Abstract
The interaction between the immune system and the brain, crucial for blood–brain barrier integrity, is a potential factor in migraine development. Although there’s evidence of a connection between immune dysregulation and migraine, a clear causal link has been lacking. To bridge this knowledge gap, we performed a two-sample Mendelian randomization (MR) analysis of 731 immune cell phenotypes to determine their causality with migraine, of which parameters included fluorescence, cell abundance, count, and morphology. Sensitivity and pleiotropy checks validated our findings. After applying a false discovery rate correction, our MR study identified 35 of 731 immune phenotypes with a significant causal link to migraine (p < 0.05). Of these, 24 showed a protective effect (inverse variance weighting : p < 0.05, odds ratio <1), and 11 were risk factors (inverse variance weighting : p < 0.05, odds ratio >1). Although limited by population sample size and potential population-specific genetic variations, our study uncovers a significant genetic link between certain immune cell markers and migraine, providing new insights into the disorder’s pathophysiology. These discoveries are crucial for developing targeted biomarkers and personalized treatments. The research enhances our understanding of immune cells’ role in migraine and may substantially improve patient outcomes and lessen its socio-economic impact.
Introduction
Migraine, a prevalent neurological disorder characterized by recurrent episodes of severe headache and associated symptoms such as nausea, vomiting, and photophobia, poses a significant burden on both individuals and healthcare systems worldwide. 1 Despite its high prevalence and impact on quality of life, the pathophysiology of migraine remains incompletely understood. In recent years, there has been a growing appreciation for the role of the immune system in the development and progression of migraine, suggesting a complex interplay between genetic, environmental, and immunological factors. 2
The immune system, traditionally viewed as a peripheral player in migraine, is increasingly recognized for its central role in the initiation and perpetuation of migraine attacks. 3 Emerging evidence implicates immune cell signatures, particularly those of innate and adaptive immunity, in the pathogenesis of migraine. In migraines, macrophages interact with trigeminal ganglion cells, suggesting a physiological relationship.4–6 Changes in macrophage morphology, like increased circularity, often indicate activation. These cells, found near dural vessels and nerve endings, respond to calcitonin gene-related peptide (CGRP) released from trigeminal afferents.7,8 CGRP increases cAMP levels in macrophages, leading to cytokine release and sensitizing nearby nerve endings, a process known as neurogenic inflammation.9–11 CGRP also prevents inflammation in pretreated macrophages, maintaining their basal functions and promoting an anti-inflammatory state. 12 Additionally, CGRP aids wound healing by promoting anti-inflammatory cytokine expression and tissue repair, making it crucial in migraine pathology.13,14 Dendritic cells could be a target for migraine treatment. A 2019 study 15 showed that culturing rat dendritic cells with interferon gamma reduced spreading depression in hippocampal slices, suggesting that mild cytokine exposure suppresses neuron firing, potentially offering a new approach for migraine therapy. Besides, individuals with migraines exhibit alterations in immune cell subsets, including increased CD4 + T helper cells and decreased CD8 + cytotoxic T cells during the interictal period.16–19 A flow cytometric study found elevated CD3 + CD16+CD56+ NK cells during ictal and interictal periods. 20 Additionally, a decrease in regulatory T (Treg) cells, crucial for preventing autoimmunity, was observed, potentially leading to autoimmune diseases.21,22 Overall, these immune changes indicate a shift toward a proinflammatory pattern, similar to cytokine changes in migraine sufferers. This paradigm shift has opened new avenues for therapeutic intervention, targeting not only the neural mechanisms but also the immunological underpinnings of the condition. 23
The application of Mendelian Randomization (MR), a powerful epidemiological approach that leverages the random assortment of genes from parents to offspring, offers a unique opportunity to investigate the causal relationship between immune cell signatures and migraine. 24 By using genetic variants as instrumental variables, MR can circumvent the limitations of traditional observational studies, such as confounding and reverse causation, providing more robust evidence for causal inference. 25
In this study, we aimed to explore the causal association between immune cell signatures and migraine using an MR approach. By analyzing large-scale genetic data, we sought to determine whether specific immune cell profiles are not only associated with migraine but also play a causal role in its etiology. Our findings have the potential to redefine the current understanding of migraine as a purely neurological disorder and highlight the importance of considering the immune system as a critical component in its management and treatment. 6
Materials and methods
Study design
In our investigation into the causal links between immune cell phenotypes and migraine, we deployed a two-sample MR approach to evaluate the relationship across 731 immune cell markers, systematically categorized into seven distinct groups. The MR method is underpinned by the use of genetic variants as instrumental variables (IVs), 26 which serve as proxies to infer causality. For these IVs to be valid, they must meet three essential criteria: (1) a robust association with the immune cell markers of interest; (2) independence from confounding factors that could bias the relationship between the exposure and the outcome; and (3) no direct pathways to the outcome that are independent of the exposure under study.
