Abstract
Chondromalacia, characterized by the softening of cartilage, is a prevalent condition affecting joint health with complex etiology. The immune system’s role in its pathogenesis has been implicated but remains to be fully elucidated. To address a critical knowledge gap, we conducted a two-sample Mendelian randomization analysis of 731 immune cell phenotypes, assessing parameters like fluorescence, cell count, and morphology. After sensitivity and pleiotropy checks, and applying a false discovery rate correction, our study linked 17 phenotypes to chondromalacia (p < .05). Among them, seven immune cell phenotypes were found to have a protective effect against chondromalacia (IVW: p < .05, OR <1), while 10 were considered risk factors (IVW:p < .05, OR >1). Despite the constraints of sample size and possible genetic differences among populations, our research has identified a notable genetic correlation between specific immune cell indicators and chondromalacia. This breakthrough sheds light on the pathophysiological mechanisms of the condition. The identification of protective and risk-associated immune cell phenotypes provides a foundation for further exploration of immunological mechanisms in chondromalacia and may pave the way for targeted interventions. Future research is warranted to validate these findings and explore their clinical implications.
Introduction
Chondromalacia, often referred to as “runner’s knee,” is a pathological condition that manifests as the softening and thinning of the articular cartilage, which is the smooth, protective tissue covering the ends of bones in synovial joints. 1 Predominantly affecting the knee joint, it is a common cause of joint pain and reduced mobility, particularly among young and active populations.2,3 The condition not only impacts the quality of life but also poses a significant socioeconomic burden due to its prevalence and the associated healthcare costs.
While chondromalacia has gained recognition in clinical circles, the precise pathophysiological mechanisms that lead to the degeneration of cartilage remain elusive. The current understanding of the disease is pieced together from a mosaic of observational studies and animal models, which provide valuable insights but fall short of offering a comprehensive picture. These studies have suggested that chondromalacia may arise from a confluence of factors, 4 including biomechanical stress, genetic predispositions, and biochemical imbalances within the joint.
The immune system’s role in joint diseases extends beyond its traditional function in combating infections and is increasingly implicated in tissue repair, inflammation, and disease progression. 5 In the context of chondromalacia, immune cells may exert both protective and detrimental effects on cartilage health, influencing the balance between anabolic and catabolic processes. 6 However, the precise nature of these effects and their causal relationship with chondromalacia remain to be elucidated.
Mendelian randomization, a powerful epidemiological tool, leverages the random assortment of genetic variants to establish causality between exposures and outcomes, bypassing many of the limitations associated with traditional observational research. 7 By employing genetic variants as instrumental variables, this approach provides a robust framework for investigating the causal pathways linking immune cell characteristics to chondromalacia.
In this study, we delve into the intricate relationship between immune cell phenotypes and chondromalacia using a two-sample, bidirectional Mendelian randomization analysis. By synthesizing data from two distinct cohorts and applying robust statistical methods, including False Discovery Rate (FDR) adjustments, we aim to uncover how specific immune cell characteristics may influence the development of chondromalacia, either protectively or risk-enhancingly. Our research offers fresh insights into the immunopathogenesis of chondromalacia, with potential to significantly impact the fields of rheumatology and musculoskeletal medicine. Identifying immune cell phenotypes causally associated with chondromalacia is crucial for developing targeted therapeutic interventions and personalized treatment approaches. Furthermore, our study contributes to the broader understanding of the immune system’s role in joint health and disease, aligning with cutting-edge research featured in premier scientific journals.
