Abstract
Human immunodeficiency virus-1 (HIV)-associated chronic pain is a debilitating comorbid condition that affects 25–85% of people with HIV. The use of opioids to alleviate pain has given rise to opioid dependency in this cohort. Therefore, there is an urgent need to understand mechanisms and identify novel therapeutics for HIV-associated chronic pain. Several animal models have been developed to study HIV-related comorbidities. HIV-1 transgenic (Tg) rats have been shown to serve as a reliable model that mimic the deficits observed in people with HIV, such as neurological and immune system alterations. However, pain-related behavior in these animals has not been extensively evaluated. In this study, we measured evoked and spontaneous behavior in HIV-1Tg male and female rats. The results indicated that HIV-1Tg rats exhibit similar behavior to those with HIV-1-related neuropathy, specifically, cold sensitivity. Consequently, HIV-1Tg rats can serve as a model of neuropathy to study pain-related mechanisms and therapeutics targeted toward individuals living with HIV-1.
Introduction
Human immunodeficiency virus-1 (HIV-1) is an infection that is currently grouped into two types, HIV-1 and HIV-2; HIV-1 virus is prevalent worldwide while HIV-2 is restricted to only some regions of Western and Central Africa. 1 HIV is classified as retrovirus because it contains reverse transcriptase, which means that these viruses use RNA to make retroviral DNA and insert it into the host’s genome which initiates viral replication. 2 Although HIV-1 and HIV-2 have slightly different organization of their genome, both include gag, pol, env, and other genes, which are important in disease pathogenesis. 1 However, unlike HIV-2 which codes for a Vpx (virus protein x), HIV-1 genome codes for a regulatory protein Vpu (virus protein unique), which increases pathogenicity of HIV-1. Consequently, due to world-wide prevalence and higher pathogenicity, 3 this study focuses on HIV-1 and associated pain.
Binding of the HIV-1 vector to CD4+ T-helper cells leads to their subsequent depletion impairing immune system function and resulting in opportunistic infections.4–6 Current treatment for HIV includes combination antiretroviral therapy (cART). 7 The adherence to cART has been shown to improve life expectancy by increasing the CD4 count and decreasing the HIV RNA levels. People living with HIV on cART have a low viral load compared to unmedicated people with HIV, which decreases the risk of HIV transmission. Additionally, cART treatment prevents HIV transmission to fetus during pregnancy, consequently, allowing people living with HIV to have an opportunity to conceive a child without a risk of passing the HIV infection. 8
However, despite increased life expectancy, people with HIV still suffer from related comorbidities, like chronic pain. People living with HIV have reported chronic or recurrent headaches, somatic pain, and visceral pain and some reporting chronic widespread pain even while being on cART.9–12 The prevalence estimate of chronic widespread pain in the general population is approximately 10%, 13 dramatically lower than in people living with HIV which has an estimated prevalence of 25–85%.9,10 However, it is important to note that pain prevalence was higher prior to the cART era, with pain prevalence in people living with HIV being as high as 80%. 14 This indicates that although cART substantially decreases the pathological pain prevalence, other factors which are not targeted by cART can contribute to enhanced pain in the HIV population.