Our analysis was designed with scrupulous adherence to ethical considerations. Each constituent study was vetted and approved by the respective institutional review boards, ensuring the highest standards of participant protection. Informed consent was obtained from all participants after they were provided with exhaustive information regarding the study’s objectives, methodologies, potential risks, and benefits. This approach not only maintains the integrity of the research but also safeguards the rights and well-being of all individuals involved.
By applying this stringent study design, we aimed to provide a clear and unbiased assessment of the causal relationship between immune cell phenotypes and migraine, contributing to a deeper understanding of the disease’s immunological basis and paving the way for novel therapeutic interventions.
Data source description for migraine GWAS
The genetic study on migraine was sourced from a robust Genome-Wide Association Study (GWAS) as reported by Donertas HM et al., 27 published in “Nat Aging” on the 8th of April, 2021, under PubMed identifier 33959723. The discovery sample comprised 13,971 individuals with migraine and 470,627 control subjects, genotyped using a genome-wide genotyping array. After a stringent quality control (QC) process, the study analyzed a substantial dataset of 9,886,868 single nucleotide polymorphisms (SNPs) on the NR platform. Although the ancestry of the discovery sample was not delineated in the provided snapshot, the research adheres to a high methodological rigor, which is imperative for dissecting the genetic architecture of migraine disorder.
Immunity-wide GWAS data sources for migraine
For our exploration of the immunological underpinnings of migraine, we turned to the rich repository of the GWAS Catalog, which archives a trove of summary statistics for a spectrum of immune traits. Our analysis encompassed accession numbers 28 ranging from GCST0001391 to GCST0002121, which correspond to a comprehensive dataset of 731 distinct immunological phenotypes. This included assessments of absolute cell (AC) counts, median fluorescence intensities (MFI) as markers of surface antigen levels, morphological parameters (MP), and relative cell (RC) counts across various immune cell types.
The inaugural GWAS for these immune traits drew upon a cohort of 3757 European individuals, 29 ensuring a non-overlapping sample set with our migraine study. Employing a high-density genotyping array, we meticulously profiled approximately 22 million single nucleotide polymorphisms (SNPs) to dissect the genetic architecture of immune-related phenotypes. The genetic data were interpreted through a reference panel anchored in the Sardinian sequence, enabling a precise estimation of genetic variations.
Our analytical approach was meticulous in controlling for confounding factors, including gender, age, and two-year age groups, which are known to modulate immune responses. This rigorous control allowed for a refined exploration of the genotype-phenotype correlations, ensuring that the genetic signals associated with immune traits were not obscured by these common demographic variables. By integrating the effects of age and gender, our study has provided a more refined and clinically relevant portrayal of the genetic influences on immune-mediated phenotypes, which are increasingly recognized as integral to the pathophysiology of migraine.
Selection of instrumental variables (IVs) for migraine study
In our migraine study, we adhered to recent methodological advancements in genetic epidemiology by setting a stringent significance threshold of 1 × 10−5 for identifying IVs associated with each immune trait.30–32 This threshold is critical for standardizing the selection of genetic factors that are likely to influence immune-related phenotypes, which are suspected to play a role in migraine pathogenesis.
To enhance the precision of single nucleotide polymorphism (SNP) selection and to mitigate issues arising from high linkage disequilibrium (LD) with neighboring SNPs, we employed PLINK software (version 1.90). 33 This tool employs a clustering algorithm that efficiently reduces genetic complexity by applying an LD r2 threshold of less than 0.001 within a 10000 kb window, thereby simplifying the genetic data and increasing the accuracy of downstream analyses.
