Abstract
Inflammatory pain is a pervasive clinical issue that severely diminishes individuals’ quality of life. AP-1 (Activating protein-1) is a transcription factor composed of Jun and Fos proteins. Upregulation of AP-1/c-Jun activity is observed in a variety of diseases, particularly in inflammatory conditions. The CCL2 (C-C Motif Chemokine Ligand 2)/CCR2 (C-C Chemokine Receptor 2) axis plays a crucial role in regulating both peripheral and central inflammation. Curcumin, a natural compound derived from the roots of turmeric, possesses anti-inflammatory, antioxidant, and analgesic properties, making it effective for treating various disorders. However, the effects of curcumin on inflammatory pain and its potential mechanisms of action remain unclear. In this study, we utilized a CFA (Complete Freund’s Adjuvant)-induced inflammatory pain model to investigate the effects of curcumin. We found that curcumin effectively reduced CFA-induced mechanical allodynia when administered via intrathecal injection. Behavioral assessments were performed using the Von Frey test. Western blot analysis was performed to detect variations in molecular expression, while immunofluorescence was employed to ascertain cellular localization. Intrathecal injection of the AP-1/c-Jun inhibitor T-5224, along with curcumin, resulted in a reduction in the levels of c-Jun, p-c-Jun, CCL2, and CCR2. Additionally, intrathecal injection of the CCR2 antagonist RS504393 also reduced the expression of CCL2 and CCR2. In summary, curcumin plays a significant role in analgesia within the CFA-induced inflammatory pain model. CCL2/CCR2 acts as a downstream mediator of AP-1/c-Jun. Curcumin can suppress the expression of AP-1/c-Jun, thereby inhibiting the expression of CCL2 and CCR2 in the spinal dorsal horn and contributing to the treatment of inflammatory pain.
Introduction
Inflammation is a crucial defense mechanism of the body in response to tissue injury, characterized by a complex biological response to harmful stimuli such as physical injury, poisoning, infection, or other trauma. 1 The onset of inflammation is often accompanied by pain, with hyperalgesia being a characteristic feature of inflammatory processes. 2 Inflammatory pain does not necessarily diminish with the resolution of inflammation. Data indicate that for many patients with inflammatory diseases, the resolution of inflammation is often insufficient to alleviate pain, which can progressively evolve into chronic pain. 3 Chronic pain resulting from inflammation can become a self-sustaining condition, evolving into evolving into a distinct clinical entity, influenced by a variety of genetic, biological, psychological, and social factors. 4 Research shows that chronic pain imposes a significant burden on individuals and economies worldwide, affecting over 30% of the global population and is a leading cause of hospital admissions. 5 Current medications for the treatment of inflammatory pain mainly include non-steroidal anti-inflammatory drugs and opioids, but both types of drugs can have significant side effects. Therefore, clinicians and patients need to seek safer alternatives.
AP-1 (Activating protein-1) consists of dimeric transcription factors composed of Jun, Fos, or ATF (Activating transcription factor) subunits that bind to a common DNA site, the AP-1 binding site. AP-1/c-Jun is involved in a wide array of biological processes, including cell differentiation, survival, apoptosis, proliferation, transformation, and migration. Upregulation of AP-1/c-Jun activity has been observed in various diseases, particularly inflammatory diseases. 6 Research has shown that in the CFA (Complete Freund’s Adjuvant)-induced inflammatory pain model, AP-1/c-Jun is upregulated and activated, which in turn promotes the expression of inflammatory factors and leads to hyperalgesia.7,8 This finding is consistent with our previous research, which integrated transcriptomic profiling of the CFA-induced inflammatory pain model by analyzing gene expression, DNA methylation, and post-transcriptional regulation. Our results indicate that AP-1 plays a central role in regulating pain-related signaling as a key mediator in the inflammatory response. 9
CCL2 (C-C Motif Chemokine Ligand 2), also known as MCP-1 (Monocyte chemoattractant protein-1), is a critical chemokine involved in regulating nociception by activating its primary receptor, CCR2 (C-C Chemokine Receptor 2). The CCL2/CCR2 axis has been extensively investigated in the context of inflammatory pain and plays a significant role in modulating both peripheral and central inflammation. 10 Research has demonstrated that AP-1, as a transcription factor, binds to the MCP-1 promoter region, and its activation can lead to the transcriptional activation of MCP-1. 11 However, it remains unclear whether AP-1/c-Jun can enhance CCL2 and CCR2 activation in inflammatory pain and how this may influence the onset and progression of inflammatory pain. This study aims to further investigate the impact of AP-1/c-Jun on the CCL2/CCR2 pathway in an inflammatory pain model and to elucidate the specific mechanisms underlying inflammatory pain.
Curcumin is a natural compound extracted from the roots of the ancient herb turmeric. 12 Most of curcumin’s medicinal effects are attributed to its anti-inflammatory, analgesic, and antioxidant properties. 13 Curcumin has been shown to exert its beneficial effects through various transcription factors, including NF-κB (Nuclear factor κB), AP-1, and Notch-1. 14 Accumulating evidence demonstrates the significant antinociceptive effects of curcumin across various pain models, including pathological pain and CFA-induced inflammatory pain in rats.15,16 Additionally, preparations of curcumin with oral bioavailability are generally considered safe for human use, 17 highlighting its therapeutic potential for pain management. However, the precise mechanisms underlying curcumin’s analgesic action in inflammatory pain remain unclear.
In this study, we aim to investigate the role of the AP-1/c-Jun-CCL2-CCR2 signaling pathway in inflammatory pain. Additionally, we assessed the analgesic effects of curcumin in rats with inflammatory pain by targeting and inhibiting the AP-1/c-Jun-CCL2-CCR2 pathway.
Material and methods
Animals
Studies have shown that chronic pain caused by inflammation does not affect everyone equally, with women exhibiting a higher prevalence than men. 5 Consequently, this research will use female rats. Adult female Sprague-Dawley (SD) rats weighing 200–220 g were obtained from Suzhou Xinuosai Biotechnology Company (Jiangsu, China). The rats were housed in a chamber with a regulated temperature (22 ± 2°C), relative humidity of 50%–60%, and a 12/12-h light/dark cycle. They had unrestricted access to food and water. The rats were given 1 week to acclimate to their new environment prior to the experiments. All experimental procedures were approved by the Animal Ethics Committee of Nanjing Medical University. We made every effort to minimize the use and suffering of animals.
