Abstract
Keywords
Introduction
Lumbar intervertebral disc herniation (LDH) is the most frequent clinical pain condition characterized by spontaneous low back pain and persistent radiating lumbar disk herniation. 1 Conventional clinical options for LDH are not effective in relieving neuropathic and radicular pain. Spinal pathologies can lead to neurological disability, resulting in repeated clinic visits, chronic pain, and even loss of function, significantly compromising the quality of life for patients worldwide. 2 Therefore, it is necessary to determine the functional molecular mechanisms underlying LDH pathophysiology to develop precise therapies for inhibiting this pathological disorder.
Although the precise pathophysiological mechanisms of LDH have not been elucidated, it is considered to be a neuroimmune disorder caused by protruded disc material. 3 Accumulating evidence suggests that the protruded tissue is regarded as a foreign invader by the immune system, and hence it might trigger neuro-immune inflammatory response in LDH. 4 Studies have proven that pro-inflammatory factors modulate the pathological plasticity associated with LDH.1,3,5 Transforming growth factor-β (TGF-β) is a relevant mediator that provides protective effects against neuroinflammatory events underlying neuropathic pain.6,7 Due to the considerable impact of LDH on people’s health, a better understanding of the neuroinflammatory reaction is likely to yield new research opportunities for the intervention of LDH pathogenesis.
As a peptide neurotransmitter, calcitonin gene-related peptide (CGRP) is primarily released from sensory nerves in numerous sites throughout the peripheral and central nervous system.8,9 It is known that nociceptors releasing CGRP are involved in the development of nociceptive signaling implicated in the chronic inflammatory pain pathway.9,10 Recently, it was reported that CGRP directly promotes proinflammatory cytokine release, contributing to neurogenic inflammation in the intervertebral disc. 11 These data imply that CGRP may represent a promising therapeutic target for the intervention of neuroinflammation-associated pain.
Silent information regulator 1 (SIRT1), belonging to the Sirtuin family, can influence the development of inflammation in numerous physiological processes.12–14 Recent studies have shown that SIRT1 is one of the most common regulators of various inflammatory genes that modulate the persistent development of inflammatory pain models.15,16 However, the precise molecular mechanisms of SIRT1-regulated neuro-inflammatory response in neuropathic pain remain to be clarified. Considering that CGRP is one promising effector protein contributing to the pathogenesis of inflammatory pain, we hypothesized that the interaction between SIRT1 and CGRP may provide a specific target for the treatment of neuropathic pain.
Recent studies have recognized special pro-resolving lipid mediators (SPMs) as biosynthetically active biomaterials that orchestrate the resolution of tissue inflammatory responses during disease.17,18 Protectin D1(PD1), one of the well-known endogenous SPMs synthesized from docosahexaenoic acid, offers a significant advantage due to its enhanced inflammatory resolution properties in the treatment of inflammatory pain.19–21 Therefore, this present study aimed to determine the potential analgesic effects of PD1 on rats with non-compressive lumbar disc herniation (NCLDH), a form of LDH. Additionally, we examined the relationship between alterations in SIRT1 activity and the expression of CGRP in the spinal dorsal horn to uncover the underlying molecular mechanisms. We highlight the potential analgesic property of PD1 by mediating SIRT1/CGRP-based pro-resolving inflammatory signaling pathways, providing an updated perspective to complement the current SPMs-based inflammatory therapeutics for LDH.
Methods
Experimental animals
Adult male Sprague–Dawley rats weighing 250–350 g were obtained from the Experimental Animal Center of Beijing Vital River (Beijing, China). The animals were housed in temperature-controlled rooms with a constant temperature of 22 ± 1°C, a 12-h light-dark cycle, and ad libitum access to food and water. Before surgery and for 14 days following surgery, all animals were acclimated to their cages. All animal experimental procedures were approved by the Institutional Animal Care, and lab. animals were cared for and used in accordance with the guidelines of the Qingdao University.
NCLDH models in rats
Rat NCLDH were carefully established as follows: The animals were intraperitoneally anesthetized with pentobarbital 40 mg/kg and placed in a prone position on a surgical board. After sterilization, a dorsal incision was made between the L4-6 spinous processes to separate the multifidus muscles. An L5 laminectomy was performed to expose the dorsal root ganglion (DRG), which was then cleaned with normal saline. In the right L5 DRG, an equivalent amount of NP was implanted without compression. Animals in the sham group underwent the same procedures except for the placement of the protruded tissues.
