven days after incubation, orthotopic tumor burdens were measured by the In Vivo Imaging System (IVIS). Specifically, D-luciferin (200 mg/kg) was intraperitoneal injected 30 min before imaging by Living Image software. Mice with successfully implanted tumors of similar size for further experiments. Notably, survival was not the endpoint of this study and given that animals in late stages of this model experienced severe pain, all mice were sacrificed at day 18 post-inoculation to maintain reasonable health conditions and minimize suffering.
Drugs and administration
To assess the role of S1PR1 signaling in pancreatic cancer-associated neuropathic pain, the S1PR1 antagonists W146 and FTY720 were employed. Intrathecal administration was utilized as an effective method for targeting structures related to the dorsal root ganglion (DRG). W146 (Cayman Chemical, CAS No. 909725-62-8) and FTY720 (Sigma Aldrich, Catalog No. SML0700) were dissolved in a 0.9% saline solution. Starting on day 12 post-MT5 cell implantation, mice received intrathecal injections of 5 μl W146 (2 nmol/day), 5 μl FTY720 (2 nmol/day), or an equivalent volume of vehicle once daily for three consecutive days. For intrathecal injection, mice were anesthetized and immobilized, and drugs were administered into the L3-L4 intervertebral space using a 30-gauge needle connected to a Hamilton syringe (Hamilton, Reno, Nevada). Accurate placement of the needle within the subarachnoid space was confirmed by observing a brisk tail-flick response upon injection, which was completed within less than 10 s. Post-injection, locomotor functions in treated mice remained normal. To evaluate the clinical applicability of an S1PR1-targeted strategy, systemic administration of the functional antagonist Fingolimod (FTY720) was performed. Treatment commenced on day 7 following tumor implantation when tumors became palpable. Mice were treated with either FTY720 (1 mg/kg diluted in saline) or an equivalent volume of vehicle via intraperitoneal injection once daily until the endpoint of the study.
Behavioral assessment
Pain behaviors were measured in mice prior to surgery (day 0, baseline, BL) and evaluated on days 6, 12, and 18 after MT5-luc cells injected. All the behavioral tests were conducted in a blinded manner and performed during between the hours of 9:00–16:00.
Abdominal hypersensitivity in the model is indicative of secondary hyperalgesia and closely parallels clinical findings in patients with pancreatic carcinoma. Abdominal mechanical hyperalgesia was performed as described previously with some modifications. 30 Graded punctate pressure was applied to the left upper abdomen using von Frey filaments (IITC Inc. Life Science, Woodland Hills, CA, USA), and each filament was tested 10 times with 5 min intervals. Licking of the abdomen, lifting, scratching, moving, or jumping immediately was considered positive response. Response frequency (%) = (positive response/10 tests) × 100.
The hunching score was utilized as a means of evaluating spontaneous visceral pain and was examined as described previously with some modifications.31,32 The mice were placed in a transparent device and their hunching behaviors were observed. The scoring criteria for hunch behavior were as follows: 0-absence of round-back posture, displaying exploratory behavior, and normal hair with a glossy sheen; 1-mild round-back posture, characterized by exploratory behavior and normal hair with a glossy sheen; 2-severe round-back posture, marked by a slight reduction in exploratory behavior, mild piloerection, and current episodes of abdominal muscle contractions; 3-severe round-back posture, marked by significantly reduced exploratory behavior, moderate piloerection, and recurrent episodes of abdominal muscle contractions; 4-severe round-back posture, characterized by minimal or no exploratory behavior, complete piloerection throughout the entire body, and lack of movement in the head region.
The open-field test was used to assess the movement of the animal which is normally influenced by motor output, exploratory drive, sickness, pain or other fear-related behavior. Clinically, a critical comorbidity in patients with pancreatic cancer pain is diminished or lost mobility, leading to functional impairment and reduced quality of life. Briefly, mice were placed in the center of a 40 × 40 cm chamber and locomotor activity was recorded by an overhead webcam connected to a laptop computer, and animals’ movements were automatically tracked for 10 min using ANY-Maze. The total distance traveled and mean speed during the 10-min period were analyzed.
Immunohistochemistry assays in human pancreatic specimens
Immunohistochemistry was conducted on paraffin-embedded sections of human pancreatic cancer tissues. For the immunofluorescence staining, consecutive 4-μm paraffin-embedded sections of human pancreatic cancer tissues were deparaffinized and rehydrated in progressively decreasing concentrations of ethanol. Antigen retrieval was achieved by boiling tissue sections in a 10 mM citrate buffer using a microwave oven for 15 min. The sections were then blocked with PBS containing 2% bovine serum albumin for 2 h. Subsequently, the sections were incubated overnight with antibodies recognizing the pan-neural marker protein gene product 9.5 (PGP9.5, 1:500, Abcam, ab108986) and S1PR1 (1:300, Abcam, ab23386). After three washes lasting 15 min, the sections were incubated with corresponding Alexa-conjugated secondary antibodies (1:500, Servicebio, GB25404; GB28301) for 1 h at room temperature. For the immunohistochemistry portion, consecutive paraffin-embedded human pancreatic cancer tissue sections (4 μm) underwent deparaffinization and rehydration similarly as described above. Antigen retrieval involved boiling tissue sections in a 10 mM citrate buffer in a microwave oven for 15 min. Endogenous peroxidase activity was inhibited using a solution of 3% hydrogen peroxide in methanol for 10 min at room temperature. The sections were subsequently blocked with PBS containing 2% bovine serum albumin for 2 h before being incubated overnight with S1PR1 antibody (1:300; Abcam; ab23386). A secondary anti-rabbit HRP-labeled polymer antibody (1:500; Servicebio; G1302) was applied for 45 min thereafter. Color development was performed by applying DAB-chromogen/H₂O₂ substrate mixture (Servicebio; G1212) to the tissue sections. Analysis and semiquantitative evaluation were performed using an Axioplan-2 microscope (Carl Zeiss, Jena). A semiquantitative analysis of S1PR1 immunoreactivity within intrapancreatic nerves was conducted based on staining intensity recorded as follows: no staining = 0 points; weak staining = 1 point; moderate staining = 2 points; strong staining = 3 points.