Materials and methods
Study design
In our research (Figure 1), we employed a two-sample Mendelian randomization approach to evaluate the causal interplay among 731 immune cell markers, divided into seven distinct groups. This Mr methodology leverages genetic variations as instrumental variables to deduce causal relationships. Credibility of these IVs hinges on fulfilling three critical conditions
8
: (1) a direct linkage between genetic variations and the exposures in question (immune cell markers); (2) independence from confounders that might skew the exposure-outcome relationship; (3) absence of influence on outcomes through pathways separate from the exposures. We ensured rigorous ethical oversight by including only studies approved by relevant institutional review boards and safeguarded participant autonomy through informed consent, predicated on a comprehensive understanding of the study’s aims, methodologies, inherent risks, and potential benefits. This approach upholds stringent ethical benchmarks while prioritizing participant rights and welfare. (1) Relevance: SNPs robustly associated wih exposure or outcome; (2) Independence: SNPs not associated with confounders; (3) Exclusion restriction: SNPs only associated with outcome through exposure; (4) Confounding Factors: Outcome and exposure may be influenced by confounding factors.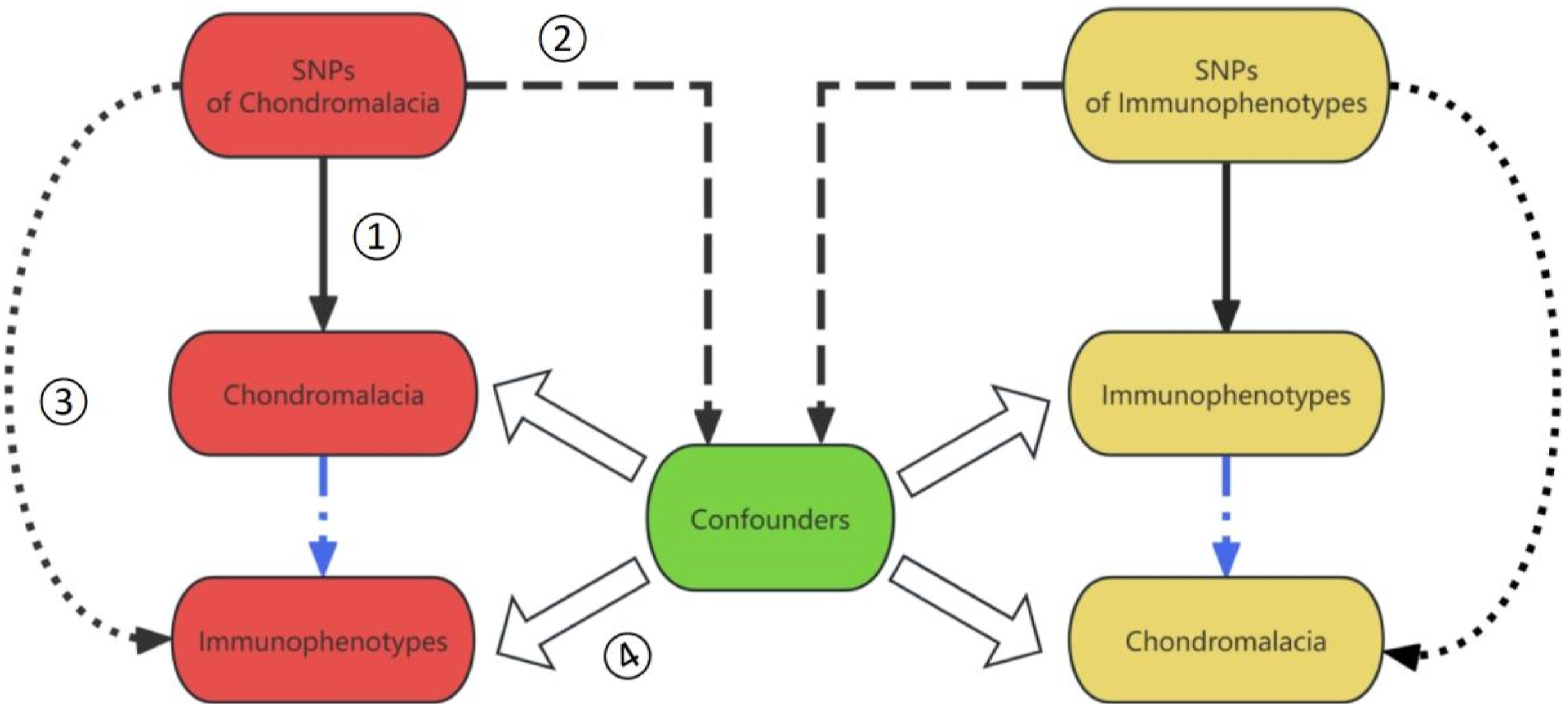
Genome-wide association study data sources for chondromalacia
In our investigation of the genetic underpinnings of chondromalacia, we leveraged genome-wide association study (GWAS) summary statistics from the FinnGen. In this data, the key details of a GWAS dataset for (https://gwas.mrcieu.ac.uk/datasets/finn-b-M13_CHONDROMALACIA/), labeled “finn-b-M13_CHONDROMALACIA,” are presented. 9 The dataset was published in 2021 and comprises a binary study type that focuses on European populations, including both males and females. The study utilized a substantial number of participants, with 432 cases and 215,335 controls, which allowed for a robust analysis of the genetic variants associated with chondromalacia.
A total of 16,380,460 single nucleotide polymorphisms (SNPs) were analyzed in the study. These genetic variations serve as markers for susceptibility to diseases. The use of the HG19/GRCh37 genome build ensures compatibility and standardization across different genomic analyses. The dataset is identified by the specific note or identifier “M13_CHONDROMALACIA,” facilitating easy reference and integration into larger genetic research endeavors.
