Abstract
Patients with preoperative pathological conditions such as anxiety, depression, and sleep disorders experience more severe postoperative pain, suggesting that preoperative pathological changes in patients may affect postoperative pain. However, the potential pathophysiological changes associated with postoperative pain remain unknown. Here, this study initially employed clinical research to investigate potential pathophysiological changes related to postoperative pain. Subsequently, animal behavioral experiments and mechanistic explorations were conducted accordingly. Pregnant women undergoing cesarean sections who could provide preoperative cerebrospinal fluid were selected as subjects. Preoperative cerebrospinal fluid proteomics, postoperative pain intensity, and neutrophil-to-lymphocyte ratio (NLR) were analyzed. Rats were used to model the corresponding preoperative pathological state. Mechanical pain thresholds were measured after plantar incision and spinal cords were harvested for analysis. Clinical studies showed that one-quarter of the proteins positively correlated with postoperative pain were related to reactive oxygen species (ROS). Furthermore, the NLR-Ratio, reflecting postoperative inflammation level, increased with the severity of postoperative pain. Establishing a preoperative ROS-increased model with oxidant t-BOOH enhanced postoperative acute mechanical hyperalgesia and spinal neuroinflammation in rats. Conversely, preoperative administration of antioxidant VE, reducing ROS, alleviated postoperative hyperalgesia and spinal neuroinflammation. galectin-3 inhibitors mitigated postoperative hyperalgesia and neuroinflammation in the preoperative ROS-increased model. Additionally, The effects of galectin-3 on pain sensitization and pro-inflammation in vitro were mediated by the TLR4 receptor. Thus, this study demonstrated that preoperative ROS exacerbated postoperative hyperalgesia via galectin-3-mediated neuroinflammation, suggesting that galectin-3 may be a potential therapeutic target for alleviating postoperative pain in clinical patients.
Introduction
Acute postoperative pain is an unpleasant sensation and emotional experience following surgery, typically manifested as hyperalgesia. 1 More than 40% of patients experience moderate to severe postoperative pain and acute postoperative pain is linked to delayed recovery, chronic pain, and analgesic abuse. 2 Opioids, the main perioperative analgesics, can cause respiratory depression and nausea/vomiting in patients.3,4 Furthermore, high doses of opioids can lead to drug tolerance and aggravate pain sensitization. 5 Therefore, it is crucial to explore therapeutic drugs targeting the pathological mechanisms of postoperative acute pain.
Numerous clinical studies have shown that the preoperative pathological condition of patients is related to acute postoperative pain. Patients who have preoperative pain, sleep disorders, anxiety, or depression are likely to experience more severe postoperative pain,6–8 suggesting that preoperative pathophysiological changes might influence postoperative pain. However, most research on the mechanisms of acute postoperative pain focuses solely on the pain resulting from surgical trauma, overlooking the impact of preoperative factors. Therefore, we initially conducted clinical research to investigate potential preoperative pathophysiological changes related to postoperative pain. Since the spinal cord is a critical hub for pain transmission, 9 we selected pregnant women undergoing cesarean sections, from whom preoperative cerebrospinal fluid could be collected, as our study subjects. NLR which reflects the acute inflammation level 10 and postoperative pain intensity were measured. Subsequently, based on protein changes in preoperative cerebrospinal fluid and the severity of postoperative pain, we inferred preoperative pathological changes related to pain and conducted animal experiments for behavioral verification and mechanistic exploration.
ROS are a group of unstable small molecular substances, such as H2O2 and OH-.11,12 Normal levels of ROS in the body can degrade mismatched or oxidized proteins and maintain homeostasis by regulating the synthesis, secretion, and assembly of the extracellular matrix. 13 However, excessive accumulation of ROS, due to their unstable molecular structure, can attack normal macromolecules in the body, such as proteins, lipids, and nucleic acids, promoting oxidative stress and contributing to various neurological diseases, including pain.14,15 Excessive ROS can contribute to the development of pain by exacerbating neuroinflammation, specifically by promoting spinal microglia activation and inducing the release of TNF-α and IL-1β.16,17 Conversely, reducing ROS can alleviate neuroinflammation, thereby relieving inflammatory pain and osteoarthritis pain.18,19 Our clinical study found that one-quarter of the cerebrospinal fluid proteins positively correlated with postoperative pain were related to ROS. Women who experienced more severe postoperative pain had higher NLR-Ratio, suggesting patients with more postoperative pain had higher levels of inflammation. When promoting the activity of NOX4, an enzyme that generates ROS, it increases the expression of galectin-3. 20 Galectin-3 is a lactose-specific lectin that can activate microglial TLR4 or TREM2 receptors, promoting the release of pro-inflammatory factors by microglia,21–24 indicating its role in regulating inflammation. Therefore, we hypothesized that preoperative ROS may affect postoperative neuroinflammation and hyperalgesia. Moreover, preoperative ROS exacerbated postoperative hyperalgesia via galectin-3-mediated neuroinflammation.
In the present study, based on clinical research results, we used animal experiments to demonstrate that increasing preoperative ROS exacerbated postoperative hyperalgesia and neuroinflammation, while reducing preoperative ROS mitigated these conditions, indicating that preoperative ROS can influence postoperative hyperalgesia and neuroinflammation. In addition, inhibiting galectin-3 may be a therapeutic strategy for preventing preoperative ROS-induced aggravation of postoperative pain by suppressing neuroinflammation.
Materials and methods
Participants and ethics
This clinical study included women who voluntarily participated and underwent elective cesarean sections with combined spinal-epidural anesthesia between December 2021 and May 2022. Exclusion criteria included chronic pain disorders, long-term use of anti-inflammatory or analgesic drugs, coagulation disorders, and central nervous system disorders. A total of 40 women met the inclusion criteria. The study was approved by the Medical Ethics Committee of the Second Affiliated Hospital of Chongqing Medical University (ethics approval number: 2021-123).