Histopathology and immunochemistry of pancreas and DRG in mice
At the end of behavioral experiments, mice were transcardially perfused with PBS and then with 4% PFA. Through retrograde labeling of pancreatic nerves and immunostaining, it has been possible to localize the pancreatic afferents in the mouse T9-T12 DRGs. 16 Referring to previous studies, we chose T9-12 segment DRG for detection.7,16 Tumor-bearing or sham-operated pancreas were dissected, and the T9-12 DRG were removed and postfixed in 4% PFA for 1 day. Pancreatic and DRG tissues were dehydrated with different concentrations of alcohol. After treating with xylene and in xylene-paraffin mixture, the tissues were immersed in paraffin. For Hematoxylin & Eosin (H&E) staining of the mouse pancreas, paraffin sections (5 μm) were re-hydrated, dipped in 1% acid water, stained in hematoxylin for 6 min, washed with water for 3 min, stained in Eosin for 20 s and then dipped in 1% acid water. For the immunofluorescence staining of the mouse pancreas and DRGs, pancreas and DRGs tissues sections were deparaffinized and rehydrated in progressively decreasing concentrations of ethanol. Antigen retrieval was achieved by boiling tissue sections in a 10 mM citrate buffer using a microwave oven for 15 min. The sections were then blocked with PBS containing 2% bovine serum albumin for 2 h. Subsequently, the sections were incubated overnight with antibodies recognizing the S1PR1 (1:300, Abcam, ab23386), cytokeratin 19 (CK19, 1:300, Abcam, ab52625), NeuN (1:500, Abcam, ab177487), GFAP (1:500, Abcam, 207165). After three washes lasting 15 min, the sections were incubated with corresponding Alexa-conjugated secondary antibodies (1:500, Servicebio, GB25404; GB28301) for 1 h at room temperature. Immunohistochemistry signals were visualized using a fluorescence microscope, and digital images were captured with a digital camera system (Nikon, Tokyo, Japan). For the immunohistochemistry portion, consecutive paraffin-embedded mouse pancreatic cancer tissue and DRGs sections (4 μm) underwent deparaffinization and rehydration similarly as described above. Antigen retrieval involved boiling tissue sections in a 10 mM citrate buffer in a microwave oven for 15 min. Endogenous peroxidase activity was inhibited using a solution of 3% hydrogen peroxide in methanol for 10 min at room temperature. The sections were subsequently blocked with PBS containing 2% bovine serum albumin for 2 h before being incubated overnight with S1PR1 antibody (1:300, Abcam; ab23386), TRPV1 antibody (1:200, Abcam; ab203103), CGRP antibody (1:400, CST; 14959), Ki 67 antibody (1:1000, Proteintech; 28074); A secondary HRP-labeled polymer antibody (1:500; Servicebio; G1302) was applied for 45 min thereafter. Color development was performed by applying DAB-chromogen/H₂O₂ substrate mixture (Servicebio; G1212) to the tissue sections. For quantification of cells, four fields (×400) were selected for each section were counted manually using NIH ImageJ software. The average of each sample was used for statistical analysis.
Western blotting assay
Western blotting analysis was conducted to quantify the levels of S1PR1, TRPV1, and CGRP in dorsal root ganglia (DRG). At the end of behavioral testing, animals were deeply anesthetized and subjected to transcardial perfusion with 0.9% saline. Subsequently, bilateral DRGs from T9 to T12 were excised and preserved at −80°C. The DRGs were homogenized in a buffer containing protease and phosphatase inhibitors (Beyotime Biotechnology, Shanghai). Protein concentrations of each homogenate were determined using a BCA assay kit (Beyotime Biotechnology, Shanghai). Equal quantities of cell lysates were separated via SDS-PAGE (Servicebio, Wuhan) and transferred onto PVDF membranes (Thermo Fisher Scientific). To reduce nonspecific binding, 3% bovine serum albumin (BSA) was used to block the membranes for 1 h at room temperature, membranes were incubated overnight at 4°C with primary antibodies: anti-S1PR1 antibody (1:1000; Abcam; ab23386), anti-TRPV1 antibody (1:2000; Abcam; ab203103), anti-CGRP antibody (1:2000; CST; 14959), and anti-GAPDH antibody (1:5000; Proteintech; 10494). Afterward, secondary antibodies were applied for an additional hour. Detection of bound antibodies was performed using the ECL western blotting substrate kit (Bridgen Biotech Inc., Beijing), while ImageJ software (NIH) was utilized for quantifying protein signal densities.
Autopsy
Autopsy was performed at the study end point. Tumor growth was monitored by measuring palpable tumor size with calipers, and volume was computed using the formula V = (W 2 × L)/2, where V represents tumor volume, W stands for tumor width, and L denotes tumor length.
Statistical analysis
Statistical analysis was performed using the GraphPad Prism 9 Software (GraphPad, San Diego, CA). All data are presented as the mean ± standard deviation. The expression levels of S1PR1 in the nerves of pancreas were compared using the Mann–Whitney U test. For behavioral test data, two-way analysis of variance (ANOVA) for repeated measures followed by Tukey test was used to determine statistical significance. Other data were statistically analyzed with two-tailed Student’s t test or a one-way ANOVA. When ANOVA showed a significant difference, pairwise comparisons between means were tested by the post hoc Tukey method. p < 0.05 was considered statistically significant.
Results
Elevated levels of S1PR1 in pancreatic nerves are correlated with pain symptoms in patients with pancreatic ductal adenocarcinoma
Recent studies have demonstrated that the S1P/S1PR1 signaling pathway plays a crucial role in the progression of various tumor diseases, including pancreatic cancer. Furthermore, emerging evidence suggests that S1PR1 is significantly involved in both the onset and maintenance of pain. In this study, we initially utilized human pancreatic cancer tissue samples from 42 patients with histologically confirmed diagnoses of PDAC to investigate the role of S1PR1 in pancreatic cancer-related pain. Among the patients included in our analysis, 14 were pain-free while 28 presented with varying degrees of abdominal or back pain upon admission. Immunofluorescence analyses conducted on human PDAC tissues revealed S1PR1 expression on cancer cells; additionally, we observed differential levels of S1PR1 expression within nerves located in pancreatic tissue (Figure 1(a) shows colocalization between S1PR1 and the neuronal marker protein gene product-9.5 (PGP9.5) in pancreatic cancer specimens). We also performed immunohistochemical studies to assess S1PR1 expression; notably, significant intra-individual variability was observed regarding its expression within nerves. A semiquantitative analysis evaluating immunoreactivity for S1PR1 within intrapancreatic nerves was conducted based on staining intensity (Figure 1(b)). Importantly, immunoreactivity for S1PR1 within nerves from PDAC patients experiencing cancer-associated pain was markedly higher compared to those who did not experience such pain (Figure 1(c); p < 0.05). These findings suggest that peripheral nervous system-associated S1PR1 may be linked to neuropathic pain experienced by individuals with pancreatic cancer.