Antiretroviral therapy aims to inhibit viral replication via various pathways. However, even though virus is not able to replicate, the expression of defective proviruses is observed even under the cART treatment. The proviruses are called “inactive” or “deffecive” since they have a mutation within the gene and therefore, were thought to be not related to the development of HIV-1 related pathogenesis. 15 Although cART inhibits viral replication, it does not address these proviruses. Recent findings indicate that the defective proviruses can be recognized by T lymphocytes and therefore, contribute to the increased inflammatory state and potentially pain.16–18 Similarly, anti-retroviral drugs can induce neuroinflammation that alters pain processing in those with HIV. 19
The pain can be inflammatory or neuropathic in nature and its treatment often relies on prescription opioids. 20 However, prolonged use of opioids to treat HIV-associated chronic pain is not reliably efficacious in this population and is associated with misuse and development of opioid tolerance.20,21 The prescription of opioids is also associated with increased risk of transmission of injection-related infection, with 9% of HIV diagnoses are among those who inject drugs and this relationship is negatively affected by increased opioid use. The lack of understanding about the underlying mechanisms of HIV-associated chronic pain and an urgent need for the development of non-opioid therapeutics requires extensive pre-clinical research which can be done using animal models. 22
Animal models are used extensively in HIV research as they allow for investigation of mechanisms required to understand important interactions between HIV-associated proteins and secondary sequelae (e.g., chronic pain). Non-human primates are one of the animal models used since these species can get infected by a similar lentivirus- Simian immunodeficiency virus (SIV); however, species-specific tropism, animal welfare concerns, and high cost are barriers to their use. 23 Consequently, rodent models have been developed to mimic human HIV infection. HIV-1 transgenic (Tg) mice require heterologous promoters for viral expression, resulting in expression in atypical tissues such as skin.24–26 HIV-1Tg rats exhibit efficient viral gene expression in lymph nodes, spleen, thymus, and blood, and therefore develop similar pathologies and immune profile as humans, including reduced number of CD4+ cells and increased T-cell apoptosis.24,27,28 Thus, HIV-1Tg rats serve as a promising tool to study HIV-associated comorbidities. However, to the best of our knowledge, no studies have been conducted to assess pain-related behaviors in this model. The present study evaluated general activity and pain-related behaviors to address the hypothesis that, compared to wild-type (WT) controls, HIV-1Tg rats will exhibit pain-related hypersensitivity similar to that reported in humans living with HIV-1.
Materials and methods
Animals
Age- and sex-matched hemizygous HIV-1Tg and non-transgenic Fisher NHsd (F344) rats were bought and licensed from the University of Maryland and then bred at the University of Alabama at Birmingham. The complete protocol of the HIV-1Tg rat construction has been previously described. 24 In short, HIV-1Tg rats were generated using a non-infectious provirus, constructed via functional deletion of gag and pol from the viral genome. When hemizygous Tg rats are bred with F344 rats, the offspring are either HIV-1Tg or wild type (WT). All rats were housed in standard polycarbonate cages (140 in2 floor space) in a temperature-controlled environment on a 12:12 h light-dark cycle and were not handled outside of routine husbandry. Food and water were provided ad libitum. All of the studies were approved by the Institutional Animal Care and Use Committee (IACUC-22344) at the University of Alabama in Birmingham.
Evoked pain-related behavior
Behavioral indices of evoked pain were used to assess mechanical allodynia, heat hyperalgesia, and cold allodynia in HIV-1Tg and WT rats. All of the evoked pain-related behavioral assessments were done in an enclosed space within the laboratory in order to decrease exposure to noise and human contact prior to testing. Outside of the regular husbandry, rats were not handled prior to the experiment. Additionally, all of the experiments were done by the same person, to minimize the suppression of nosifensive behaviors which is often observed in rodents.
Mechanical allodynia
To measure cutaneous mechanical allodynia, rats were placed on a mesh floor inside a plexiglass chamber (23 × 23 × 14.5 cm) for at least 30 min to acclimatize prior to testing. After the acclimatization period, a simplified up-down method 29 was used to assess mechanical sensitivity with a set of calibrated von Frey monofilaments based on the Semmes Weinstein monofilament set. A range of monofilaments were applied to the glabrous skin of the hind paw to assess the withdrawal threshold. The starting monofilament was always number 4.93. If the monofilament did not elicit a withdrawal, the next highest monofilament was applied. If a withdrawal occurred, the next lowest monofilament was applied. A total of five trials occurred per hind paw and the average score was recorded. After converting to force in grams, withdrawal thresholds were averaged to obtain a single value for each animal. This procedure was repeated daily over 3 days and the average withdrawal threshold across 3 days for each rat was used in analyses.
Heat hyperalgesia
Thermal hyperalgesia was assessed using a hot plate (Maze Engineers). The testing temperature was set to 52.5°C. The heat sensitivity measures were always done following the von Frey assessment. Therefore, rats had time to acclimate to the new room environment for at least 30 min prior to any evoked behavior testing. Following the von Frey, rats were placed on the surface of the hot plate surrounded by a glass cylinder (10 × 25 cm) and the average withdrawal latency in seconds was recorded. The withdrawal latency was characterized by the time until complete withdrawal or licking of the hind paws occurred, a measure often used in nociceptive tests in animal models. If the rats did not respond within 30 s, they were removed from the hot plate. This was repeated daily for 3 days to generate a single average withdrawal latency for each rat across 3 days.
