Abstract
Resolvin D1 (RvD1) suppresses inflammatory, postoperative, and neuropathic pain. The present study assessed the roles and mechanisms of RvD1 in mechanical allodynia after burn injury. A rat model of burn injury was established for analyses, and RvD1 was injected intraperitoneally. Pain behavior and the expression levels of spinal dorsal horn Iba-1 (microglia marker), GFAP (astrocyte marker), p-p38 mitogen-activated protein kinase (MAPK), brain-derived neurotrophic factor (BDNF), and tropomyosin-related kinase B (TrkB) were detected by behavioral and immunocytochemical assays. The results showed that RvD1 attenuated mechanical allodynia after burn injury, prevented microglial and astroglial activation, and downregulated p-p38 MAPK in microglia and BDNF/TrkB following burn injury. Similarly, inhibition of p38 MAPK and BDNF/TrkB signaling attenuated mechanical allodynia after burn injury. In addition, inhibition of p38 MAPK prevented spinal microglial activation and downregulated BDNF/TrkB following burn injury. Furthermore, inhibition of BDNF/TrkB signaling prevented spinal microglial activation and downregulated p-p38 MAPK within spinal microglia. Taken together, this study demonstrated that RvD1 might attenuate mechanical allodynia after burn injury by inhibiting spinal cord glial activation, microglial p38 MAPK, and BDNF/TrkB signaling in the spinal dorsal horn.
Introduction
Burns are very common and sometimes fatal, and the associated pain is difficult to relieve. Worldwide, 11 million individuals are affected yearly by burns with sufficient severity to seek medical care. 1 Due to the heavy burden of burn wounds, related therapeutics are being developed. 2 However, treatment options for such sensory ailments remain limited and often ineffective.
Resolvins (Rvs), comprising RvD and RvE, represent endogenous lipid modulators produced during the termination of acute inflammation, with strong anti-inflammatory and pro-resolution effects demonstrated in multiple experimental studies.3,4 RVs may constitute a novel class of analgesics for managing inflammatory pain in arthritis. 5 Recent evidence suggests that RvD1 markedly dampens postsurgical pain and neuropathic pain by modulating the activities of a variety of transient receptor potential channels and regulating proinflammatory and anti-inflammatory mediators in the development of central sensitization.6–12 Burn injury–associated pain likely comprises inflammatory and neuropathic elements. 13 However, whether RvD1 relieves burn pain remains unknown.
Glial cell activation and neuron–glial cell interactions are currently considered important mechanistic pathways of chronic pain. 14 Upon nerve damage, inflammation, bone malignancy, and chronic exposure to opioids, activated glial cells may produce and release glial mediators,14–19 which control the activities of neurons and synapses as well as pain sensitivity, contributing to pain persistence and morphine tolerance.14–19 A recent study revealed that burn injury has a significant association with elevated spinal cord’s glial cell activity. 13 In vivo and in vitro studies have indicated that RVs inhibit microglia and astrocyte activation, and reduce the secretion of inflammatory factors.20,21 However, the effects of RVs on burn injury–induced glial activation remains unknown.
Recent studies assessing central neuropathic pain (CNP), peripheral neuropathic pain, and central sensitization suggested that induction of important signaling proteins, including MAPK family members, contributes to dorsal horn hyperexcitability and affects the occurrence and maintenance of mechanical allodynia.12,22–28 Crown and colleagues showed that occurrence of at-level CNP upon minor spinal cord damage correlates with elevated amounts of activated/phosphorylated extracellular signal–related kinases (ERKs) and p38 MAPK.26,27 Inhibiting p38,s enzymatic activity concentration-dependently reverses at-level mechanical allodynia and reduces hyperexcitability of thoracic dorsal horn neurons upon minor spinal cord injury (SCI). 29 Hains and colleagues found that rats with SCI administered minocycline show reduced below-level pain associated with decreased glial activation of p38 MAPK. 30 Burn injury–induced pain probably has inflammatory and neuropathic components, 13 and RVs can also inhibit glial activation, attenuate MAPK signaling, and reduce the secretion of inflammatory mediators in neuropathic and inflammatory pain.20,21,31 However, the associations of p38 MAPK with burn pain and RVs remain unknown.