Measurement of pain-related behavior
Behavioral analysis tests were performed at predetermined time points: preoperative day 1 and postoperative days 1, 3, 5, 7, 10, and 14. All experimental animals were allowed to acclimate to the test conditions for at least 30 min before each behavioral assessment. Mechanical hyperalgesia and thermal allodynia were assessed using the mechanical paw withdrawal threshold (PWT) and thermal paw withdrawal latency (PWL), respectively, as previously described.22,23
To assess mechanical allodynia, PWT was measured using calibrated von Frey filaments (Stoelting, Wood Dale, Illinois, USA) applied to the bilateral plantar surface of the hind paws, following the “up-down” method to calculate responses to mechanical stimuli. Thermal hyperalgesia of the bilateral hind paw plantar surfaces was evaluated using the Hargreaves test in response to heat. To prevent sensitization, five trials were performed on each hind paw with a 10-min interval between them. Behavioral tests were conducted in a blinded manner to the groups and were performed at approximately the same time of the day.
Drug administration
The rats were randomly divided into the following groups: sham group, sham + PD1 group, NCLDH group, NCLDH + PD1 group, sham + CGRP, sham +CGRP + PD1 group, NCLDH + CGRP8-37 group (CGRP inhibitor), sham + EX-527 group (SIRT1 inhibitor), NCLDH + SR1720 group (SIRT1 activitor), and NCLDH+ PD1+SR1720 group. The drugs of PD1 (25 μm, Cayman Chemical Company), CGRP (1 μM, MCE), CGRP8-37 (2 μM, MCE), SRT1720 (10 μM, Selleck) and EX527 (1 mM, Selleck) were dissolved in sterile phosphate buffer saline (PBS) to prepare the stock solution, following the manufacturer’s instructions. Starting from the first three consecutive days after NCLDH surgery and following the behavior tests, the drugs were administered intrathecally as a single dose administration. The same volume (10 μL) of the vehicle was injected into rats in the sham groups as a control.
Western blotting
To assess the underlying mechanisms, the expression of SIRT1 and CGRP proteins was examined in rats implanted with autologous protruded materials. After killing the rats, we harvested and stored the ipsilateral spinal dorsal horn tissues from different groups in liquid nitrogen. The tissues were homogenized using RIPA lysis buffer (Beyotime Biotechnology, CHN) following the manufacturer’s protocol. Total protein levels in the extracts were determined using a BCA Protein Assay kit (Pierce Biotechnology, Rockford, IL, USA). Protein lysates from each sample (30 μg) were subjected to electrophoresis on an 8%–15% sodium dodecyl sulfate (SDS)-polyacrylamide gel electrophoresis (PAGE) gel. Subsequently, the proteins were transferred to a PVDF membrane (Merck Millipore, Massachusetts, USA) for 1 h at room temperature. The membranes were blocked with 5% nonfat milk in 0.1% Tween-20 in 0.5% tris buffered saline (TBS) and incubated for 2 h at room temperature. The membranes were then incubated overnight at 4°C with primary antibodies against CGRP (1:1000, Cell Signaling Technology, #14959) and SIRT (1:1000, Abcam, ab110304, Cambridge, MA, USA). The membranes were then incubated with the appropriate anti-rabbit secondary antibody (1:5000, Santa Cruz Biotechnology, Texas, USA) for 2 h at room temperature. Protein bands were visualized using an enhanced chemiluminescence assay (Merck Millipore, Massachusetts, USA) after washing. Protein bands were developed using Pierce ECL Western Blotting reagents (ThermoFisher, Rockford, IL, USA) to detect the targeted proteins. Subsequently, the densities of the target proteins were analyzed and quantified. The expression level was normalized to that of the controls.
Measurement of cytokine levels by enzyme-linked immunosorbent assay (ELISA)
Spinal tissues were collected and homogenized using PBS buffer pH7.4 containing a protease inhibitor cocktail. An aliquot of the supernatant was stored at −80°C following centrifugation at 10,000g at 4 C for 30 min. The levels of TGF-β, TNF-α, and IL-1β in DRGs lysates were quantified according to the manufacturer’s instructions.
Quantification of immunofluorescence
After killing the rats, a cautious harvest of ipsilateral spinal dorsal horns (L4-6) was performed. The harvested sections were immersed in a 30% sucrose solution in phosphate buffer overnight at 4°C for cryoprotection after overnight post-fixation with 4% paraformaldehyde. Subsequently, rabbit anti-SIRT1 antibody (18,725-1-AP; 1:500; Proteintech, CHN) and rabbit anti-CGRP antibody (1:400, Cell Signaling Technology, #14959) were added to the sections at 4°C and incubated overnight. Following three washes with TBS, the sections were incubated with goat anti-rabbit secondary antibody (PV-9000; 1:100; ZSGB, CHN) for 30 min at 37°C. After three additional washes with TBS, the sections were counterstained with DAPI (Abcam). To detect immune-positive reactions, a diaminobenzidine kit (ZSGB, CHN) was used, followed by staining with hematoxylin and sealing. Pictures were taken using a Leica DM4000 B microscope and a digital camera (Germany). The Image Pro-Plus program was used to conduct quantitative analyses of CGRP or SIRT1 immunostaining.