Establishment of CFA-induced inflammatory pain model
In this experiment, CFA was injected into the left plantar region of the rats to establish an inflammatory pain model. CFA was obtained from Sigma (Beijing, China). After anesthetizing the rats with tribromoethanol (10 mL/kg), they were positioned in a supine position. The left posterior plantar skin was then disinfected with a cotton ball soaked in 75% alcohol. A needle containing 100 µL of CFA was inserted at a diagonal angle into the metatarsal region of the rat’s hind paw, nearly parallel to the foot pad. The 100 µL CFA solution was injected, and immediately after withdrawing the needle, gentle pressure was applied to the injection site using a thumb to seal the pinhole. The rat was kept in the supine position for 3 min to prevent drug leakage before being returned to its cage. Following the injection, signs of successful modeling included swelling, redness of the skin, agitation, foot licking, and lameness in the left foot. The vehicle-injected group received an equal volume of normal saline solution, with all other procedures being identical.
Drugs and administration
T-5224, an AP-1 inhibitor, and RS 504393, a CCR2 antagonist, were purchased from MedChemExpress (Shanghai, China). Curcumin was obtained from Selleck Chemicals (Shanghai, China), and recombinant rat CCL2 protein was sourced from Novoprotein (Jiangsu, China). T-5224 was dissolved in dimethyl sulfoxide (DMSO) and diluted to a concentration of 500 µg/20 µL. RS 504393 was also dissolved in DMSO and diluted to 20 µg/20 µL. Curcumin was diluted in DMSO to achieve three concentrations: 100 µg/20 µL, 500 µg/20 µL, and 1 mg/20 µL. The recombinant rat CCL2 protein was reconstituted in sterile PBS with 0.1% bovine serum albumin to a concentration of 1 µg/10 µL.
Experimental grouping
Experiment 1
To establish a rat model of inflammatory pain using CFA and to investigate changes in the mechanical pain threshold over time, we divided the rats into two groups: the Vehicle-injected group and the CFA-injected group. The mechanical pain threshold was measured at 1, 3, 5, 7, 10, and 14 days after the injection (n = 6).
Experiment 2
To investigate the endogenous expression and cellular localization of c-Jun, p-c-Jun, CCR2, and CCL2 in CFA-injected rats, the rats were randomly divided into five groups: Vehicle-injected group, CFA1d group, CFA3d group, CFA7d group, and CFA14d group, for Western blot analysis (n = 6). Effects of intrathecal injection on 50% PWT in left hind foot of rats were detected by Von Frey filament (n = 6). Moreover, rats from the CFA3d group were selected for immunofluorescence assays to determine the cellular localization of c-Jun, p-c-Jun, CCL2, and CCR2.
Experiment 3
To further elucidate the roles of AP-1/c-Jun, CCL2, and CCR2 in CFA-injected rats, the rats were randomly divided into four groups: Vehicle-injected group, CFA-injected group, CFA + RS 504393 group, and CFA + T-5224 group. T-5224 (500 μg/20 μL) and RS 504393 (20 μg/20 μL) were administered intrathecally once daily from the second postoperative day to the fourth postoperative day to evaluate alterations in the mechanical pain threshold (n = 6). Effects of intrathecal injection on 50% PWT in left hind foot of rats were detected by Von Frey filament. At the end of the study, samples were collected for Western blot and immunofluorescence analyses to detect the expressions of c-Jun, p-c-Jun, CCL2, and CCR2 (n = 6).
Experiment 4
To further investigate the upstream and downstream relationship between AP-1/c-Jun and CCL2/CCR2, the rats were randomly divided into four groups: CFA-injected group, CFA + CCL2 group, CFA + CCL2 + T-5224 group, CFA + T-5224 group. T-5224 (500 μg/20 μL) and recombinant rat CCL2 protein (1 μg/10 μL) were administered intrathecally once daily from postoperative day 2 through 4 to assess changes in the mechanical pain threshold (n = 6). Effects of intrathecal injection on 50% PWT in left hind foot of rats were detected by Von Frey filament. At the conclusion of the study, samples were collected for Western blot and immunofluorescence analyses to detect the expression and localization of c-Jun, p-c-Jun, CCL2, and CCR2 (n = 6).
Experiment 5
To investigate whether repeated intrathecal injections of curcumin can alleviate established inflammatory pain in CFA-injected rats by inhibiting the AP-1/c-Jun-CCL2-CCR2 pathway, the rats were randomly divided into five groups: Vehicle-injected group, CFA-injected group, CFA + Cur100 μg group, CFA + Cur500 μg group, and CFA + Cur1 mg group (n = 6). Effects of intrathecal injection on 50% PWT in left hind foot of rats were detected by Von Frey filament. Western blot analysis was performed to detect the expression of c-Jun, p-c-Jun, CCL2, and CCR2 in these groups, and Immunofluorescence was used to assess the content and localization of these proteins (n = 6).
Intrathecal injection
SD rats were anesthetized with pentobarbital, and their backs were shaved and sterilized with 75% ethanol. The rats were then positioned on a plastic tube to widen the intervertebral space. A 25-µL micro-syringe needle filled with the drug was inserted into the L5-6 intervertebral space. The correct subarachnoid placement of the needle tip was confirmed by observing tail-flick or paw-flick responses immediately after needle insertion. The needle was then left in place for an additional 15 s to ensure complete drug delivery.
Pain behavior test
The mechanical paw withdrawal threshold was measured using the method described by Chaplan et al. 18 Rats were placed in a glass box and allowed to acclimate for 30 min. After this adaptation period, the middle plantar regions of the hind limbs were stimulated vertically with von-Frey filaments of varying forces (1.0, 1.4, 2.0, 4.0, 6.0, 8.0, 10.0, 15.0, 26.0 g). A positive response was defined as lifting or licking of the foot; otherwise, the reaction was considered negative. If a 2.0 g filament produced a negative response, the next higher force filament was applied. Each stimulation lasted for 3 s, with a 25-s interval between stimuli of different forces. After the first crossover from negative to positive responses, four additional consecutive measurements were taken. The series of data were recorded and analyzed to determine the Dixon-defined 50% paw withdrawal threshold (PWT). Measurements were repeated three times for each rat, and the mean of the three results was calculated to provide the final 50% PWT.
