Abstract
Bone cancer pain (BCP) is severe chronic pain caused by tumor metastasis to the bones, often resulting in significant skeletal remodeling and fractures. Currently, there is no curative treatment. Therefore, insight into the underlying mechanisms could guide the development of mechanism-based therapeutic strategies for BCP. We speculated that Rac1/PAK1 signaling plays a critical role in the development of BCP. Tumor cells implantation (TCI) into the tibial cavity resulted in bone cancer-associated mechanical allodynia. Golgi staining revealed changes in the excitatory synaptic structure of WDR (Wide-dynamic range) neurons in the spinal cord, including increased postsynaptic density (PSD) length and thickness, and width of the cleft. Behavioral and western blotting test revealed that the development and persistence of pain correlated with Rac1/PAK1 signaling activation in primary sensory neurons. Intrathecal injection of NSC23766, a Rac1 inhibitor, reduced the persistence of BCP as well as reversed the remodeling of dendrites. Therefore, we concluded that activation of the Rac1/PAK1 signaling pathway in the spinal cord plays an important role in the development of BCP through remodeling of dendritic spines. Modulation of the Rac1/PAK1 pathway may be a potential strategy for BCP treatment.
Introduction
Bone cancer pain (BCP), is a kind of chronic pain caused by primary or metastatic cancer, and that is currently incurable. 1 With the dramatic improvement in the detection and treatment, the survival rate of cancer patients has improved significantly, allowing more cancer patients to live with a tumor. 2 However, cancer-related pain has become a problem that greatly affects quality of life. Cancer pain occurs in 70% of patients with advanced cancer, and more than 80% of it is due to metastatic cancer-induced bone pain.3–5 BCP is one of the most severe pains and is difficult to manage because it is generally not confined to a single site of metastasis. However, the efficacy of common analgesics used to treat BCP such as NSAIDs and opioids, is limited by significant side effects.6–9 Therefore, an in-depth investigation of the pathophysiology of BCP may help identify a mechanism-based therapeutic approach.
Several studies have shown that significant pathological changes in the central nervous system (CNS) occur in animals with cancer pain and contribute to the development and maintenance of BCP.10–13 Central sensitization is a recognized mechanism underlying the occurrence and development of BCP. 1 Changes in the type and number of dendritic spines are the fundamental structural basis for the phenomenon of central sensitization.14,15 Dendritic spines are small finger-like protrusions on the surface of the dendrites of neurons, distributed throughout the nervous system and represent the synaptic connections through which neurons receive information. 16 More than 90% of the excitatory synapses in the central nervous system are located on dendritic spines. 17 Therefore, changes in the number, shape, and distribution of dendritic spines would strongly influence the process of synaptic plasticity.
Rho GTPases regulate cytoskeleton assembly, and mediate the growth and development of dendritic spines, which may be related to central sensitization. 18 As one of the main members of the Rho GTPase family, Rac1 is a molecular switch in the receptor-mediated cytoskeletal signal transduction pathway of the neuronal cell membrane. It binds to GTP through a functional domain and switches between active GTP-Rac1 and inactive GDP-Rac1. 19 In addition, as a downstream effector of Rac1, p-PAK1, links Rac1 signaling to cytoskeletal reorganization which underlies dendritic spine plasticity. 20 Previous studies have reported that Rac1 signaling plays a crucial role in the initiation and development of acute and chronic pain from burns, neuropathy, etc.21–24 Neuropathic pain can lead to some degree of dendritic spine remodeling and promote the increase of neuronal activity in WDR neurons in the deep lamellar layer of the dorsal horn of the spinal.25,26 The enhancement of neuronal synaptic plasticity is the basis of central sensitization in neuropathic pain. 27 Dendritic spine remodeling has been observed in a variety of neuropathic pain models including spinal cord injury (SCI),25,28,29 chronic constriction, 30 and burns, 31 as well as diabetic neuropathic pain. 26 NSC23766 can reverse dendritic spine remodeling and increase excitability of WDR neurons25,30 by downregulating Rac1/PAK1 signaling. The present study aims to investigate whether Rac1 signaling is involved in the initiation and development of BCP by regulating the plasticity of spinal cord dendritic spines. Understanding the pathophysiological mechanisms underlying BCP is important for developing effective treatment strategies.