Statistical analysis
IBM SPSS 21.0 statistical software (United States) and GraphPad Prism (United States) was used to graph the results. Two-way analyses of variance (ANOVA) were performed on behavioral data collected from rats. Data differences between groups for western blot results, ELISA , and immunohistochemistry assays were compared using one-way ANOVA and Tukey's multiple comparisons test with time points as independent factors. P<0.05 was considered significant, while P<0.01 was considered statistically highly significant.
Data are presented as the mean ± SEM. Statistical analyses were performed using IBM SPSS 21.0 statistical software (United States) and GraphPad Prism (United States) was used to graph the results. Two-way analyses of variance (ANOVA) were performed on behavioral data collected from rats. Data differences between groups for western blot results, ELISA, and immunohistochemistry assays were compared using one-way ANOVA and Tukey’s multiple comparisons test with time points as independent factors.
Results
PD1 attenuates allodynia and neuro-inflammation induced by autologous protruded materials implantation
The mechanical and thermal paw-withdrawal thresholds (PWT and PWL) of the left hind paw were evaluated 1 day before surgery, as well as 1, 3, 5, 7, 10, and 14 days post-operatively (Figure 1(a)). Autologous NP implantation into the DRG significantly decreased the PWT and PWL compared to the sham groups ( PD1 alleviated hyperalgesia and neuro-inflammation in the NCLDH rat models. (a) Time course of the effects of PD1 treatment on paw withdrawal threshold and latency in rats receive the autologous protruded tissues implantation. 