BDNF modulates synaptic transmission and plasticity in multiple brain areas via binding to TrkB.32,33 BDNF is considered an effector in aberrant pain signaling between spinal microglia and neurons.12,16,18,34 Following peripheral nerve injury, BDNF secreted by induced microglia participates in neuropathic pain.35–38 In addition, BDNF and TrkB are upregulated under both inflammatory39–41 and neuropathic42–44 pain conditions. Numerous reports have indicated that induced BDNF/TrkB signaling contributes to neuropathic pain.45–47 A recent study indicated BDNF could activate spinal long-term potentiation (LTP) at C-fiber synapses as well as mechanical hypersensitivity upon peripheral nerve damage via microglial activation, 48 which could be suppressed by RVs.20,21
We hypothesized that RvD1 attenuates mechanical allodynia after burn injury, with the involvement of spinal glia, p38 MAPK, and BDNF/TrkB signaling.
Material and methods
Animals
Male Sprague–Dawley (SD) rats (aged 4–5 weeks and weighing 140–150 g) were purchased from the Experimental Animal Center, the First Affiliated Hospital of Zhengzhou University (Zhengzhou, China). Housing was performed in individual cages under a 12h–12h light/dark cycle at ambient temperature with food and water at will, for 1 week before initiating the study. According to the International Association for the Study of Pain guidelines for pain research in animals, experiments involving animals were approved by the Animal Care and Use Committee of the First Affiliated Hospital of Zhengzhou University (No. 2014-002).
The effects of RvD1 were investigated by randomly dividing the rats into five groups (n = 6): sham group (Sham + Veh1), burn group (Burn + Veh1), small-dose (100 ng) RvD1 treatment group [Burn + R(S)], large-dose (300 ng) RvD1 treatment group [Burn + R(L)], and sham + large-dose RvD1 treatment group [Sham + R(L)]. The vehicle [Veh1, PBS containing 0.1% ethanol] or RvD1 (100 or 300 ng in 50 μL of Veh1) was administered intraperitoneally (ip) 30 min before burn injury and subsequently twice a day for 7 days. Less than 1 h before injection, the aqueous solution of RvD1 (Cayman Chemical, USA) was prepared by evaporating the stock RvD1 to dryness under a gentle steam of nitrogen and immediately adding the Veh1, while minimizing exposure to light.
The effects of p38 MAPK were investigated by randomly dividing the animals into five groups (n = 6): sham group (Sham + Veh2), burn group (Burn + Veh2), SB203580(Abcam, UK, which inhibits the enzymatic activity of p38 MAPK on its downstream targets) treatment group (Burn + SB203580), sham + SB203580 treatment group (Sham + SB203580), and large-dose RvD1 treatment group [Burn + R(L)]. SB203580 (10 μ
The effects of BDNF/TrkB were investigated by randomly dividing the rats into five groups (n = 6): sham group (Sham + Veh3), burn group (Burn + Veh3), TrkB-Fc (Abcam, UK, a specific BDNF inhibitor)
48
treatment group (Burn + TrkB-Fc), sham + TrkB-Fc treatment group (Sham + TrkB-Fc), and large-dose RvD1 treatment group [Burn + R(L)]. TrkB-Fc (100 μg, 10 μL) or vehicle (Veh3, 0.01
In these experiments, TrkB-Fc and RvD1 doses were based on Zhou et al. 48 and Lima-Garcia et al., 49 respectively.
Burn injury
Burn injury was induced as reported previously. 13 Briefly, the animals were administered anesthesia by isoflurane inhalation, and the hind paw’s dorsum was immersed in hot water (85°C) for 12 s. In the sham group, immersion was performed in warm water (35°C) instead. Iodine application was used daily to prevent infection.
Intrathecal catheter
Seven days before burn injury, the animals were administered inhalation anesthesia with isoflurane, followed by insertion of a polyethylene-10 catheter into the lumbar subarachnoid space as previously described. 13 Only the animals with no neurological deficits preceding lidocaine administration but showing paralysis of both hind limbs underwent further investigation.