Materials and methods
Animals, anesthesia, drugs, and administration
This study was conducted in strict accordance with the recommendations of the National Institutes of Health Guide for the Care and Use of Laboratory Animals. The experimental protocol was approved by the Animal Experimentation Ethics Committee of Xuzhou Medical University (permitting number: 202112A149).
Adult female Sprague-Dawley rats weighing 180–200 g (n = 96) were obtained from the Experimental Animal Research Center of Xuzhou Medical University. We chose females for this study because Walker 256 breast cancer cells can only grow in female rats. 32 Animals were kept in separate cages at a stable temperature range (22–25°C), free access to food and water, and a 12h light/dark cycle. The animals were randomly divided into 4 groups, including the sham group (Sham), sham + NSC23766 group (Sham + NSC), TCI + vehicle group (TCI + Veh), and TCI + NSC23766 group (TCI + NSC). The experiment was performed after a 3-days acclimation period. All surgeries were done under anesthesia with sodium pentobarbital (50 mg/kg, intraperitoneal). NSC23766 (1 mg/1 ml) (Selleck Chemicals, USA) was administrated intrathecally by lumbar puncture. The dose was chosen based on a previous study. 33
Model of bone cancer pain
The protocol was performed as described previously.34,35 Tumor cells were extracted from ascites fluid of rats inoculated with Walker 256 mammary gland carcinoma cells. To induce BCP, tumor cells were implanted into the medullary cavity of the right tibia by TCI. In brief, after pentobarbital anesthesia, rats were placed on the operating table with their abdomen up. After sterilization with 75% ethanol, an incision approximately 1 cm long was made in the tibial plateau area. Tumor cells were then injected into the medullary cavity of the right tibia via a micro syringe. The injection site was sealed with bone wax to prevent tumor cell leakage after completion of the injection. The wound was dusted with antibiotics and then sutured. The sham group was injected with an equal volume of boiled cells.
Behavioral test
Mechanical allodynia on the right hind paw was observed on days −3, −1, 1, 3, 5, 7, 9, 13, 17, 19, and 21 after surgery. A positive response was noted by a significant decrease in hind paw withdrawal threshold to mechanical stimuli at the plantar surface of the foot. During testing, after acclimating to the devices for at least 30 min, the rat was tested with a series of calibrated Von Frey filaments which were presented from below and held in the flexed position on the medial plantar surface of the hind paw for 3 s. The initial stimulus intensity was 2 g, and the stimulus intensity was gradually increased. Each intensity was repeated five times at 5-min intervals, and vigorous withdrawal of the paw, licking, or shaking was considered a positive response. Mechanical withdrawal threshold (MWT) was defined as the minimum force that could elicit at least three withdrawal responses from the right hind paw.
Western blotting
To determine the temporal expression of Rac1 signaling, fresh spinal cord tissue of the lumbar enlargement (L4-L6) was collected from deeply anesthetized rats and then homogenized in pre-cooled lysis buffer containing a cocktail of protease inhibitors. Total proteins were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred to a 0.2 mm polyvinylidene difluoride membrane (Millipore, IPV H00010). The following primary antibodies were used: anti-p-PAK1 (1:1,000, Affinity, AF3424), anti-Rac1 (1:200, Affinity, AF4200), and anti-GAPDH (1:1,000, Servicebio, GB12002). Horseradish peroxidase-conjugated antibodies were used as secondary antibodies. An ECL detection system (Beyotime) and ImageJ software were used to quantify the protein bands.