Immunity-wide GWAS data sources
The GWAS Catalogue is a comprehensive repository that archives GWAS summary statistics for a plethora of immune traits, with accession numbers ranging from GCST0001391 to GCST0002121. 10 This expansive dataset encompasses 731 immunological phenotypes, including absolute cell (AC) counts (n = 118), median fluorescence intensities (MFI) that reflect surface antigen levels (n = 389), morphological parameters (MP) (n = 32), and relative cell (RC) counts (n = 192). The MFI, AC, and RC phenotypes represent a spectrum of immune cell types, encompassing B cells, conventional dendritic cells (CDCs), mature T cells, monocytes, myeloid cells, T cells, natural killer cells (NK), and regulatory T cells (Tregs). The MP phenotype is particularly focused on CDCs and NK panels.
The pioneering GWAS for immune traits was executed with data from 3757 European individuals, 11 ensuring no cohort overlap with this analysis. This approach allowed for a detailed exploration of the genetic underpinnings of immune phenotypes. To refine the genetic analysis, a high-density array was utilized to genotype approximately 22 million single nucleotide polymorphisms (SNPs), and the data were analyzed using a reference panel anchored in the Sardinian sequence to estimate genetic variations.
After controlling for potential confounders, including gender, age, and 2-year age groupings, the study delved into the associations between genotyped SNPs and the immune traits of interest. By integrating the modifying impacts of age and gender, the study offered a more sophisticated delineation of the genetic factors influencing immune phenotypes. This rigorous adjustment for confounders was pivotal in elucidating the genetic variants linked to immune traits, thereby reinforcing the robustness and broader relevance of the study’s conclusions.
Selection of IVs
In concordance with the latest strides in genetic epidemiology,12,13 we have delineated a stringent significance threshold of 1 × 10^−5 for IVs associated with each immune trait. This benchmark is pivotal for the standardized recognition of genetic elements that could modulate immune phenotypes.
To refine the precision of SNP selection and meticulously filter out those in high linkage disequilibrium (LD) with neighboring SNPs, we harnessed PLINK software (version 1.90). 14 This tool employs a sophisticated clustering algorithm, applying an LD r2 threshold of less than 0.001 within a 10,000 kb window, thereby effectively simplifying the genetic landscape and augmenting the fidelity of downstream analyses.
For the Chondroma trait, the threshold was further tightened to a more conservative level 15 of 1 × 10^−5. To rigorously assess the potency of the IVs and mitigate the impact of potential weak instrumental variables, we meticulously calculated the proportion of phenotypic variance explained and the F statistic for each IV. These quantitative measures are crucial for fortifying the integrity of our genetic instruments and for curtailing the likelihood of skewed causal inferences in our Mendelian randomization analysis.
Statistical analysis
All statistical analyses were performed using R 4.4.0 software, available on the R Project website (httpa://www.rproject.org/). To investigate the causal links between 731 immune phenotypes and chondromalacia, we deployed the inverse-variance weighted (IVW) method 16 alongside the weighted median 17 and pleiotropy-based approaches, 18 facilitated by the “MendelianRandomization” R package (version 0.4.4). 19
The IVW method adjusts the influence of each data point based on its statistical significance, ensuring a balanced analysis by proportionally weighting the contributions akin to the culinary principle of ingredient impact. We applied Cochran’s Q statistic and its associated p-value to evaluate the concordance of our instrumental variables (IVs), transitioning to a random-effects IVW model if significant heterogeneity was detected.
To address the possibility of horizontal pleiotropy, we integrated the MR-Egger method 20 as a sentinel for influential outliers and employed the MR-PRESSO method 21 to excise genetic variants that might skew our outcomes. Scatter and funnel plots were engaged for visual data scrutiny, identifying outliers and ensuring the robustness of our findings.
Results
Exploring the causal effects of chondromalacia onset on immunological phenotypes
In our study, we employed a two-sample Mendelian randomization analysis to explore the underlying causal influences of chondromalacia onset on immune phenotypes, with the inverse-variance weighted method serving as our principal analytical technique. Post-adjustment for multiple comparisons via the false discovery rate approach, no immune traits demonstrated significant correlations at the 0.2 threshold (Figure 2). The forest plot presents the causal associations between chondromalacia and diverse immune cell phenotypes. This forest plot illustrates the outcomes of an inverse-variance weighted Mendelian Randomization analysis, scrutinizing the causal links between Chondromalacia and a spectrum of immune cell phenotypes. Key elements depicted for each phenotype within the plot include: Trails: List of immunological phenotypes examined in the study; nsnp: The tally of Single Nucleotide Polymorphisms (SNPs) examined per phenotype; pval: The statistical measure representing the significance of the association; Odds Ratio (95% CI): The odds ratio accompanied by its 95% confidence interval, denoting the magnitude and precision of the estimated effect; FDR Adjustment: The False Discovery Rate correction applied to mitigate the impact of multiple comparisons.