PD1 attenuates CGRP expression in NCLDH rats
Based on the outcomes of behavior tests and ELISA results, we selected day 7 post-surgery for the subsequent examination, considering it to be the day with the peak pain level and the most opportune time for treatment. To elucidate the mechanisms responsible for NCLDH-induced neuropathic pain, the expression level of CGRP was measured using western blotting and immunofluorescence assays. Western blot analysis revealed a significant increase in CGRP protein expression in the NCLDH group compared to the sham group (Figure 2(a), PD1 decreased the expression of CGRP protein level in the NCLDH rat models. (a) After implantation of autologous protruded tissue, Western blotting was conducted to determine CGRP expression changes. (b) Immunohistochemistry analysis showing the expression of CGRP. 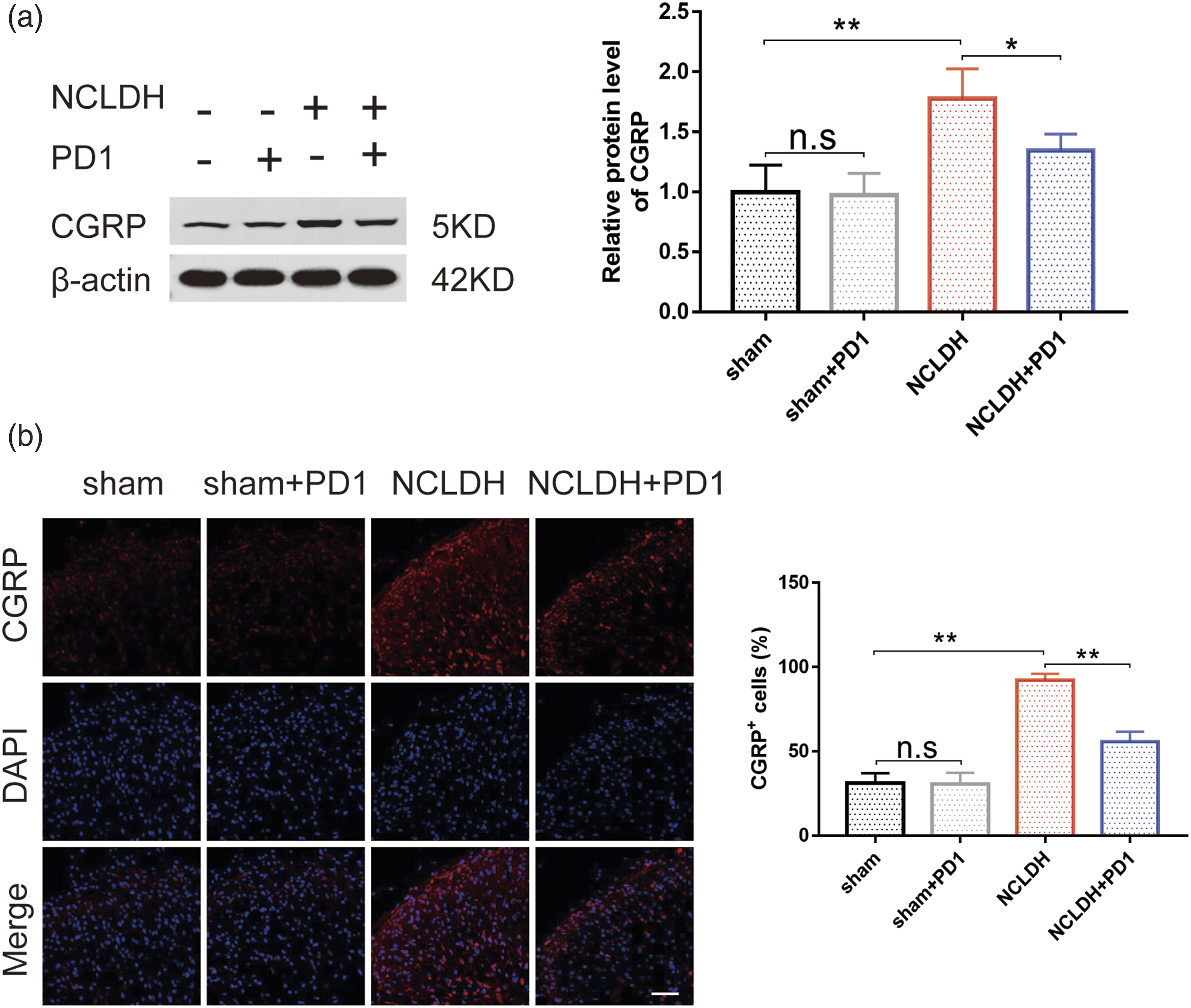
PD1 alleviates NCLDH-induced inflammatory pain partly by activating SIRT1
The alteration in SIRT1 protein levels in spinal cord tissues after NCLDH was detected using Western blotting and immunofluorescence. The results demonstrated a significant decrease in SIRT1 following NCLDH and a dramatic upregulation after PD1 intervention (Figure 3(a) and (b), PD1 decreased the expression of SIRT1 protein level in the NCLDH rat models. (a) Western blot was used to detect the changes in SIRT1 expression. (b) Immunohistochemistry analysis was carried out to evaluate changes in SIRT1 expression in different groups. 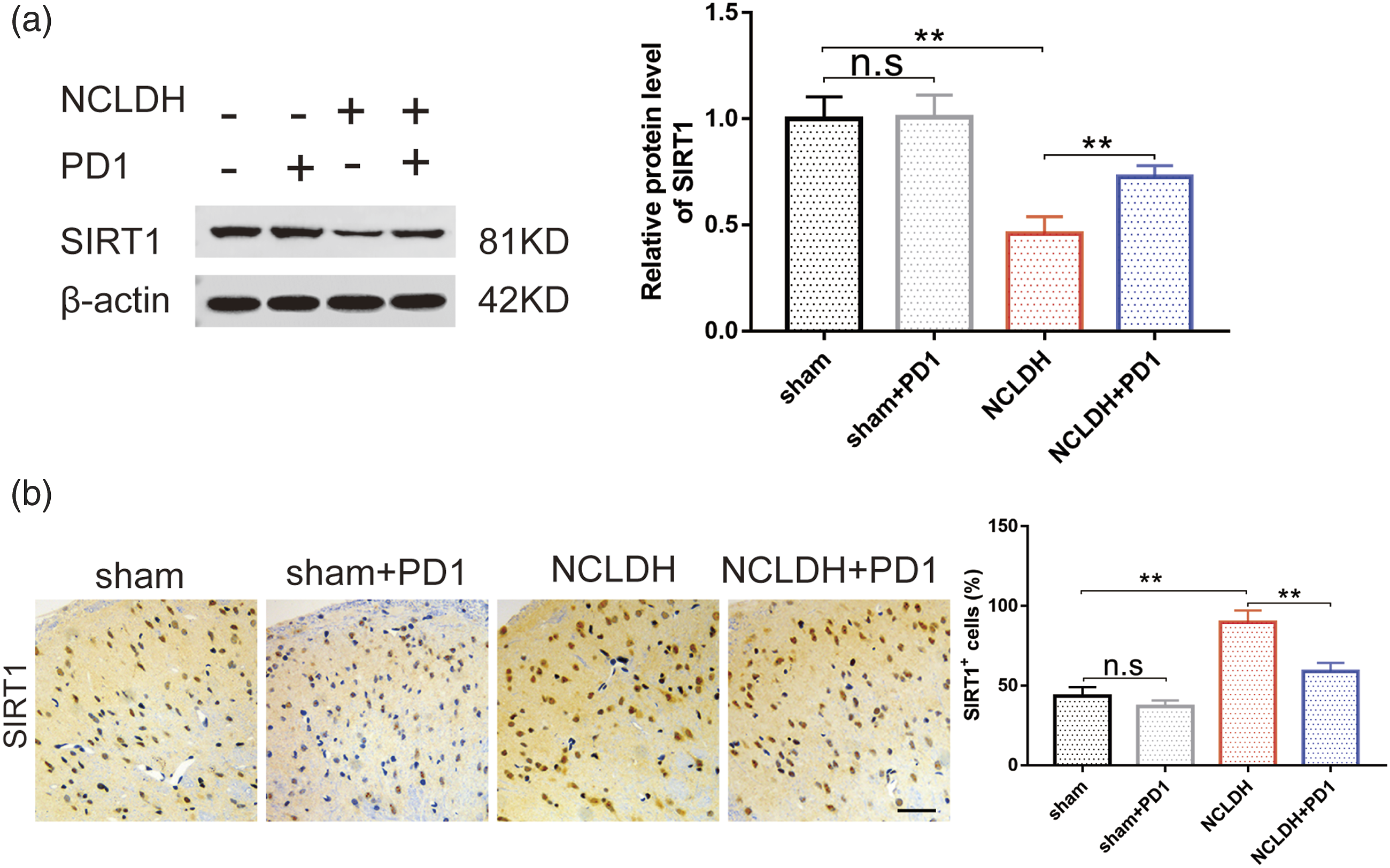