Immunofluorescence and image analysis
After deep anesthesia, the rats received 200 ml of 0.9% saline followed by 200 ml of 4% paraformaldehyde. The lumbar enlargement (L4-L6) was harvested and postfixed in 4% paraformaldehyde for 6–8 h, then transferred to a 30% sucrose solution and stored at 4°C, dehydrated and allowed to sink to the bottom. Twenty micrometer-thick frozen sections were cut with a freezing microtome and blocked in 10% donkey serum and 0.4% Triton X-100 in 0.01
Spinal cord subarachnoid catheterization
After anesthesia, the rats were placed in the prone position on the operating table. The dorsal skin was prepared and disinfected, a 1–1.5 cm longitudinal incision was made in the region of the spinous process (L3–L4), and muscle and fascia were bluntly separated to fully expose the spinous process space. The PE-10 catheter with a built-in guide wire was slowly inserted into the subarachnoid space. After observing the tail swing of the rat, which indicated that the catheter had reached the subarachnoid space, the guide wire was taken out and the catheter was slowly inserted into the subarachnoid cavum. A clear leakage of cerebrospinal fluid indicated a successful insertion. After ensuring that the depth of insertion of the catheter was 3 cm and the outer end of the catheter extended 2 cm posterior to the neck, the catheter was fixed and the incision was sutured. The exposed portion of the catheter was closed with hot melt adhesive. 20 μl lidocaine was injected through the catheter, and the motor behavior of the hind limbs of the rats was observed within 5 min to confirm whether the catheter was successfully placed.
Golgi-cox staining and dendritic spine counting
To observe synaptic structural plasticity, we used Golgi-Cox staining and counting of dendritic spines as previously described. 37 Staining was performed according to the manufacturer’s instructions of the FD Rapid Golgi Stain TM Kit (FD Neurotechnologies, Inc., Columbia, USA). Briefly, the spinal cord tissue was immersed in solutions A and B for 24 h, then the solution was changed and the samples stored in the dark at room temperature for 2 weeks. The spinal cord tissue was then stored in solution C in the dark at 4°C for another 72 h. A cryostat was used to prepare spinal cord slices and an optical microscope to image and analyze dendrites and dendritic spines in the dorsal horn of the spinal cord. Assessment of the overall complexity of WDR neurons by Sholl analysis. First, we used the Neuron ImageJ software to track the WDR neurons, got a 2D distribution map of neurons, then drew a concentric circle every 20 μm with the neuron cell body as the center, recorded the number of intersection points between each concentric circle and dendrites. We also measured the perimeter of the cell body, the total length of dendrites, the number of primary branches, and the percentage of primary branches with secondary branches to analyze the changes in neuron cell body and dendritic branches.
Transmission electron microscope
For transmission electron microscopy, spinal cords were divided into small fragments and fixed overnight at room temperature in 4% cacodylate-buffered glutaraldehyde (pH 7.40). Then, the spinal cord was cut into 1 mm thick slices, and the deep lamellar dorsal horn of the spinal cord (IV-V) was harvested according to the atlas. After rinsing three times in 0.1
Statistical analysis
All experimental data are presented as mean ± SEM. GraphPad Prism (version 8.0.2; GraphPad Software, Inc., USA) was used for statistical analyses. Comparison of values between different groups was done using one-way ANOVA followed by Bonferroni post hoc tests. Sholl analysis of dendrite distribution and time-series data between different groups were analyzed by repeated measures two-way ANOVA. Statistical results were considered significant if the p-value was less than 0.05.