Clinical sample collection
Upon admission, a 3 mL peripheral blood sample was collected. The patient was positioned in the lateral decubitus position, and combined spinal-epidural anesthesia was performed at the L2–L3 interspace. After standard surgical disinfection of the puncture site, 3 mL of 2% lidocaine was used for local anesthesia. Following successful dura mater and lumbar puncture, 2 mL of cerebrospinal fluid was collected. Then, 1.5 mL of 0.75% bupivacaine was mixed with the cerebrospinal fluid to a total volume of 2.5 mL and administered into the subarachnoid space. After withdrawing the lumbar puncture needle, an epidural catheter was placed through the epidural needle and advanced 3–5 cm cephalad. After securing and dressing the puncture site and epidural catheter, the patient was positioned supine, and 3 mL of 2% lidocaine was administered epidurally, with the level of anesthesia assessed. Peripheral blood samples (3 mL) were collected 60–90 min post-cesarean section.
Cerebrospinal fluid samples were used for proteomic analysis. Peripheral blood samples were analyzed for complete blood counts, including neutrophils and lymphocytes, to calculate the NLR. The NLR-Ratio, representing the ratio of postoperative to preoperative NLR, was used to reflect the acute inflammatory state post-operation. 10
Clinical outcome analysis
The Dynamic Numeric Rating Scale (NRS) was used to assess pain during 0–6, 6–12, 12–24, and 24–48 h postoperatively, where 0 indicates no pain and 10 indicates unbearable pain. A line graph was plotted with time points on the x-axis and NRS scores on the y-axis. The area under the curve (AUC) was calculated between the endpoints of the graph and the x-axis. AUC-moving pain NRS and maximum pain NRS were calculated based on NRS assessments at four postoperative time points. Participants were categorized into severe pain (maximum pain NRS score ≥6) and non-severe pain (maximum pain NRS score <6) groups based on their highest reported pain scores.
Experimental animals
Adult female and male Sprague-Dawley rats (200–250 g) were obtained from Beijing Vital River Laboratory Animal Technology. The rats were maintained on a 12-hour light-dark cycle with ad libitum access to food and water. Experimental protocols were approved by the Animal Care and Use Committee of the Second Affiliated Hospital of Chongqing Medical University (ethics approval number: IACUC-SAHCQMU-2024-0002).
Drug preparation
For intrathecal injection (i.t.), t-BOOH (a ROS donor, 50 μg/kg, Sigma-Aldrich, 458139), GB1107 (a galectin-3 inhibitor, 50 μM, MCE, HY-114409), and Gal-3 (rat recombinant galectin-3, 4 μg/kg, Cloud-Clone Corp, APA303Ra01) were administered via catheter. Vitamin E (an antioxidant, 100 mg/kg, Sigma-Aldrich, 258024) was administered orally via a gavage needle.
Intrathecal catheterization
The intrathecal catheter was implanted under anesthesia as previously described. 25 Briefly, a PE-10 catheter was placed in the lumbar enlargement. Rats with a positive lidocaine test and no paralysis or infection after a 7-day recovery period were included in subsequent intrathecal drug administration experiments.
ROS measurement
After 5 days of intrathecal t-BOOH injections and 6 days of Vitamin E gavage, the lumbar enlargements (L3–L5 segments) of female rats were harvested. The lumbar enlargements were embedded in O.C.T. (Tissue-Tek) and rapidly frozen in liquid nitrogen. Tissue sections (15 µm) were incubated with the DHE probe (10 μM, MedChemExpress, HY-D0079) at 37°C in the dark for 30 minutes. After incubation, sections were washed three times with PBS, mounted with an anti-fade solution containing DAPI, and the fluorescence intensity in the dorsal horn of the spinal cord was immediately observed using confocal microscopy (Olympus SpinSR). DHE fluorescence intensity was used as a measure of ROS levels. At least 2 slices per rat and 3–4 rats per group were used for the quantification of immunostaining.
Plantar incisional pain model
Rats were anesthetized with isoflurane (2%–3%), and a longitudinal incision was made on the plantar surface of the right hindpaw. The underlying plantaris muscle was elevated and incised longitudinally, and the skin was closed with two sutures.
Assessment of mechanical hyperalgesia
Mechanical pain threshold, represented as the 50% paw withdrawal threshold (PWT), was assessed using von Frey filaments. Rats were placed on a metal mesh floor in individual transparent compartments for at least a 30-min habituation period before testing. The von Frey filament was pressed vertically against the mid-plantar aspect of the incised hind paw. The 50% PWT was determined using the up-down method. 26 A line graph was plotted with time points on the x-axis and 50% PWT on the y-axis. The area enclosed by the endpoints of the postoperative time points and the line graph reflected the average postoperative pain intensity in rats.
Immunofluorescence
Female rats were perfused with PBS and 4% paraformaldehyde. Spinal cord (L3–L5) segments were taken and treated with 20% and 30% sucrose for dehydration after overnight fixation with 4% paraformaldehyde. A sliding microtome was used to cut the spinal cord transversely into 15-μm-thick sections. After washing with PBS, frozen sections were permeabilized with 0.1% Triton X-100 and blocked with 10% goat serum and 1% BSA at room temperature for 2 h. Sections were then incubated with primary antibodies against Iba-1 (a microglial marker, 1:400, Oasis Biofarm, OB-PGP049-01) or galectin-3 (1:60, Abcam, ab76245) at 4°C. After overnight incubation and washing with PBS, the slices were incubated with secondary antibodies for 2 hours at room temperature and mounted with DAPI-containing anti-fade agent and glass slides. Fluorescence signals from the dorsal horn of the spinal cord were acquired using a confocal laser scanning microscope (Olympus SpinSR). At least 2 slices per rat and 3–4 rats per group were used for quantifying fluorescence signals.