Exploring the causal links between immunological phenotypes and chondromalacia
Following multiple testing corrections with the false discovery rate (FDR) approach and setting a stringent significance threshold at P FDR <0.05, our analysis identified 17 immune cell phenotypes within four categories that are causally linked to chondromalacia (p < .05) (Figure 3). Forest plot illustrating the causal relationships between immune cell traits and chondromalacia. This forest plot illustrates the outcomes of an inverse-variance weighted Mendelian Randomization analysis, scrutinizing the causal links between Chondromalacia and a spectrum of immune cell phenotypes. Key elements depicted for each phenotype within the plot include: Trails: List of immunological phenotypes examined in the study; nsnp: The tally of Single Nucleotide Polymorphisms (SNPs) examined per phenotype; pval: The statistical measure representing the significance of the association; Odds Ratio (95% CI): The odds ratio accompanied by its 95% confidence interval, denoting the magnitude and precision of the estimated effect; FDR Adjustment: The False Discovery Rate correction applied to mitigate the impact of multiple comparisons.
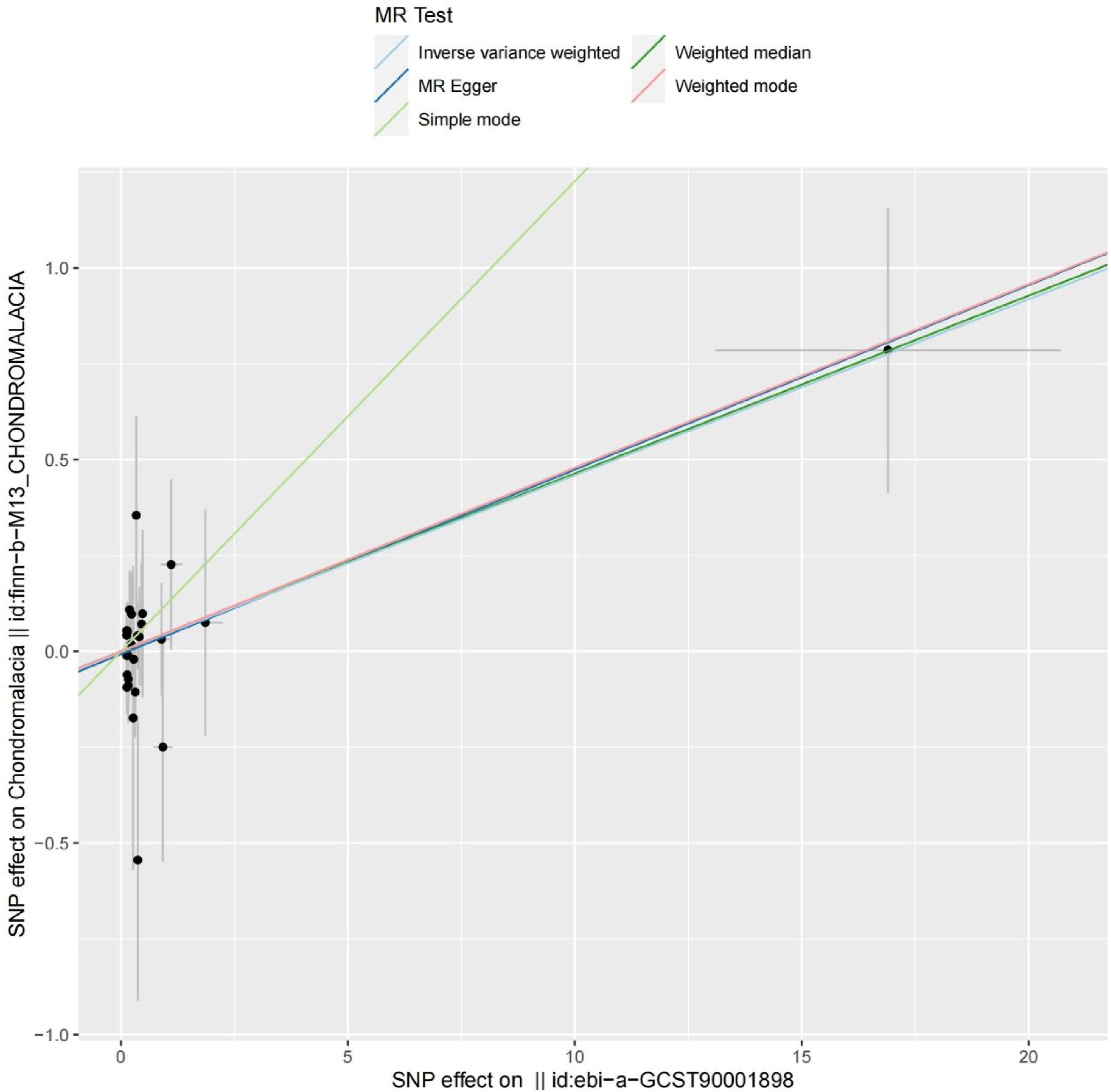
The MR-Egger regression intercept and the global test from MR-PRESSO provided no evidence of horizontal pleiotropy affecting the four significant associations, thus substantiating the robustness of our study outcomes. Additional support for the robustness of the observed causal links was furnished by consistency analyses, as illustrated in Figure 4. Scatter plots demonstrated the relative uniformity of effects across diverse analytical techniques, and the odds ratios (ORs) derived from multiple models were in close agreement. This methodological concordance underscores the reliability of our discoveries, as further detailed in Figure 5 for an encompassing perspective. Funnel plot analysis for the association between CD28 on CD4+ and chondromalacia. MR: Mendelian randomization. Scatter plot depicting the relationship between CD28 on CD4+ and chondromalacia. MR: Mendelian randomization; SNP: Single nucleotide polymorphism.