Cold allodynia
An acetone spray test was used to assess cold allodynia. Rats were placed on mesh floor inside a plexiglass chamber and were allowed to acclimatize for at least 30 min. Following the acclimatization period, a drop of room temperature water was applied via syringe as a negative control to the top of each hind paw. The response was scored from 0 to 2, where 0 is no response, 1 is brisk withdrawal or flick of the paw, and 2 is repeated licking or flicking of the paw. Following the application of water, a drop of room temperature acetone was applied in a similar fashion. This procedure was repeated daily for 3 days for each rat to get an average response score across 3 days.
Activity monitoring
Spontaneous behavior of HIV-1Tg and WT rats was measured using a PhenoTyper instrument observation cage equipped with a top unit containing an infrared sensitive camera and motion sensors and EthoVision XT video tracking software (Noldus) located in the Behavioral Core at the University of Alabama at Birmingham. Rats were individually placed in an observation cage and remained there for 3 days prior to the experiment to acclimatize. Testing began on day 4, during which the velocity and total distance traveled within the chamber, and the frequency and duration of eating and drinking were recorded. A total of 12 trials (6 h each) over 3 consecutive days were performed. Between trials, a one-minute pause was programmed to indicate the start of the next trial. Rats were not handled during the combined 6-day period of acclimatization and testing. Prior to the acclimatization, two arenas were selected to outline the areas with food and water, which were provided ad libitum for the 6-day period. The nose and tail of each rat was specified using the acquisition software to ensure the maximize the accuracy of food and water intake detection. A maximum of six rats of the same sex were observed at any given time.
Statistical analysis
The statistical analyses were performed in RStudio version 2022.07.2 (RStudio Team, 2020) and in SPSS version 28.0.1.1 (IBM SPSS Statistics, 2021). All of the statistical analyses were two sided with an α level of 0.05. A two-way ANCOVA was conducted for each of the measures of spontaneous behavior (distance traveled, velocity, eating and drinking duration, and eating and drinking frequency) and each of the evoked behavior measures (mechanical allodynia, thermal hyperalgesia, and thermal allodynia). Age was used as a covariate during these statistical procedures. Sex (male and female) and genotype (HIV-1Tg and WT) were used as independent variables. ANCOVA was chosen as a statistical test instead of multivariate ANCOVA (MANCOVA) due to several reasons. First, MANCOVA is not designed to test whether the groups differ on a specific dependent variable rather than on a variate. Additionally, most often MANCOVA is followed up by an ANCOVA to find which dependent variables generate the observed effect. Consequently, MANCOVA does not prevent type I error which might arise once multiple ANCOVAs are generated as a follow-up. 30 One of the variables (eating frequency) followed a Poisson distribution and therefore, a binary logistic regression was performed as well. Where there was a significant interaction, it was followed by a simple main effects analysis. The simple main effects analysis is a priori analysis usually performed following a significant interaction and represents the differences among the means within the design. Since the design depicted in the study had only two independent variables (sex and genotype), and post-hoc tests (e.g., Tukey’s honest significant difference test, Bonferroni’s test, Scheffe’s test) are often less powerful due to chances of an alpha inflation, simple main effects analysis was chosen instead of the post-hoc analysis. The Bonferroni adjustment was applied to all of the simple main-effect p values. Significant effects are represented as: *p < .05, **p < .01, or ***p < .001.
Results
Evoked pain-related behavior
Descriptive statistics for evoked behavior (N = 43).