Immunohistochemical analysis of S1PR1 expression in resected biopsy sections from human PDAC. (a) Immunofluorescence assay for S1PR1 and protein gene product-9.5 (PGP 9.5, a marker for peripheral nerves) in human pancreatic tissue specimens, with representative images illustrating the colocalization of S1PR1 and PGP 9.5 within pancreatic cancer tissues. Scale bar: 50 μm. (b) Assessment of S1PR1 immunoreactivity in nerves associated with pancreatic cancer, categorized as No (a), Low (b), Moderate (c), and Strong (d) immunostaining for S1PR1 in nerve fibers within pancreatic cancer tissues. (c) A semiquantitative analysis comparing the immunoreactivity of S1PR1 between PDAC patients experiencing cancer-associated pain and those who do not experience such pain; Among the patients included in analysis, 14 were pain-free, while 28 presented with varying degrees of abdominal or back pain. Scale bar: 50 μm; * p < 0.05, as determined by Mann-Whitney U test.
Establishment of a model of pain associated with pancreatic ductal adenocarcinoma in mice
To further investigate the role of S1PR1 in neuropathic pain associated with pancreatic cancer, a mouse model of PDAC-related pain was established using C57BL/6J mice through orthotopic transplantation of MT5 KPC-luc cells. Seven days post-implantation, orthotopic tumors were assessed using the In Vivo Imaging System (IVIS). In vivo fluorescence imaging confirmed successful implantation and tumor development from the injected cells within the mouse pancreas (Figure 2(a)). PDAC-related pain was evaluated via abdominal mechanical hyperalgesia tests, hunching scores, and open-field assessments conducted one day prior to surgery (baseline) and on days 6, 12, and 18 following MT5-luc cell injection. Abdominal pain and lower perception threshold in epigastric skin are common symptoms of patients with PDAC. Abdominal hypersensitivity is indicative of secondary hyperalgesia and closely parallels clinical findings in patients with PDAC. 33 Compared with mice in sham operation group, mice receiving MT5-luc cell injections exhibited significantly heightened abdominal mechanical sensitivity on days 12 and 18 post-operation (Figure 2(b), p < 0.05). Referring to Sevcik et al.’s 31 study, mice can be scored according to their hunching backs, which can be a simple and effective evaluation of the occurrence of cancer pain in pancreatic cancer. Compared with mice in sham operation group, spontaneous visceral pain became evident after orthotopic transplantation of MT5-luc cells; these mice displayed increased hunching scores characterized by rounded-back posture on days 12 and 18 post-operation (Figure 2(c), p < 0.05). In keeping with the known decrease in quality of life in pancreatic cancer pain patients, we also assessed locomotor activity during open field testing. The distance of movement and speed of movement were similar in exploring the open field across test days with mice in sham operation group, the distance and speed were significantly and progressively reduced on day 12 and 18 in the MT5-luc cells injected mice (Figure 2(d), p < 0.05). The results showed that the mouse model of pain with PDAC was successfully established.

Establishment of a mouse model of pain associated with pancreatic ductal adenocarcinoma in mice. (a) In vivo fluorescence imaging of tumors 7 days after implanted. (b) Responses to von Frey filaments applying mechanical pressure to the abdomen of mice prior to and following implantation of MT5-luc cells. Left panel shows responses to von Frey filament of strength of 0.02 g and right panel shows responses to filament strength of 0.16 g. (c) The left panel shows the hunching silhouette day 18 after sham operation or MT5-luc cells injected. No signs of pain were present in sham operation mice (hunching silhouette with a score of 0), signs of pain were observed in MT5-luc cells injected mice with a severe rounded-back posture (hunching silhouette with a score of 4); The right panel shows hunching score in sham operation mice and MT5-luc cells injected mice. (d) Open field testing at several days post-surgery to determine the distance of movement (m) and mean speed of movement (cm/s) over a 10 min duration in sham-operated mice or MT5-luc cells injected mice. Left: the distance of movement. Right: the speed of movement. *p < 0.05, two-way ANOVA for repeated measures followed by Tukey test. n = 8 mice/group.
MT5 cells form differentiated pancreatic ductal adenocarcinoma and demonstrate neural invasion within the pancreas, accompanied by an increased expression of S1PR1 in the DRG during the progression of PDAC
Hematoxylin and eosin (HE) staining revealed that the histological morphology of the pancreas in the sham operation group was comparable to that of normal pancreas tissue. In contrast, tumors derived from MT5 cell orthotopic implantation in mice exhibited moderate to well-differentiated ductal adenocarcinoma morphologies (Figure 3(a)). A hallmark feature of PDAC is the neural invasion by cancer cells, which directly contact nerve cells and promote nerve damage associated with PDAC. 34 Cancer cells were identified through immunoreactivity to cytokeratin 19 (CK19), a surface protein expressed by pancreatic cancer cells, while nerves were labeled using immunoreactivity to the peripheral nerve marker PGP-9.5. Consistent with human PDAC pathology, we observed neural invasion within tumor tissue 12 days post-MT5 cell transplantation (Figure 3(b)). Consequently, this neural invasion emerges as one of the primary contributors to PDAC-related neuropathic pain. Pancreatic neuropathy develops alongside PDAC progression, correlating with increased expression of nociceptive genes in sensory ganglia. 7 Whether S1PR1 in the DRG contributes to PDAC associated neuropathic pain is unknown. We explore the expression of S1PR1 in the DRGs (T9-12) after orthotopic transplantation of MT5 cells in mice. Accompany with behavioral changes, the expression of S1PR1 was time-dependently upregulated in the DRGs after MT5 cells injected. Western blotting analysis demonstrated that the level of S1PR1 protein in DRGs was significantly higher in the MT5 cells injected group on days 12 and 18, as compared to the sham operation mice (Figure 3(c), p < 0.05). Moreover, immunohistochemistry for S1PR1 showed the percentage of positive immunoreactivity cells in DRG was significantly higher on day 18 after MT5 cells injected as compared to the sham treated mice (Figure 3(d), p < 0.05).

MT5 cells form differentiated PDAC and show neural invasion in pancreas, and increased expression of S1PR1 in the DRG accompany PDAC progression. (a) Hematoxylin and eosin staining of pancreatic tissue from sham operation mice and MT5 cells orthotopic implantation mice. The left panel shows HE-stained section of sham-operated pancreas (black arrowheads: ducts; black arrow: acini of the pancreas), the right panel shows the morphology of tumors developed from MT5 cells injected mice (arrowhead denotes the duct-like differentiated structures in the tumor). Scale bar: 50 μm. (b) Perineural invasion of cancer cells in the mouse orthotopic MT5 cells of PDAC. PDAC cells were identified via immunoreactivity to Cytokeratin 19, the nerve trunks were labeled via immunoreactivity to peripheral nerve marker PGP 9.5. Invasion of tumor cells in nerves is denoted by arrows. Scale bar: 10 μm. (c) Expression of S1PR1 protein in the T9-12 DRGs on baseline, day 6, 12, and 18 after MT5 cells injection or sham surgery. Two-way ANOVA followed by post hoc Tukey test. p < 0.05 versus sham group at the corresponding time points. n = 6 mice/group. (d) Immunohistochemistry for S1PR1 in the DRGs 18 days after MT5 cells injected or sham operation. p < 0.05 by unpaired Student t test. Scale bars: 50 μm. n = 6 mice/group. Eighteen images per group were acquired and quantified. (e) Double-labeling immunohistochemistry for S1PR1 and NeuN (neuronal markers) or glial fibrillary acidic protein (GFAP; a satellite glial cell marker) in the DRGs 18 day after MT5 cells injected. Scale bars: 50 μm. (f) Size distribution of S1PR1-positive neuronal somata in DRGs 18 days after MT5 cells injected. small: 62%; medium; 24%; Large 14%.
