Abstract
Previous studies have confirmed the relationship between chloride homeostasis and pain. However, the role of sodium potassium chloride co-transporter isoform 1 (NKCC1) in dorsal horn and dorsal root ganglion neurons (DRGs) in spinal cord injury (SCI)-induced neuropathic pain (NP) remains inconclusive. Therefore, we aimed to explore whether suppression of NKCC1 in the spinal cord and DRGs alleviate the NP of adult rats with thoracic spinal cord contusion. Thirty adult female Sprague-Dawley rats (8 week-old, weighing 250–280 g) were randomly divided into three groups with ten animals in each group (sham, SCI, and bumetanide groups). The paw withdrawal mechanical threshold and paw withdrawal thermal latency were recorded before injury (baseline) and on post-injury days 14, 21, 28, and 35. At the end of experiment, western blotting (WB) analysis, quantitative real-time Polymerase Chain Reaction (PCR) and immunofluorescence were performed to quantify NKCC1 expression. Our results revealed that NKCC1 protein expression in the spinal cord and DRGs was significantly up-regulated in rats with SCI. Intraperitoneal treatment of bumetanide (an NKCC1 inhibitor) reversed the expression of NKCC1 in the dorsal horn and DRGs and ameliorated mechanical ectopic pain and thermal hypersensitivities in the SCI rats. Our study demonstrated the occurrence of NKCC1 overexpression in the spinal cord and DRGs in a rodent model of NP and indicated that changes in the peripheral nervous system also play a major role in promoting pain sensitization after SCI.
Keywords
Introduction
Neuropathic pain (NP) is highly prevalent following spinal cord injury (SCI) and problematic in approximately 61%–85% of patients.1,2 NP is a kind of intractable chronic pain induced by injury or dysfunction of the nervous system. 3 It has been characterized as continuous and paroxysmal spontaneous pain caused by mechanical, thermal/cold stimulation, and hyperalgesia (increased pain response to a noxious stimulus)/ectopic pain (pain caused by non-pain stimulation). 4 The characteristics of NP can negatively interfere with the rehabilitation of patients, affect their quality of life, and increase the family and socio-economic burden.
NP is defined formally as “pain caused by a lesion or disease of the somatosensory system”. 5 The various manifestations of NP are notoriously resistant to the actions of nonsteroidal anti-inflammatory drugs and opioids. 6 However, there is no similar panacea for the treatment of NP, which continues to promote research on the underlying mechanism of pain. Acute traumatic SCI causes neurogenic inflammation, excitotoxicity, and neurochemical reactions, which lead to drastic changes in neural activities. 4 The adaptive response of the nervous system to injury will trigger a series of pathological mechanisms related to NP in the spinal cord and primary afferent neurons, including central sensitization, neuronal excitability, and nociceptive de-inhibition. 7
Following nerve injury, attenuation of γ-aminobutyric-acid (GABA) and/or glycinergic transmission leads to aberrant processing of sensory information within the spinal dorsal horn, including changes in chloride gradient and decreased excitatory drive to inhibitory neurons.8–11 Sodium potassium chloride co-transporter isoform 1 (NKCC1) is responsible for establishing chloride concentrations across nerve cell membranes. 12 Because of the importance of the electrochemical Cl− gradient in determining the strength of inhibition mediated by GABAA and glycine receptors, the change in protein levels or the activities of NKCC1 has been predicted to lead to NP. In the state of pathological pain, the expression and activity of NKCC1 in primary sensory neurons increases, resulting in a large amount of extracellular Cl− influx and an accumulation of intracellular Cl−. 13 At the central process terminals, due to the Cl− concentration gradient inside and outside the cells, GABA may be activated to produce primary afferent depolarization (PAD) and play a presynaptic inhibitory role. 13 After exceeding a certain threshold, action potential will be generated, resulting in an increase in the excitability of second-order sensory neurons in the spinal dorsal horn. The low threshold afferent can excite nociceptive neurons, lead to hyperalgesia and promote the production of NP.
Most research into the mechanisms underlying SCI-induced pain has focused on alterations within the spinal cord associated with reflex hypersensitivity. In NP, Cl− channel dysfunction is associated with hyperexcitability in dorsal root ganglion neurons (DRGs). However, whether analogous changes mediate SCI-induced pain sensitization, which does not involve direct injury of first-order sensory neurons, remains to be determined. In the present study, we elucidated a novel mechanism involving alleviation of NP induced by inhibition of NKCC1 in spinal dorsal horn and DRGs following SCI.