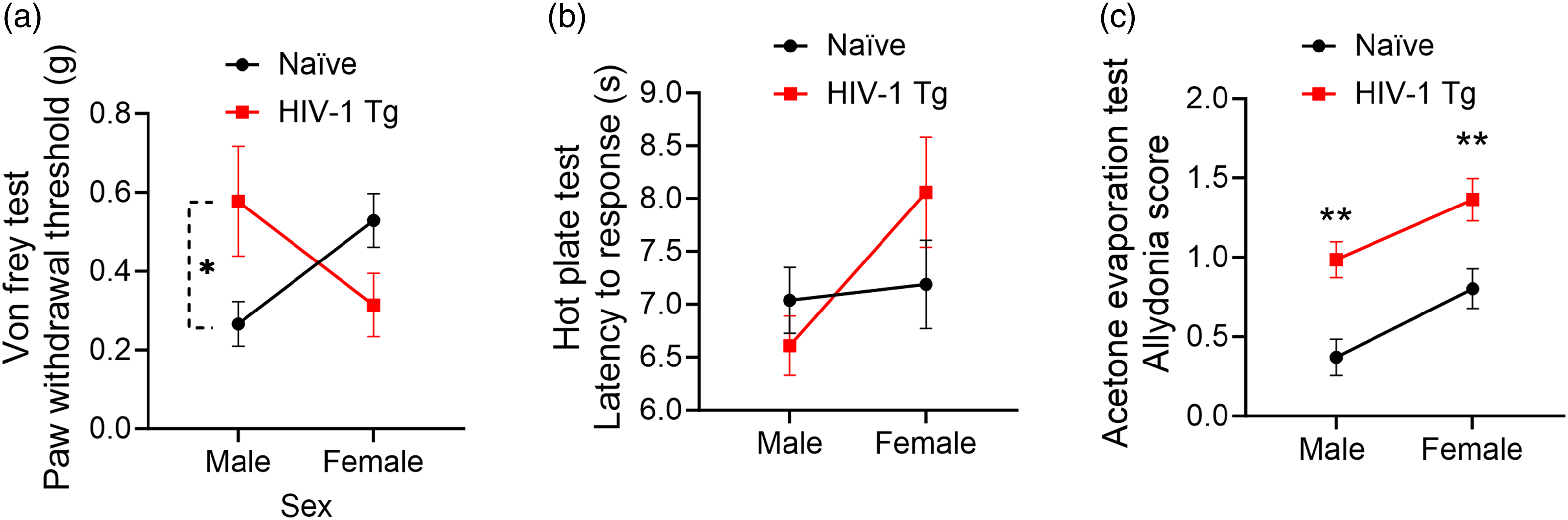
Evoked behavior in HIV-1Tg and naïve female and male rats. All graphs present a non-log transform data for better interpretation. A significant sex by genotype interaction was observed for the paw withdrawal threshold upon Von frey test with male HIV-1Tg rats having less mechanical allodynia than HIV-1Tg females (p = 0.010). A simple main effect of sex was observed following a Bonferroni adjustment (p = 0.0381) (n = 9–11) (a). A significant main effect of genotype was observed during a hot plate test with HIV-1Tg females having higher withdrawal latency than HIV-1Tg males (p = 0.047) (n = 9–11) (b). A significant main effect of genotype (p <0.0001) and sex (p = 0.003) was observed for acetone test with male and female HIV-1Tg rats having a higher allodynia score compared to naïve animals (c) (n = 9–12). The data are represented with mean and SEM error bars. The significance is represented by * (p < 0.05), ** (p < 0.01), *** (p < 0.001).
A significant interaction effect was observed between age and sex for the withdrawal latency during hot plate assessment, indicating that the assumption of homogeneity of regression slopes was violated [F (1,35) = 5.79, p = 0.022]. The hot plate assessment revealed a significant main effect of genotype [F (1,38) = 2.21, p = 0.047] (Figure 1(b)). The rats did not respond to the application of room temperature water (results not shown). However, a significant main effect of genotype and sex was observed during application of acetone [F (1,38) = 22.16, p <0.001; F (1,38) = 10.15, p = 0.003] (Figure 1(c)).
Locomotor activity
Descriptive statistics for spontaneous behavior (N = 24).