CGRP participates in NCLDH by activating SIRT1 and inducing neuro-inflammation
Behavioral analysis indicated that both CGRP and NCLDH alone drastically decreased the paw withdrawal threshold of rats to mechanical and thermal stimulation. However, administration of CGRP 8-37 (CGRP antagonist) significantly increased PWT and PWL (Figure 4(a), CGRP participates in NCLDH by activating SIRT1 and inducing neuro-inflammation. (a) Time course of the effects of PD1 treatment on PWT and PWL in rats of different treated groups. 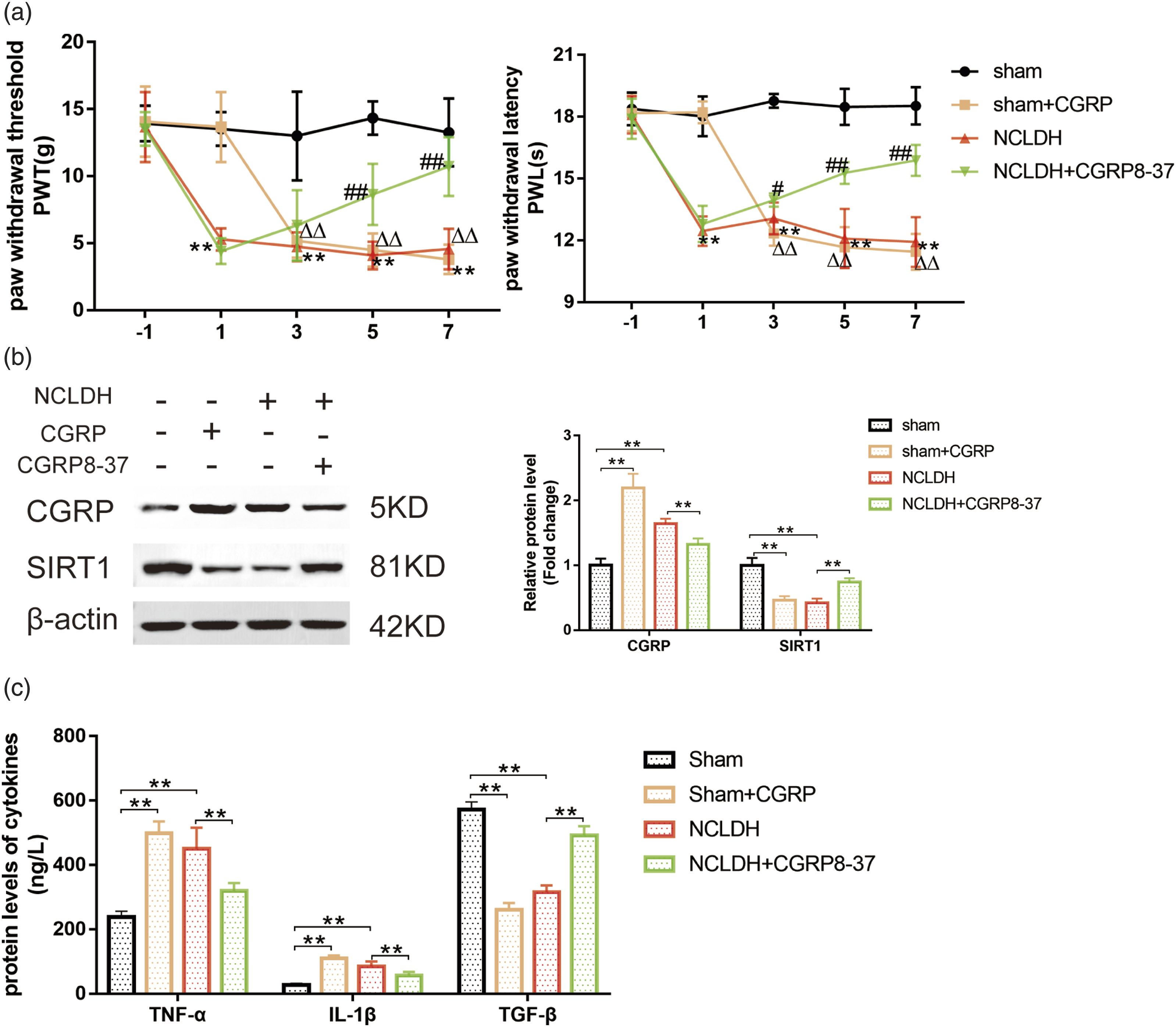
Western blot analysis showed that both CGRP and NCLDH alone inhibited the protein level of SIRT1 (Figure 4(b),
To investigate the relationship between CGRP and neuro-inflammation, CGRP8-37 (a CGRP antagonist) was used to treat NCLDH rats, and the levels of pro-inflammatory and anti-inflammatory factors in the tissues were examined. As shown in Figure 4(c), stimulation with CGRP elevated the protein levels of the inflammation-related cytokines TNF-α and IL-1β, as well as down-regulated the anti-inflammatory protein TGF-β (Figure 4(c)). Furthermore, compared to NCLDH alone, co-treatment with CGRP 8-37 markedly reversed the NCLDH-induced increase in TNF-α and IL-1β expression and the decrease in TGF-β protein expression (Figure 4(c),
These results confirm that CGRP may mediate NCLDH-induced SIRT1 activation and neuro-inflammation in the spinal dorsal horn following autologous protruded tissue implantation.
SIRT1 participates in NCLDH by activating CGRP and mediating neuro-inflammation