Western blot
After extracting the L4-L6 segments of the rat spinal cord, we prepared lysates using protease inhibitor complexes and RIPA lysis buffer. The tissues were mixed at a ratio of 100 mg of tissue to 1 mL of lysis buffer. The tissue was homogenized using a freezing grinder, and the homogenate was prepared and kept on ice. After vortexing and centrifugation, the supernatant was collected to obtain the protein. Protein content in the samples was quantified using the BCA Protein Assay Kit (Beyotime, Shanghai, China). The samples were then denatured by boiling for 10 min with 5X loading buffer. Equal amounts of protein were subjected to SDS-PAGE for electrophoresis. After transferring the proteins to a membrane, the membrane was blocked with 5% skim milk for 2 h at room temperature. Primary antibodies were then incubated overnight at 4°C: rabbit anti-c-Jun antibody (9165S, Cell Signaling, 1:1000), rabbit anti-p-c-Jun antibody (91952S, Cell Signaling, 1:1000), rabbit anti-CCR2 antibody (16153-1-AP, Proteintech, 1:1000), rabbit anti-CCL2 antibody (26161-1-AP, Proteintech, 1:500), and mouse anti-tubulin antibody (1:3000). The membranes were washed three times with TBST. After washing, secondary antibodies conjugated to horseradish peroxidase were incubated with the membranes for 2 h at room temperature. Following another round of washing, band signals were visualized using enhanced chemiluminescence substrates and quantified using Image-Pro Plus. Tubulin was used as an internal reference for quantification.
Immunofluorescence
After the L4-L6 segments of the rat spinal cord were extracted, they were placed in 4% paraformaldehyde overnight. This was followed by alcohol gradient dehydration and paraffin embedding of the tissues. Sections were blocked with PBS containing 1% bovine serum albumin for 2 h at room temperature after sectioning and subsequently incubated overnight at 4°C in different antibodies: rabbit anti-c-Jun antibody (9165S, Cell Signaling, 1:100), rabbit anti-p-c-Jun antibody (91952S, Cell Signaling, 1:100), rabbit anti-CCR2 antibody (16153-1-AP, Proteintech, 1:100), rabbit anti-CCL2 antibody (26161-1-AP, Proteintech, 1:500), goat anti-iba1 antibody (microglia marker, ab5076, Abcam, 1: 100), mouse anti-NeuN antibody (neuronal marker, GB11138, Servicebio, 1: 100), and mouse anti-GFAP antibody (astrocyte marker, 3670S, Cell Signaling, 1:100). The sections were then incubated with fluorescent secondary antibodies for 1 h at room temperature. We observed the resulting images of the left spinal dorsal horn under a confocal fluorescence microscope (NIKON ECLIPSE C1, Japan). The intensity was analyzed using Zeiss Zen.
Statistical analysis
All data analyses were conducted using GraphPad Prism 8 software, and results are presented as the mean ± standard deviation (SD). Repeated measures two-way analysis of variance (two-way ANOVA) was employed to compare pain behavior test results among different groups of rats. One-way ANOVA was used to analyze Western blot and immunofluorescence results. A p-value of < 0.05 was considered statistically significant.
Results
CFA induces inflammatory pain
We used CFA to establish an inflammatory pain model and investigated the behavioral performance related to inflammatory pain in rats that were in either the vehicle-injected group or the CFA-injected group at the specified time points. The results showed that the mean paw-withdrawal thresholds remained relatively stable in the vehicle-injected group throughout the observation period. In contrast, the thresholds significantly decreased in CFA-injected rats as early as day 1, reached their lowest point by day 3, and remained at this low level until day 14, the final day of measurement (Figure 1(a)).
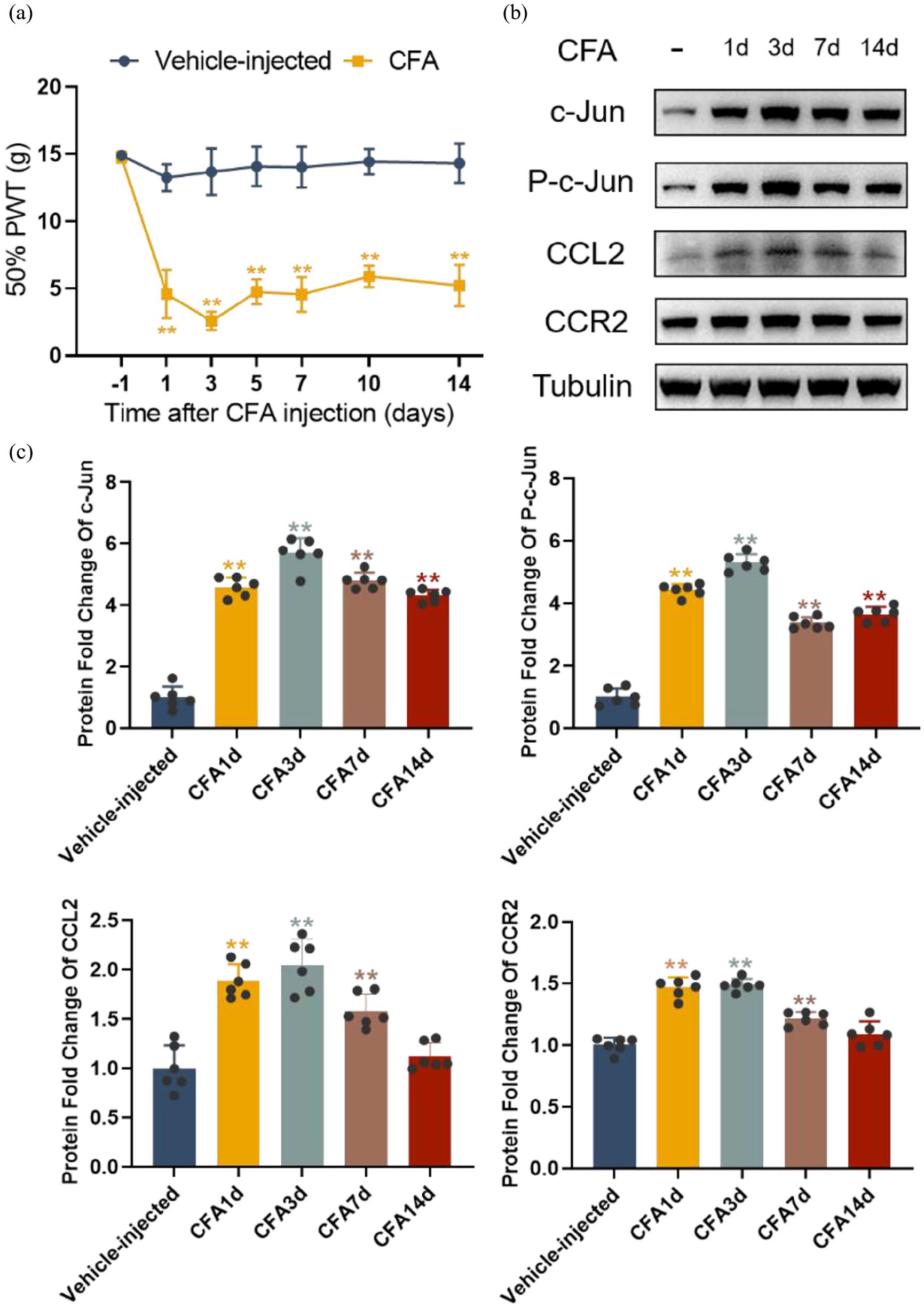
CFA induces mechanical hyperalgesia and alters spinal cord protein expression in rats. (a) Paw withdrawal thresholds (PWTs) of rats in each group on different days (n = 6). (b) Western blot analysis of c-Jun, p-c-Jun, CCL2, and CCR2 in the spinal cord of vehicle-injected and CFA-injected rats on days −1, 1, 3, 5, 7, 10, and 14 after CFA injection. (c) Quantitative analysis of the relative expressions of c-Jun, p-c-Jun, CCL2, and CCR2 in the spinal cords of vehicle-injected and CFA-injected rats on days 1, 3, 7, and 14 after CFA injection (n = 6).