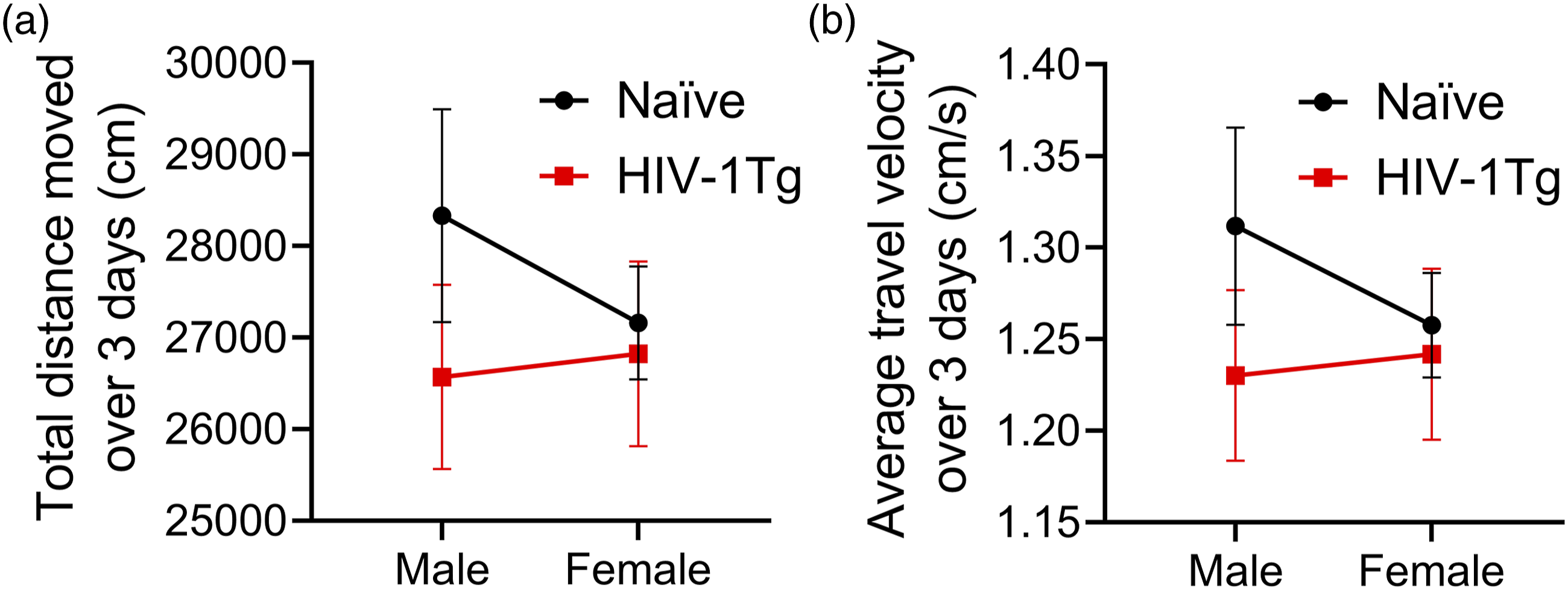
Spontaneous locomotor behavior in HIV-1Tg and naïve female and male rats. All graphs present a non-log transform data for better interpretation. No significant difference was observed in average total distance travelled across all trials between sexes and genotypes in rats (p > 0.05) (n = 5–7) (a). No difference in average velocity of travel across all trials was observed between sexes and genotypes in rats (n = 5–7) (p > 0.05) (b). The data data represented with mean and SEM error bars. The significance is represented by * (p < 0.05), ** (p < 0.01), *** (p < 0.001).