Nociceptive behavioral testing
The animals were allowed a 2-day adaptation period before burn injury. The nociceptive behaviors of the rats in the Sham + Veh2, Burn + Veh2, Burn + SB203580, Sham + SB203580, and Burn + R(L) groups (n = 6 per group) were tested before the intraspinal injection, and 15, 30, 60, and 120 min upon the intraspinal injection 7 days after burn injury. The nociceptive behaviors of the other groups (n = 6 per group) were tested before burn injury and at 1, 3, 5, 7, and 14 days post-burn injury. Upon adaptation (30 min), mechanical thresholds were measured by hind paw withdrawal to a graded series of von Frey filaments (Stoelting, USA) (4–26 g) applied to the plantar area of each hind paw. The modified Dixon “up–down” technique was applied to determine the mechanical nociceptive threshold, as described previously. 22 The experimenter was blinded to the groups of rats.
Immunofluorescence
Immunofluorescence detecting Iba-1 and GFAP (microglial and astroglial activation, respectively), and BDNF/TrkB was carried out in distinct juvenile animals 7 days after burn injury (n = 3 per group). Immunofluorescent staining for p-p38 MAPK was performed after measuring the mechanical withdrawal threshold (MWT) following SB203580 administration. For spinal cord immunofluorescent staining, the rats were terminally administered anesthesia by isoflurane inhalation, followed by transcardial perfusion with heparinized saline and 4% formalin in PBS. After excision, spinal cord lumbar segments underwent postfixation overnight, storage in 20% sucrose (3 days), and sectioning using a cryostat (25 μm). After blocking (3% bovine serum albumin in 0.3% Triton X-100) for 1 h ambient, incubation were carried out overnight at 4°C with primary antibodies targeting GFAP, Iba1, BDNF, TrkB (Abcam, UK), and p-p38 MAPK (Santa Cruz, USA). This was followed by incubation with appropriate fluorescein isothiocyanate- or cyanine 3 (CY3)-conjugated secondary antibodies (Jackson, USA). Spinal cord cells that expressed p-p38 MAPK were examined by double staining with anti-NeuN (Abcam; neuronal marker), GFAP (astrocytic marker), or Iba1 (microglial marker). GFAP was stained red to distinguish it from p-p38 MAPK. Laser confocal microscopy (Zeiss LSM 510; Zeiss, Germany) was used to observe and image the dorsal horn area of the spinal cord (1 field per section). Image Pro Plus 6.0 was used for quantifying GFAP, Iba1, p-p38 MAPK, BDNF, and TrkB, based on the IOD (integrated optical density) values of immunofluorescence intensity. Measurement of signal co-localization was performed with NIH Image J program. Five spinal cord sections of each animal were randomly selected. The experimenter who performed the immunofluorescent staining was blinded to the groups of rats.
Statistical analysis
All data are mean ± standard error of the mean (SEM), and were assessed by one-way analysis of variance (ANOVA) followed by Tukey HSD test for between-group comparisons. The sample size is indicated in each figure legend. SPSS 17.0 (SPSS, USA) was employed for data analysis. p < 0.05 indicated statistical significance.