Western blotting
Spinal cord proteins from female rats were lysed and extracted with a mixture of RIPA buffer and protease inhibitor, and protein concentration was determined using the BCA method. After denaturation with SDS-PAGE Sample Loading Buffer, the proteins were separated by electrophoresis on a 12% precast gel (ACE, ET15012) and then transferred to a PVDF membrane. Membranes were blocked with 5% non-fat milk for 2 h, then incubated with primary antibodies against β-actin (1:20,000, R&D Systems, MAB8929), TNF-α (1:1,000, Abcam, ab205587), IL-1β (1:1,000, Abcam, ab254360), and galectin-3 (1:5,000, Abcam, ab76245) at 4°C overnight. After washing with TBST, membranes were incubated with HRP-coupled secondary antibody for 90 min at room temperature. After incubation and washing with TBST, protein bands were visualized using ECL luminescent solution, and the gray values of the bands were analyzed using ImageJ.
BV2 cell treatment and ELISA
BV2 cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM) with 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin at 37°C and 5% CO2. After 1 h of starvation treatment with serum-free culture medium and TAK-242 (1 μM, MedChemExpress, HY-11109) pretreatment for 1 h, Gal-3 (mouse recombinant Galectin-3, 3 μg/mL, Abcam, ab224635) stimulation was performed for 24 h. After stimulation, cellular morphology was observed under an optical microscope, and supernatants were centrifuged at 2,000 × g for 10 min at 4°C. TNF-α and IL-1β concentrations in the supernatants were determined using ELISA kits (ExCell Bio, Suzhou, China) following the manufacturer’s instructions.
Statistical analysis
Clinical data were presented as mean ± SD, and basic research data as mean ± SEM. Comparisons between two groups were analyzed using t-tests or non-parametric Mann-Whitney U tests, and comparisons among three or more groups were performed using one-way ANOVA followed by Dunnett’s post hoc test. Pearson correlation analyses were used to explore correlations. For animal behavior data, 2-way ANOVA followed by the Sidak or Holm–Sidak multiple comparison tests was used. P values less than 0.05 were considered statistically significant.
Results
Post-cesarean section pain in pregnant women correlated positively with the NLR-Ratio
This study initially included 52 pregnant women undergoing elective cesarean sections. However, 12 participants were excluded, leaving 40 women whose preoperative cerebrospinal fluid was collected and postoperative pain was assessed using the NRS during 0–6, 6–12, 12–24, and 24–48 hours after surgery. NLR analysis was conducted for 38 participants (Figure 1(a) and (b)). Based on the NRS scores at these four time periods, we calculated the Area Under the Curve for moving pain (AUC-moving pain NRS) score to reflect average postoperative pain intensity and the maximum NRS score to represent peak pain intensity. Results showed that most participants had AUC-moving pain NRS scores ranging from 10 to 25, with an average of 15.6 and a standard deviation of 4.6 (Figure 1(c)). Maximum pain NRS scores ranged between 2–6 and 6–10, with an average of 5.5 and a standard deviation of 1.8 (Figure 1(c)). Both AUC-moving pain NRS scores and maximum pain NRS scores were positively correlated with the NLR-Ratio (Figure 1(d)). Participants were categorized into severe pain (NRS score ≥6) and non-severe pain (NRS score <6) groups based on maximum pain NRS scores. No significant differences were observed in demographic characteristics or other confounding factors between the two groups (Figure 1(e)). However, the severe pain group exhibited significantly higher AUC-moving pain NRS scores and maximum NRS scores compared to the non-severe pain group (Figure 1(f)). Additionally, the NLR Ratio was significantly higher in the severe pain group (Figure 1(f)). These findings suggest that higher postoperative pain intensity is associated with a higher NLR-Ratio, indicating that women with more severe postoperative pain may experience higher levels of inflammation.

A positive correlation between post-cesarean section pain and NLR ratio in pregnant women. (a) and (b) Flowchart and inclusion/exclusion criteria of the clinical study. (c) Analysis of postoperative AUC-moving pain NRS and maximum pain NRS based on NRS scores in post-cesarean pregnant women. The distribution of AUC and maximum pain scores were depicted by frequency diagrams. (d) Correlation analysis between AUC-moving pain NRS or maximum pain NRS and NLR-Ratio. Pregnant women were categorized into the severe pain group (maximum pain NRS score ≥6) and the non-severe pain group (maximum pain NRS score <6). (e) and (f) Demographic factors and other confounding variables affecting postoperative pain were statistically analyzed, and a comparison of AUC-moving pain NRS or maximum pain NRS and NLR-Ratio was conducted between the two groups.
Correlation analysis of preoperative cerebrospinal fluid proteins and postoperative pain intensity
We conducted proteomic analysis on preoperative cerebrospinal fluid samples from pregnant women to examine protein correlations with postoperative AUC-moving pain NRS scores and maximum pain NRS scores (Figure 2(a)). Thirty-six proteins positively correlated with both postoperative AUC-moving pain NRS scores and maximum pain NRS scores were selected for Gene Ontology (GO) functional enrichment analysis (Figure 2(b)). This analysis revealed that six categories were related to ROS (Figure 2(c)). Among these 36 proteins, nine were specifically enriched in functions related to ROS, accounting for 25% of the proteins positively correlated with postoperative pain intensity (Figure 2(d)). This suggests that preoperative ROS may influence the intensity of postoperative acute pain.