For the migraine trait, we adopted a more conservative significance level of 1 × 10−5 to align with the most rigorous standards of genetic association studies. 34 To ascertain the robustness of our IVs and to safeguard against the potential confounding effects of weak instruments, we calculated the proportion of phenotypic variation explained and the F statistic for each IV. These quantitative measures are indispensable for verifying the strength of our genetic instruments and for reducing the likelihood of obtaining biased causal estimates in our Mendelian randomization analysis.
Our meticulous approach to IV selection is designed to provide a solid genetic foundation for understanding the complex interplay between immune cell phenotypes and migraine, ultimately contributing to the discovery of novel preventive and therapeutic strategies for this debilitating neurological condition.
Statistical analysis
For our migraine study, all statistical analyses were conducted using R 3.5.3 software, which is available through the comprehensive resources provided by the R Project (https://www.R-project.org). To investigate the causal links between 731 immunological phenotypes and migraine, we employed a suite of MR methods, including the inverse variance weighting (IVW) method, 35 weighted median, 36 and mode-based approaches. 37 These techniques were facilitated by the ‘Mendelian Randomization’ package (version 0.4.3), 38 ensuring a robust analytical framework.
The IVW method is analogous to a culinary recipe, where each ingredient’s contribution is weighted by its statistical significance, thereby balancing the overall impact and mitigating the influence of outliers. This approach ensures that the analysis is both comprehensive and nuanced.
To ensure the consistency of our selected instrumental variables (IVs), we applied Cochran’s Q statistic and its associated p value. These statistical tools are adept at discerning genuine heterogeneity among IVs. In the event that the null hypothesis of homogeneity is rejected, indicating variability, we transitioned to a random-effects model to accommodate this diversity.
Considering the possibility of horizontal pleiotropy, which can confound MR analyses, we incorporated the MR-Egger regression method 39 to identify potential outliers that may disproportionately sway the results. Additionally, we deployed the MR-PRESSO method 40 to systematically detect and exclude outlier genetic variants that could bias our findings. The meticulous exclusion of such variants is essential for preserving the integrity of our conclusions.
Data visualization was facilitated through scatter plots and funnel plots, which are invaluable for revealing outliers and assessing the uniformity of correlations among data points. These graphical representations are crucial for validating the accuracy and dependability of our research outcomes.
Result
Investigation of causal influences on immunological phenotypes in migraine
In our research, we conducted a two-sample MR analysis to explore the potential causal effects of migraine on a spectrum of immunological phenotypes. Utilizing the IVW method as our primary analytical technique, we aimed to detect any substantial associations between migraine and the immune cell phenotypes under investigation.
After applying the False Discovery Rate (FDR) correction to rigorously account for multiple testing, we observed no significant associations between migraine and the immune phenotypes at the conventional level of significance (Figure 1). This outcome points to a lack of substantial causal effects of migraine on the immune cell phenotypes examined within the context of our study. The forest plot presents the causal associations between migraine and diverse immune cell phenotypes.
This forest plot displays the results of the inverse variance weighted Mendelian Randomization analysis, assessing the causal associations between migraine and various immune cell phenotypes. The plot includes the following key elements for each immune cell phenotype: Trails: List of immunological phenotypes examined in the study;nsnp: The number of Single Nucleotide Polymorphisms (SNPs) analyzed for each phenotype;pval: The statistical p-value indicating the strength of the association; OR (95% CI): The odds ratio with its 95% confidence interval, showing the estimated effect size and precision;FDR: The False Discovery Rate correction applied to adjust for multiple testing.
Investigation of the causal relationship between immunological phenotypes and migraine
Following the adjustment for multiple comparisons using the False Discovery Rate (FDR) method, with a significance threshold defined as p FDR <0.05, our analysis determined that migraine significantly influences a subset of immunological phenotypes across various immune trait categories. These included Median Fluorescence Intensity (MFI), Relative Cell (RC) counts, Absolute Cell (AC) counts, and Morphological Parameters (MP), with each phenotype demonstrating a p value of less than 0.05 (Figure 2). The forest plot illustrates the causal relationships between immune cell traits and migraine.