To further assess the distribution and cellular localization of S1PR1 within DRGs, co-staining for S1PR1 was performed alongside NeuN (a neuronal marker) and glial fibrillary acidic protein (GFAP; a satellite glial cell marker) in MT5-injected mice. Double immunofluorescence staining confirmed that both neurons and satellite glial cells within DRGs express S1PR1 (Figure 3(e)). Cross-sectional area analysis of neuronal somata indicated that approximately 62% of S1PR1-labeled neurons were small (<500 μm²), while medium-sized neurons constituted about 24% (500–1000 μm²), and large neurons represented around 14% (>1000 μm²) within the DRGs (Figure3(f)). Collectively, these findings suggest that both the distribution pattern of S1PRl and increases in its protein levels induced by pancreatic injection of MT5 cells implicate its involvement in neuropathic pain associated with PDAC.
Intrathecal administration of S1PR1 antagonist alleviated pancreatic carcinoma induced nociceptive hypersensitivity and spontaneous pain
Intrathecal administration is an effective method for interfering with target sites related to dorsal root ganglion, therefore, intrathecal injection was used for intervention. To ascertain the involvement of S1PR1 in DRGs in the development and maintenance of neuropathic pain behaviors induced by pancreatic cancer, we initially aimed to evaluate whether intrathecal administration of S1PR1 antagonist would yield an analgesic effect in our mouse model of pancreatic cancer. We used the S1PR1 antagonists W146 and FTY720 to investigate the role of S1PR1 in pancreatic cancer pain. W146 is a potent and highly selective competitive S1PR1 antagonist that competitively binds to S1PR1, inhibits its binding with S1P (ligand), and completely blocks downstream signals; while FTY720 is a functional S1PR1 antagonist that, after short-term activation of the receptor, induces its persistent internalization and degradation. 35 To strengthen our research results and enhance rigor, we also conducted a comparative analysis of the analgesic effects of W146 and FTY720. Following the establishment of orthotopic pancreatic cancer in mice on day 12, the male animals received intrathecal injections of either a vehicle or W146 or FTY720 for three consecutive days (Figure 4(a)). Compared to vehicle-treated mice, intrathecal injections of W146 and FTY720 significantly reduced abdominal mechanical hyperalgesia on day 15 following MT5 cell injection (Figure 4(b), p < 0.05) and diminished hunching behavior (Figure 4(c), p < 0.05). Furthermore, W146 and FTY720 treatment markedly improved both the distance and speed of movement in pancreatic cancer-bearing mice on day 15 (Figure 4(e), p < 0.05). Further observation revealed that, compared with the vehicle-treated mice, on day 18 post-MT5 cell transplantation (3 days after cessation of drug administration), no statistically significant difference was observed in the pain behavior data of mice receiving intrathecal W146 injection. In contrast, mice receiving intrathecal FTY720 injection continued to exhibit improved pain behavior. These findings suggest that the analgesic effect of FTY720 persists longer than that of W146 (Figure 4(b), (d), (f), p < 0.05). We further investigated the analgesic effect of FTY720 in female mice with pancreatic ductal adenocarcinoma and found that intrathecal administration of FTY720 significantly improved pain-related behavioral outcomes in female mice with pancreatic cancer following MT5 cell transplantation (Figure 4(g)–(i), p < 0.05). These results indicate that targeting the S1PR1 pathway exerts analgesic effects in both males and females. These behavioral results suggest that targeted inhibition of S1PR1 signaling partially alleviated pain behaviors associated with pancreatic cancer.

Intrathecal administration of S1PR1 antagonists, W146 and FTY720, alleviates PDAC-induced nociceptive hypersensitivity and spontaneous pain. (a) Experimental design to evaluate the antinociceptive effects of W146 and FTY720 in a mouse pancreatic cancer orthotopic transplantation model. On day 12 post-orthotopic tumor establishment, mice received intrathecal injections of either vehicle, W146 (2 nmol/day), or FTY720 (2 nmol/day) for three consecutive days. (b) Von Frey testing to assess cancer-induced mechanical allodynia on day 15 and day 18 post-operation, as measured by withdrawal frequency under 0.02 g stimulus (left panel) or 0.16 g stimulus (right panel) in male mice treated with vehicle, W146 (2 nmol/day, i.th.), or FTY720 (2 nmol/day, i.th.). (c–d) Hunching score on day 15 and day 18 post-operation in male orthotopic pancreatic cancer mice following injection of W146, FTY720, or vehicle. (e–f) Open field testing to measure distance traveled (m) and mean speed (cm/s) over a 10-min period in male mice treated with vehicle, FTY720, or W146 on day 15 and day 18 post-operation. (g) Von Frey testing to assess cancer-induced mechanical allodynia on day 15 post-operation, as measured by withdrawal frequency under 0.02 g stimulus (left panel) or 0.16 g stimulus (right panel) in female mice treated with vehicle or FTY720 (2 nmol/day, i.th.). (h) Hunching score on day 15 post-operation in female orthotopic pancreatic cancer mice following injection of FTY720 or vehicle. (i) Open field testing to measure distance traveled (m) and mean speed (cm/s) over a 10-min period in female mice treated with vehicle or FTY720 on day 15 post-operation. n = 8 mice/group. All data are presented as mean ± SD and were analyzed statistically using two-tailed Student’s t-test or one-way ANOVA. When ANOVA revealed significant differences, pairwise comparisons between means were performed using the post hoc Tukey method. *p < 0.05, compared with mice treated with the corresponding solvent. #p < 0.05, compared with mice treated with W146.