We conducted various experiments to determine whether SIRT1 activity influences the analgesic effect of PD1 in NCLDH pathology. Behavioral tests revealed a decrease in ipsilateral mechanical and thermal sensitivity in rats treated with the SIRT1 antagomir (EX527) and NCLDH alone (Figure 5(a), SIRT1 participates in NCLDH by activating CGRP and mediating neuro-inflammation. (a) Time course of the effects of PD1 treatment on PWT and PWL in rats of different treated groups. 
To investigate the protective mechanisms of PD1 against NCLDH-induced hyperalgesia, we measured the effect of SIRT1 on CGRP protein expression. Figure 5(b) demonstrated that EX527 treatment significantly upregulated CGRP expression, while SIRT1 activation by SRT1720 significantly downregulated CGRP expression in NCLDH rats, indicating that it negatively modulated CGRP expression by SIRT1 in tissues (Figure 5(b),
To assess the biological effects of SIRT1 activity on neuro-inflammation, we further investigated whether EX-527 and SRT1720 could modulate the pro-inflammatory/anti-inflammatory response following autologous NP implantation. As expected, administration of EX-527 and NCLDH alone significantly increased TNF-α and IL-1β expression, as well as reduced TGF-β protein expression compared to the sham groups (Figure 5(c)). Moreover, data shown in Figure 5(c) showed that the NCLDH + STT1720 treatment restored the NCLDH-induced effects on pro-inflammatory and anti-inflammatory factors, further confirming the anti-inflammatory effects of SIRT1 in NCLDH (Figure 5(c),
These findings suggest that PD1 may alleviate NCLDH–induced hyperalgesia by improving the neuro-inflammatory response through the upregulation of SIRT1 activity.