Furthermore, the MR-Egger regression intercept and the global test derived from MR-PRESSO did not suggest the presence of horizontal pleiotropy across the significant associations detected. This absence of pleiotropy bolsters the reliability of our findings. The consistency analysis, as illustrated in Figure 3, offered additional support for the robustness of the observed causal links. The scatter plot showcased a general consistency in the effects observed across different analytical approaches, and the odds ratios (ORs) derived from various models were in substantial agreement. This methodological consistency substantiates the reliability of our results, as further visualized in Figure 4, which presents a broader perspective of the data. Funnel plot analysis for the association between CD33 on Mo MDSC and migraine. MR: mendelian randomization. Scatter plot depicting the relationship between CD33 on Mo MDSC and Migraine. MR: Mendelian randomization; SNP: Single nucleotide polymorphism.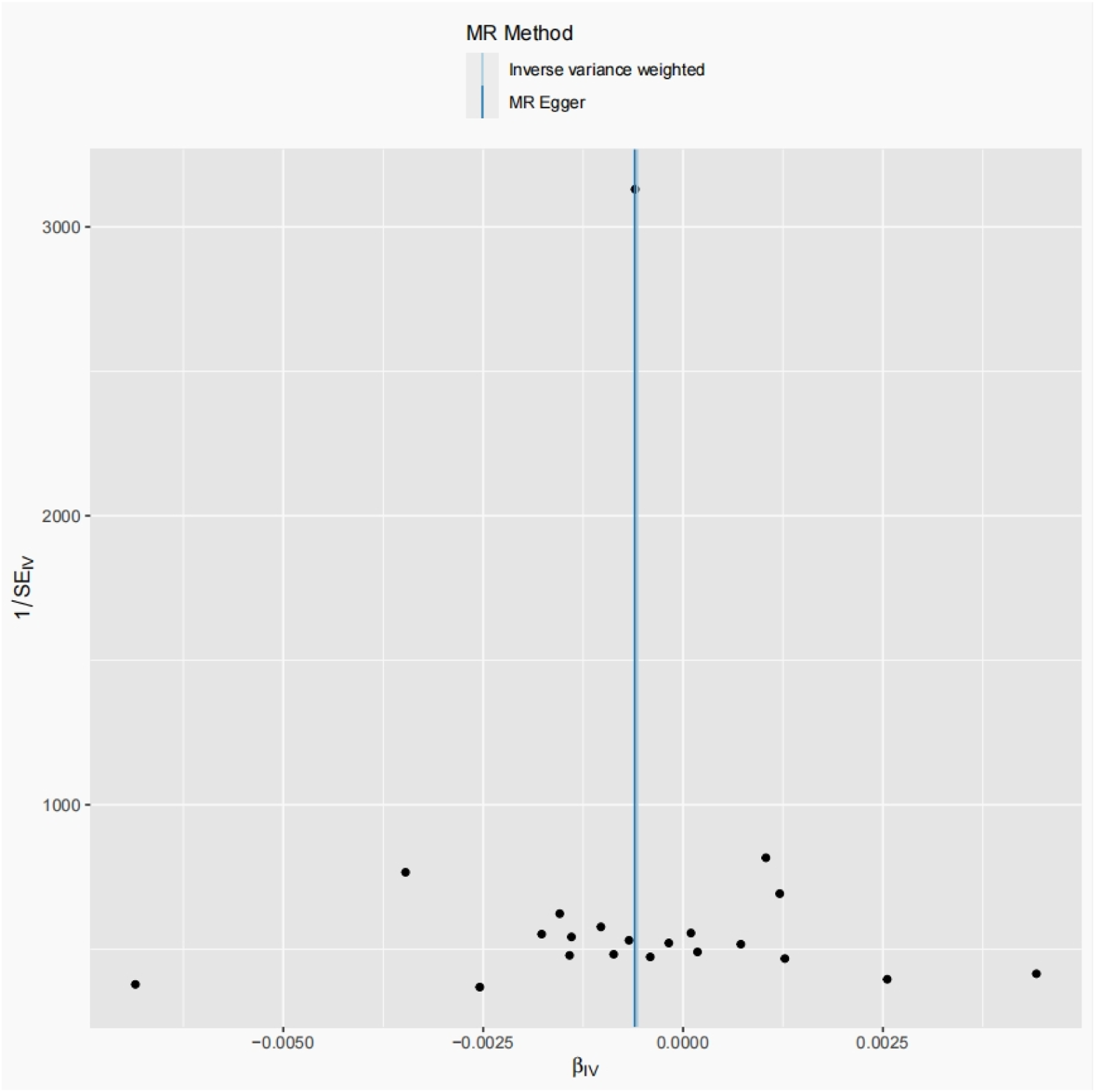

This forest plot displays the results of the inverse variance weighted Mendelian Randomization analysis, assessing the causal associations between various immune cell phenotypes and migraine. The plot includes the following key elements for each immune cell phenotype: Trails: List of immunological phenotypes examined in the study;nsnp: The number of Single Nucleotide Polymorphisms (SNPs) analyzed for each phenotype;pval: The statistical p-value indicating the strength of the association; OR (95% CI): The odds ratio with its 95% confidence interval, showing the estimated effect size and precision;FDR: The False Discovery Rate correction applied to adjust for multiple testing.