Materials and methods
Ethical approval
All procedures and methods were performed in accordance with the Experimental Animal Committee of Capital Medical University (Beijing, China). Surgical interventions and postoperative animal care were conducted in strict compliance with the ethical principles of animal welfare specified in the National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH Publications No.80-23) revised 1996. All efforts were made to minimize the number of animals used and their suffering. Animal protocols were approved by the Animal Care and Use Committee of Capital Medical University (Ethical code: AEEI-2021-190).
Animals
Thirty adult female Sprague-Dawley (SD) rats (8 week-old, weighing 250–280 g) were used for all of the animal studies performed, which were purchased from the Institute of Rehabilitation Medicine China Rehabilitation Science Institute (Beijing, China). They were kept under standard conditions maintained at 22 ± 2°C, 55% ± 10% humidity, 12:12 h light/dark cycle with free access to food and water. All animals were randomly divided into three groups for the current study (sham, SCI, and bumetanide (BU) groups). Pharmacological studies involving BU group were conducted with SCI animals. At the end of experiments and before tissue harvesting, rats were deeply anesthetized by intraperitoneal injection of 10% chloral hydrate (mg kg−1).
Spinal cord contusion
Rats were anesthetized with gaseous isoflurane in oxygen (5% for induction, 2% for maintenance) throughout the duration of the surgery. After shearing off about 4 × 2 cm hair and disinfecting with 75% ethanol, a 2–3 cm incision was made at the T10 spinal segment, skin and fascia were opened and muscles around the spinous process were separated. The spinal cord was exposed by T9-T10 laminectomy and the canalis spinalis opened to expose dura mater spinalis. The T8 and T11 spinous processes were clamped to ensure the location of the injury. An Infinite Horizons Spinal Cord Impactor (IH-400, Precision Systems and Instrumentation, Lexington, KY, USA) was used to induce a contusive SCI force corresponding to the T10 spinous process.
The impact force level selected was 150 kilodyne, which was based on data collected from exploratory experiments that were conducted with this impactor. In these preliminary studies, contusion injuries were induced with increasing impact forces to optimize the spinal cord contusion force needed to produce rats. Rats impacted at 150 kilodyne produced incomplete SCI with ideal NP behavior at 2-3 weeks post-injury. The impact was applied by using a standard rat tip (diameter 2.5 mm).
Afterward, overlying muscles and skin were sutured and the surgical site was disinfected with iodophor to prevent infection. The rats in the sham group received the same procedure except for the spinal cord contusion. During the recovery from anesthesia, each rat received a number and was placed on a warming pad (30°C) until completely awake. Postoperatively, animals were individually housed and hydrated with 2 mL 0.9% normal saline and antibiotics once daily for 3 days. Bladders were emptied manually at least twice per day or as needed until automatic bladder function recovered. Due to the relative ease of bladder expression and lower risk of bladder infections and other complications (e.g., urethral blockages) in female rats compared to males, only female rats were used in the current study. 14
Experimental design
In total, 30 rats were randomly assigned to three groups with ten animals in each group. (1) rats in the sham group were subjected to T10 laminectomy without SCI and given an equal control vehicle at the third week; (2) rats in the SCI group were subjected to T10 spinal cord contusion and given an equal vehicle at the third week; and (3) rats in the BU group were subjected to T10 spinal cord contusion and intraperitoneally treated with BU (30 mg kg−1) at the third-week post-injury.
Locomotor and pain behavioral analyses were used to reveal the analgesic effect of NKCC1 inhibition in SCI rats. In addition, quantitative real-time PCR (qRT-PCR), western blotting (WB), and immunofluorescence were performed to quantify NKCC1 expression and confirm the causal relationship between the inhibition of NKCC1 in spinal dorsal horn/DRGs and alleviation of NP.
The following was the number of rats allocated for various experiments in each group. Immunofluorescence of spinal cords and DRGs, n = 4 in each group; WB analysis of spinal cords and DRGs, n = 5 in each group; qRT-PCR of spinal cords and DRGs, n = 5 in each group.
Drug preparation
For drug treatment, 30 mg/kg of the NKCC1 antagonist BU stock (B129942, Aladdin, Shanghai, China), dissolved in 10 mg/mL normal saline containing 0.25% NaOH was administered intra-peritoneally in rats exhibiting neuropathic hyperalgesia on the third-week post-SCI, performed as described previously. 15 An equal amount of control vehicle, prepared as 0.25% NaOH in saline, was administered intraperitoneally into two groups (sham and SCI) of rats 3 weeks after injury.