Discussion
Chondromalacia, a condition characterized by the softening of articular cartilage,2,22 predominantly impacts the knee joint and presents a significant clinical challenge due to its potential to cause persistent pain, functional impairment, and reduced quality of life. 23 Despite its prevalence and impact, the pathophysiological mechanisms that drive chondromalacia are not fully understood, and the condition remains a subject of ongoing research and debate within the medical community. 24
The relationship between immune cells and chondromalacia is complex and multifaceted. 25 Immune cells, such as T cells, B cells, and macrophages, are known to be present in the joint environment and are thought to contribute to the disease process through the production of cytokines, chemokines, and matrix-degrading enzymes. 26 These immune mediators can influence the balance between the synthesis and degradation of cartilage matrix components, potentially leading to the characteristic softening and degeneration observed in chondromalacia. 27
Moreover, the immune system’s involvement in chondromalacia may not be limited to the promotion of cartilage degradation. 5 There is evidence to suggest that certain immune responses may also play a protective role, 28 modulating the inflammatory process and promoting tissue repair. This duality in the immune system’s function highlights the need for a nuanced understanding of the specific immune cell phenotypes and their interactions with cartilage tissue. 29
Building on this premise, our Mendelian randomization study meticulously investigates the causal pathways that may link immune cell characteristics to the development of chondromalacia. By employing genetic variants as instrumental variables, our research transcends the traditional constraints of observational studies, 30 providing a clearer lens through which to examine the potential causal relationship between the immune cell profile and the risk of chondromalacia. This approach not only enhances our understanding of these genetic correlations but also fortifies the causal inference of the observed associations, 31 establishing a solid foundation for future work. In the following sections, we will delve into the mechanisms by which immune cell phenotypes may exert their influence on the pathogenesis of chondromalacia, offering insights into novel avenues for prevention and therapeutic intervention.
Classic dendritic cells (cDCs), as pivotal members of the immune system, play a crucial role in immune surveillance and antitumor immune responses through their unique cellular phenotypes. 32 Subpopulations of cDCs, particularly cDC1 and cDC2, participate in cell recognition and antigen presentation processes by expressing different surface markers, such as CD62L and HLA-DR. In this study, we discovered that CD62L- HLA-DR++ monocytes, CD62L on granulocytes, and CD80 on CD62L + myeloid DCs can inhibit the occurrence of chondromalacia. Based on existing immunological theories and literature, we explored the mechanisms by which these immune cell phenotypes inhibit the onset of chondromalacia. 1, Immune Modulation: cDC cells, especially monocytes expressing high levels of HLA-DR, may play a key role in the immune modulation of chondromalacia. 33 These cells activate the adaptive immune response by presenting antigens to T cells. The high expression of HLA-DR molecules enhances the antigen-presenting capacity of cDCs, which may help regulate inflammatory responses and prevent autoimmune attacks; 2, Anti-inflammatory Effects: CD80 (B7-1), an important provider of T cell co-stimulatory signals, is expressed on CD62L + myeloid DCs and may help regulate the activation threshold of T cells. This regulation could suppress excessive inflammatory responses and reduce damage to chondrocytes 33 ; 3, Cell Interaction: The expression of CD62L (L-selectin) may affect the homing of immune cells and their interactions with other cells. On granulocytes, the expression of CD62 L may regulate their recruitment and activity at inflammatory sites, impacting the inflammatory process; 4, Cartilage Protection: cDC cells may secrete factors with chondroprotective effects, such as IL-10, to promote the proliferation and matrix synthesis of chondrocytes and inhibit cartilage degradation 34 ; 5, Maintenance of Immune Tolerance: CD62 L + myeloid DCs may be involved in maintaining immune tolerance and preventing the occurrence of autoimmune reactions. 35 In chondromalacia, this could help prevent the immune system from mistakenly attacking its cartilage tissue; 6, Regulation of Cell Death and Apoptosis: Immune cells, particularly DC cells, may influence the survival of chondrocytes and the progression of the disease by regulating cell death pathways, such as apoptosis36,37; 7, Regulation of Chondrocyte Dedifferentiation: The dedifferentiation process of chondrocytes is closely related to the development of chondromalacia. cDC cells may regulate the regenerative capacity and functional state of chondrocytes by affecting their dedifferentiation process. 38