Expression and localization of c-Jun, p-c-Jun, CCL2, and CCR2 in the spinal dorsal horn of the CFA – induced inflammatory pain model
To detect the endogenous expression and cellular localization of c-Jun, p-c-Jun, CCL2, and CCR2 in the spinal dorsal horn, lumbar spinal cord segments (L4-L6) from rats were collected at 1, 3, 7, and 14 days following the complete establishment of the CFA model and analyzed by Western blotting and immunofluorescence. Western blotting results showed a significant increase in c-Jun and p-c-Jun levels on days 1, 3, 7, and 14 after CFA injection compared to the vehicle-injected group. The expression levels began to increase on day 1, reached a peak on day 3, and remained at an elevated state until day 14. The upregulation of CCL2 and CCR2 in the spinal cord followed a similar temporal pattern to that of c-Jun and p-c-Jun (Figure 1(b) and (c)). To determine the cellular localization of c-Jun, p-c-Jun, CCL2, and CCR2 in the spinal dorsal horn, we performed co-staining with neuronal and glial markers (NeuN, Iba1, and GFAP). The findings indicated that c-Jun, p-c-Jun, CCL2, and CCR2 were primarily localized in neurons, with CCL2 and CCR2 also being expressed in astrocytes (Figures 2 and 3). These results suggest that the AP-1/c-Jun, CCL2, and CCR2 pathways play a role in CFA-induced inflammatory pain.
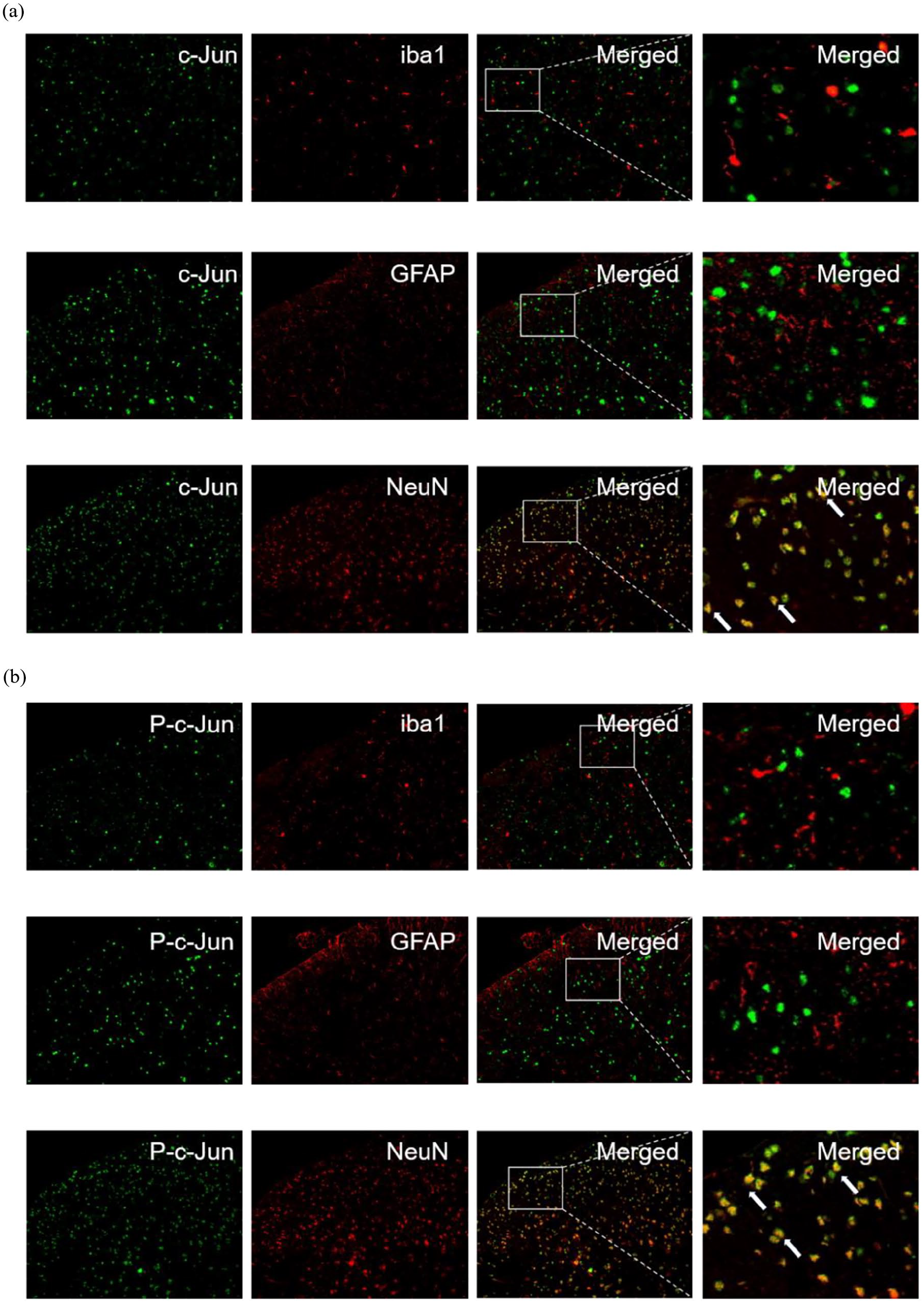
Endogenous expression and cellular localization of c-Jun and p-c-Jun in the spinal cord of CFA-injected rats. (a) and (b) Double-label immunofluorescence assays were used to detect the colocalization of c-Jun and p-c-Jun with NeuN, Iba1, and GFAP in CFA-injected rats.

Endogenous expression and cellular localization of CCL2 and CCR2 in the spinal cord of CFA-injected rats. (a) and (b) Double-label immunofluorescence assays were used to detect the colocalization of CCL2 and CCR2 with NeuN, Iba1, and GFAP in CFA-injected rats.