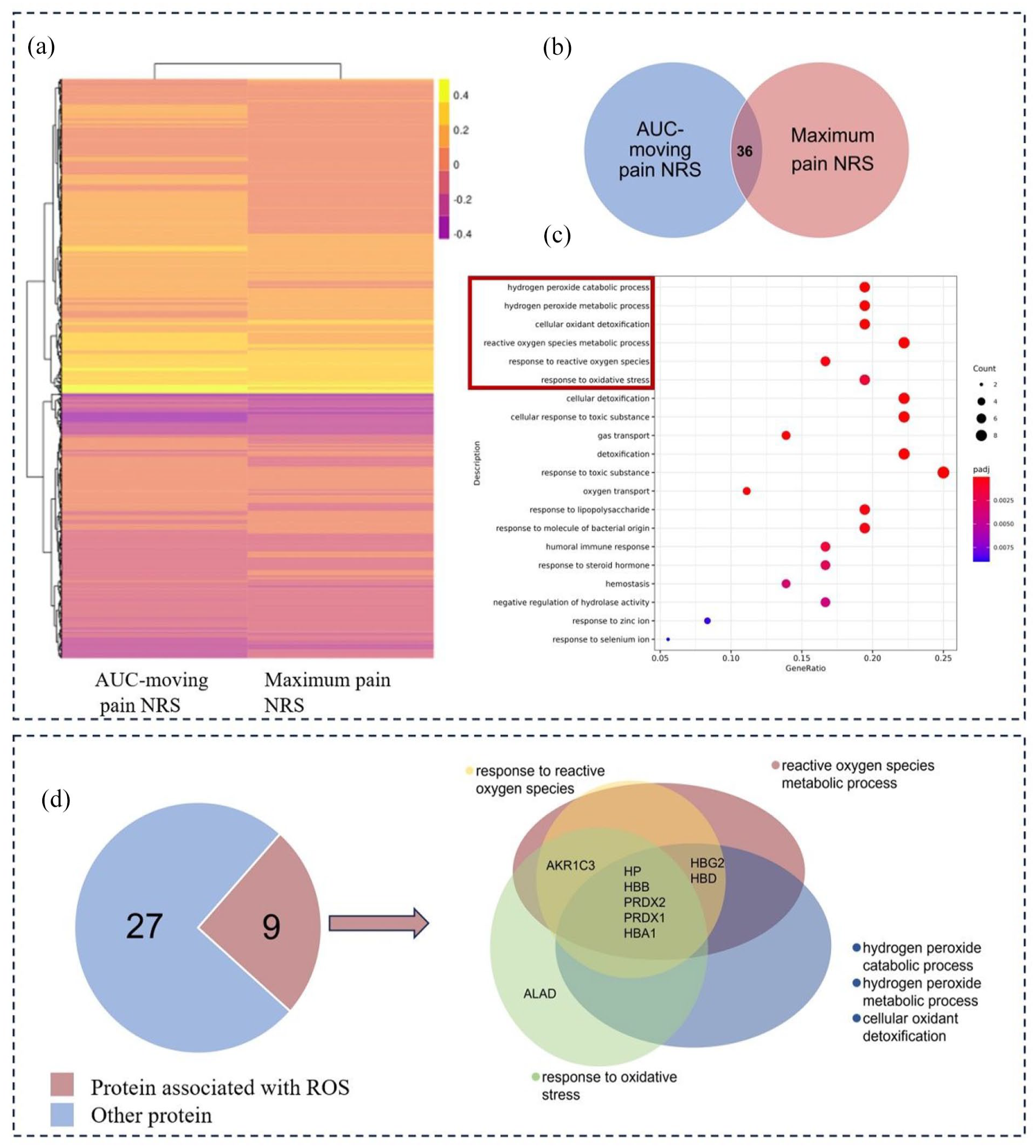
1/4 of proteins positively correlate with postoperative pain severity were linked to ROS. (a) Correlation analysis between AUC-moving pain NRS score or maximum pain NRS score and proteins in preoperative cerebrospinal fluid. (b) and (c) GO functional analysis was conducted on 36 proteins positively correlated with AUC-moving pain NRS and maximum pain NRS scores. (d) Analysis of the number and percentage of proteins enriched in relation to ROS.
Postoperative hyperalgesia was influenced by preoperative ROS levels
To investigate the impact of preoperative ROS on postoperative hyperalgesia, we established models with increased and decreased ROS using the oxidant t-BOOH and the antioxidant VE, respectively, and measured mechanical pain thresholds after plantar incision (Figure 3(a)). DHE, a commonly used fluorescent probe for detecting superoxide radicals, undergoes dehydrogenation in the presence of intracellular superoxide radicals, producing ethidium, 27 which binds to RNA or DNA to generate red fluorescence. Higher levels of intracellular superoxide radicals result in more ethidium and stronger red fluorescence, while lower levels produce weaker fluorescence. Superoxide serves as a precursor to other ROS, 28 making DHE suitable for detecting ROS levels. Intrathecal injection of t-BOOH increased preoperative spinal cord ROS levels (Figure 3(b) and (c)). Following plantar incision, increased ROS levels resulted in reduced mechanical pain thresholds at 6, 12, and 24 h, as well as 2 and 3 days postoperatively in female rats, and decreased the AUC of postoperative mechanical pain thresholds (Figure 3(f) and (g)). Conversely, reducing preoperative ROS levels with VE (Figure 3(d) and (e)) increased mechanical pain thresholds at 6 h to 2 days and improved the AUC of postoperative pain thresholds (Figure 3(h) and (i)). These behavioral results indicate that preoperative ROS levels affect postoperative mechanical pain thresholds, with increased levels exacerbating postoperative hyperalgesia and decreased levels alleviating it. Similar results were observed in male rats (Fig. S1 in Supplemental Material), suggesting that the effect of preoperative ROS on postoperative hyperalgesia is consistent across genders.

Preoperative ROS influenced postoperative hyperalgesia in rats. (a) Flowchart of ROS levels detection and mechanical pain threshold measurement. Following treatment with oxidant t-BOOH or antioxidant VE, rat spinal cords were sectioned and stained with DHE probe to measure ROS levels through fluorescence intensity. (b) and (d) Representative fluorescence images from DHE probe staining in each experimental group. (c) and (e) Statistical analysis of DHE fluorescence intensity in each group (n = 4–5). (f) and (g) After the establishment of an incision model following pre-treatment with t-BOOH, 50% PWT at various postoperative time points were measured and the AUC of 50% PWT from 6 h to 3 d postoperatively was analyzed (n = 8–9). (h) and (i) 50% PWT at various postoperative time points were measured following pre-treatment with VE, and the AUC of 50% PWT from 6 h to 3 d postoperatively was analyzed (n = 10–12). Data were presented as mean ± SEM; *P < 0.05, **P < 0.01, ***P < 0.001 versus respective control group.