Myeloid cells, as integral components of the immune system, may play a pivotal role in the pathogenesis and progression of chondromalacia. Specific myeloid cell phenotypes, such as CD33dim HLA-DR + CD11b- AC, may reflect the unique functional states of these cells in the microenvironment. The phenotypic characteristics of these cells may indicate their multifaceted roles in immune regulation, angiogenesis, immune evasion, cell proliferation, and differentiation, as well as invasion and metastasis. The mechanisms by which this phenotype induces chondromalacia are as follows: 1, Promotion of Inflammatory Responses: Myeloid cells with the phenotype CD33dim HLA-DR + CD11b- AC may play a key role in the inflammatory process. 39 Upon recognition of injury or inflammatory signals, these myeloid cells can release a variety of inflammatory mediators, such as cytokines and chemokines, which recruit more immune cells to the site of injury, exacerbating the inflammatory response; 2, Degradation of Cartilage Matrix: Myeloid cells may directly participate in the degradation of the cartilage matrix by secreting matrix metalloproteinases (MMPs) and other proteases. 40 These enzymes can disrupt the extracellular matrix of cartilage, leading to the destruction of its structure; 3, Immune Regulation Imbalance: The expression of CD33 and HLA-DR may be related to the immune regulatory functions of myeloid cells. Changes in the expression levels of these molecules may affect the interactions between myeloid cells and T cells, leading to an imbalance in immune tolerance and promoting the occurrence of autoimmune reactions 38 ; 4, Alteration of Cellular Signaling: CD11b is an important molecule on the surface of myeloid cells, involved in various cellular signaling processes. 41 The absence or downregulation of CD11b may affect the functions of myeloid cells, including their adhesion, migration, and phagocytic capabilities, thereby influencing the course of chondromalacia; 5, Cell Differentiation and Phenotypic Shift: Myeloid cells exhibit high phenotypic plasticity and can undergo differentiation and phenotypic shifts based on signals from the microenvironment. 42 Myeloid cells with the phenotype CD33dim HLA-DR + CD11b- AC may play a different role in chondromalacia than conventional myeloid cells, such as affecting the inflammatory process by differentiating into specific subsets of immune cells; 6, Interference with Tissue Repair and Regeneration: Myeloid cells also play a role in tissue repair and regeneration. Myeloid cells with the phenotype CD33dim HLA-DR + CD11b- AC may interfere with normal tissue repair mechanisms, affecting the regenerative and reparative capacity of cartilage. 43
The maturation phases and phenotypes of T cells are crucial in immune responses, yet their role in chondromalacia remains unclear. Drawing from studies on arthritis and other autoimmune diseases, we hypothesize that specific T cell phenotypes, such as “TD CD8bright AC” and “Naive DN (CD4-CD8-) AC,” may facilitate the development and progression of chondromalacia through several mechanisms: 1, Maturation Stages of T Cells: T cell maturation initiates with lymphoid precursors in the thymus, undergoing various stages including the double-negative (DN), double-positive (DP), and single-positive (SP) stages, culminating in the differentiation into mature T cells44–46; 2, TD CD8bright AC Phenotype: TD (Thymus Dependent) denotes mature T cells reliant on the thymus, CD8bright refers to the intensity of CD8 molecule expression, and AC likely signifies an activated state. These cells may contribute to chondromalacia by directly interacting with chondrocytes or by secreting cytokines that influence inflammatory responses and cartilage metabolism47,48; 3, Naive DN (CD4-CD8-) AC Phenotype: This phenotype describes a group of immature, unactivated T cells that have experienced positive and negative selection in the thymus before entering peripheral circulation. These DN T cells, categorized into different subsets based on specific protein markers, may significantly modulate autoimmune responses in chondromalacia44–46; 4,Immune Modulation: T cells regulate the vigor and trajectory of immune responses through the secretion