AP-1/c-Jun, CCL2, and CCR2 play an important role in CFA-injected rats
For a deeper understanding of the influence of AP-1/c-Jun in CFA-injected rats, T-5224 was administered once daily for three consecutive days starting from day 2 to day 4. The outcomes indicated that T-5224 significantly mitigated the mechanical pain developed in CFA-injected rats (Figure 4(a)). We further investigated whether inhibition of AP-1/c-Jun influenced the expression of CCL2 and CCR2 in CFA-injected rats. Western blot analyses revealed that repeated intrathecal injections of T-5224 significantly lessened the upregulation of CCL2 and CCR2 in the spinal cord (Figure 4(b) and (c)). Immunofluorescence findings further demonstrated a decrease in the expression of CCL2 and CCR2 in the spinal dorsal horn (Figure 5(a) and (b)). Additionally, we utilized the CCR2 antagonist RS 504393 to observe its effects on CFA-induced inflammatory pain. RS 504393 was administered intrathecally once a day for three consecutive days starting from day 2 to day 4. A comparison of the PWTs of the treatment and control group rats indicated that RS 504393 reduced pain behavior (Figure 6(a)). Our Western blot results showed that repeated intrathecal injections of RS 504393 significantly reduced the up-regulation of CCL2 and CCR2 in the spinal cord without affecting the expression of c-Jun and p-c-Jun (Figure 6(b) and (c)), and immunofluorescence results corroborated these findings (Figure 7(a) and (b)). These results suggest that AP-1/c-Jun, CCL2, and CCR2 play crucial roles in the development of CFA-induced inflammatory pain, with CCL2 and CCR2 potentially functioning as downstream effectors of AP-1/c-Jun that regulate the progression of CFA-induced inflammatory pain.

Intrathecal injection of the AP-1 inhibitor T-5224 attenuates CFA-induced inflammatory pain and suppresses the expression of c-Jun, p-c-Jun, CCL2, and CCR2. (a) Effect of intrathecal injection of T-5224 on PWT (n = 6). (b) Western blot analysis of c-Jun, p-c-Jun, CCL2, and CCR2 in spinal cords following intrathecal injection. (c) Quantitative analysis of the relative expressions of c-Jun, p-c-Jun, CCL2, and CCR2 in the spinal cord (n = 6). T-5224 (500 μg/20 μL, i.t.) was administered once daily on days 2, 3, and 4 after CFA injection. PWTs were measured 4 h after each injection. L4-L6 spinal tissues were collected 4 h after the last injection.
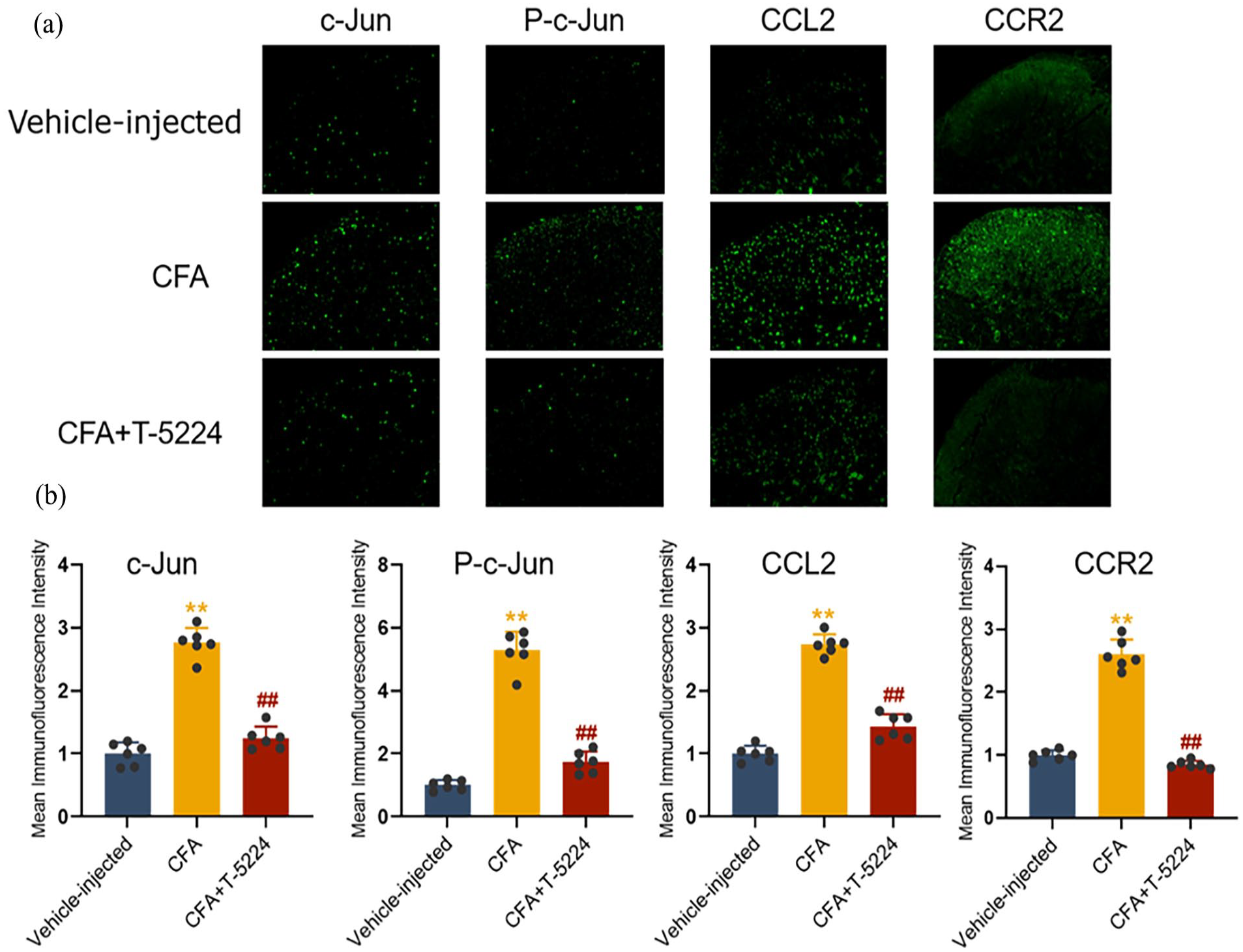
Intrathecal injection of the AP-1 inhibitor T-5224 suppresses the expression of c-Jun, p-c-Jun, CCL2, and CCR2 in the spinal cord. (a) Immunofluorescence analysis of c-Jun, p-c-Jun, CCL2, and CCR2 in the spinal cord following intrathecal injection. (b) Mean fluorescence intensity of c-Jun, p-c-Jun, CCL2, and CCR2 in the spinal cord (n = 6).