Blocking S1PR1 signaling reduced the expression of TRPV1 and CGRP in the DRGs
In human PDAC, pancreatic neuropathy and perineural invasion are associated with PDAC associated pain, suggesting that pancreatic nerves are damaged and sensitized by aggressive tumor cells. Previous research indicates that TRPV1 and CGRP in DRGs are closely associated with the onset and progression of various types of pain. Moreover, in a mouse model of spontaneous pancreatic cancer, it was noted that during pronounced pain symptoms, nociception-related genes such as TRPV1 and CGRP in the DRG were significantly upregulated. In our experiment, the western blot and immunohistochemistry detection results revealed a significant increase in TRPV1 and CGRP levels in the DRG at 18 days after MT5 cell transplantation, compared to the sham group (Figure 5(a) and (b), p < 0.05). Notably, when compared to vehicle-treated mice, intrathecal administration of the S1PR1 antagonist FTY720 significantly mitigated the elevation of S1PR1 expression in DRG induced by PDAC (Figure 5(c), p < 0.05), however, intrathecal administration of W146 did not modify the expression levels of S1PR1(Figure 5(c)). Importantly, we found that compared to vehicle-treated mice, both intrathecal injection of W146 and FTY720 reduced the expression of TRPV1 and CGRP in the DRG after MT5 cell transplantation. Moreover, the effect of FTY720 was more significant than that of W146 (Figure 5(d), p < 0.05). These results indicate that intrathecal administration of S1PR1 antagonists alleviates pain through the inhibition of S1PR1 signaling, leading to the suppression of TRPV1 and CGRP expression in DRG.
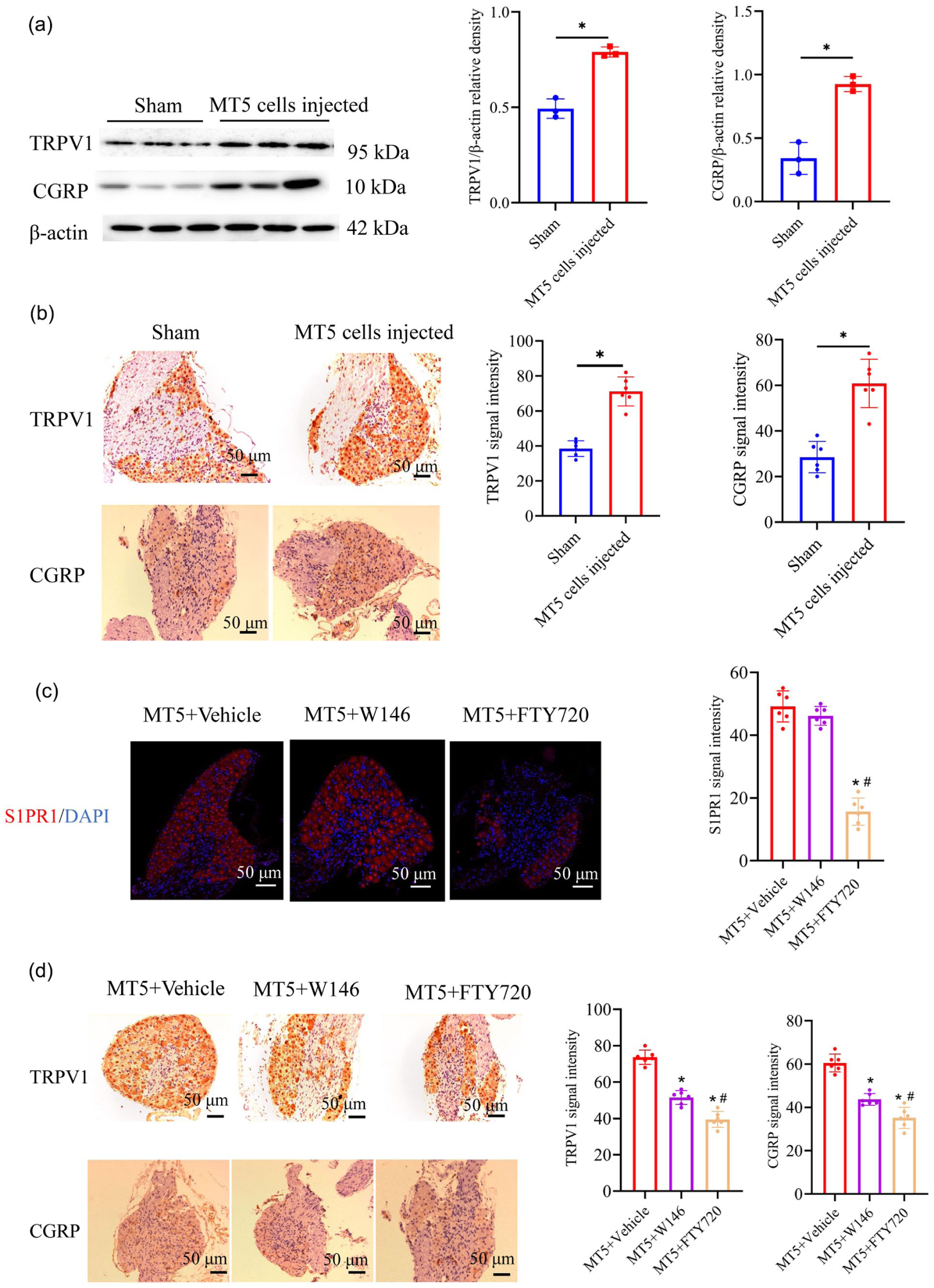
Blocking S1PR1 signaling reduced the expression of TRPV1 and CGRP in the DRGs. (a) The expression alterations of TRPV1 and CGRP proteins in the DRG of mice 18 days after sham operation or pancreatic transplantation of MT5 cells. Representative Western blots (left) and a summary of the densitometric analysis (right) are shown. n = 3 mice/group. (b) Representative TRPV1 and CGRP immunohistochemical staining (left) and quantification of the TRPV1 and CGRP staining (right) in the DRG of mice 18 days after sham operation or pancreatic transplantation of MT5 cells. n = 6 mice/group. Scale bar: 50 μm. (c) Representative S1PR1 immunofluorescence staining (left) and quantification of the S1PR1 staining (right) in the DRG of mice in vehicle, W146 or FTY720 treated mice. n = 6 mice/group. Scale bar: 50 μm. (d) Representative TRPV1 and CGRP immunohistochemical staining (left) and quantification of the TRPV1 and CGRP staining (right) in the DRG of mice in vehicle, W146 or FTY720 treated mice. n = 6 mice/group. Scale bar: 50 μm. All data displayed represent the mean ± SD, data were statistically analyzed with two-tailed Student’s t test or a one-way ANOVA. When ANOVA showed a significant difference, pairwise comparisons between means were tested by the post hoc Tukey method. *p < 0.05, compared with the mice treated with the corresponding solvent. #p < 0.05, compared with the mice treated with W146.