Impact of preoperative ROS on postoperative spinal neuroinflammation
Neuroinflammation is a key mechanism underlying hyperalgesia, characterized by the proliferation or activation of glial cells and increased expression of pro-inflammatory cytokines.9,29 To explore the impact of preoperative ROS on postoperative spinal neuroinflammation, we harvested the lumbar enlargement of the spinal cord from female rats 24 h after surgery. We used immunofluorescence to assess Iba1 fluorescence intensity, a marker for microglial cells, in the dorsal horn of the spinal cord, and Western blot to evaluate the expressions of TNF-α, precursor IL-1β (pro-IL-1β), and mature IL-1β (Figure 4(a)). Compared to the saline group, the t-BOOH group exhibited significant microglia activation (Figure 4(b) and (c)) and increased levels of TNF-α, pro-IL-1β, and IL-1β (Figure 4(f)). Conversely, reducing preoperative ROS levels with VE led to decreased microglia activation (Figure 4(d) and (e)) and lower expressions of TNF-α, pro-IL-1β, and IL-1β post-surgery (Figure 4(g)). These findings indicate that changes in preoperative ROS levels can affect postoperative spinal neuroinflammation, suggesting that preoperative ROS may influence postoperative hyperalgesia through neuroinflammation.

Preoperative ROS influenced postoperative spinal neuroinflammation. (a) Flowchart of modeling and tissue sampling. Spinal cord segments L3-L5 were taken 24 h postoperatively from models with increased or decreased preoperative ROS. The immunofluorescence intensity of Iba-1, a marker of microglia and expressions of inflammatory factors were analyzed. Representative images of Iba-1 fluorescence, scale bar = 50 μm, and statistical analysis of fluorescence intensity after increasing preoperative ROS ((b) and (c), n = 3) or after decreasing ROS ((d) and (e), n = 3–4). Western blot analysis was used to assess postoperative expression of inflammatory factors following changes in preoperative ROS levels. Expressions of TNF-α, pro-IL-1β, and IL-1β after increasing preoperative ROS ((f) n = 5–6) or decreasing preoperative ROS ((g) n = 4–5). Data were presented as mean ± SEM; *P < 0.05, versus respective control group.
Preoperative ROS exacerbated postoperative hyperalgesia via galectin-3
To investigate how ROS affects postoperative pain thresholds, we first identified a positive correlation between galectin-3 levels and AUC-moving pain NRS scores in clinical data (Figure 5(a)). The correlation between galectin-3 and maximum pain NRS scores approached significance with a p-value of 0.054 (Figure 5(a)). Galectin-3 expression was significantly higher in the postoperative severe pain group compared to the non-severe pain group (Figure 5(b)), suggesting that galectin-3 may be a potential nociceptive target. To test this, we initially validated in animal experiments that increased ROS promoted galectin-3 expression (Figure 5(c)). Western blot analysis confirmed that increased ROS enhanced galectin-3 expression (Figure 5(d)), and immunofluorescence analysis revealed elevated galectin-3 levels in the dorsal horn of the spinal cord under ROS stimulation (Figure 5(e) and (f)). Behavioral experiments in a female rat model with increased preoperative ROS, were followed by intrathecal injection of the galectin-3 inhibitor GB1107 (Figure 5(g)). The results showed that GB1107 significantly alleviated postoperative hyperalgesia, and this analgesic effect was not due to direct inhibition of incisional pain (Figure 5(h)). These molecular and behavioral experiments suggest that galectin-3 is involved in the development of postoperative hyperalgesia in the preoperative ROS-increased model. Similar findings were observed in male rats (Fig. S2 in Supplemental Material), indicating that the analgesic effects of GB1107 are consistent across genders.

Preoperative ROS exacerbated postoperative hyperalgesia via galectin-3. (a) Positive correlation between galectin-3 and AUC-moving pain NRS scores or maximum pain NRS scores. (b) Galectin-3 expression was higher in the severe pain group compared to the non-severe pain group (n = 16–23). (c) Spinal cord of the preoperative ROS-increased model was harvested at 24 h postoperatively to assess changes in galectin-3 expression. Changes in galectin-3 expression were detected using Western blot ((d) n = 5–6) and immunofluorescence, scale bar = 50 μm ((e) and (f) n = 3). 50% PWT were tested after intrathecal injection of galectin-3 inhibitor gb1107 ((g) and (h) n = 6–9). Data were shown as mean ± SEM; in molecular experiments, *P < 0.05, **P < 0.01 versus Saline group; in behavioral data, *P < 0.05, #P < 0.05 versus Saline+PINC, &P < 0.05 versus t-BOOH+ 0.5%DMSO.
Galectin-3 inhibitor alleviated postoperative neuroinflammation in the preoperative ROS-increased model
To explore the mechanism of galectin-3-induced hyperalgesia, we administered GB1107 intrathecally for 5 consecutive days following t-BOOH injection and plantar incision. During this period, mechanical pain thresholds in female rats were measured, and spinal cord lumbar enlargement was harvested on the 5th day (Figure 6(a)). Continuous administration of GB1107 significantly alleviated mechanical hyperalgesia in rats at 1, 3, and 5 days postoperatively (Figure 6(b)) and increased the AUC of postoperative mechanical pain thresholds (Figure 6(c)). With the reduction of hyperalgesia, the activation of microglia (Figure 6(d) and (e)) and the expressions of TNF-α, pro-IL-1β, and IL-1β (Figure 6(f)) were significantly decreased under the influence of the galectin-3 inhibitor, indicating that postoperative neuroinflammation in the preoperative ROS-increased model may be exacerbated by galectin-3.