Effects of PD1 on SIRT1 and CGRP activation in NCLDH rat models
To gain deeper insights into the effect of CGRP-targeted therapies on NCLDH, we investigated the potential interrelationship between CGRP and SIRT1 in the development of neuropathic pain. The results of behavioral experiment indicated that the hind limb of the PD1 and CGRP combination group had better mechanical allodynia and thermal hyperalgesia compared to the CGRP group (Figure 6(a), Effects of PD1 on SIRT1 and CGRP activation in NCLDH rat models. (a) Time course of the effects of PD1 treatment on PWT and PWL in rats of different treated groups. 
To further investigate whether PD1 alleviated hyperalgesia by influencing the relationship between CGRP and SIRT1, we explored the changes in CGRP and SIRT1 expression by western blot. Treatment with CGRP increased the level of CGRP and down-regulated SIRT1 in sham rats. However, when combined with PD1 and CGRP, these alterations were reversed (
Next, we evaluated the effect of neuro-inflammation on the relationship between CGRP with SIRT1. Figure 6(c) shows that CGRP treatment elevated the synthesis of TNF-α and IL-1β while decreasing the induction of TGF-β. However, injection of PD1 effectively mitigated these pro-inflammatory and anti-inflammatory reactions. Similarly, SRT1720 treatment down-regulated the production of TNF-α and IL-1β and up-regulated the induction of TGF-β. Co-administration of PD1 further strengthened the effects of SRT1720 (Figure 6(c)). Therefore, these results collectively indicate that PD1 treatment could alleviate the inflammation severity of NCLDH-induced neuropathic pain by regulating the interaction between CGRP and SIRT1.
Discussion
This study aimed to investigate the anti-nociceptive effect of PD1 and investigate its molecular mechanism involving neuro-inflammatory processes. Our findings strongly suggest that PD1 exerts its analgesic effect by regulating the interaction between spinal SIRT1 activity and CGRP protein expression in NCLDH–induced hyperalgesia. This study sheds light on the interplay between SIRT1 activity and CGRP expression in the development of LDH.
Neuro-inflammation has been widely implicated in the pathogenesis of LDH.3,24 Herniated tissue, acting as a foreign nociceptive antigen, triggers strong immune neuro-inflammatory responses around the lumbar herniated disc (4). Stimulation of protruded tissues leads to central sensitization, which is considered the primary cause of neuropathic pain (5). In our study, observed a persistent decrease in PWT and PWL in the ipsilateral paw from the first day to the 14th day following autologous protruded materials implantation in the DRG.
Numerous studies have demonstrated the involvement of immune neuro-inflammatory responses in the generation of inflammatory and neuropathic pain.3,5,24 Inflammatory cytokines participate in the initiation of immune-inflammatory processes by recruiting immune cells to the discal tissues in LDH pathophysiology.1,6,25 Our results showed that the implantation of protruded materials led to a significant decrease in PWT and PWL, accompanied by a chronic immune-inflammatory response as characterized by increased spinal inflammatory cytokines (TNF-α, IL-1β, and TGF-β). These findings are consistent with previous studies, highlighting the contribution of neuroinflammation to the pathogenesis of LDH.
PD1, a newly discovered member of SPMs has been shown to inhibit neural inflammation in traumatic nerve injury models.19,26 Moreover, it can control over-active neuro-inflammation in various inflammatory diseases.16,27 Therefore, strategies that mimic the action of PD1 can alleviate nociceptive sensitivity induced by inflammatory pain. Our previous studies have demonstrated that specialized pro-resolving mediators (SPMs) improve mechanical allodynia by reducing the expression of TNF-α and IL-1β in spinal nerve ligation models.28,29 In this study, we observed that the administration of PD1 significantly suppressed the levels of TNF-α and IL-1β and enhanced the protein expression of TGF-β in the spinal dorsal horns, confirming the key role of PD1 in mediating the resolution of inflammation. Therefore, our findings indicate that the analgesic effects of intraperitoneal injection of Protectin D1 may be associated with the attenuation of neuro-inflammatory responses induced by NCLDH. However, the specific mechanisms mediating the anti-inflammatory and nociceptive sensitivity-alleviating effects of PD1 in LDH remain largely unexplored.