Results
Rac1/PAK1 signaling in the nociceptive pathway is upregulated during BCP
Changes in the spinal cord induced by tumor growth are critical for BCP. We first examined the expression of Rac1 and PAK1 in the spinal cord. Western blot showed that although the total levels of Rac1 and PAK1 in the spinal cord did not change significantly after TCI treatment, the expression of their activated forms GTP-Rac1 and p-PAK1 increased significantly in a time-dependent manner (Figure 1(a) and (b)). The increased expression of GTP-Rac1 and p-PAK1 began at day5 after TCI, and peaked at day 7–14, and then decreased slightly at day 21 after surgery. At the same time, immunofluorescence also showed that the increase in p-PAK1 was distributed throughout the dorsal horn of the spinal cord (Figure 1(c)). The Rac1/PAK1 signaling is activated in the spinal cord after TCI treatment. (a, b) The expression of GTP-Rac1 and p-PAK1 in the spinal cord after TCI. (c) Immunofluorescence shows the expression of p-PAK1 in the spinal cord. Tissues were collected on postoperative day 14. Four samples were included in each group. *p < 0.05, **p < 0.01, ***p < 0.001 indicates significant differences compared with sham group. Scale bar: 200 μm.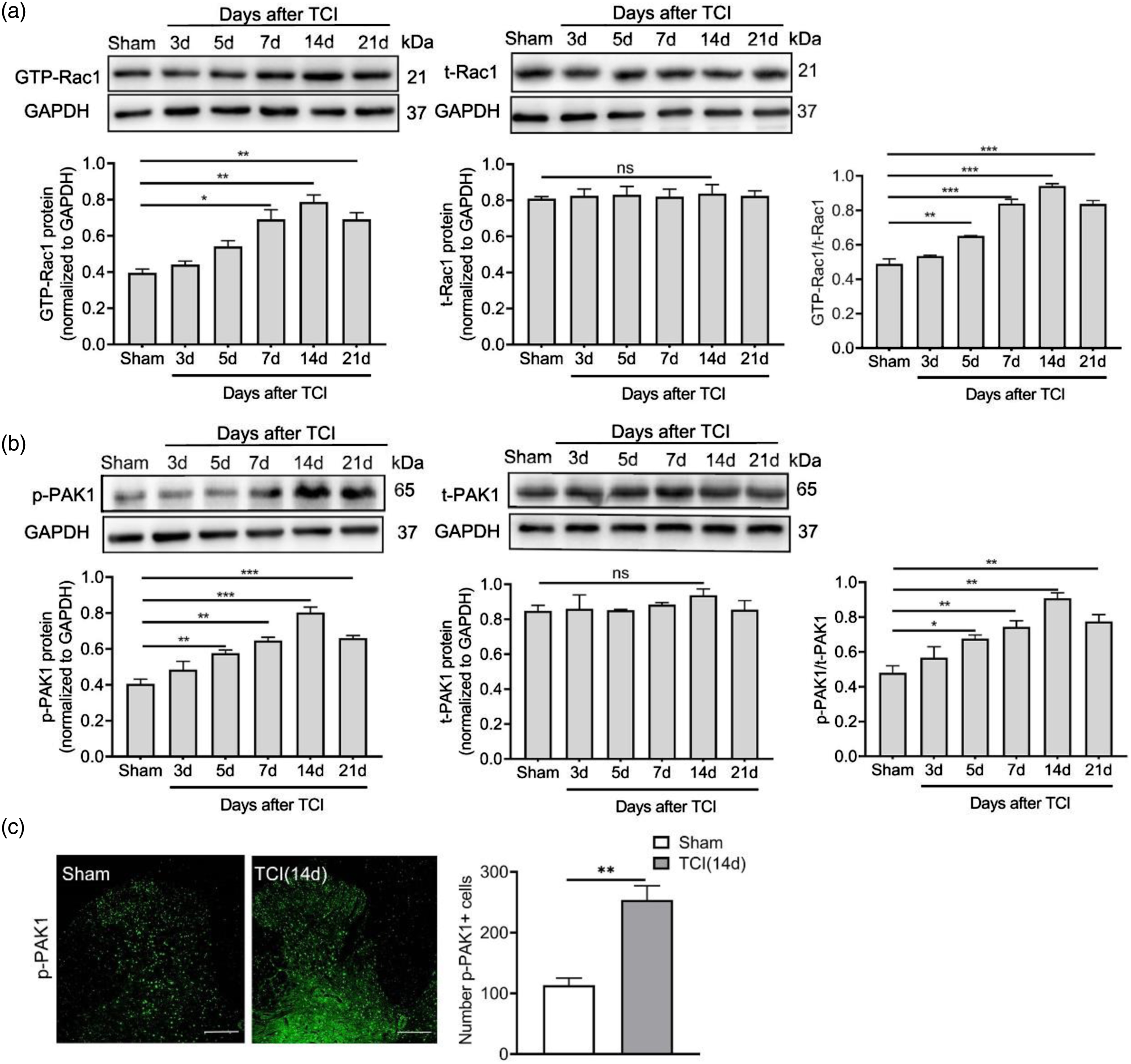
Rac1 and PAK1 are mainly expressed in neurons of the dorsal horn of the spinal cord
To explore the location of Rac1 and PAK1 in dorsal horn of the spinal cord, we performed co-staining of Rac1 and PAK1 with neuronal and glial markers, respectively. As shown in Figures 2(a) and (b), double immunofluorescence staining revealed that Rac1 is mainly present in the superficial layer of the spinal dorsal horn, and co-localized mainly with neurons (NeuN), but hardly co-localized with microglial cells (Ibal) or astrocytes (GFAP). The deep dorsal horn of the spinal cord, showed negligible expression levels of Rac1 in astrocytes and microglia, and low levels in neurons (Figure 2(c)). Rac1 is mainly expressed in neurons. (a) Immunofluorescence showing the colocalization of Rac1 (green) with neurons (NeuN, red), astrocytes (GFAP, red), and microglial cells (Iba1, red) in the dorsal horn of the spinal cord (Scale bar 150 μm, n = 4). a1-c1 and a2-c2 are magnifications of the superficial and deep dorsal horns of the spinal cord, respectively (Scale bars 50 μm). Tissues were collected on postoperative day 14. (b) Quantitative statistics of co-localization of Rac1 with NeuN, Iba1 and GFAP. (c) Co-localization of Rac1 with NeuN in the superficial and deep layers of the dorsal horn of the spinal cord. ***p < 0.001 indicate significant differences.
The expression and distribution of PAK1 were consistent with that of Rac1. Compared with the Sham group, the expression of p-PAK1 was up-regulated in the superficial and deep layers of the dorsal horn of the spinal cord (Figure 3(a) and (b)). p-PAK1 was predominantly co-labeled with neurons but rarely co-labeled with microglia or astrocytes (Figure 3(a) and (c)). At the same time, the number of activated microglia and astrocytes also increased (Figure 3(d) and (e)). The increase of PAK1 after TCI was accompanied by the activation of glial cells and was mainly co-stained with neurons. (a)Immunofluorescence showed the colocalization of PAK1 (green) with neurons (NeuN, red), astrocytes (GFAP, red), and microglial cells (Iba1, red) in the dorsal horn of the spinal cord (Scale bar 150 μm, n = 4). a1-f1, a2-f2 are local magnifications of the superficial and deep dorsal horns of the spinal cord (Scale bar 50 μm). (b) The number of p-PAK1 in the dorsal horn of spinal cord in sham group and TCI group; (c) the number of p-PAK1 co-labeled with neurons; (d, e) the number of Iba1+ and GFAP+ cells in the dorsal horn of spinal cord in sham group and TCI group. **p < 0.01, ***p < 0.001 indicate significant differences compared with Sham group.