Results
RvD1 attenuates mechanical allodynia after burn injury
In order to examine whether RvD1 relieves burn pain, the mechanical thresholds were measured. Sham rats which received 300 ng RvD1 (ip) and vehicle had similar pain responses. However, in rats with burn injury receiving i.p. vehicle treatment, mechanical allodynia was obvious on the ipsilateral side at 1–14 days (Figure 1); meanwhile, 100 or 300 ng RvD1 administered i.p. attenuated mechanical allodynia at 1, 3, 5, 7, and 14 days upon burn injury in comparison with the vehicle group (Figure 1). These data suggested that RvD1 had persistent beneficial effects up to day 14 after burn injury, although the drug was discontinued at 7 days following burn injury. In comparison with the burn group receiving 100 ng RvD1, the MWT in animals receiving 300 ng RvD1 was significantly increased upon burn injury (p < 0.05). These findings suggested that RvD1 attenuated burn injury–associated mechanical allodynia without interfering with normal pain perception. MWT values in different groups at the indicated times after burn injury. The vehicle or RvD1 was administered intraperitoneally (ip) 30 min before burn injury and subsequently twice a day for 7 days: ap < 0.05 versus pre-burn; bp < 0.05 versus Sham + Veh1; cp < 0.05 versus Burn + Veh1; dp < 0.05 versus Burn + R(S). R(S): small-dose (100 ng) RvD1 treatment; R(L): large-dose (300 ng) RvD1 treatment. n = 6 rats from a single experiment.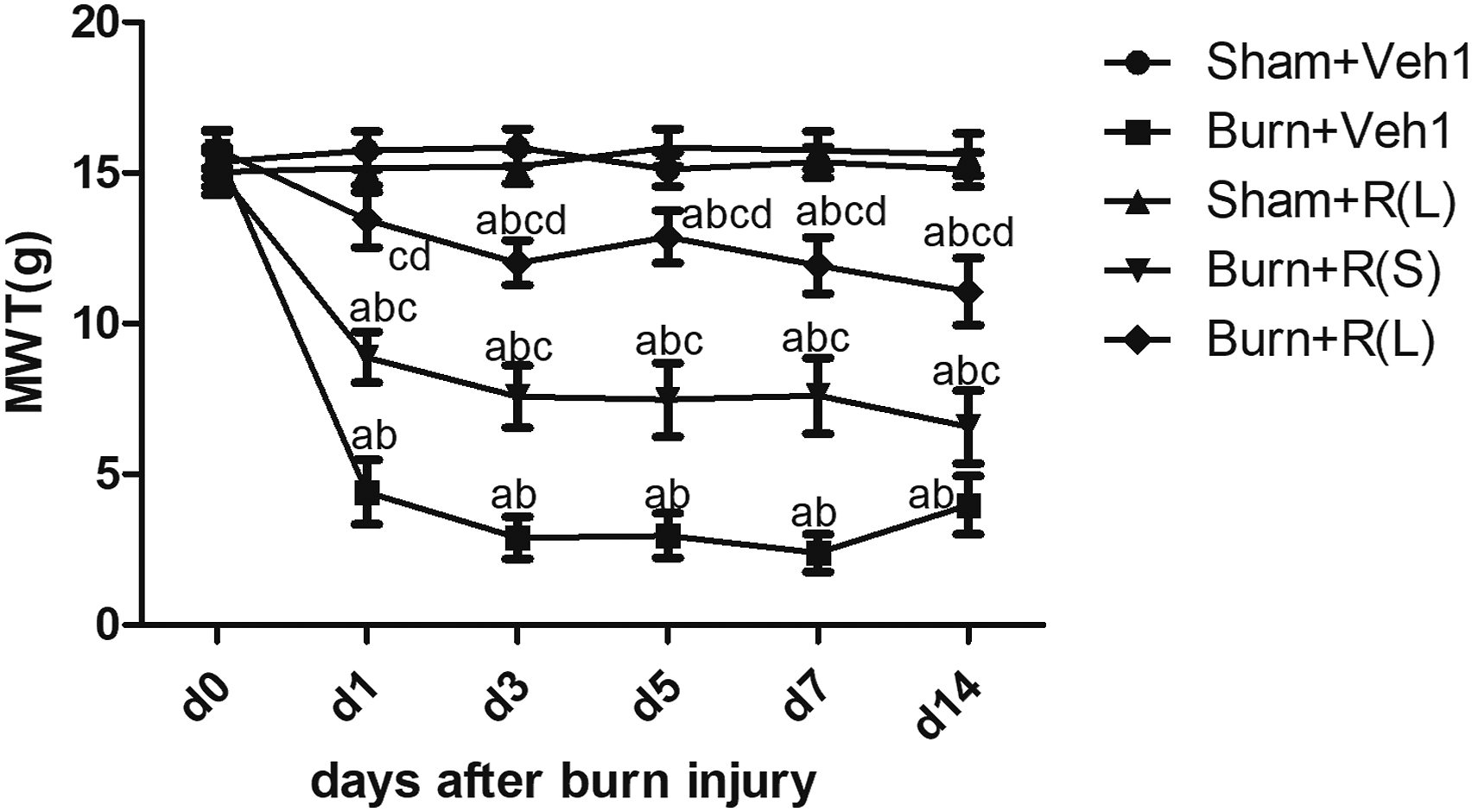
RvD1 prevents the activation of spinal microglia and astroglia, and downregulates p-p38 MAPK and BDNF/TrkB following burn injury
In order to examine the associations of spinal microglia and astroglia, p38 MAPK and BDNF/TrkB with burn pain and RvD1, immunofluorescent staining for Iba-1 and GFAP (microglial and astroglial activation, respectively), p-p38 MAPK and BDNF/TrkB were performed 7 days following burn injury. Dorsal horn immunoreactive signals for Iba1, GFAP, p-p38 MAPK, BDNF, and TrkB on the ipsilateral side of burn injury were significantly increased (p < 0.05) in the burn group in comparison with sham burn (Figure 2, Figure 3). RvD1 administration to burn-injured rats prevented burn injury–associated upregulation of Iba1, GFAP, p-p38 MAPK, BDNF, and TrkB (Figure 2). Next, spinal cord cells expressing p-p38 MAPK were examined by double staining with Iba-1 GFAP, or NeuN. Furthermore, p-p38 MAPK was found to be mostly co-localized with Iba-1-positive microglia (86%), but rarely with GFAP-expressing astrocytes (0.6%) and NeuN-positive neurons (5.9%) (Figure 3). These findings suggested that RvD1 prevented the activation of spinal microglia and astroglia, and downregulated p-p38 MAPK in microglia and BDNF/TrkB following burn injury. RvD1 prevents the activation of spinal microglia and astroglia and downregulates p-p38 MAPK and BDNF/TrkB following burn injury. The Immunofluorescence