Locomotor function
The 21 scores (0, no hind limb movement; 21, normal movement coordinated gait with parallel paw placement) of the Basso, Beattie, and Bresnahan (BBB) locomotor rating scale were used to evaluate the functional neurological deficits of rats before surgery (baseline) and on days 0, 3, 7, 14, 21, 28, and 35 after injury, which was based on the observation of hindlimb movements. 16 A single-blinded reviewer scored the locomotor function of the animals using the BBB locomotor rating scale. During the evaluation, rats were allowed to walk freely on the open field for 4 min. The performance of left and right hindlimbs was rated separately and averaged to generate BBB scores. Within 3 days after injury, the BBB score continued to be 0 or 1, indicating that the SCI model was successfully established.
Paw withdrawal mechanical threshold
Mechanical sensitivity was measured by the von Frey test, following previously reported methods. 17 The rats were put in a cage with an elevated metal mesh floor in a quiet environment for 30 minutes before the experiment. The von Frey calibrated monofilaments (Stoelting 58011, USA) of different bending forces (0.4, 0.6, 1, 2, 4, 6, 8, 10, and 15 g) were applied manually on the mid-plantar area of hind paws for about 5 seconds, which was repeated three times at 30 seconds intervals on each hind paw to eliminate the influence of the previous stimulus. Brisk withdrawal or paw flinching were considered positive responses. The measurement started with the 2 g filament. The PWMT was estimated by the up-down method. If the stimulation caused a negative reaction, we gave a higher stimulation. In contrast, if there was a positive reaction, we gave a smaller stimulation. If a positive reaction was not elicited by a von Frey filament with a force of less than 15 g, the PWMT of the rat was considered as 15 g. The PWMT was first performed 1 day before the injury to get a baseline and repeated post-injury on days 14, 21, 28, and 35 and post BU injection at 1, 2, 4, 6, and 24 h. As defined in the present study, in an animal with ideal NP behavior the intensity of the von Frey test was two or more levels lower than the baseline and rapid foot retraction or licking was shown.
Paw withdrawal thermal latency
Thermal hyperalgesia was assessed using an infrared thermal stimulator (RWD Life Technology Inc, Shenzhen, China). Rats were placed inside the test cage and allowed to acclimatize for 30 min before testing. The experimental environment was quiet to avoid frightening the rats. An infrared thermal stimulus (intensity = 30%) was focused onto the plantar surface of the hind paw. The stimulator could automatically capture the characteristic lifting or licking of the hind paw and record the latency without visual scoring. There were three repeats of the trial in each rat, and 5 min intervals between trials. The mean PWTL was obtained from the three recordings, which represented the reaction time of the rat to infrared thermal stimulation. A safety cut-off time of 30 s was adopted to avoid possible tissue damage. Specifically, the PWTL was recorded before injury (baseline) and on post-injury days 14, 21, 28, and 35. A decrease in the PWTL of greater than 2 s from baseline was considered the development of neuropathic hyperalgesia. The onset of thermal hyperalgesia approximately 14–21 days following SCI, represented the formation of post-SCI NP in animals.
Spinal cord and dorsal root ganglion neurons harvesting
Five weeks after injury, euthanasia was performed on rats after evaluating all pain behaviors. The epicenters of the contusion spinal cords (7–10 mm sections) were harvested from three groups. DRGs were extracted from both sides of the fourth lumbar vertebra (L4) and the fifth lumbar vertebra (L5) and both sides of the L5 and the sixth lumbar vertebra (L6). Rat DRGs was acutely isolated, dissociated, and maintained in short-term primary culture, as previously described. 18 All sections were immediately submerged in liquid nitrogen and stored at −80°C
Quantitative real-time PCR
Total RNA from rat spinal cords and DRGs were extracted with a TRNzol Kit (Kangwei Biotech, China) according to the manufacturer’s manual. Ultramicro spectrophotometer (Nano Drop 2000) was used to detect the concentration and purity of extracted nucleic acid. First-strand cDNA was synthesized with a reverse transcription kit from Kangwei Biotech (CW2569M, Beijing, China) according to instructions and diluted five times as the test sample. The PCR reaction system was 20 μl in volume, which consisted of 400 ng mRNA, 4 μL dNTP mix, 2 μL primer mix, 300 ng RNA template, 4 μL 5× RT buffer, 2 μL DTT, 1 μL HiFiScript, and up to 20 μL RNase-free water. The reaction procedures occurred in a 42°C warm bath for 15 min and an 85°C warm bath for 5 min. Quantitative real-time PCR was performed with Kangwei (CW0957 M, Beijing, China) on a LineGene 9600 (Bori Biotech, Hangzhou, China) with primers GGGTCAGGCTGGAATAGGTCTCTC and ATGCTCCTCCTCCTCTCACGAATC for NKCC1; and ACAGCAACAGGGTAATGGAC and TTTGAGGGTGCAGCGAACTT for GAPDH. The reaction program was 95°C for 10 min followed by 40 cycles of 95°C for 15 s and 60°C for 30 s. The 2−ΔΔCt method was used to calculate the relative expression levels of NKCC1 mRNA with GAPDH as the internal control.