Blockade of Rac1/PAK1 signaling significantly reversed pain behaviors caused by BCP
TCI treatment could induce significant hyperalgesia and allodynia. Intrathecal injection of NSC23766 (5 μl, 1 mg/ml, daily for 3 consecutive days) on postoperative days 3, 4, and 5 (the early phase) and on postoperative days 7, 8, and 9 (the later phase) significantly suppressed bone cancer-related pain behavior, but showed no effects on normal pain perception (Sham + NSC group) (Figure 4(a)–(d)). The early administration cannot completely inhibit the production of BCP, but it can delay the development of BCP. It is worth noting that, regardless of the intensity of analgesia or the persistence of analgesia, the effect of the late administration is better than that of the early stage. Western blot showed that the expression of GTP-Rac1 and p-PAK1 in the spinal cord was significantly increased after TCI treatment and intrathecal injection of NSC23766 effectively reduced the increases (Figure 4(e) and (f)). Immunofluorescence results showed that the expression of p-PAK1 was up-regulated in the TCI group compared with the Sham group, while down-regulated after NSC23766 treatment (Figure 4(g)). These results suggest that Rac1/PAK1 signaling may play an important role in the development of BCP induced by TCI. Blockade of the Rac1/PAK1 signaling suppresses bone cancer-induced pain behaviors. Behavioral changes after intrathecal injection of NSC23766 in the later phase (postoperative days 3, 4. and 5 or 7, 8, and 9) (a, b). (b, d) are the areas under the curves of a, c respectively. (e, f)The expression of GTP-Rac1 and p-PAK1 after injection of NSC23766. (f) The expression of p-PAK1 in the dorsal horn of the spinal cord (Scale bar 200 μm, n = 4). *p < 0.05, **p < 0.01, ***p < 0.001 indicate significant differences compared with Sham group; #p < 0.05, ##p < 0.01, ###p < 0.001 indicate significant differences compared with TCI + Veh group.
Blockade of Rac1/PAK1 signaling reversed bone cancer-related spinal cord dendritic spine remodeling
Golgi-Cox staining and dendritic spine counts were used to examine the effects of TCI and Rac1/PAK1 pathway on WDR neurons. The results of Sholl analysis (Figure 5(b)) showed that neither the TCI + Veh group nor the TCI + NSC group had a significant effect on the overall complexity of WDR neurons. Furthermore, neuron cell body circumference, total dendritic length, number of primary branches, percentage of primary branches with secondary branches, and other related parameters were statistical analyzed. The results showed that both TCI + Veh group and the TCI + NSC group had no obvious changes on the above parameters. Although the overall complexity of WDR neurons had not changed significantly, there were obvious changes in the density of dendritic spines of WDR neurons. We counted the number of spines in the dendrites per unit length (μm) and found 911 dendritic spines in Sham group, 1468 dendritic spines in TCI + Veh group, and 1146 dendritic spines in TCI + NSC group. They were then classified into the following standard categories
38
stubby, mushroom, and thin spines (Figure 5(c), c1–c3). The overall density of dendritic spines increased after TCI treatment. The density of thin and mushroom dendritic spines increased significantly, while the density of stubby dendritic spines showed no variation. NSC23766 (i.t.) reversed the increase in dendritic spine density (Figure 5(d)). Effects of blocking Rac1/PAK1 signaling on WDR neuron development. (a) 2D schematic diagrams of neurons in the sham group, TCI + Veh group, and TCI + NSC group. Scale bar 100 μm. (b) The result of the Sholl analysis (n = 16). Above is a schematic diagram of the Sholl analysis. (c) Representative dendritic spines images of Sham group, TCI + Veh group, and TCI + NSC group. Scale bar 10 μm. (c1-c3) Schematic diagrams of thin, mushroom, and stubby dendritic spines respectively. Scale bar 1 μm. (d) The changes in dendritic spine density of various types and the effects of NSC23766 (i.t.) after TCI (n = 6). *p < 0.05, **p < 0.01 indicate significant differences compared with Sham group; #p < 0.05, ##p < 0.01 indicate significant differences compared with TCI + Veh group.