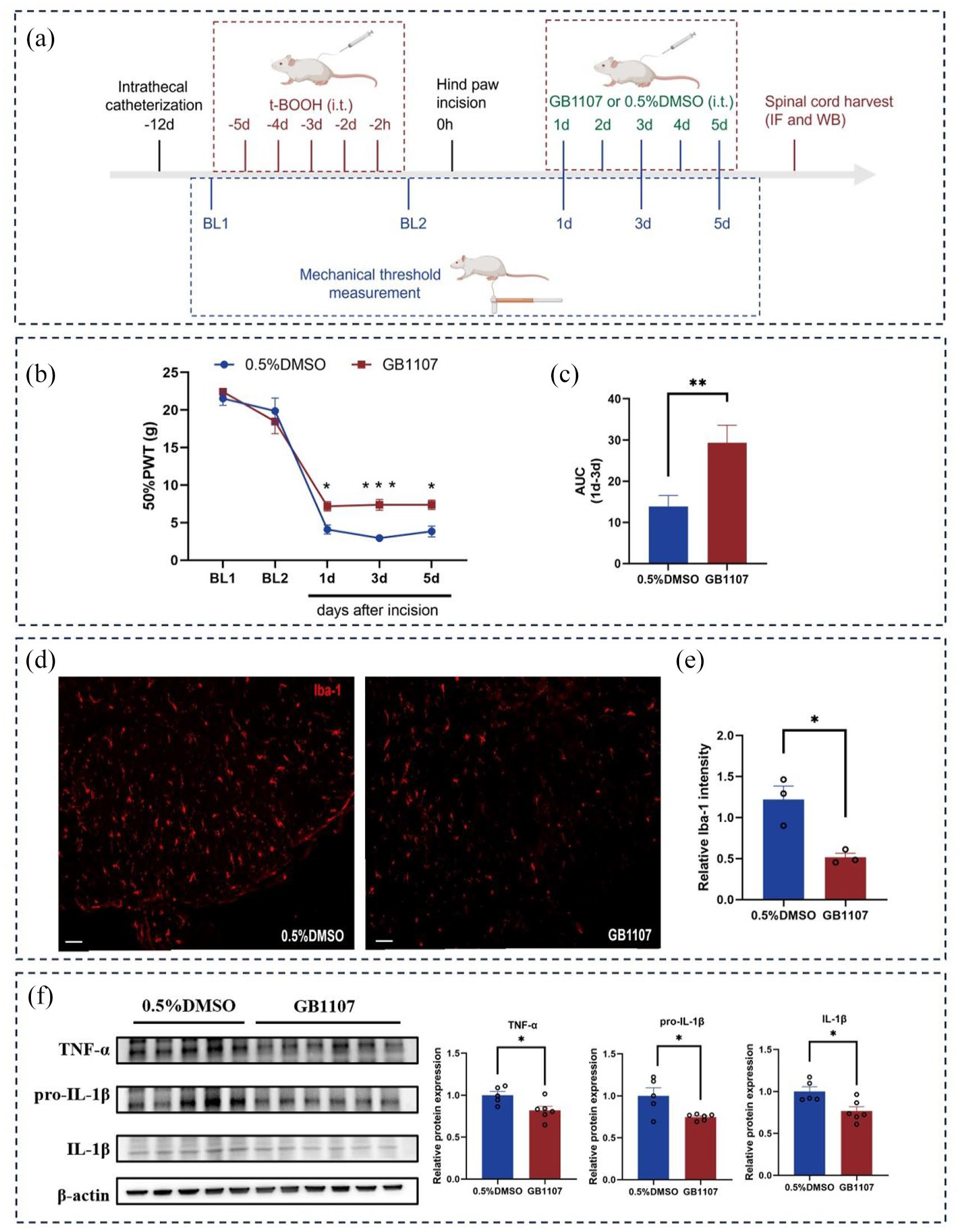
Galectin-3 participated in the formation of postoperative spinal neuroinflammation in the preoperative ROS-increased model. (a) Flowchart of mechanical pain threshold measurement and tissue sampling. (b) and (c) 50% PWT and AUC from day 1 to day 3 postoperatively after continuous injection of GB1107 (n = 6-10). Iba-1 fluorescence intensity was assessed in the spinal cord after continuous intrathecal injection of GB1107. Representative images of Iba-1 fluorescence, scale bar = 50 μm, and statistical analysis of fluorescence intensity in the GB1107 group and control group ((c) and (e) n = 3). (f) Changes in spinal TNF-α, pro-IL-1β, and IL-1β expressions after continuous intrathecal injection of gb1107 (n = 5–6). Data were shown as mean ± SEM; *P < 0.05, **P < 0.01, ***P < 0.001 versus 0.5% DMSO group.
Galectin-3 recombinant protein induced mechanical hyperalgesia and spinal neuroinflammation
To further investigate the nociceptive and pro-inflammatory effects of galectin-3, we intrathecally injected galectin-3 recombinant protein into female rats and measured mechanical pain thresholds and spinal neuroinflammation levels (Figure 7(a)). Repeated intrathecal injections of galectin-3 recombinant protein significantly decreased mechanical pain thresholds and the AUC of mechanical pain thresholds (Figure 7(b) and (c)), demonstrating a notable hyperalgesia-inducing effect. Galectin-3 also increased microglial activation (Figure 7(d) and (e)) and the expression of TNF-α, pro-IL-1β, and IL-1β (Figure 7(f)), indicating its significant role in promoting neuroinflammation.

Galectin-3 induced hyperalgesia and neuroinflammation. (a) Intrathecal injection of galectin-3 recombinant protein, assessing mechanical pain thresholds and tissue sampling. (b) and (c) Effects of repeated intrathecal injection of galectin-3 on 50% PWT and statistical analysis of AUC from day 1 to day 4 (n = 10). Spinal cord collection after the fifth intrathecal injection of galectin-3. Representative image of Iba-1 fluorescence, scale bar = 50 μm (c), and statistical analysis of Iba-1 immunofluorescence intensity in the Gal-3 group and solvent control group ((e) n = 3–4). (f) Impact of galectin-3 on spinal TNF-α, pro-IL-1β, and IL-1β expressions. Data were presented as mean ± SEM; *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001 versus saline group.