of cytokines, such as interferon-gamma (IFN-γ), tumor necrosis factor-alpha (TNF-α),49,50 and interleukins (ILs), potentially impacting chondrocyte metabolism and the synthesis and degradation of the cartilage matrix 51 ; 5, Autoimmune Reactions: T cells may erroneously identify joint cartilage as foreign, inciting autoimmune reactions that result in chondrocyte damage and cartilage matrix degradation; 6,Intercellular Interactions: Interactions between T cells and other immune cells, including B cells, monocytes, and dendritic cells, may influence the release of inflammatory mediators and the recruitment of immune cells within the joint, thereby affecting the course of chondromalacia; 7,Dedifferentiation of Chondrocytes: T cells may affect the phenotype and function of chondrocytes, promoting their dedifferentiation and reducing their numbers and functionality, contributing to the progression of chondromalacia, as highlighted by Professor Ouyang Hongwei’s team regarding the molecular landscape of chondrocyte dedifferentiation. 38
The TBNK cell consortium, including T cells, B cells, NK cells, and NKT cells, plays a pivotal role in immune responses and the pathogenesis and progression of diseases. This study investigates the potential connections between specific TBNK cell phenotypes and the development of chondromalacia, proposing a series of immunological mechanisms: 1, CD4 + T Cell Percentage: CD4 + T cells, also known as helper T cells, regulate immune responses through cytokine secretion, aiding B cells in antibody production to enhance the immune response. In chondromalacia, CD4 + T cells may mitigate inflammation by releasing anti-inflammatory cytokines such as IL-4 and IL-10, potentially preventing the disease’s onset1,52; 2, TCRgd T Cell Percentage: TCRgd T cells, with their unique T cell receptors, contribute to mucosal immunity and antitumor responses. They may protect against chondromalacia by directly targeting damaged or stressed chondrocytes or modulating the local immune environment via cytokine secretion1,52; 3, Double-Negative (DN) NKT Activated Cells: DN T cells, lacking CD4 and CD8 expression, may modulate immune responses. However, when combined with NKT cells, they may intensify inflammatory responses through the release of pro-inflammatory cytokines such as IFN-γ and TNF-α, causing chondrocyte damage and potentially advancing chondromalacia1,52; 4, Forward Scatter Area (FSC-A) on CD4+: FSC-A, a flow cytometry parameter for cell size measurement, may indicate increased cell volume on CD4 + T cells, associated with activation or abnormal proliferation. Activated CD4 + T cells could exacerbate inflammation and cartilage damage by secreting pro-inflammatory cytokines, thus promoting the development and progression of chondromalacia.1,52
Regulatory T cells (Treg cells) are essential immunomodulators that sustain immune tolerance and avert the development of autoimmune diseases. They exert a suppressive effect on the immune system through cell-to-cell contact and the secretion of inhibitory cytokines, thereby inhibiting immune responses and promoting active tolerance to self-antigens, preventing the onset of autoimmune conditions. The following explores various Treg cell phenotypes and their potential roles in chondromalacia, highlighting the intricacies of immune regulation. 1, CD28- CD25++ CD8bright %T cell: This Treg cell phenotype may enhance responsiveness to IL-2 by highly expressing CD25, allowing for local expansion and survival. CD8bright T cells, typically cytotoxic, may have been repurposed with regulatory functions in this context. They may prevent chondromalacia by secreting anti-inflammatory cytokines such as IL-10 and TGF-β, and by inhibiting the activity of other immune cells through contact-dependent mechanisms53,54; 2, CD28 on CD28+ CD4+: CD28 is a co-stimulatory molecule on T cells that, upon binding to B7 molecules on antigen-presenting cells, provides the necessary secondary signal for T cell activation. CD28+ CD4 + T cells may include effector T cells that, under CD28 co-stimulation, differentiate into Th1 or Th17 cells producing substantial inflammatory cytokines, potentially contributing to the occurrence and progression of chondromalacia 54 ; 3, CD28 on CD4+: CD4 + T cells, the primary subset of helper T cells, receive co-stimulatory signals in a CD28-dependent manner, potentially driving inflammatory responses. 54 In the context of chondromalacia, these cells may intensify cartilage damage by activating other immune cells like macrophages and B cells, and by secreting inflammatory cytokines, thus facilitating the development of chondromalacia.