images show the expression of GFAP (astrocytic marker), Iba1 (microglial marker), p-p38 MAPK, BDNF(Brain-derived neurotrophic factor) and TrkB (tropomyosin-related kinase B) in spinal dorsal horn of rats. The IOD (integrated optical density) values of immunofluorescence intensity are shown in P-T. ap < 0.05 versus Sham + Veh1 group; bp < 0.05 versus Burn + Veh1 group. Scale bar = 20 μm. n = 3 rats per group. P–p38 MAPK (green) mostly co-localizes with Iba-1-positive microglia (red, 86%) but rarely with GFAP-positive astrocytes (red, 0.6%) and NeuN-positive neurons (red, 5.9%). ap < 0.05 versus p-p38/NeuN; bp < 0.05 versus p-p38/GFAP. Scale bar = 20 μm. n = 3 rats per group.

Suppression of p38 MAPK with SB203580 attenuates mechanical allodynia after burn injury
To investigate the role of p38 MAPK in the mechanism of RvD1 relieving burn pain, the mechanical thresholds were measured after p38 MAPK inhibition with SB203580. As found with RvD1, SB203580 had no impact in the sham group (Figure 4). In contrast, in rats with burn injury, SB203580 significantly attenuated mechanical allodynia (Figure 4, p < 0.05), similar to RvD1 (Figure 4, P > 0.05). In addition, the vehicle-treatment group with burn injury had markedly greater responses (p < 0.05) to von Frey stimuli compared with the remaining groups with burn injury (Figure 4). The above data indicated that p38 MAPK inhibition attenuated burn injury–induced mechanical allodynia. MWT values in different groups at the indicated times upon the intraspinal injection 7 days after burn injury: ap < 0.05 versus pre-injection; bp < 0.05 versus Sham + Veh2; cp < 0.05 versus Burn + Veh2; dp < 0.05 versus Burn + SB203580. R(L): large-dose (300 ng) RvD1 treatment. n = 6 rats from a single experiment.
Suppression of p38 MAPK with SB203580 prevents spinal microglial activation and downregulates BDNF/TrkB upon burn injury
Then we assessed the expression levels of Iba-1, BDNF and TrkB after p38 MAPK inhibition with SB203580. The expression levels of Iba-1, BDNF and TrkB in SB203580 treated rats with burn injury were markedly reduced in comparison with the vehicle-treatment group with burn injury, but still elevated compared with sham animals without injury (Figure 5, p < 0.05). The above data showed that induction of spinal microglia and BDNF/TrkB expression following burn injury were prevented by SB203580. Inhibition of p38 MAPK by SB203580 prevents spinal microglial activation and BDNF/TrkB downregulation following burn injury. The Immunofluorescence images show the expression of Iba1 (microglial marker), BDNF(Brain-derived neurotrophic factor) and TrkB (tropomyosin-related kinase B) in spinal dorsal horn of rats. The IOD (integrated optical density) values of immunofluorescence intensity are shown in J-L. ap < 0.05 versus Sham + Veh2 group; bp < 0.05 versus Burn + Veh2 group. Scale bar = 20 μm. n = 3 rats per group.