TLR4 receptor inhibitors mitigated the nociceptive effect of galectin-3 and the pro-inflammatory effect in vitro experiments
The TLR4 receptor is crucial in mediating pro-inflammatory effects, 30 and inflammation is a key mechanism in hyperalgesia development. Therefore, the TLR4 receptor plays a significant role in the development of hyperalgesia. To determine if galectin-3-induced hyperalgesia is mediated by TLR4, we administered the TLR4 receptor inhibitor TAK-242 intrathecally before galectin-3 injection (Figure 8(a)). Behavioral results showed that mechanical pain thresholds significantly decreased in female rats at 1 and 3 hours after intrathecal galectin-3 recombinant protein injection, while TAK-242 alleviated galectin-3-induced hyperalgesia (Figure 8(b)). The BV2 microglial cell line was used for in vitro experiments (Figure 8(c)). When stimulated, BV2 cells exhibited shortened and thickened processes, and enlarged cell bodies resembling an amoeboid shape, indicative of activation. 31 Activated BV2 cells can increase the expression and release of pro-inflammatory cytokines. 32 Galectin-3 activated BV2 cells, while TAK-242 mitigated galectin-3-induced activation of BV2 cells (Figure 8(d)). Correspondingly, galectin-3 promoted the release of TNF-α and IL-1β from BV2 cells, while pretreatment with TAK-242 significantly reduced its pro-inflammatory effect (Figure 8(e) and (f)). Thus, in vivo and in vitro experiments suggest that the nociceptive and pro-inflammatory effects of galectin-3 are mediated through the TLR4 receptor.

TLR4 receptors were involved in the nociceptive and in vitro pro-inflammatory effects of galectin-3. (a) Experimental flowchart explored the relationship between galectin-3-induced hyperalgesia and TLR receptors. (b) Effect of TLR4 receptor inhibitor pretreatment on galectin-3-induced hyperalgesia (n = 10–12). (c) Experimental flowchart illustrated the association between pro-inflammatory effect of galectin-3 and TLR receptors in vitro. (d) Effects of galectin-3 and TAK-242 pretreatment on BV2 cell morphology, scale bar = 50 μm. (e) Impact of galectin-3 and TAK-242 pretreatment on TNF-α and IL-1β release from BV2 cells (n = 3). Data were shown as mean ± SEM; for behavioral data, ****P < 0.0001 versus Vehicle group, ###P < 0.001, ####P < 0.0001 versus Gal-3; in vivo experiments, *P < 0.05, **P < 0.01.
Discussion
In the present study, animal experiments were used to validate clinical inferences and explore potential mechanisms. Clinical studies suggested a link between postoperative pain and preoperative ROS, and patients with more postoperative pain had a higher NLR-Ratio which reflected elevated levels of inflammation. Accordingly, animal experiments confirmed that preoperative ROS can affect postoperative hyperalgesia and spinal neuroinflammation. Furthermore, Inhibition of galectin-3 mitigated postoperative hyperalgesia and neuroinflammation in the preoperative ROS-increased model. In addition, the TLR4 receptor inhibitor reduced the nociceptive effects of galectin-3 and galectin-3-induced inflammation in vitro. These studies demonstrated preoperative ROS increasing, a preoperative pathological change, could worsen postoperative hyperalgesia and identified galectin-3 as a corresponding therapeutic target.
Numerous studies have shown that ROS play a crucial role in the development of pain.33–35 In our clinical study, we also found that 25% of the proteins associated with post-cesarean section pain were related to ROS, suggesting that an increase in preoperative ROS may exacerbate postoperative pain. To test this hypothesis, we used the oxidant t-BOOH and the antioxidant VE to alter preoperative ROS levels in female rats. Pre-administration of t-BOOH increased ROS and further reduced the postoperative mechanical pain thresholds of rats, whereas VE treatment reduced preoperative ROS and increased postoperative mechanical pain thresholds, thereby demonstrating that preoperative ROS can affect postoperative hyperalgesia. When the body tissues are subjected to harmful stimuli, inflammatory cells such as neutrophils, microglia, and macrophages proliferate significantly, activating the NF-kB pathway in inflammatory cells to promote the expression of TNF-α and pro-IL-1 which is further cleaved by inflammasomes to generate mature IL-1β. 36 Neuroinflammation activation is a crucial factor in hyperalgesia, as the increase in inflammatory factors can enhance neuronal excitability by increasing the presynaptic release of the excitatory neurotransmitter glutamate and reducing the release of inhibitory neurotransmitters.37,38 It was found that the NLR-Ratio in post-cesarean section women was positively correlated with pain, indicating that the more severe the postoperative pain, the higher the level of inflammation. Furthermore, increasing preoperative ROS activated microglia and increased the expressions of TNF-α, pro IL-1, and IL-1β, while reducing ROS decreased the activation of microglia and the expressions of inflammatory factors. Therefore, preoperative ROS may affect postoperative pain sensitivity by altering neuroinflammation.