Next, the total time spent in the eating and drinking zones across all trials (eating and drinking duration) was evaluated. The data for eating duration violated the assumption of homogeneity of variance. A significant sex by genotype interaction was observed for the average time spent at the eating zone [F (1,19) = 5.610, p = 0.029] (Figure 3(a)). However, no significant simple main effects were observed at α of 0.025 following a Bonferroni adjustment (p > 0.025). It is important to note that violation of the assumption of the homogeneity of variance could explain the lack of simple main effects. No significant main effects or interaction were observed for the average time spent at the drinking zone (p > 0.05) (Figure 3(b)). However, the data did have a large variance associated with it which could explain the lack of significance even in the presence of seemingly distinguishable means. Spontaneous eating and drinking in HIV-1Tg and naïve female and male rats. All graphs present a non-log transform data for better interpretation. A significant sex by genotype interaction was observed for the average time spent in the eating zone across all trials with HIV-1Tg males spending more time in the eating zone than HIV-1Tg females (p = 0.029). However, no significant simple main effects were observed which indicates that although an interaction was present (the effect of one variable is dependent on another variable) there were no individual differences between cell means, hence, no significance asterisks were added to the graph (n = 5–7) (a). No significant main effect or interactions were observed for the average total time animal spent in the drinking zone across all trials (p > 0.05) (n = 5–7) (b). No significant main effects or interactions were observed for the number of times animals was observed in the eating zone across all trials (p > 0.05) (n = 5–7) (c). A significant interaction was observed for the frequency of drinking zone visits across all trials (p = 0.039). The simple main effect analysis indicated that although the drinking zone frequency was higher for male HIV-1Tg rats than for HIV-1Tg female rats, this difference was not statistically significant following the Bonferroni adjustment (p > 0.025). A simple main effect of genotype in males was observed following a Bonferroni adjustment with HIV-1Tg rats spending more time in the drinking zone compared to the WT rats (p = 0.013) (n = 5–7) (d). The data are represented with mean and SEM error bars. The significance is represented by * (p < 0.05), ** (p < 0.01), *** (p < 0.001).
Lastly, the number of times an animal was detected in a food or water zone across all trials (eating and drinking frequency) was quantified. Data for eating frequency followed a Poisson distribution, therefore, a binary logistic regression was computed. To construct a binary variable, eating frequency was split into high frequency (more than 3000 occurrences at the food zone), and low frequency (less than 3000 occurrences in a food zone). The final distribution consisted of 12 animals with high food frequency and 12 animals with low food frequency. The VIF score for the multicollinearity assumption was between 1 and 3.2 and therefore, the assumption was not violated. Both ANCOVA and a logistic regression indicated that no significant main effects or interactions were present (p > 0.05). Since both of the analyses demonstrated similar results, a line graph from a linear model was chosen to present the results (Figure 3(c)). The drinking frequency was not normally distributed and therefore a log transformation was used to achieve normality. No significant main effects were observed for the amount of time an animal was detected in the drinking zone (p > .05) (Figure 3(d)). However, a significant sex by genotype interaction was observed (p = .039). After α was adjusted to 0.025 using Bonferroni method, a significant simple main effect of genotype was observed. Specifically, there was a significant difference between WT and HIV-1Tg male rats for the drinking frequency [F (1,19) = 5.276 p = 0.033]. No simple main effect was observed for female rats (p > .025) (Figure 3(d)). Additionally, age was a significant covariate for the number of times an animal was detected in a drinking zone [F (1,19) = 7.262, p = 0.013].
Discussion
This study examined spontaneous locomotor activity and pain-related behaviors in a non-infectious transgenic rat model of HIV-1. The HIV-1Tg rat has been promoted as a model of long-term exposure to HIV-1 viral proteins that occurs in people with HIV who are on long-term cART. A clinical subpopulation of people living with HIV experience persistent or ongoing pain characterized by mechanical and thermal sensitivity, but evidence of increased nociception has not previously been examined in this HIV-1Tg rat model. In the current study, behavioral responses to presentation of thermal stimuli revealed the presence of cold allodynia in HIV-1Tg rats, consistent with previous findings that a clinical subpopulation of people living with HIV and pain experience cold hypersensitivity. 31 This is also consistent with cold sensitivity reported in a murine model of HIV gp120-induced neuropathic pain. 32 However, that study reported cold sensitivity was greater in females than males, 32 which could be a species-specific effect. Alternatively, the lack of a sex difference in cold sensitivity among HIV-1Tg rats may indicate that the genes gag and pol, which are deleted from the HIV-1Tg genome, partially contribute to sex differences in cold sensitivity although this possibility has not been explored to our knowledge.