Intrathecal injection of TrkB-Fc attenuated mechanical allodynia after burn injury
To investigate the role of BDNF/TrkB in the mechanism of RvD1 relieving burn pain, the mechanical thresholds were measured after BDNF/TrkB signaling inhibition with TrkB-Fc. As shown in Figure 6, burn injury decreased the MWT on the ipsilateral side in vehicle-treated rats (vs pre-burn and sham injury groups, p < 0.05), but not in the TrkB-Fc-treatment group at 3 and 14 days (vs pre-burn and sham injury groups, p > 0.05). After burn injury, the MWT was markedly elevated in TrkB-Fc-treated animals with burn injury in comparison with the vehicle-treatment group with burn injury (p < 0.05), but similar to the MWT in RvD1-treated animals (Figure 6, p > 0.05). These findings suggested TrkB-Fc alleviated burn injury–induced mechanical allodynia. MWT values in different groups at the indicated times. TrkB-Fc treatment was initiated 1 h before burn injury and continued daily for 1 week post-burn injury: aP<0.05 versus pre-burn; bp < 0.05 versus Sham + Veh3; cp < 0.05 versus Burn + Veh3; dp < 0.05 versus Burn + TrkB-Fc. R(L): large-dose (300 ng) RvD1 treatment. n = 6 rats from a single experiment.
Intrathecal TrkB-Fc prevents spinal microglial activation and downregulates p-p38 MAPK within spinal microglia
Then we assessed the expression levels of Iba-1 and p-p38 MAPK after BDNF/TrkB signaling inhibition with TrkB-Fc. In vehicle-treated animals, Iba1 and p-p38 MAPK amounts in the ipsilateral spinal dorsal horn were significantly higher in the burn injury group in comparison with the sham injury group (Figure 7, p < 0.05). However, pretreatment with TrkB-Fc alleviated Iba1 and p-p38 MAPK upregulation associated with burn injury, with Iba1 and p-p38 MAPK levels considerably lower in the TrkB-Fc-treated group with burn injury compared with counterparts administered the vehicle (Figure 7, p < 0.05), but were still elevated in comparison with the vehicle-treated sham group (Figure 7, p < 0.05). These data suggested that microglial activation and p38 MAPK upregulation within spinal microglia were remarkably suppressed by intrathecally delivered TrkB-Fc. Intrathecal injection of TrkB-Fc prevents spinal microglial activation and p-p38 MAPK downregulation within spinal microglia. The Immunofluorescence images show the expression of Iba1 (microglial marker) and p-p38 MAPK in spinal dorsal horn of rats. The IOD (integrated optical density) values of immunofluorescence intensity are shown in G and H. ap < 0.05 versus Sham + Veh3 group; bp < 0.05 versus Burn + Veh3 group. Scale bar = 20 μm. n = 3 rats per group.
Discussion
The present work demonstrated that burn injury in rats resulted in hyperalgesia and allodynia on the ipsilateral side of burn injury, which was consistent with reports by others. 13 Recent findings demonstrated that RvD1 could potently dampen inflammatory, postoperative, and neuropathic pain. The analgesic properties of RvD1 are partly attributed to its ability to regulate the activities of transient receptor potential (TRP) channels, proinflammatory mediators and anti-inflammatory cytokines, and development of central sensitization.3,4,20,50 This study demonstrated that young rats administered RvD1 intraperitoneally starting right after burn injury showed alleviated/reversed burn injury–induced nociceptive behaviors. Furthermore, the beneficial effects of RvD1 persisted for up to day 14 after burn injury, although the drug was discontinued on day 7 post-burn injury. Importantly, RvD1 had no effect on normal pain perception, which was consistent with previous reports.3,4,24,50 These findings suggest that RvD1 might represent a novel group of analgesics that act to reduce abnormal pain and restore normal sensitivity, rather than altering pain transmission as described for commonly used analgesics.