Systemic administration of S1PR1 antagonists alleviated visceral pain induced by PDAC
We then sought to evaluate the clinical applicability of a S1PR1 targeted strategy, FTY720 was administered systemically to PDAC mice. FTY720, a functional S1PR1 antagonist, binds to S1PR1, activating the receptor and inducing endocytosis of the receptor-ligand complex. The internalized S1PR1 is eventually degraded. Through this “activation-internalization-degradation” mechanism, FTY720 chronically downregulates S1PR1 expression on the cell membrane, thereby blocking the S1PR1 signaling axis. The functional S1PR1 antagonist FTY720 has been approved by the US Food and Drug Administration for the treatment of multiple sclerosis. Vehicle or FTY720 (1 mg/kg) was intraperitoneally (i.p.) injected daily to these mice from day 7 to day 18 after tumor implantation. PDAC associated pain was assessed by abdominal mechanical hyperalgesia test, hunching score and open-field test on 1 day before surgery (baseline), 6, 12, and 18 days after injection of the MT5-luc cells (Figure 6(a)). Compared with vehicle-treated mice, FTY720 treatment significantly reduced mechanical allodynia on day 12 and day 18 after MT5 cells injected (Figure 6(b), p < 0.05). FTY720 treatment also significantly reduced hunching score on day 12 and day 18 after MT5 cells injected (Figure 6(c), p < 0.05). Moreover, mice treated with FTY720 exhibited greater overall distance of movement and increased speed of movement on day 12 and day 18 after tumor inoculation (Figure 6(d), p < 0.05). These behavioral results suggest that targeted blocking of S1PR1 signaling with FTY720 can also reduce neuropathic pain associated with pancreatic cancer.

Systemic administration of S1PR1 antagonists alleviated visceral pain induced by PDAC. (a) Experimental design to test the antinociceptive effects of FTY720 in the PDAC mice model on 1 day before surgery (baseline), 6, 12, and 18 days after injection of the MT5 cells. FTY720 (1 mg/kg/day, i.p.) or vehicle (i.p.) was administered once daily from postsurgical days 7 to 18. (b) Von Frey testing to determine cancer-induced mechanical allodynia, as assessed by positive response frequency (0.02 g stimulus, left; 0.16 g stimulus, right) in mice treated with vehicle or FTY720. (c) Assessment of hunching score after MT5 cells inoculation in mice treated with vehicle or FTY720. (d) Open field testing post-inoculation to determine distance traveled (m) and mean speed (cm/s) over a 10 min duration in vehicle or FTY720 treated mice (Left: representative traces. Right: quantification). Data are presented as mean ± SD. n = 8 mice/group. *p < 0.05, two-way ANOVA followed by Tukey test.
Systemic administration of S1PR1 antagonists alleviated the expression of S1PR1 in DRG and pancreatic tumors, and inhibit tumor progression
Similar to the previous research findings that FTY720 can alleviate neuropathic pain induced by chemotherapy and cancer-induced bone pain, we found that FTY720 can also alleviate neuropathic pain in mice with pancreatic cancer. As discussed in the previous studies, S1PR1 signaling appears to play an important role in cancer development, we further explored the FTY720 treatment regimen that alleviated neuropathic pain in pancreatic cancer mice and its impact on tumor progression. Our research further found that, compared with the vehicle-treated mice, continuous intraperitoneal injection of FTY720 not only significantly reduced the expression level of S1PR1 in pancreatic tissue, but also reduced the content of S1PR1 in DRG (Figure 7(a) and (b), p < 0.05). Moreover, FTY720 treatment suppressed tumor progression determined by decreases in primary tumor volumes (Figure 7(c), p < 0.05). Consistent with the effects on tumor size, there was a significant decrease in tumor cell proliferation as determined by Ki67 staining after treatment with the drugs (Figure 7(d), p < 0.05). These results indicate that systemic administration of FTY720 can facilitate pain relief by targeting S1PR1 signaling in the DRGs, while concurrently inhibiting tumor progression. It may be feasible to concurrently target S1PR1 in pancreatic tissue, thereby attenuating tumor progression.

Systemic administration of S1PR1 antagonists alleviated the expression of S1PR1 in DRG and pancreatic tumors, and inhibit tumor progression. Mice were treated with vehicle or FTY720, as described in Figure 6. Mice were sacrificed on day 18. (a) Representative S1PR1 immunofluorescence staining (left) and quantification of the S1PR1 staining (right) in the DRG of mice in vehicle or FTY720 treated mice. n = 6 mice/group. Eighteen images per group were acquired and quantified. Scale bar: 50 μm. (b) Representative S1PR1 immunohistochemical staining (left) and quantification of the S1PR1 staining (right) in the pancreatic tumor of mice in vehicle or FTY720 treated mice. n = 6 mice/group. Scale bar: 100 μm. (c) Representative images of tumors (left) total tumor volumes (right). n = 8 mice/group. (d) Representative Ki67 immunohistochemical staining (left) and quantification of the Ki67 staining (right) in the pancreatic tumor of mice in vehicle or FTY720 treated mice. Scale bar: 100 μm. n = 8 mice/group. *p < 0.05, two-tailed Student’s t-test.
Discussion
The main findings in this study are that: The level of S1PR1 in the pancreatic nerves is associated with the pain experienced by pancreatic cancer patients. During the progression of PDAC, tumor invasion of nerve tissue and result in nerve damage, leading to an increase in the expression of S1PR1 in the DRG. Inhibiting the S1PR1 signaling alleviate pain behaviors by suppressing the expression of TPRV1 and CGRP in the DRG. Our findings suggest that targeting S1PR1 signal may be a promising strategy for alleviating pancreatic cancer neuropathic pain and adjuvant treatment of pancreatic cancer.
Many patients with pancreatic cancer complain of intractable abdominal or back pain. 4 Hyperalgesia in PDAC patients implies alterations of pain transmission in the peripheral and central nervous systems. The administration of conventional analgesics, non-steroidal anti-inflammatory drugs (NSAIDs), and opioids, demonstrates partial effectiveness in the management of pancreatic cancer-related pain. 36 However, they are not always effective, and the severe side effects can significantly impact patients’ quality of life. Perineural invasion (PNI) is a prominent characteristic of pancreatic cancer.6,37 PNI is a process whereby cancer cells invade the surrounding nerves, it is identified in approximately 80%–100% of pancreatic cancer patients. 2 A prominent consequence of PNI in pancreatic cancer is the generation of neuropathic pain.1,2 Consequently, investigating the mechanisms underlying neuropathic pain in pancreatic cancer is crucial for effective pain management.