Discussion
The application of Mendelian randomization in our study has provided a robust framework to investigate the causal relationships between specific immune cell phenotypes and migraine. This genetic epidemiological approach leverages the random allocation of genes from parents to offspring, serving as a natural experiment that circumvents many of the biases inherent in traditional observational research. 3
By utilizing validated genetic variants as instrumental variables, we have been able to establish associations that are less susceptible to confounding factors and reverse causality—a common challenge in migraine epidemiology. 41 This methodological rigor has allowed us to propose potential causal links between immune cell phenotypes and migraine susceptibility with greater confidence.
The identification of both protective and risk-associated immune cell phenotypes underscores the nuanced role of the immune system in the pathogenesis of migraine. 42 Our findings indicate that the immune response may be a double-edged sword, with certain cellular profiles exacerbating the condition while others appear to mitigate its effects.
B cells exhibit a complex role in the onset and progression of migraine, with certain phenotypes such as Unsw mem AC, IgD− CD27− AC, CD20− CD38− AC, CD20− CD38− % lymphocyte, CD19 on IgD + CD38−, CD24 on IgD + CD38br, and CD38 on transitional B cells demonstrating a protective effect, while IgD + B cells are considered a risk factor for migraine. These findings challenge the previous understanding of the singular role of B cells and highlight the diversity of B cell functions across different immunological environments. In terms of B cell phenotypes reducing the likelihood of migraine onset, our study suggests that specific B cell subsets may exert a protective role by modulating immune responses and promoting an anti-inflammatory milieu. For instance, Unsw mem AC and IgD− CD27− AC may inhibit inflammatory processes by producing inhibitory cytokines such as interleukin-10 (IL-10), 43 thereby reducing the incidence of migraine. Additionally, the elevated expression of CD20− CD38− % lymphocyte may be associated with regulatory B cells (Bregs), which maintain immune tolerance and prevent the onset of autoimmune reactions. 44 In contrast to the protective B cell phenotypes, the IgD + AC phenotype is linked to an increased risk of migraine. This association may be related to the pro-inflammatory capabilities of these cells, such as the secretion of pro-inflammatory cytokines or the enhancement of immune responses through interactions with T cells. 45 To comprehend the dual roles of these B cell phenotypes, future research must delve into the underlying molecular mechanisms. It is particularly important to study how these B cell subpopulations respond to various immunological signals and how they interact with other immune cells to modulate immune responses.
Myeloid cell phenotypes play a dual role in migraine, with certain subtypes such as Gr MDSC AC, CD33dim HLA DR + CD11b− AC, CD34 on HSC, CD33 on CD33dim HLA DR + CD11b+, CD33 on Mo MDSC, CD33 on CD33br HLA DR+, and CD33 on CD33br HLA DR + CD14− exhibiting protective effects, while CD45 on basophil is considered a risk factor for migraine. Protective Myeloid cell phenotypes may mitigate the pathological processes of migraine through various mechanisms. For example, Gr MDSC AC might function by suppressing inflammatory responses and promoting vascular stability. The expression of CD33dim HLA DR + CD11b− AC may be associated with anti-inflammatory and immunomodulatory functions, contributing to the maintenance of immune homeostasis and preventing excessive immune activation. 46 In contrast to the protective phenotypes, an increase in CD45 on basophil is linked to a higher risk of migraine. This association may relate to the role of these cells in promoting inflammatory reactions, releasing inflammatory mediators, and participating in immune-mediated vascular regulation. 47 These findings provide new insights into the complex role of Myeloid cells in migraine and offer potential targets for future therapeutic interventions.