HIV-1Tg rats (specifically females) were less sensitive than their WT counterparts to heat stimulation. This is in contrast with reports that people living with HIV display increased sensitivity to heat.33,34 This could be dependent on CD4+ count and viral load which has been previously associated with increased mechanical sensitivity possibly due to the decrease in intraepidermal nerve fiber (IENF) density and subsequent neuropathy.33,35 The CD4+ count was not measured in the following study and high CD4+ count could potentially explain the lack of a significant difference in mechanical sensitivity between WT and Tg rats in females. In any case, sex differences and interactions between sex and genotype that were observed in this study are not surprising, since there is substantial evidence demonstrating that sex is a biological variant in pain. 36 It was surprising that no difference between HIV-1Tg rats and WT female rats was observed in mechanical sensitivity, since this is a common manifestation of neuropathy that has been observed in some people living with HIV.33,34,37 This is also discordant with the observation of mechanical allodynia in the aforementioned mouse model of gp120-induced neuropathic pain. 32 It is important to note that lifestyle factors, socioeconomic burden, stress, and comorbid conditions are all known contributors to pain sensitivity. The prevalence of mental disorders is higher for those with HIV, and the risk for comorbid mental disorders is further increased with a history of substance use.38,39 Additionally, people living with HIV face negative stigma and discrimination, factors that contribute to negative health outcomes. 40 The lack of significant pain behaviors in HIV-1Tg rats that have been reported in people living with HIV may be at least partially explained by the highly controlled environment in which rats were maintained. Future studies that assess the potential impact of stress exposure and comorbidities in HIV-1Tg rats are warranted to better model the clinical population they are intended to represent.
In general, there was no impact of HIV-related viral protein expression on normal activities assessed in this study, such as eating, drinking, and mobility. It has been shown previously that HIV-1Tg rats perform equally well as their WT counterparts on basic motor tasks, 41 and do not differ from WT animals in exercise-based tasks such as time spent running and swim speed.41,42 However, HIV-1Tg rats have been reported to exhibit differences in exploratory behavior, and neurocognitive decline in these animals becomes evident with more complex tasks. 43 Measures of distance and velocity of travel might not be descriptive of the complex motor behavior previously shown to be impacted by HIV infection.
The group differences in eating and drinking duration and frequency in the present study is partially consistent with a previous study that reported a difference in food and water intake between HIV-1Tg and WT rats. 44 This could potentially be explained by the spontaneous behavior setup in this study. Although the authors tried to maximize the accuracy of food and water intake by identifying the tail and nose points on the software, the set-up itself that was used for the experiments, cannot record whether the animal actually drank or ingested the water. Therefore, every time the nose of the animal was detected within the “water” or “food” zone, the software would count this as an incident even if the animal did not actively engage in drinking/eating. This serves as a limitation, since frequency and duration within each zone are not indicative of eating/drinking behavior. However, since the animal has to be within each of these zones to eat/drink, frequency, and duration, in theory, have a high correlation with the actual ingestion of food and water. Consequently, authors believe that measuring frequency/duration can serve as an alternative equivalent to food/water ingestion. It is necessary for future research to use an alternative setup that can measure eating and drinking directly. For example, PhenoTyper device manufacturer offers mouse feeding and drinking monitors which can measure eating and drinking behaviors directly.
Untreated chronic pain is associated with malnutrition which can increase with age. 45 Furthermore, malnutrition and lack of necessary nutrients increases the risk of opportunistic infection in people living with HIV. People living with HIV who are on cART, are often advised to have a balanced diet to enhance the effectiveness of the medications, 46 Therefore, people with HIV and chronic pain have potentially higher risk for malnutrition. Consequently, the evaluation of eating and drinking habits in the HIV-model is necessary to ensure that the eating and drinking behaviors do not contribute to the nociceptive findings described above.
The observed interaction in this study between sex and genotype in the context of eating zone duration suggests that sex may be an important consideration in the impact of HIV. Male HIV-1Tg rats spent more time in the food zone than their female counterparts, mirroring the increased likelihood of malnutrition in females living with HIV compared to males. 47 Alternatively, other research suggests that males are more prone to undernutrition, although this finding is probably explained by the fact that males present late for HIV treatment. 48 It is important to note that the following study did not find a significant difference between individual cell means during the simple main effects analysis (i.e., male HIV-1Tg vs female HIV-1Tg, male HIV-1Tg vs female HIV-1 WT etc.). These results indicate that data follows a cross-over interaction pattern, meaning that there is no overall effect of either genotype or sex, but there is a significant cross-over interaction or a significant effect of sex and genotype together on the time spent in a food zone. This is again consistent with the aforementioned study, where males with HIV-1 had lower scores of malnutrition compared to the females living with HIV-1. It would be beneficial to conduct a similar clinical study but to include healthy controls when measuring malnutrition. This would, in theory, result in a similar cross-over interaction observed here.