Sphingosine-1-phosphate (S1P), a metabolic product of cell membrane sphingolipids, is known to interact with five S1P receptors (S1PR1–S1PR5), which respond to extracellular S1P to regulate physiological homeostasis and pathogenic processes across multiple organ systems. 18 Studies have shown that, in somatosensory nerves, S1PR1, S1PR2, and S1PR3 are the major receptors expressed.20,38 Activation of these receptors has been reported to exhibit regulatory effects on sensory nerve functions. In the peripheral nervous system, S1PR1 is predominantly expressed in sensory neurons and nociceptors.20,23 And increasing evidence suggests that S1PR1 plays a crucial role in the initiation and maintenance of pain. Notably, previous studies have established the prominent expression of S1PR1 in pancreatic cancer cells and human pancreatic cancer tissues, and current evidence indicates that the S1P/S1PR1 signaling pathway plays a critical role in the progression of various tumors, including pancreatic cancer.39,40 To investigate the association between S1PR1 and neuropathic pain in pancreatic cancer, we collected histopathological sections and pain-related data from patients with PDAC. Intrapancreatic nerves were labeled using protein gene product 9.5 (PGP9.5), a neuronal marker. Our results indicated that, in addition to pancreatic ductal epithelial cells, prominent S1PR1 expression was also detected in the neural tissue of the pancreas. Notably, immunoreactivity for S1PR1 within nerves from PDAC patients experiencing cancer-associated pain was significantly higher compared to those without such pain. These findings suggest that peripheral nervous system-associated S1PR1 may be linked to neuropathic pain experienced by individuals with pancreatic cancer.
An appropriate animal model is essential for studying the pain mechanisms in pancreatic cancer. Wang et al. 41 transplanted the human pancreatic cancer cell line SW1990 into the pancreas of BALB/c nude mice to investigate cancer-related pain; while Miura et al. 33 injected the Capan-1 cell line into the sciatic nerve of SCID mice to explore neuropathic pain associated with pancreatic cancer. However, these models face challenges due to the absence of an immune component or inadequate simulation of the pancreas’s complex local environment. Several genetically engineered mouse models have been developed to study pain associated with pancreatic cancer; however, their high costs and challenges in large-scale production, coupled with delayed onset and variability in tumor latency, limit their applicability in pain behavioral research. 42 Refer to the method of Selvaraj et al. 30 and our laboratory experience, 28 we established a syngeneic murine pancreatic cancer pain model by inoculating MT5 cells into the pancreatic of C57BL/6 mice, the MT5 cell line from mouse PDAC was obtained by dissociating tumor cells from KPC transgenic mice. PDAC-related pain was evaluated via abdominal mechanical hyperalgesia tests, hunching scores, and open-field assessments. Abdominal pain and lower perception threshold in epigastric skin are common symptoms of patients with PDAC. 33 Abdominal hypersensitivity is indicative of secondary hyperalgesia and closely parallels clinical findings in patients with PDAC. Referring to Sevcik et al.’s 31 study, mice can be scored according to their hunching backs, which can be a simple and effective evaluation of the occurrence of cancer pain in pancreatic cancer. The open-field test was used to assess the movement of the animal which is normally influenced by motor output, exploratory drive, sickness, pain, or other fear-related behavior. Clinically, a critical comorbidity in patients with pancreatic cancer pain is diminished or lost mobility, leading to functional impairment and reduced quality of life. Through these behavioral tests, we found that mice experienced significant behavioral changes in pain perception after MT5 cell transplantation within 12–18 days. Moreover, HE staining revealed that tumors derived from MT5 cell orthotopic implantation in mice exhibited moderate to well-differentiated ductal adenocarcinoma morphologies. We identified tumor cells by their immunoreactivity to the surface protein CK19 (cytokeratin 19) in pancreatic cancer cells, and marked neural tissue by the immunoreactivity to the nerve marker PGP-9.5. We found that, consistent with human PDAC pathology, neural invasion was observed in the tumor tissue 12 days post-MT5 cell transplantation. This neural invasion becomes one of the main inducers of neuropathic pain associated with PDAC.
The DRG is responsible for receiving nociceptive signals and transmitting them to the spinal cord, thereby initiating the pain conduction pathway. Certain mediators within the DRG or injured nerves are recognized to be associated with pain-related behaviors. 10 Through retrograde labeling of pancreatic nerves and immunostaining, it has been possible to localize the pancreatic afferents in the mouse T9-T12 DRGs. 16 The dorsal root ganglions are extremely sensitive to pancreatic disease. Zhu et al. 17 previously demonstrated that hyperexcitability of DRG neurons contributes to both the onset and persistence of pain related to pancreatic cancer. In the tumor microenvironment (TME), the DRG undergoes structural and functional reorganization.7,9 Therefore, we further examined whether neural invasion-induced damage in pancreatic cancer mouse models results in alterations of S1PR1 expression within the DRG. Accompany with behavioral changes, the expression of S1PR1 was time-dependently upregulated in the DRGs after MT5 cells injected. Double immunofluorescence staining confirmed that both neurons and satellite glial cells within DRGs express S1PR1. Further analysis showed that S1PR1 was mainly localized in small and medium diameter neurons. As we know, most nociceptive neurons are small to medium-sized neurons.11,14 This suggests that both myelinated Aδ fiber nociceptive neurons in the DRG and unmyelinated C fiber nociceptive neurons may be involved in the neuropathic pain caused by PDAC. Our findings are consistent with previous research, as Mair et al. 22 have shown S1PR1 is expressed in subpopulations of sensory neurons including nociceptors, and S1PR1 was predominantly in small neurons in DRG sections, and a small amount is expressed in medium diameter neurons. Study from Chi and Nicol 21 demonstrated that sphingosine 1-phosphate (S1P) increased the excitability of small-diameter sensory neurons, and S1PR1 plays a prominent role in mediating the enhancement of excitability produced by S1P. Moreover, S1P induced hypersensitivity was significantly reduced in mice with a conditional nociceptor-specific deletion of the S1PR1. 23 Collectively, these findings suggest that the increased expression of S1PR1 in DRGs induced by PDAC is closely related to the PDAC neuropathic pain.