Western blotting analysis
The total protein of the spinal cords was extracted using RIPA lysis buffer (Beyotime Institute of Biotechnology, Beijing, China), which was added in advance with protease inhibitors. The homogenate was cracked for 15 min at 4°C and centrifuged at 12,000 r/min for 10 min. The supernatant was retained and the protein content of the supernatant was determined by the BCA protein assay (Kangwei Biotech, China) according to the manufacturer’s manual. The concentration of all samples was adjusted with RIPA lysis buffer for consistency, 5× loading buffer was added to the corresponding volume, then boiled and denatured for 5 min, centrifuged at 12,000 r/min for 5 min at 4°C and stored at 4°C for later use. Total proteins of each group (15 μg/lane) were electrophoretically concentrated on 6% SDS-polyacrylamide gels at 80 V for 25 min and separated on 8% SDS-polyacrylamide gels at 120 V for 70 min. Then the resolved proteins were transferred to 0.22 polyvinylidene fluoride membranes (Millipore, Bedford, MA) at 400 mA for 70 min. The corresponding region of the target protein was cut for antibody incubation. After the membranes were blocked in 5% non-fat milk at room temperature for 90 min and washed three times with TBST for 10 min, they were incubated with anti-NKCC1 (14,581, 1:1000, CST, USA) or anti-GAPDH (5174, 1:1000, CST, USA) antibody at 4°C overnight and reacclimatized to room temperature for 60 min. After three washes with TBST, the membranes were incubated with horseradish peroxidase-conjugated goat anti-rabbit IgG antibody (1:10,000; Kangwei Biotech, China) at room temperature for 60 min. After three washes with TBST, Tianneng 5200 muti was used for chemiluminescence imaging, and the image analysis software GIS 1D was used to analyze the exposure results. The images were scanned and analyzed with Image J (NIH, Bethesda, MD). Background density was subtracted from the NKCC1 receptor band density and normalized to GAPDH, which was used as the loading control.
Immunofluorescence staining
Rats were anesthetized with a 10% chloral hydrate intraperitoneal injection. The aorta was perfused with 500 mL 0.9% normal saline to wash the blood away quickly after opening the chest, followed by 250 mL 4% paraformaldehyde-phosphate buffer (pH 7.4), and continuously perfused for about 1 h. The epicenter of the contusion spinal cords and L4–L6 DRGs were removed and immersed in the same fixative overnight at 4°C.
Paraffin-embedded spinal cord tissue at the injury site was cut at a thickness of 6 microns. Slides were deparaffinized and rehydrated by washing in xylene and passing through an ethanol gradient. Endogenous peroxidase activity was quenched with freshly made 0.3% hydrogen peroxide and antigens were heat retrieved in citrate buffer. Slides were blocked with normal goat serum for 30 min at 37°C, incubated with antibody against NKCC1 (#85403, CST, USA) overnight at 4°C, rinsed with 0.01 M phosphate buffered saline (PBS) 3 min for three times each, incubated with FITC-(for NKCC1) conjugated goat anti-rabbit IgG antibodies for 30 min at 37°C, and co-stained with DAPI. The slides were dehydrated with alcohol gradient, cleared with xylene, and mounted with neutral gum. The images were obtained with an Olympus IX71 Microscope.
The L4–L6 DRGs were sliced into 30 μm-thick sections with a cryotome (CN1800, Leica, Wetzlar, Germany) at −20°C. The sections were collected in sterile 0.01 mol/L phosphate buffer (pH 7.4) at 4°C and processed for NKCC1 immunofluorescence staining. The free-floating sections were rinsed in 0.01 mol/L PBS (pH 7.4) three times (10 min each) and then sliced into 0.3% TritonX-100 and 2% sheep serum 0.01 PBS mol/L at room temperature for 1 hour and then incubated overnight at 4°C with monoclonal mouse anti-NKCC1 antibody (#85403, diluted 1:200 CST, USA). The sections were then washed with PBS (3 × 10 min) and incubated with FITC-(for NKCC1) conjugated goat anti-rabbit IgG antibodies (diluted 1:200) for 4 h. Avoiding light, the sections were washed with PBS (3 × 10 min). After staining, all of the sections were mounted onto glass slides and cover-slipped with 50% (v/v) glycerol and mounted onto glass slides and cover-slipped with 2.5% (w/v) triethylenediamine (anti-fading agent) in 0.05 M PBS. After being dried with a fluorescent mounting medium, the sections were mounted onto gelatin-coated glass slides.