The eating zone frequency demonstrated that there is no difference among any of the groups in the number of times the animal was present in the eating zone. This indicates that the HIV-1Tg males and females and naïve animals visit the eating zone at the same frequency, even though there is a difference in how much time they spent within this zone. Although not significant, the male and female HIV-1Tg rats were observed less frequently in the food zone. However, male HIV-1Tg rats spend more time in the eating zone compared to the female counterparts (not significant relationship). Increased food consumption is sometimes associated with analgesic effects in rats and since male HIV-1Tg rats had an increased sensitivity to cold, this might indicate that these animals consume more food to cope with pain. 49
When looking at the drinking zone duration results, the interaction was not statistically significant. The lack of significant results for the average time spent in the drinking zone could be explained by the small sample size which produced a large variance within the groups. Further research should address this phenomenon by increasing the sample size. The previous characterization of HIV-1Tg rat model demonstrated that HIV-1Tg rats consumed significantly less water compared to the controls. 44 However, the study did not examine the effect of sex on this relationship which could explain why we are seeing a different trend in this study with naïve male rats spending less time in the water zone, and the opposite relationship in females. The drinking zone frequency demonstrated a significant interaction effect with male HIV-1Tg rats visiting the drinking zone more than the naïve male animals. In addition, the age, which ranged from 2 to 14 months in this study, was a significant covariate of drinking frequency as described earlier in the result section. This relationship suggests that the effect of sex on the drinking frequency depends on the genotype of the rat, after controlling for age. This may be explained by the fact that expression of viral proteins in the peripheral and central nervous systems in HIV-1Tg rats changes with age. 44 Specifically, the expression of Tat, gp120, nef, and vif genes is the highest in the spleen at earlier stages of life (i.e., 2–3 months). However, once the HIV-1Tg rats age, the highest gene expression is in the cerebellum (Tat, gp120, vif) and in the spinal cord (nef). Furthermore, the expression of these genes was downregulated in spleen and prefrontal cortex as the rats age. 44 On the contrary, the expression of genes was upregulated with age in the cerebellum, spinal cord, and striatum. Different protein distribution across various brain regions can explain the development of various neurocognitive deficits in clinical population. 44 Furthermore, difference in the protein expression across the lifespan in HIV-1Tg rats can potentially also contribute to spontaneous behavior, such as drinking frequency. Heightened expression of viral proteins is associated with the recruitment of cytokines and chemokines that can ultimately result in oxidative stress. 50 Since striatum and cerebellum are associated with thirst, increased protein expression within these brain regions can have an impact on water intake frequency in HIV-1Tg rats since they can feel thirsty more often. 51 However, since animals have been drinking water more often and the mechanism for satiety is intact, animals might feel fool from drinking small quantities of water and therefore, have no difference in total duration spent drinking compared to the non-HIV-1 animals. In conclusion, our findings indicate that HIV-1Tg rats are a good model to study cold sensitivity in people with HIV. The lack of significant findings for some of the behavioral assessments indicate that the role of gag and pol genes may be compulsory for this aspect of long-term HIV infection. Additionally, future studies should consider manipulations that mimic exposure to adverse events and/or pre-existing or comorbid conditions in clinical populations that are known to contribute to the development and progression of chronic pain.
Footnotes
Author contributions
The authors confirm contributions to the paper as follows: study conception and design – AEG, SA; data collection – AG; analysis and interpretation of results – AEG, JJD, SA; draft manuscript preparation – A G, TC, JJD. All authors reviewed the results and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIDA R01DA049657 (SA), NHLBI HL143958 (SA), and support from the UAB Center for AIDS Research (TC & AG).