An increasing body of evidence suggests that S1PR1 signaling is associated with the development and maintenance of nociceptive behaviors related to inflammation, chemotherapy-induced neuropathy, and morphine-induced hyperalgesia.22,24,27 To ascertain the involvement of S1PR1 in DRGs in the development and maintenance of neuropathic pain behaviors induced by pancreatic cancer, we used the S1PR1 antagonists W146 and FTY720 to investigate the role of S1PR1 in pancreatic cancer pain. W146 is a potent and highly selective competitive S1PR1 antagonist that competitively binds to S1PR1, inhibits its binding with S1P (ligand), and completely blocks downstream signals, and the dosage of W146 refers to previous studies 27 ; while FTY720 is a functional S1PR1 antagonist that, after short-term activation of the receptor, induces its persistent internalization and degradation. 35 We found that intrathecal administration of the S1PR1 antagonist W146 and FTY720 significantly reduced abdominal mechanical hyperalgesia, diminished hunching behavior, and improved both distance traveled and movement speed in pancreatic cancer-bearing mice, and the analgesic effect of FTY720 persists longer than that of W146. These behavioral results indicate that targeting the S1PR1 pathway exerts analgesic effects in both males and females. W146 is a highly selective antagonist that competitively binds to the ligand-binding site of S1PR1, thereby blocking the interaction between endogenous S1P and its receptor, and inhibiting the activation of the S1P/S1PR1 signaling pathway. It exhibits a relatively short half-life. In contrast, FTY720 acts as a functional antagonist of S1PR1 by initially activating the receptor and subsequently inducing its persistent internalization and degradation, leading to a more sustained effect. Our observations indicate that intrathecal administration of W146 alleviates pain without significantly altering S1PR1 expression, whereas FTY720 markedly suppresses S1PR1 expression. Three days after discontinuation of treatment, the analgesic effects of W146 were no longer detectable, while FTY720 continued to exhibit partial analgesic activity. TRPV1 is a non-specific cation channel that is not only activated by heat but also by vanilloid agonists like capsaicin, and various lipids.13,43 The activation of TRPV1 ion channels results in opening of the channel and subsequent membrane depolarization of nociceptive neurons and the release of pain-related neurotransmitters such as calcitonin gene-related peptide (CGRP) and substance P. Previous research indicates that TRPV1 and CGRP in DRGs are closely associated with the onset and progression of various types of pain.11,13 Stopczynski et al. 7 using a genetically engineered mouse model of spontaneous PDAC, found that neuronal damage was correlate with reduced exploratory behavior related to pain and increased levels of nociceptive genes (TRPV1 and CGRP) in DRGs, leading to the stimulation of nociceptive nerve fibers. In our experiment, the immunohistochemistry and western blot detection results revealed a significant increase in TRPV1 and CGRP levels in the DRG at 18 days after MT5 cell transplantation. Intrathecal injections of S1PR1 antagonist, W146, and FTY720 both significantly reduced the increase in TRPV1 and CGRP induced by PDAC. It has been shown that S1PR1 activation in DRG neurons enhances activity of TRPV1 via a Gαi-phosphoinositide-3-kinase (PI3K)-protein kinase C (PKC)-p38 signaling cascade. Langeslag et al. 44 reported that S1P-induced heat sensitization is strongly attenuated in mice lacking TRPV1 channels. Moreover, pharmacological and genetic evidence indicate that the S1P/S1PR1 axis is also a critical contributor to DRG and nociceptor hyperexcitability. Conditional knockout mice lacking S1PR1 specifically in nociceptive DRG neurons exhibited reduced thermal hypersensitivity and hyperalgesia induced by inflammation following S1P exposure. 22 Xie et al. 23 demonstrated that the knockdown of sphingosine-1-phosphate receptor S1PR1 in rat sensory ganglion cells significantly reduces pain-related behaviors induced by local inflammation within the sensory ganglia. These results indicate that intrathecal administration of S1PR1 antagonists alleviates pain through the inhibition of S1PR1 signaling, leading to the suppression of TRPV1 and CGRP expression in DRG.
The first S1PR-targeted drug, FTY720/fingolimod, has been approved for the treatment of multiple sclerosis since 2010. FTY720 is phosphorylated in the body to a S1P mimic that binds to S1PRs, except S1PR2. 45 It is now established that FTY720-P acts as a S1PR1 functional antagonist. FTY270-P leads to sustained depletion of S1PR1 from the cell surface by inducing its irreversible proteasomal degradation.35,45 Recent studies have shown that FTY720 could block neuropathic pain states of diverse etiologies.46,47 Intrathecal or systemic administration of FTY720 in mice with cancer-induced bone pain reduced pain behaviors and increased the levels of the anti-inflammatory cytokine IL-10 in the lumbar spinal cord. The dosage of FTY720 refers to previous studies. 25 Similarly, we also found that continuous intraperitoneal administration of FTY720 significantly alleviates neuropathic pain behaviors in pancreatic cancer mouse models and inhibits the expression of S1PR1 in DRGs. Furthermore, Available evidence strongly suggests that S1PR1 plays a pivotal role in tumor development, affecting different tumorigenesis stages and modulating the significant pathways involved. 39 Lankadasari et al. reported that treatment of murine and human pancreatic cancer cell lines with FT720 resulted in decreased cell proliferation, increased apoptosis, and cell cycle arrest. Importantly FTY720 targets S1PR1 abrogates desmoplasia and chemosensitizes pancreatic cancer to gemcitabine in mouse pancreatic cancer model. 40 Similar to this studies, continuous intraperitoneal administration of FTY720 significantly reduces tumor volume and suppresses indicators of tumor proliferation. More recently, Singh et al. demonstrated that in naive and breast tumor-bearing immunocompetent mice, a clinically relevant dose of FTY720/Fingolimod targeting S1PR1 effectively alleviated neuropathic pain induced by paclitaxel. Furthermore, the administration of FTY720 significantly enhanced the anti-tumor effects of paclitaxel, resulting in reduced tumor progression and lung metastasis. 46 These results suggest that targeting the S1PR1 signal may be used for the treatment of cancer pain.
In conclusion, our findings suggest that S1PR1 in the dorsal root ganglia plays a crucial role in neuropathic pain associated with pancreatic ductal adenocarcinoma. Inhibition of S1PR1 signaling may alleviate neuropathic pain associated with pancreatic cancer, and targeting S1PR1 represents a promising strategy for adjuvant management of pancreatic cancer-related pain.
Footnotes
Author contributions
S-Q Cai, Y-X Zhang, C Wang: Writing-original draft preparation, Methodology; Y Gao, T-Y Wang, F-N Xu: Formal analysis and investigation, Writing-original draft preparation, Conceptualization; Q-Z Liu, J Yin, Z-J Zhang: Writing-review and editing, Methodology; S Zhang, Y Huang: Writing-review and editing, G-J Tao, M-L Duan: review and editing, supervision, resources, Supervision. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Data availability statement
The data that support the findings of this study are available from the corresponding authors, upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Nature Scientific Fund of China (No. 82102309) and Nanjing Medical University Science and Technology Development Foundation (NMU20230143).
Ethical approval
This study was approved by the Animal Care and Use Committee of Nanjing Jinling Hospital, Affiliated Hospital of Medical School, Nanjing University (ID: DZYGKTZY240003), in accordance with the ethical guidelines of the International Pain Research Association and the National Institutes of Health. All human PDAC specimens were obtained from patients who underwent pancreaticoduodenectomy for pancreatic cancer at the Affiliated Huaian No.1 People’s Hospital of Nanjing Medical University, and was approved by the Institutional Review Board of the hospital (ID: KY-2023-141-01).