TBNK cells, owing to the expression of their phenotypes, assume a dual role in migraine, with certain subtypes such as CD8br % leukocyte, TCRgd AC, TCRgd % lymphocyte, FSC−A on CD4+, and SSC−A on B cell demonstrating protective effects, while NKT AC, Granulocyte AC, and FSC−A on NKT are considered risk factors for migraine. Protective TBNK cell phenotypes may ameliorate the pathological processes of migraine through various mechanisms. For instance, the elevated expression of CD8br % leukocyte may be associated with the anti-inflammatory actions of cytotoxic T cells, which can help reduce neuroinflammation. TCRgd T cells, being integral to gut and mucosal immunity, may indirectly influence the onset of migraine by maintaining the balance of gut microbiota. 48 In opposition to the protective phenotypes, an increase in NKT AC and Granulocyte AC is linked to a heightened risk of migraine. This association may pertain to the role of these cells in promoting inflammatory responses, releasing inflammatory mediators, and engaging in immune-mediated vascular regulation. The aberrant expression of FSC−A on NKT may indicate the activation state of NKT cells, a phenomenon often observed in autoimmune and inflammatory diseases. 49
Specific phenotypes of Tregs have been identified to have a causal relationship with migraine, with phenotypes such as CD25 on CD4 Tregs and CD25 on CD39+ secreting Tregs being associated with a protective effect against migraine, while CD4 Treg % CD4, CD28− CD8br AC, and CD127 on granulocyte are linked to an increased risk of migraine. These results offer new perspectives on the complex immunomodulatory role of Treg cells in migraine. In terms of reducing the incidence of migraine, Treg cell phenotypes may play a role by maintaining immune tolerance and suppressing inflammatory responses. The high expression of CD25 on CD4 Tregs likely indicates the activated state of these cells, which can inhibit inflammation by secreting anti-inflammatory cytokines such as interleukin-10 (IL-10) and transforming growth factor-beta (TGF-β). Additionally, CD39+ secreting Tregs may contribute to vascular stability and reduce neuroinflammation by producing adenosine, which could be one of the mechanisms underlying their protective effects. 50 In contrast, a high proportion of CD4 Treg % CD4 may reflect a dysfunction of Treg cells, potentially leading to an imbalance in immune regulation and exacerbation of inflammatory responses. 51 The expression of CD28− CD8br AC may be associated with the instability and/or functional defects of Treg cells, which could diminish their ability to suppress autoimmune reactions. The expression of CD127 on granulocytes may indicate the activation state of these cells, a phenomenon often observed in inflammatory and autoimmune diseases.
Monocyte phenotypes exhibit complex associations with migraine. Our study has linked the HLA DR on CD14− CD16+ monocyte phenotype to a protective effect against migraine, whereas CD40 on monocytes and CD64 on monocytes are considered risk factors for the condition. The protective role of the HLA DR on CD14− CD16+ monocyte phenotype may be related to its functions in anti-inflammatory and immunomodulatory activities. This subset of monocytes likely participates in antigen presentation through the expression of HLA DR molecules, which can promote the activation of Tregs, thereby helping to maintain immune homeostasis and prevent excessive inflammatory responses. 52 In contrast, increased expression of CD40 on monocytes and CD64 on monocytes may be associated with pro-inflammatory activities of monocytes. Activation of CD40 can stimulate the release of inflammatory cytokines through various signaling pathways and may enhance the adhesion of monocytes to endothelial cells, facilitating the migration of inflammatory cells and the localization of inflammatory responses. 53 CD64, also known as FcγRI, when upregulated, may augment the antibody-dependent cellular phagocytosis by monocytes, which could exacerbate inflammatory processes in certain contexts.