Using transmission electron microscopy, we found that TCI treatment caused dorsal remodeling of the excitatory synaptic structure in the deep stratum corneum (IV -V) compared with the Sham group (spinal cord tissue was harvested on day 14 after TCI) (Figure 6(b)). This remodeling was mainly reflected in an increase in postsynaptic density (PSD) length (Figure 6(e)), widening of the cleft (Figure 6(f)), and an increase in PSD thickness (Figure 6(g)), whereas the curvature of the synapses did not change significantly (Figure 6(h)). Interestingly, NSC23766 (i.t.) was able to reverse the observed remodeling of excitatory synapses (Figures 6(c), (e)–(g)). Intrathecal injection of NSC23766 significantly inhibits TCI-induced remodeling of spinal dorsal cord dendritic spines. (a–c) Image of excitatory synapse in Sham group, TCI + Veh group, and TCI + NSC group. Magnification: 30,000 ×, Scale bars 200 nm, Sv: synaptic vesicle; Pre-: presynaptic membrane; Cleft: synaptic cleft; PSD: post-synaptic density. (d) PSD thickness calculation method is PSD area S/PSD length L, PSD area is hand-drawn with ImageJ software, synaptic curvature is postsynaptic membrane arc length a/chord length b, and the synaptic gap width is measured by multi-point measurement and average method. (e–h) The changes of synaptic PSD length, Cleft width, PSD thickness, and synaptic curvature after TCI and after TCI + NSC23766 (i.t.) respectively. n = 16 pre-group. *p < 0.05 indicate significant differences compared with Sham group; #p < 0.05, ##p < 0.01 indicate significant differences compared with TCI + Veh group.
Discussion
This study reveals to a critical role of Rac1/PAK1 signaling pathway in the development of BCP induced by TCI through the regulation of dendritic spine remodeling. In short, (1) TCI treatment caused upregulation of GTP-Rac1 (activated form of Rac1), and phosphorylated PAK1 in the spinal dorsal horn. Moreover, these activated molecules mainly co-labeled with neurons; (2) Blocking of Rac1/PAK1 signaling decreased the expression of GTP-Rac1 and PAK1 and inhibited the pain behavior induced by TCI treatment; (3) Spinal dendritic spines were remodeled during the development of BCP, and this phenomenon could be reversed by blocking Rac1/PAK1 signaling. These results partly uncovered the mechanism of BCP and provide a potential target for treatment.
Dendritic spines are small, specialized protrusions of the dendrites of excitatory neuron. Classification of spines is largely based on their shape, with four categories generally accepted: filopodial, thin, stubby, and mushroom spines. 39 The major components of spines include cytoskeletal structures (actin), cell membrane receptors, other cytoskeletal proteins, small GTPase and associated proteins, PSD region, etc. 40 Morphology and number of dendritic spines have been reported to play an important role in the development, maintenance and plasticity of synapses under physiological and pathological conditions. 41 Actin regulatory proteins are controlled by Rho GTPases, which switch between active and inactive states via the hydrolysis of GTP. 42 Rac1, an important member of the GTPase family,43,44 is involved in neurite outgrowth and in the maintenance of mature dendritic spines.45,46 Rac1 signaling is known to be important in neuronal development, 47 degenerative diseases of the nervous system,48,49 tumor invasion and migration,50–52 synaptic function,30,53 and learning and memory.54,55 In this study, we found that TCI caused a time-dependent increase in GTP-Rac1 and phosphorylated PAK1. Activation of Rac1 signaling in the spinal cord correlated well with the temporal pattern of BCP after TCI treatment. Rac1 and PAK1 signaling occurred primarily through neurons, where they were mainly expressed. Intrathecal injection of NSC23766 in the early phase of BCP (postoperative day 3, 4, 5) and in the later phase of BCP (postoperative day 7, 8, 9) can relieve pain, but in the later phase analgesic intensity and durability are better than early administration. This difference may be related to dendritic spine remodeling and Rac1 expression level during BCP. It has been reported in the literature that during STZ-induced diabetic neuralgia and chronic constriction injury (CCI),26,30 dendritic spine remodeling occurs on pain days 7-14. As one of the important members of the GTPase family, Rac1 is mainly involved in maintaining the function of mature dendritic spines.43–46 These results suggested that the Rac1/PAK1 signaling was activated during BCP. Activation of Rac1/PAK1 signaling can phosphorylate cofilin, which facilitates retraction and stabilization of dendritic spines.56,57 The signaling molecules that mediate dendritic spine morphogenesis may also be involved in the regulation of BCP.