Statistical analysis
All quantitative data were presented as the mean values with the standard error of the mean (SEM). SPSS 25.0 (IBM, Chicago, IL) was used for the difference in analysis between data. GraphPad Prism version 8.0.0 (GraphPad Software, San Diego, California USA, www.graphpad.com) was used for rendering images from the collected data. BBB scores and PWMT of the groups at the different time points were analyzed using two-way repeated-measure analysis of variance (ANOVA) followed by post-hoc Bonferroni correction, while one-way ANOVA with post-hoc Fisher’s LSD test was used to compare differences between treatment groups at the same time points. Changes in the PWTL were analyzed by two-way repeated measure ANOVA with a within-subject factor of time (Five levels: before surgery and on days 14, 21, 28, and 35 after surgery) and a between-subjects factor of group (Three levels: the treatment groups) with post-hoc Bonferroni correction. One-way ANOVA followed by post-hoc LSD analysis was performed to compare the expression of NKCC1 in three groups. The comparison was considered statistically significant when the two-sided p value was <0.05.
Results
Inhibition of sodium potassium chloride co-transporter isoform 1 improves basso, beattie, and bresnahan scores in spinal cord injury rats
Figure 1 showed that all SD rats were allowed to acclimate for 1 week before experiments and euthanized at the 6th week. To investigate the effects of NKCC1 inhibition on SCI rats, we administered intraperitoneal BU injections to rats that had been subjected to SCI. After acclimatization for one week, rats were randomly assigned to three groups. All rats were housed and sacrificed at the end of week 6. We assessed locomotor behavior using BBB scores to verify a similar degree of injury in all animals (Figure 2). The sham group showed stable and comparable BBB scores over 4 weeks of observation. Locomotor improvement was observed during 5 weeks post-injury in SCI groups. NKCC1 inhibition significantly increased hindlimb locomotor function starting from 28 days after injury compared to that of the SCI group (p = 0.049). In addition, we assessed joint mobility, plantar movement, and stepping based on the BBB score at week 5 post-injury. The percentage of plantar movement in BU rats was higher than that in SCI rats and the joint mobility percent was significantly decreased in the BU group (Figure 3). As shown in Figure 4, the plantar movement coordination was significantly elevated with BU treatment in SCI animals. Our results suggested that BU rats exhibited plantar movement improvement and coordination compared to SCI rats, which indicated inhibition of NKCC1 improved locomotor behavior to a certain extent. Schematic representation of the experiments. Locomotor behavior in indicated groups. BBB scores of the groups at the different time points were analyzed using two-way repeated-measure ANOVA followed by post-hoc Bonferroni correction, while one-way ANOVA with post-hoc Fisher’s LSD test was used to compare differences between treatment groups at the same time points. ****p < 0.0001 compared to Sham. Error bars, SEM. The percentage of rats in joint mobility (BBB 0–7 scores), plantar movement (BBB 8–13 scores), and stepping (BBB 14–21 scores) at week 5 post-injury. Coordination of plantar movement.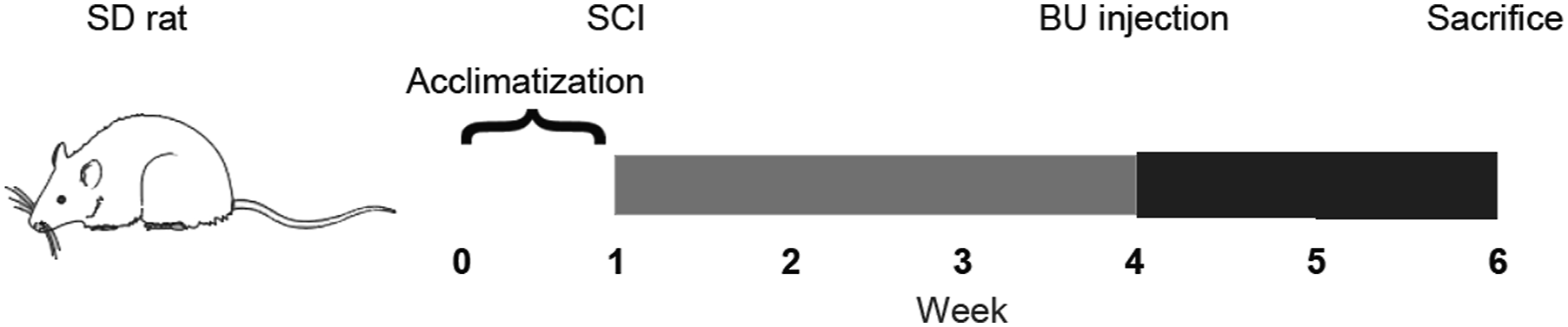


Inhibition of sodium potassium chloride co-transporter isoform 1 alleviated mechanical ectopic pain and thermal hyperalgesia in spinal cord injury rats
To evaluate the degree of SCI-triggered pain and the analgesic effect of NKCC1 inhibition on rats with mechanical ectopic pain and thermal hyperalgesia, we recorded the PWMT and PWTL in the indicated groups.