Additionally, we observed specific T cell maturation stage phenotypes, including CM CD4+ % T cell and CM DN (CD4−CD8−) % T cell, to be associated with a protective effect against migraine. These findings provide new insights into the immunomodulatory role of T cells in migraine and may significantly impact the development of preventive and therapeutic strategies for the condition. CM CD4 + T cells, as mature helper T cells, may exert their protective effect by promoting the production of anti-inflammatory cytokines and modulating the activity of other immune cells. These cells could contribute to the maintenance of immune homeostasis, preventing overactivation of the immune system, and thereby reducing the occurrence of migraines. 54 CM DN (CD4−CD8−) T cells, also known as double-negative (DN) T cells, constitute a population with regulatory T cell (Treg) functions. They may reduce the incidence of migraine attacks by directly suppressing inflammatory responses or by modulating the function of dendritic cells. Furthermore, CM DN T cells may play a critical role in regulating autoimmune responses and neuroinflammation.55,56
Conventional dendritic cells (cDCs), a pivotal component of the immune system, are tasked with the capture, processing, and presentation of antigens to T lymphocytes, thereby initiating immune responses. Our research has identified a specific phenotype of cDCs—CD62L− monocyte AC—that is significantly correlated with the occurrence of migraine. This discovery sheds new light on the role of cDCs in the pathophysiology of migraine and may have important implications for the prevention and treatment of the condition. The mechanism of action involves several pathways through which CD62L− monocyte AC cDCs may contribute to the onset of migraine. These cells could play a critical role in local inflammatory responses by presenting antigens to autoreactive T cells, thereby activating downstream immune reactions. 57 Additionally, the reduced expression of CD62 L may be associated with enhanced maturation and migratory capacity of cDCs, potentially leading to their accumulation at sites of inflammation and exacerbating neuroinflammation. 58
These findings challenge the previously simplistic understanding of the immunological mechanisms of migraine, emphasizing the diversity and complexity of immune cell functions. The discovery of these immune cell phenotypes provides new targets for the prevention and treatment of migraine. For instance, therapeutic strategies could involve modulating the activity of protective immune cell phenotypes to enhance their anti-inflammatory effects, or suppressing phenotypes that are risk factors to reduce inflammatory responses.
Moreover, our study points to directions for future research. Firstly, it is essential to further elucidate how these immune cell phenotypes influence the onset of migraine through specific molecular mechanisms. Secondly, exploring how individual genetic backgrounds affect the relationship between immune cell phenotypes and migraine may aid in the development of personalized treatment plans. Lastly, long-term follow-up studies are necessary to assess the long-term efficacy and safety of immunomodulatory therapies in preventing migraine attacks.
The limitations of this study include the fact that, although the Mendelian randomization approach reduces the impact of confounding factors and reverse causality, it cannot entirely exclude all potential biases. Additionally, our findings require validation across different ethnic and geographical populations to ensure their generalizability. Future research should consider these limitations and aim to replicate and verify the results in a broader demographic.
Our study has underscored the pivotal role of specific immune cell phenotypes in the pathophysiology of migraines. The identification of these cell types offers a novel perspective for therapeutic intervention, potentially leading to more targeted and effective treatment strategies for migraine sufferers. Our findings not only contribute to a deeper understanding of the immunological mechanisms underlying migraine but also set the stage for future clinical trials aimed at validating the safety and efficacy of these targeted therapies.
The implications of our research are twofold: firstly, by elucidating the immunological basis of migraine, we provide a foundation for the development of new therapeutic approaches that could significantly improve the management of this condition. Secondly, our work opens up new horizons for immunological research in migraine, encouraging further exploration of the complex interplay between the immune system and the neurological processes that give rise to migraine symptoms. The clinical implications of our findings are significant. Understanding the specific immune cell phenotypes involved in migraine can lead to the development of targeted therapies. For example, therapeutic strategies that enhance the protective effects of certain B cell subsets or suppress the pro-inflammatory activities of specific T cell phenotypes could offer new avenues for treatment.
As we look towards the future, it is imperative that our findings are corroborated through rigorous clinical trials. These trials will be essential in confirming the potential of our proposed therapeutic strategies and ensuring that they meet the highest standards of safety and efficacy. By doing so, we aim to enhance the quality of life for those affected by migraines and contribute to the broader field of immunological research.
In summary, our study marks a significant step forward in the quest for more effective migraine treatments. With the insights gained from our research, we are optimistic about the prospects of advancing the clinical care of migraine patients and fostering innovative approaches within the realm of immunological medicine.
Conclusion
Our study has underscored the pivotal role of specific immune cell phenotypes in the pathophysiology of migraines. The identification of these cell types offers a novel perspective for therapeutic intervention, potentially leading to more targeted and effective treatment strategies for migraine sufferers. Our findings not only contribute to a deeper understanding of the immunological mechanisms underlying migraine but also set the stage for future clinical trials aimed at validating the safety and efficacy of these targeted therapies.
Footnotes
Acknowledgements
I sincerely appreciate the support and assistance provided by Dr. Wei Hao Chai in writing and data analysis.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is funded by “Tianshan Elite” Talent Development Program for Medical and Health Professionals (Grant No. TSYC202301B146).