WDR neurons relay information through the spinothalamic tract, which is the main pain pathway of the brain. These cells exhibit high excitability in a variety of neuropathic pain injuries and diseases. 58 We observed the ultrafine structure of WDR neurons and synapses by Golgi staining, fluorescence and electron microscopy. The results showed that the complexity of WDR neurons had not changed, but the morphology of neuronal dendritic spines had changed significantly. The thin and mushroom spines as well as the total density of dendritic spines increased significantly after TCI treatment. The increase in the density of mushroom spines indicates that the strength of the synapses increased, and the activity of neurons increased accordingly. 59 Besides, the PSD length and the thickness of excitatory synapses in the deep dorsal horn of the spinal cord (IV-V) increased, and the synaptic space widened. Changes in the number, morphology, and distribution of dendritic spines leaded to increased excitability of the postsynaptic membrane. These changes resulted in remodeling of somatosensory neurons and enhanced neuronal function and signaling in nociceptive circuits. Intrathecal injection of NSC23766 was able to reverse the above changes and relieved BCP. These results suggested that Rac1 signaling is involved in the regulation of BCP by mediating dendritic spine remodeling.
In recent years, research on the relationship between Rac1 and pain has made great progress. In several neuropathic pain models25,29 remodeling of the dendritic spine has been observed, as evidenced by an increase in the density of the neuron’s dendritic spines, an altered spatial distribution, and an increase in the length of the dendritic spines and the diameter of the head. Typically, spinal cord dendritic spine remodeling is accompanied by high neuronal excitability. The enhancement of neuronal synaptic plasticity is the basis of neuropathic central sensitization. 27 Central sensitization leads to hyperalgesia and paresthesias, which aggravate pain. In this neuropathic pain, the use of NSC23766 can significantly reduce pain by decreasing neuronal hyperexcitability and reversing dendritic spine remodeling. Activation of Rac1 was also observed in acute inflammatory pain induced by bee venom, which was also significantly relieved by NSC23766. 33 However, whether this peripheral neuritis pain is accompanied by remodeling of spinal cord dendritic spines is not addressed.
Conclusions
In summary, this study reveals a role of Rac1/PAK1 signaling in BCP. Activation of Rac1/PAK1 signaling is critical for the maintenance of BCP. The underlying mechanism may involve sensitization of WDR neurons and regulation of dendritic spine plasticity. Therefore, inhibition of Rac1/PAK1 signaling activation may be a potential effective therapy for BCP.
Footnotes
Acknowledgements
We thank the Key Laboratory of Anesthesiology, Xuzhou Medical University for providing technical support and experimental platform.
Author Contributions
SL and Y-PL as the primary and corresponding authors supervised all experiments and funded the work; L-FX and X-XW. wrote the first draft and designed the research; L-FX, LY, WY and X-XW performed the experiments and analyzed data. SL, L-FX, X-HT, Y-QX, and Q-SC revised the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article. This work was supported by the grants from Natural Science Research Fund of Higher Education Institutions in Jiangsu Province (22KJA320007); Jiangsu Province Postgraduate Research and Practice Innovation Program (KYCX22_2954); Qing Lan Project of Jiangsu Province (53041605); Key R&D Program of Xuzhou (Social Development) (KC20153).
Correction (June 2023):
The funding details have been updated.