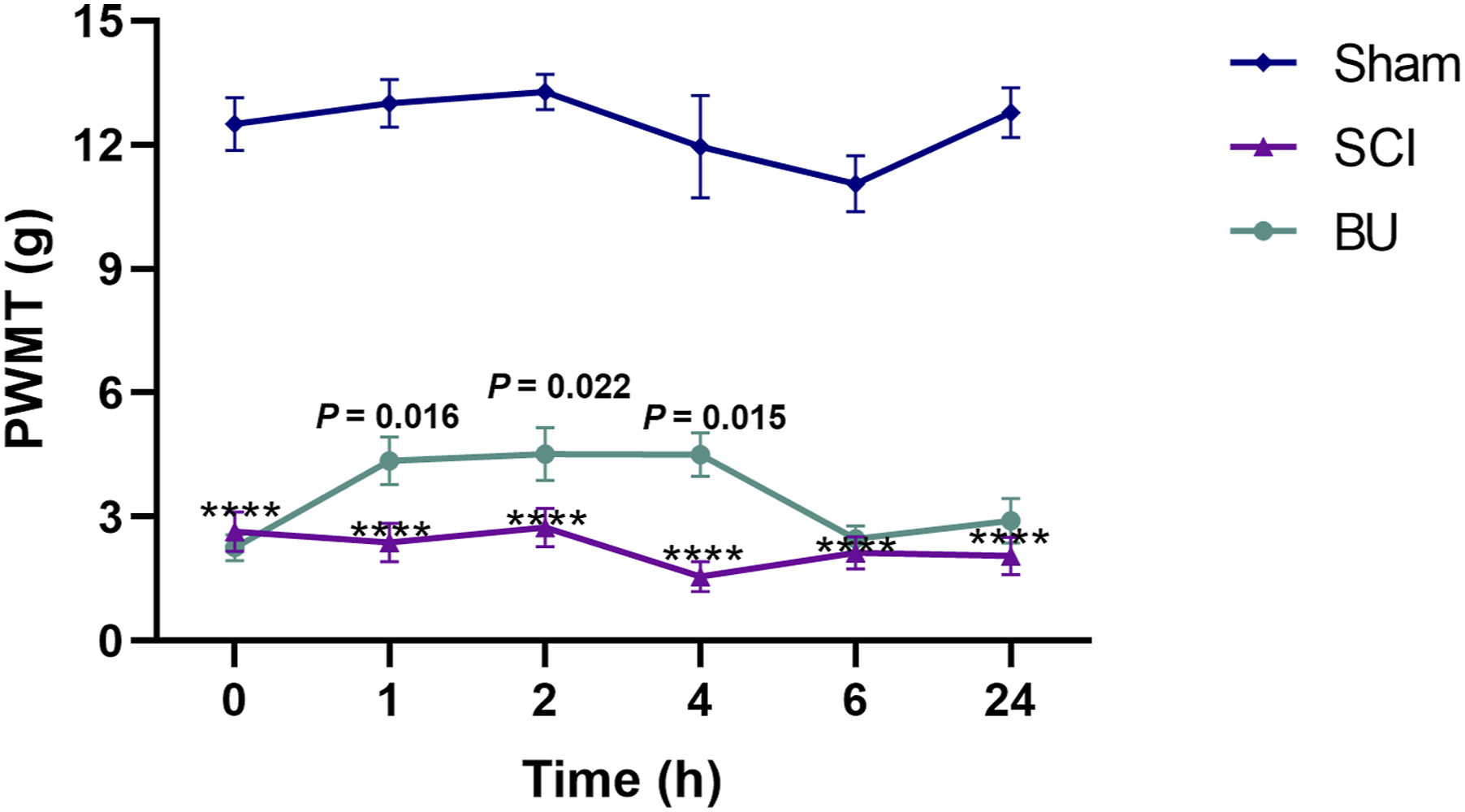
As shown in Figure 5, within 21 days, the PWMT in the SCI group was statistically lower than at baseline (p < 0.0001), indicating that the spinal cord contusion model gave rise to obvious NP in rats. There was no significant difference in the PWMT in rats after laminectomy without SCI between baseline and post-surgery. Next, we evaluated the PWMT changes over a 24 h period after administration when the neuralgia was most obvious in the third-week post-injury (Figure 6). Before treatment, the baseline PWMT between the SCI and BU groups showed no significant difference. Compared with the SCI group, the PWMT of BU rats gradually increased and showed a statistically significant difference at 1, 2, and 4 h after the BU injection (p = 0.016, p = 0.022, p = 0.015, respectively). Consistent with these results, the improvement in mechanical threshold was observed in the BU group compared to the SCI group and continued until the end of the experiment. The above results suggested that rats with spinal cord contusion developed obvious NP at about 2-3 weeks, and inhibition of NKCC1 could alleviate mechanical ectopic pain in rats with SCI. PWMT in rat with operation. Two-way repeated measure ANOVA followed by Bonferroni post-hoc test. n = 20 in SCI group, n = 10 in sham group. n.s.: Not significant. PWMT after intraperitoneal BU injection of rats in indicated groups. One-way ANOVA followed by Fisher’s LSD. ****p < 0.0001 compared to sham. Error bars, SEM.