Intrathecal injection of the CCR2 antagonist RS 504393 attenuates CFA-induced inflammatory pain and suppresses the expression of CCL2 and CCR2. (a) Effect of intrathecal injection of RS 504393 on PWT (n = 6). (b) Western blot analysis of c-Jun, p-c-Jun, CCL2, and CCR2 in spinal cords following intrathecal injection. (c) Quantitative analysis of the relative expressions of c-Jun, p-c-Jun, CCL2, and CCR2 in the spinal cord (n = 6). RS 504393 (20 μg/20 μL, i.t.) was administered once daily on days 2, 3, and 4 after CFA injection. PWTs were measured 4 h after each injection. L4-L6 spinal tissues were collected 4 h after the last injection.
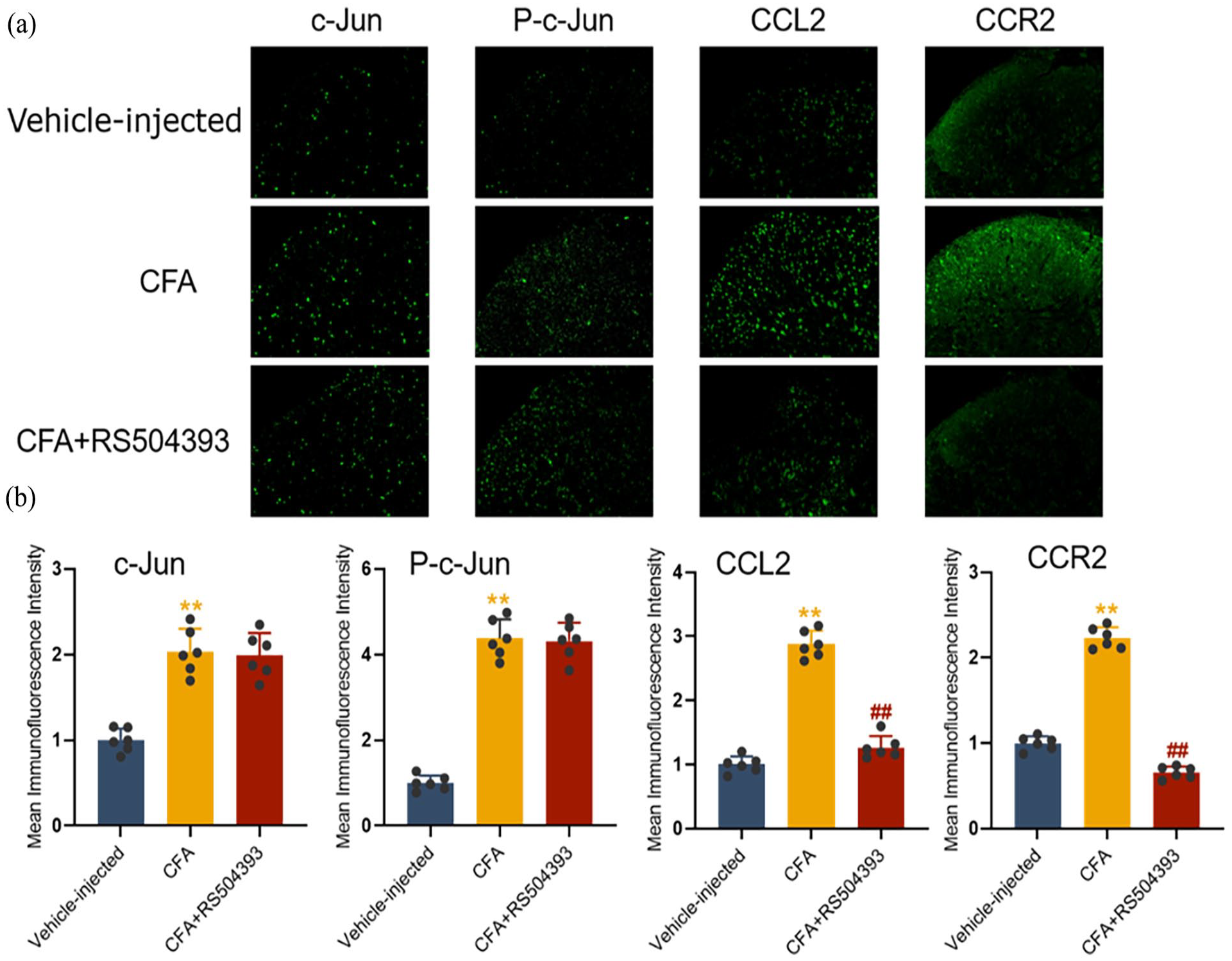
Intrathecal injection of the CCR2 antagonist RS 504393 suppresses the expression of CCL2 and CCR2 in the spinal cord. (a) Immunofluorescence analysis of c-Jun, p-c-Jun, CCL2, and CCR2 in the spinal cord following intrathecal injection. (b) Mean fluorescence intensity of c-Jun, p-c-Jun, CCL2, and CCR2 in the spinal cord (n = 6).
The relationship of AP-1/c-Jun, CCL2, and CCR2 in CFA-injected rats
To further investigate the upstream and downstream relationship between AP-1/c-Jun and CCL2/CCR2, recombinant rat CCL2 protein and T-5224 were administered once daily for 3 consecutive days, from day 2 to day 4. Repeated intrathecal injections of recombinant rat CCL2 protein significantly increased mechanical pain in rats compared to the CFA-injected group, while T-5224 treatment reversed this effect (Figure 8(a)). Our Western blot and immunofluorescence results demonstrated that repeated intrathecal injections of recombinant rat CCL2 protein significantly elevated the expression of CCL2 and CCR2 in the spinal cord, whereas the expression of c-Jun and p-c-Jun remained unaffected (Figure 8(b) and (c)–10). These results strongly suggest that AP-1 in the spinal cord of rats influences CFA-induced pain by regulating the expression of the functional downstream targets CCL2 and CCR2.

Intrathecal injection of recombinant rat CCL2 protein enhances CFA-induced inflammatory pain and increases the expression of CCL2 and CCR2, whereas T-5224 treatment reverses these effects. (a) Repeated intrathecal injections of recombinant rat CCL2 protein significantly increased mechanical pain in rats compared to the CFA-injected group, while T-5224 treatment reversed this effect (n = 6). (b) Western blot analysis of c-Jun, p-c-Jun, CCL2, and CCR2 in spinal cords following intrathecal injection. (c) Quantitative analysis of the relative expressions of c-Jun, p-c-Jun, CCL2, and CCR2 in the spinal cord (n = 6). Recombinant rat CCL2 protein (1 µg/10 µL, i.t.) or T-5224 (500 µg/20 µL, i.t.) was administered once daily on days 2, 3, and 4 after CFA injection. PWTs were measured 4 h after each injection. L4-L6 spinal tissues were collected 4 h after the last injection.