CGRP, a neuropeptide that acts as an irritant to the immune system, has been shown to regulate spontaneous neuro-inflammatory responses and sensitize nociceptive information.10,30 Recent studies have demonstrated the involvement of CGRP in the pathophysiology of various neurogenic inflammatory-associated diseases, such as migraine, 9 colitis, 31 and diabetic sensory neuropathy. 32 Functional blockade targeting the CGRP receptor has been developed as a therapeutic approach for migraine prophylaxis. 33 In our study, we observed a significant increase in CGRP protein expression following the implantation of autologous protruded tissues, which was reversed upon PD1 intraperitoneal injection. Moreover, the elevated release of CGRP was accompanied by overexpression of TNF-α and IL-1β, along with down-regulation of TGF-β. Moreover, CGRP induced pain-related behavior and promoted the formation of inflammatory reactions. Therefore, we hypothesized that the regulation of CGRP might directly affect the balance of neurogenic inflammation, leading to peripheral and central pain sensitization in LDH rat models. Furthermore, intrathecal administration of CGRP8-37, a specific antagonist for CGRP receptor 1, 34 strongly inhibited the activation of TNF-α and IL-1β while increasing the activation of TGF-β in the NCLDH rat model. Similarly, intrathrecally administration of CGRP8-37 alleviated mechanical and thermal allodynia induced by implanted nucleus pulposus in rats. Based on these findings, we conclude that CGRP activation is crucial for initiating inflammatory pain induced by NCLDH.
SIRT1, an endogenous lipid-derived mediator, has been shown to function as a ‘‘braking signal’’ in mediating neuro-inflammation and participating in various pathophysiology processes. 13 Accumulating evidence supports the protective properties of SIRT1 in chronic pain conditions such as low back pain (LBP), 15 Chronic prostatitis/chronic pelvic pain syndrome (CP/CPPS), 35 and osteoarthritis. 12 Our study revealed a significant decrease in spinal SIRT1 protein expression with the progression of NCLDH-induced neuropathic pain. Intrathecal injection of PD1 induced the activation of spinal Sirt1 protein, demonstrating an inflammation resolution effect that correlated with the alleviation of mechanical and thermal hyperalgesia. The application of SRT1720, a specific agonist of SIRT1, 36 alleviated NCLDH-induced mechanical allodynia and promoted inflammation resolution, further confirming the potential anti-inflammatory and analgesic function of spinal SIRT1. Therefore, targeting SIRT1 may be a promising neuroprotection strategy for LDH. However, the specific mechanism linking SIRT1 with CGRP remains largely unknown, and further research is needed to elucidate this relationship.
To investigate the relationship between SIRT1 and CGRP, we applied EX-527, a classical SIRT1 enzymatic activity inhibitor. 37 Interestingly, we found that intrathecal injections of EX-527 effectively inhibited SIRT1 activity, which resulted in increased activation of CGRP and a significant nociceptive effect compared to the sham group. Consistently, we observed elevated levels of TNFα and IL-1β, along with reduced TGF-β levels following EX-527 injection. Additionally, we employed SRT1720, a specific agonist of SIRT1(36), to further evaluate the effect of SIRT1 on CGRP expression in NCLDH rats. Results indicated that SIRT1720 significantly decreased the expression of NCLDH-induced pro-inflammatory factors, upregulated the expression of anti-inflammatory factors, and attenuated the development of allodynia in rats with NCLDH-induced neuropathic pain. Importantly, the SIRT1720 injection also reduced the expression of CGRP in spinal cords. Furthermore, CGRP administration was shown to significantly decrease the release of SIRT1 and induce an allodynic response. However, these responses were significantly blocked when CGRP was co-injected with PD1, which exhibited inflammatory resolution properties. Moreover, the administration of a CGRP antagonist ameliorated radicular inflammation, and allodynia behavior, and reversed the impaired SIRT1 activity induced by NCLDH. These findings indicate clear crosstalk between SIRT1 and CGRP signaling in spinal cord tissues. In LDH rat models, PD1 may exert analgesic and pro-resolving effects through SIRT1 mediated CGRP signaling.
In conclusion, our study demonstrates that PD1 is an effective analgesic strategy for treating lumbar disc herniation due to its anti-inflammatory effects. This research provides the first exploration of the interplay between SIRT1 and CGRP in the nociceptive pathways related to pain processing. The findings suggest that PD1 could potentially serve as a promising pharmacological therapy, targeting the SIRT1/CGRP axis for effective pain management.
Footnotes
Author contributions
Conception and design: Y.H.W. and Y.R.T.; Administrative support: L.X.L.; Provision of study materials: Y.C.Z. and Y.Z.; Collection and assembly of data: Y.Z. and X.G.; Data analysis and interpretation: Y.Z., Y.R.T. and X.G.; Manuscript writing: Y.C.Z.; Final approval of manuscript: All authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from Natural Science Foundation of Shandong Province (No. ZR2022MH110), Qingdao Medical and Health Research Program (No. 2021-WJZD015), Qingdao Chinese Medicine Science and Technology Program (No. 2022-zyym13), Medical Health and Technology Development Plan of Shandong Province (No. 202120010670).
Ethical Statement
Data Availability Statement
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.