The development of chronic pain, such as post-herpetic neuralgia and neuropathic pain, is accompanied by increased galectin-3 expression.39,40 In this study, ROS also promoted Galectin-3 expression, as demonstrated by immunofluorescence and Western blot results. In addition, the intensity of post-cesarean section pain in pregnant women was positively correlated with galectin-3, indicating that galectin-3 may be a crucial protein involved in pain formation. To verify this hypothesis, we first intrathecally injected the galectin-3 inhibitor GB1107 in female rats with preoperative increased ROS and found it significantly improved postoperative hyperalgesia. However, to rule out that GB1107 alleviated hyperalgesia by inhibiting incisional pain, we injected an equivalent dose of GB1107 into the incision-only group and found that the postoperative mechanical pain threshold did not change. Meanwhile, intrathecal injection of exogenous galectin-3 recombinant protein revealed that galectin-3 significantly reduced the pain thresholds. These behavioral results together showed galectin-3 was a nociceptive substance and participated in the formation of postoperative hyperalgesia in the model of increased preoperative ROS. Pain behavior exhibits gender differences,41,42 yet most basic research focuses on a single gender, which is one of the major reasons for the poor clinical translation of analgesics. Similar to females, behavioral results in male rats demonstrated that preoperative ROS were associated with postoperative hyperalgesia, and inhibiting galectin-3 can ameliorate postoperative hyperalgesia in the preoperative ROS-increased model. Moreover, clinical patients frequently suffer from preoperative anxiety, depression, or sleep disorders,43,44 and these preoperative pathological states can lead to an increase of ROS,27,45 suggesting that increased preoperative ROS may be a common pathological change in clinical patients. Therefore, GB1107, the galectin-3 inhibitor, may serve as an analgesic to improve postoperative pain in clinical patients.
Increased preoperative ROS were associated with increased galectin-3 expression and aggravated postoperative neuroinflammation. Studies have shown that galectin-3 can participate in musculoskeletal chronic pain developments by promoting microglial proliferation and inflammatory mediator release. 22 Therefore, to explore the pro-inflammatory role of galectin-3 in the model of increased preoperative ROS, we intrathecally injected GB1107 and found a significant reduction in the activation of microglia and expression of pro-inflammatory factors and exogenous galectin-3 recombinant protein increased the activation of microglial cells and expression of pro-inflammatory factors, indicating preoperative ROS exacerbated postoperative hyperalgesia by aggravating neuroinflammation through galectin-3. The C-type carbohydrate recognition domain (CRD) structure of galectin-3 protein can bind to TLR4 receptor, and GB1107 can block its binding to TLR4 receptors by binding to the CRD domain.46,47 TLR4 receptors are common nociceptive and pro-inflammatory factors, suggesting that Galectin-3’s nociceptive and pro-inflammatory effects may be triggered by TLR4 receptors. Using TLR4 receptor knockout mice, it has been found that Galectin-3’s nociceptive and pro-inflammatory effects are alleviated. 38 Similarly, in our study, pretreatment with TLR4 receptor inhibitors significantly alleviated the hyperalgesia induced by galectin-3. The effects of galectin-3 on promoting microglial release of TNF-α and IL-1β can be inhibited by TAK-242 in vitro.
There are some limitations in this study. Firstly, since the preoperative cerebrospinal fluid and peripheral blood samples were exhausted in the preliminary analysis and these samples could not be obtained again due to ethical rules, we were unable to directly or indirectly measure the preoperative ROS levels. Secondly, although we used Western blot and immunofluorescence to demonstrate that ROS can promote galectin-3 expression, the mechanism by which ROS enhances galectin-3 expression and the specific cellular origin of galectin-3 remain unknown, which should be further studied. Thirdly, 40 participants were primarily included to explore the potential mechanism of postoperative pain using proteomics, but the sample size was not calculated for other analysis (e.g. correlative analyses galectin-3 vs. pain scores). Thus, the potential Type II errors are unavoidable and this correlation should be further validated in the future. Finally, studies have reported that there are gender differences in the degree or type of inflammation under the same external stimuli.48–50 The mechanistic exploration in this study was conducted in female rats. Therefore, it is still necessary to investigate whether ROS and galectin-3 exacerbate postoperative hyperalgesia by stimulating microglial activation and the expressions of TNF-α and IL-1β in male rats.
Conclusions
In conclusion, our clinical research indicated that acute postoperative pain may be associated with preoperative ROS and pain intensity was positively correlated with the NLR-Ratio. The animal models confirmed that the severity of postoperative hyperalgesia and neuroinflammation were influenced by preoperative ROS levels. Moreover, preoperative ROS exacerbated postoperative hyperalgesia by promoting neuroinflammation via galectin-3. Thus, galectin-3 may be a promising target for treating postoperative pain in clinical patients.
Supplemental Material
sj-docx-1-mpx-10.1177_17448069251353026 – Supplemental material for Preoperative reactive oxygen species exacerbate postoperative hyperalgesia by aggravating neuroinflammation through galectin-3
Supplemental material, sj-docx-1-mpx-10.1177_17448069251353026 for Preoperative reactive oxygen species exacerbate postoperative hyperalgesia by aggravating neuroinflammation through galectin-3 by Xuemei Zheng, Bin Shu, Ai Yan, Yonggang Liang, Niannian Huang, Houping Wang, Weilong Hong, Yongqin Chen, He Huang and Guangyou Duan in Molecular Pain
Footnotes
Acknowledgements
We would like to appreciate the participants for their selfless contribution to this study.
Authors contributions
XZ, BS, YL, AY, WH, and HW contributed to the data collection. XZ and GD contributed to data analysis. XZ contributed to the draft writing. BS, HH, and GD contributed to draft revision. All authors gave final approval for the version to be published and agreed to be accountable for all aspects of the work.
Data Availability Statement
The data used and analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (82171210) and Chongqing Natural Science Foundation (Grant No. CSTC2021JCYJ-MSXMX0188 and CSTB2022NSCQ-BHX0015), and the Kuanren Talents Project of The Second Affiliated Hospital of Chongqing Medical University.
Ethics approval and consent to participate
The clinical study was approved by the Medical Ethics Committee of the Second Affiliated Hospital of Chongqing Medical University (ethics approval number: 2021-123). All patients provided informed consent before the study. Animal experimental protocols were approved by the Animal Care and Use Committee of the Second Affiliated Hospital of Chongqing Medical University (ethics approval number: IACUC-SAHCQMU-2024-0002).
Informed Consent Statement
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