Immunofluorescence analysis of c-Jun, p-c-Jun, CCL2, and CCR2 in the spinal cord following intrathecal injection.

Mean fluorescence intensity of c-Jun, p-c-Jun, CCL2, and CCR2 in the spinal cord (n = 6).
Intrathecal injection of curcumin attenuates CFA-induced inflammatory pain via inhibition of the expression of AP-1/c-Jun, CCL2, and CCR2
The data suggest that curcumin exerts a strong analgesic effect on CFA-induced inflammatory pain. 15 To investigate the potential mechanisms underlying the antinociceptive effects of curcumin in CFA-injected rats, we examined its impact on the expression of AP-1/c-Jun, CCL2, and CCR2 in the spinal cord. Intrathecal injection of curcumin for three consecutive days reversed the established paw withdrawal thresholds (PWTs), demonstrating its efficacy in attenuating inflammatory pain (Figure 11(a)). Furthermore, we explored whether curcumin mediates its analgesic effects through the AP-1-CCL2/CCR2 signaling pathway. Western blot analysis revealed that repeated intrathecal injections of curcumin significantly reduced the upregulation of c-Jun, p-c-Jun, CCL2, and CCR2 in the spinal cord (Figure 11(b) and (c)). Immunofluorescence results showed that curcumin treatment significantly decreased the expression of c-Jun, p-c-Jun, CCL2, and CCR2 in the spinal dorsal horn (Figure 12(a) and (b)). These findings indicate that curcumin effectively alleviates CFA-induced inflammatory pain by markedly inhibiting the AP-1-CCL2/CCR2 signaling pathway.

Intrathecal injection of curcumin attenuates CFA-induced inflammatory pain and suppresses the expression of c-Jun, p-c-Jun, CCL2, and CCR2. (a) Effect of intrathecal injection of curcumin on paw withdrawal thresholds (PWTs; n = 6). (b) Western blot analysis of c-Jun, p-c-Jun, CCL2, and CCR2 in spinal cords following intrathecal injection. (c) Quantitative analysis of the relative expressions of c-Jun, p-c-Jun, CCL2, and CCR2 in the spinal cord (n = 6). Curcumin (100 µg/20 µL, 500 µg/20 µL, and 1 mg/20 µL, i.t.) was administered once daily on days 2, 3, and 4 after CFA injection. PWTs were measured 4 h after each injection. L4-L6 spinal tissues were collected 4 h after the last injection.
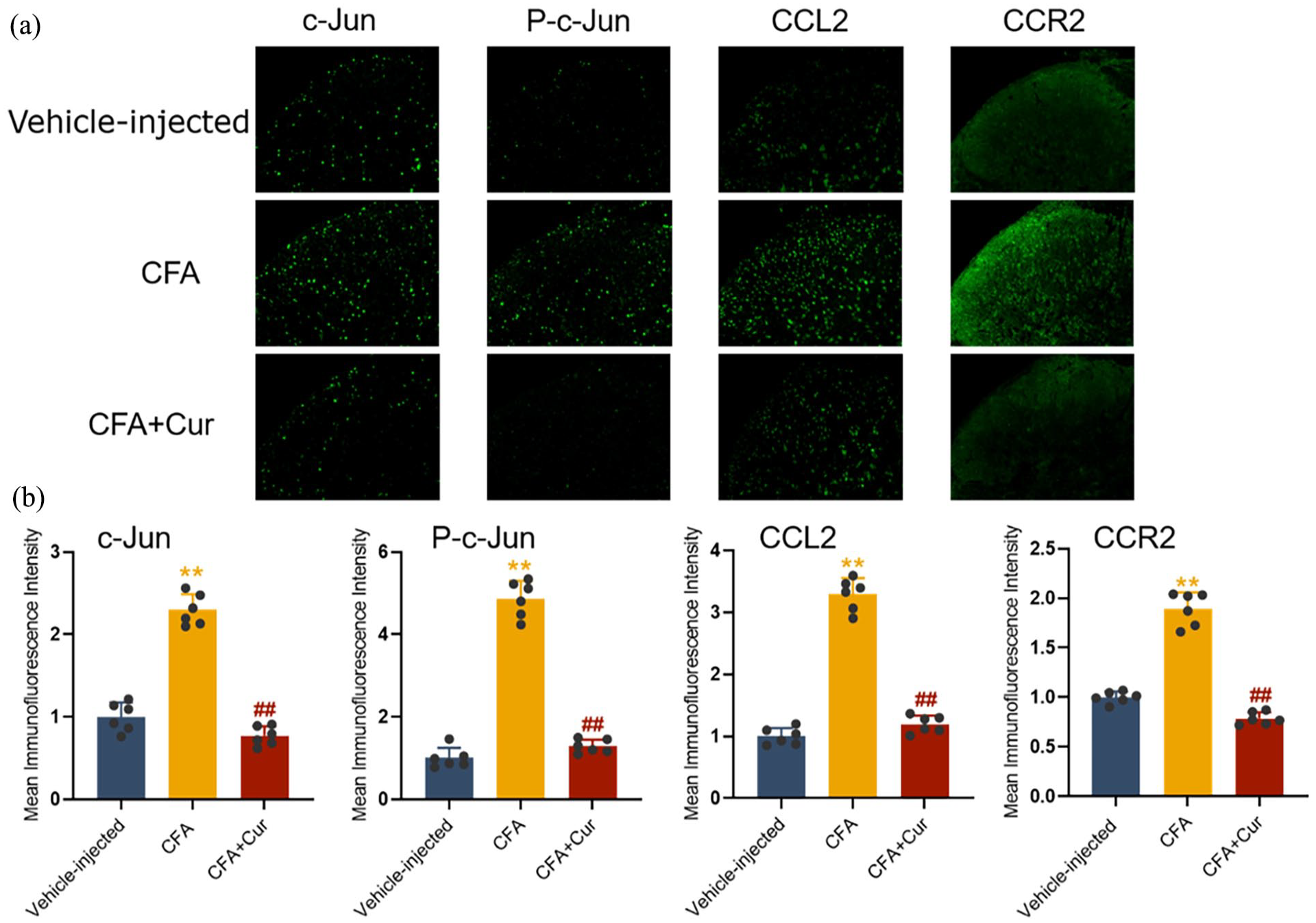
Intrathecal injection of curcumin suppresses the expression of c-Jun, p-c-Jun, CCL2, and CCR2 in the spinal cord. (a) Immunofluorescence analysis of c-Jun, p-c-Jun, CCL2, and CCR2 in the spinal cord following intrathecal injection. (b) Mean fluorescence intensity of c-Jun, p-c-Jun, CCL2, and CCR2 in the spinal cord (n = 6).