B cells are instrumental in immune responses, particularly in the production of antibodies and the establishment of immune memory. Employing Mendelian randomization techniques, we have discovered a causal link between distinct B cell phenotypes and the onset and progression of chondromalacia. Drawing on current research, we have investigated the underlying mechanisms. 1, IgD + CD38- Unswitched Memory B Cells: This B cell phenotype likely represents unswitched memory B cells that express IgD without CD38. Possessing self-renewal and long-term viability, these cells can swiftly respond to the re-encounter of the same antigen. In chondromalacia, they may prevent disease onset by generating regulatory antibodies or through interactions with T cells that mitigate inflammation 55 ; 2, CD20 Expression on CD20- CD38- B Cells: CD20, a surface marker on B cells, is implicated in B cell activation and signaling. The presence of CD20 in this phenotype may be indicative of B cell maturity and differentiation status. B cells devoid of CD20 may be in a resting or early activation phase, and the lack of CD38 could impact their proliferation and differentiation. Such B cells might facilitate chondromalacia progression by differentiating into plasma cells that generate inflammatory antibodies, thus exacerbating the inflammatory response 56 ; 3, CD20 Expression on Unswitched Memory B Cells: Unswitched memory B cells with CD20 expression may engage in rapid immune responses. Upon initial antigen recognition, these cells can quickly expand and differentiate into plasma cells, producing a substantial quantity of antibodies. In the context of chondromalacia, these antibodies might target cartilage constituents, triggering an autoimmune response and cartilage damage57,58; 4, CD38 Expression on IgD + B Cells: CD38, a polysaccharide hydrolase enzyme involved in B cell signaling, is expressed on IgD + B cells in this phenotype, potentially signifying early switched B cells or plasma cells. These cells could contribute to chondromalacia progression by secreting inflammatory cytokines or antibodies, thereby intensifying joint inflammation and cartilage damage. 59
Our study has delved deeply into four pivotal immune-related parameters—Mean Fluorescence Intensity (MFI), Regulatory Cells (RC), Activation Markers (AC), and Memory Populations (mp). By employing bidirectional multifactorial regression (Mr), we have dissected the intricate interactions between these factors and chondromalacia. The outcomes of our research have forged causal links between chondromalacia and 17 distinct immune phenotypes, identifying seven that confer protective effects against the disease and 10 that correlate with heightened vulnerability.
This research has forged a pioneering path in investigating the nexus between immune cell phenotypes and chondromalacia, yet we recognize with clarity the limitations intrinsic to our study. The constraints of our sample size may impede the universality of our insights, and the intricate nature of immune cell phenotypes necessitates a more expansive sampling framework and profound analytical depth in subsequent inquiries. We recommend that subsequent studies employ multi-centric and multi-ethnic samples and apply more advanced analytical techniques, such as multivariate Mendelian randomization analysis, to comprehensively understand the relationship between immune cell phenotypes and chondromalacia. Additionally, although the Mendelian randomization approach provides us with a robust analytical tool, we must still adhere strictly to the principles of scientific rigor when selecting and utilizing instrumental variables. To this end, we suggest that future studies should screen instrumental variables more rigorously and consider potential confounding factors in the analysis, such as by using sensitivity analyses to assess the impact of different instrumental variables on the results. Lastly, we acknowledge that there may be other unidentified limitations in the current study, such as biases in data collection and analysis methods. We will further explore these potential issues in future research and employ appropriate statistical methods to correct or mitigate these biases.
As we look to the future, we are eager to transcend the existing constraints by expanding our research to encompass a diverse global population, further exploring the profound links between immune cell phenotypes and chondromalacia. We are confident that the convergence of cutting-edge technologies like genomics, proteomics, and metabolomics will offer a holistic view to decipher the intricate interactions within the disease microenvironment. Additionally, the advancement of precision medicine and the development of individualized therapeutic approaches will allow us to deliver more accurate and personalized treatment regimens for patients.
Moreover, our study’s findings on the immune system’s dual role in chondromalacia contribute to a nuanced understanding that aligns with evidence from studies highlighting the role of MSCs in joint diseases and repair. 60 The advancement of precision medicine and the development of individualized therapeutic approaches, as seen in the use of MSCs for treating joint disorders, 61 will allow us to deliver more accurate and personalized treatment regimens for patients.
In the realm of clinical practice, the evolution of precision medicine aligns with our findings, advocating for personalized treatment strategies that are informed by a deeper genetic and immunological understanding of each patient’s condition. This is a significant departure from a one-size-fits-all treatment approach, which has been the norm in many clinical settings.
Conclusion
In conclusion, our study represents a pivotal landmark, offering fresh insights into the immunological research domain of chondromalacia and charting new avenues for technological progress and clinical practice. As our comprehension of the sophisticated interactions between immune cells and pathological processes continues to evolve, in tandem with the advancement of innovative technologies, we are optimistic that the findings of our research will confer substantial improvements in patient outcomes and quality of life. These strides hold the promise of new horizons for global health and the collective well-being of humanity.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