The PWTL was measured and compared at indicated time points. There was no significant difference in baseline PWTL between the three groups (Figure 7). For both the SCI and BU groups, the PWTL gradually decreased during the first 3 weeks after surgery (p < 0.001), indicating that SCI resulted in significant thermal hyperalgesia in rats. The SCI rats demonstrated a rapid continued increase of the PWTL after BU administration in the following days. Accordingly, after BU administration a further significant increase in the PWTL was observed at 35 days after injury compared to that of the SCI group (p = 0.014). These observations indicated that the inhibition of NKCC1 was beneficial and could ameliorate the thermal hyperalgesia induced by SCI. Postoperative PWTL of rats in indicated groups. Two-way repeated-measure ANOVA followed by post-hoc Bonferroni. Error bars, SEM.
Inhibition of sodium potassium chloride co-transporter isoform 1 in the dorsal root ganglion neurons of spinal cord injury rats
Thirty-five days after SCI, we analyzed the expression of the NKCC1 gene in DRGs in the different groups by qRT-PCR. GAPDH mRNA was probed with the same conditions to serve as a control. PCR analysis showed that the relative expression levels of NKCC1 mRNA in DRGs in the sham, SCI, and BU groups were 1.08 ± 0.43, 1.45 ± 0.35, and 0.47 ± 0.4, respectively. The results indicate that a significantly higher level of NKCC1 mRNA was observed post-injury, which means that spinal cord contusion could greatly promote the expression of NKCC1 mRNA when compared with laminectomy alone. Moreover, administration of BU could significantly reduce the concentration of NKCC1 mRNA compared with the SCI group (p = 0.0227). In other words, inhibition of NKCC1 exerted more beneficial effects compared with the control (Figure 8(a)). Analysis by WB confirmed these results (Figures 8(b) and (c)). Furthermore, we evaluated the number of NKCC1 cells/field following SCI in different groups using the NKCC1 for single immunofluorescent staining together with DAPI, which detected the NKCC1 distribution in the DRGs (Figures 8(d) and (e)). As shown in Figure 8(f), a marked increase in the NKCC1 cells/field following SCI and lower level of NKCC1 cells/field was observed in DRGs at day 14 after BU administration compared with the SCI group, but there were no significant differences among the three groups. Consequently, these results demonstrated that BU had a significant effect on inhibiting NKCC1 expression in DRGs after SCI. Inhibition of NKCC1 in DRGs. (a): The mRNA expression level of NKCC1 gene was detected by qRT-PCR. (b): The protein level of NKCC1 was detected by WB analysis. (c): NKCC1 protein concentration in the three groups using the BCA assay. (d): DAPI staining of DRGs section with outlined spinal nerve dorsal root area and a squared inset of observed ganglion. (e-f): Quantification of NKCC1 immunoreactivity (green) with representative immunofluorescence images of DRGs sections. DAPI, blue; scale bar, 50 μm.