Immunoreactive signals corresponding to astroglial (GFAP) and microglial (Iba-1) activation were assessed 7 days following burn injury, and both markers showed elevations on the ipsilateral side of burn injury relative to sham burn injury, corroborating previous observations. 13 Administering RvD1 to animals with burn injury decreased astroglial and microglial activation in the dorsal horn on the ipsilateral side of burn injury. In view of the important role of glial activation in inducing and maintaining chronic pain,14,15 it is likely that the effect of RvD1 in attenuating burn injury–associated mechanical allodynia is linked to the inhibition of glial activation.
In the current burn injury model, activation of p-p38-expressing microglia occurred in the spinal cord dorsal horn on the ipsilateral side of burn injury, as also shown by others.51–58 Suppression of p38 MAPK with SB203580 attenuated mechanical allodynia after burn injury. This finding suggested that, following burn injury, p38 MAPK up-regulation within spinal microglia plays an essential role in the pathogenesis of burn pain. Administering RvD1 to animals with burn injury prevented injury-induced upregulation of p-p38 MAPK, suggesting that RvD1 inhibits p38 MAPK activation in the dorsal horn upon burn injury. Using behavioral testing and immunohistochemical analysis, we found that suppression of p38 MAPK within spinal microglia contributed to the analgesic potency of RvD1. In addition, the present study revealed that the p38 MAPK inhibitor SB203580 had no effects on nociceptive reactivity in sham animals, suggesting p38 MAPK signaling might only alter reactivity in case of existing pathology, corroborating previous reports.59–62 A previous study demonstrated that fish oil treatment prevents mechanical allodynia as well as depressive-like behaviors in diabetic rats, likely in association with its ability to restore the levels BDNF, 63 which exerts beneficial effects on neurogenesis. 64
This study revealed significantly increased BDNF and TrkB immunoreactive signals in the ipsilateral spinal dorsal horn following burn injury. Meanwhile, intrathecal administration of TrkB-Fc, a scavenger of endogenous BDNF, attenuated mechanical allodynia after burn injury, which was similar to previous observations that BDNF and TrkB are remarkably upregulated in the ipsilateral spinal dorsal horn after spared nerve injury (SNI), with TrkB-Fc administered intrathecally attenuating mechanical hypersensitivity on the ipsilateral side produced by SNI. 48 However, administering RvD1 to animals with burn injury prevented the injury-associated BDNF and TrkB upregulation following burn injury. These findings suggested that the analgesic potency of RvD1 involves blocking of the BDNF/TrkB receptor signaling pathway.
The present study further showed that p38 MAPK inhibition with SB203580 prevented spinal microglial activation and down regulated BDNF/TrkB following burn injury. However, blocking the BDNF/TrkB receptor signaling pathway by TrkB-Fc administered intrathecally blunted spinal microglial activation and down regulated p-p38 MAPK within spinal microglia. Thus, p38 MAPK activation in spinal microglia and BDNF/TrkB receptor signaling seem to promote each other, playing important and joint roles in the progression of burn pain.
As a study limitation, some critical parameters of the burn model were not assessed, including the release of neuropeptides, TRPV1 activation spontaneous locomotor activity, shaking or licking and Grimace scale score. In addition, the sample was relatively small.
In summary, RvD1 may attenuate burn injury–associated mechanical allodynia by inhibiting spinal cord glial induction and p38 MAPK activation within microglia as well as BDNF/TrkB signaling in the spinal dorsal horn. Its analgesic properties might provide novel therapeutic approaches for managing burn pain.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The present work was funded by the First Affiliated Hospital of Zhengzhou University, Zhengzhou, China and Chinese Natural Science Foundation (NO. 81200909).
Ethics approval
According to the International Association for the Study of Pain guidelines for pain research in animals, experiments involving animals were approved by the Animal Care and Use Committee of the First Affiliated Hospital of Zhengzhou University (No. 2014-002).
Data availability
All data generated or analyzed during this study are included in this published article.