Discussion
Acute inflammation is intimately linked with the development of acute pain, acute inflammation is essential for protecting our bodies from invading pathogens, as well as promoting tissue remodeling and repair.2,19 However, quite a number of acute inflammatory pain will turn into chronic pain. 3 Pain associated with chronic inflammation is a globally – focused health problem and seriously affects people’s quality of life. 5 Consequently, effective treatment of inflammatory pain is of utmost importance. The mechanisms underlying inflammatory pain are highly complex, involving neuronal sensitization and alterations in various signaling pathways.20,21 Numerous studies have indicated that chemokines, their receptors, and the AP-1/c-Jun pathway play significant roles in the onset and progression of inflammatory pain.8,9 Our study has demonstrated that the AP-1/c-Jun pathway activates CCL2, which then interacts with CCR2 in neurons following CFA-induced injury. The AP-1/c-Jun -CCL2-CCR2 axis may serve as a target for alleviating CFA-induced inflammatory pain. Additionally, we investigated the specific mechanism through which curcumin exerts its analgesic effects on inflammatory pain via the AP-1/c-Jun-CCL2-CCR2 signaling pathway in the spinal cord.
AP-1 is a transcription factor that consists of different components, such as the Jun family, Fos family, and activating transcription factor (ATF) family. 22 The Jun and Fos subfamilies are the most predominant. While Jun proteins can form both homo- and heterodimers with other proteins, Fos proteins can only heterodimerize with Jun proteins. 23 AP-1 can be activated as a proinflammatory factor, the activation of AP-1 can regulate the transcription of downstream target genes. 24 In our study, the expression levels of c-Jun and p-c-Jun proteins began to increase 1 day after CFA injection, peaked on day 3, and remained elevated in the spinal cord until day 14. On day 3 post-CFA injection, c-Jun and p-c-Jun were co-localized with neurons. Furthermore, inhibiting AP-1/c-Jun activation also alleviated CFA-induced inflammatory pain. Thus, our results suggest that inhibiting AP-1/c-Jun in the neurons of the spinal cord alleviates central sensitization and CFA-induced inflammatory pain.
CCL2, also known as MCP-1, is from the family of chemokines. It has a vital role in the process of inflammation, where it attracts inflammatory cells and enhances the expression of other inflammatory factors. It leads to the advancement of many disorders by this main mechanism of migration and infiltration of inflammatory cells like monocytes and other cytokines at the site of inflammation. 25 CCR2 plays important roles in the extravasation and transmigration of monocytes under inflammatory conditions, and CCL2 is the most potent activator of CCR2 signaling. 26 In our study, we observed that CCL2 and CCR2 levels were significantly elevated in the spinal cords of CFA-injected rats, and this elevation was linked to the activation of AP-1/c-Jun. Previous research has demonstrated that AP-1 can mediate CCL2 activation in adipocytes. 27 Our immunofluorescence results indicate that c-Jun, p-c-Jun, CCL2, and CCR2 were predominantly co-localized with neurons in CFA-injected rats. Moreover, inhibiting AP-1/c-Jun activation led to a decrease in CCL2 and CCR2 expression in the spinal dorsal horn, whereas injections of recombinant rat CCL2 protein or RS 504393 did not affect AP-1/c-Jun expression. These findings suggest that activated AP-1/c-Jun in spinal cord neurons in CFA-injected rats signals to promote CCL2 activation, which in turn binds to the CCR2 receptor. Therefore, the AP-1/c-Jun-CCL2-CCR2 signaling axis in the spinal cord appears to regulate the development of CFA-induced inflammatory pain.
Curcumin, the major component of the Curcuma longa rhizome, has various pharmacological actions, including anti-inflammatory, immune-regulatory, anti-oxidative, and analgesia properties. 28 Studies show that chemokines and chemokine receptors can be modulated by curcumin, which mediates regulatory effects on inflammatory conditions in the systems of the body. 29 Curcumin can also adjust the AP-1 in the respiratory system, cancer, and inflammation, thereby playing its extensive pharmacological effects.14,30 However, little is known about the analgesic effect of curcumin through regulating AP-1 and chemokines. Therefore, we explored whether curcumin can inhibit the expression of AP-1/c-Jun-CCL2-CCR2 signaling pathways and alleviate inflammatory pain. Our results show that intrathecal injections of curcumin relieved the CFA-induced pain and also significantly reduced the expression of c-Jun, p-c-Jun, CCL2, CCR2 in the spinal dorsal horn of CFA-injected rats. On the basis of these results, we propose that curcumin alleviates inflammatory pain by inhibiting the AP-1/c-Jun-CCL2-CCR2 signaling pathway.
Conclusion
In summary, our findings show that curcumin can significantly alleviate CFA-induced inflammatory pain. This effect was achieved by inhibiting the activation of AP-1/c-Jun. Curcumin can alleviate CFA-induced inflammatory pain by inhibiting the activation of AP-1/c-Jun and decreasing the activation of CCL2, which further reduces the binding of CCL2 to the CCR2 receptor in the spinal cord. These findings highlight the therapeutic potential of curcumin in inflammatory pain. By targeting the AP-1/c-Jun-CCL2-CCR2 signaling pathway, curcumin provides a novel therapeutic option for inflammatory pain, indicating its feasibility for future clinical applications.
Footnotes
Author contribution
Yi Zhu: Writing – review & editing, Writing – original draft, Methodology, Investigation. Yinhong Jiang: Writing – review & editing, Methodology, Investigation. Xinyu Lu: Supervision, Investigation, Conceptualization. Siyu Li: Supervision, Investigation, Conceptualization. Fujiaying Liu: Methodology, Investigation, Conceptualization. Yidan Xu: Methodology, Investigation, Conce-ptualization. Liangliang Gao: Formal analysis. Yue Tian: Formal analysis. Lei Wei: Writing – review & editing, Methodology, Investigation, Funding acquisition, Conceptualization. All authors approved the final version of the manuscript. All authors listed have made a substantial, direct, and intellectual contribution to the work and approved it for publication.
Data availability
All the data used to support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding: This work was supported by Suzhou Gusu Medical Youth Talent (GSWS2023013).
Ethical considerations
All experimental procedures were approved by the Animal Ethics Committee of Nanjing Medical University.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