Inhibition of sodium potassium chloride co-transporter isoform 1 in spinal cords in spinal cord injury rats
To determine whether BU exerts similar therapeutic effects in spinal cords to those observed in DRGs, spinal cord tissues were harvested 35 days following the injury. We investigated the gene expression of NKCC1 with GAPDH mRNA as a control. As shown in Figure 9(a), the relative expression levels of NKCC1 mRNA in the spinal cords of the sham, SCI, and BU groups were 0.94 ± 0.25, 0.74 ± 0.35, and 0.78 ± 0.12, respectively. However, we found no significant change in the expression of the NKCC1 gene in spinal cords. WB analysis also confirmed the qRT-PCR results (Figures 9(b) and (c)). We found that spinal cord contusion could increase the concentration of NKCC1 protein in spinal cords, which was similar to the results in DRGs. BU administration did not appear to reduce the expression level of the NKCC1 protein in the epicenter of the contusion spinal cords. Using immunofluorescence (Figures 9(d) to (f)), we showed that the mean intensity of NKCC1 in the spinal dorsal horn in the SCI group was higher than in the sham group (p = 0.0087), and administration of BU significantly affected the expression level of NKCC1 (p = 0.0124). These combined results suggest that BU plays a role in regulating NKCC1 expression in spinal cords, which confirmed the results observed in DRGs. Inhibition of NKCC1 in spinal cords. (a): The mRNA expression level of NKCC1 gene was detected by qRT-PCR. (b): The protein level of NKCC1 was detected by WB analysis. (c): NKCC1 protein concentration in the three groups using the BCA assay. (d): DAPI staining of spinal cord section with outlined gray matter area and a squared inset of observed ventral horn. (e): Immunostaining of NKCC1 (green) in the T10 region of spinal cords in different groups. DAPI, blue; sale bar, 50 μm. (f): Quantification of NKCC1 immunoreactivity. n.s.: Not significant between the three groups.
Discussion
SCI results in motor and sensory deficits and disabling chronic NP. 19 The research related to SCI in recent years mainly focuses on nerve repair, but rarely involves improving the NP.20–23 Effective analgesic therapy has been hampered by a lack of knowledge about the mechanisms underlying NP following SCI. Accumulating evidence suggests that the dysregulation of chlorine homeostasis is involved in the pathogenesis and clinical manifestations of NP after SCI.24–26 Chloride homeostasis through the function of ionotropic GABAA-receptors (GABAAR) is an important mechanism in the regulation of nociceptive processing. 27 In the present study, we analyzed the expression changes of NKCC1 in spinal cords and DRGs following thoracic spinal cord contusion in rats. In addition, we explored the effects of NKCC1 inhibition on the alleviation of NP. Importantly, we have provided evidence to confirm that NKCC1 is involved in NP after spinal cord contusion in rats. This conclusion was based on the following evidence. NKCC1 was significantly up-regulated in the dorsal horn and DRGs of SCI rats. Intraperitoneal injection of BU, an inhibitor of NKCC1, significantly attenuated mechanical ectopic pain and thermal hyperalgesia, indicating that NKCC1 was involved in allodynia and hyperalgesia in SCI rats. These findings revealed that both central and peripheral alterations appear to be important for inducing and maintaining SCI pain.
Previous studies have shown that the suppression of NKCC1 expression may account for the attenuation of pain. As a Cl− transporter, NKCC1 plays an important role in NP and bone cancer pain.28–32 Cramer et al. suggested that changes in NKCC1 protein expression were involved in the development of chronic NP following SCI. In their study, NKCC1 protein level was elevated on day 7 post- SCI and increased by 60% on day 14 post-SCI. 15 In the present study, we demonstrated that the presence of mechanical ectopic pain and thermal hyperalgesia can be detected in rats on days 14–21 post-SCI. Moreover, inhibition of NKCC1 with its potent antagonist BU significantly reduced pain behavior in these rats, which was consistent with published articles. Importantly, we showed that rats in pain displayed a sustained and significant increase of the PWMT at 1, 2, and 4 h after the BU injection, which was consistent with the duration of its pharmacological effects. Moreover, we observed that the pain threshold of rats in the BU group was still higher than that in the SCI group after 4 h, considering the results of early postoperative spinal suppression or reduction of the formation of dorsal root reflexes. 33 Therefore, it did not rule out that another endogenous analgesia system is at work. It should be noted that the PWTL was significantly increased in SCI rats after the injection of BU. Future studies are necessary to assess the long-term influence of NKCC1 inhibition on allodynia and hyperalgesia.
To further investigate the expression level of NKCC1 in DRGs, morphological and immunological assays were carried out. On day 14 after drug treatment, a two-thirds reduction of NKCC1 mRNA in the L5–6 DRGs occurred. The hyperalgesic rats displayed an increase of NKCC1 protein in DRGs at day 35 post-SCI. Immunofluorescence results showed that a greater than threefold increase in NKCC1 cell number was detected in DRG neurons of rats exhibiting NP following SCI. The above data suggest that spinal cord contusion was associated with an increase in NKCC1 mRNA and protein levels and cell number in DRGs. To determine the relationship between NP and NKCC1, the intraperitoneal injection of NKCC1 inhibitors was used. The injection of BU significantly reversed NKCC1 expression, indicating that inhibition of NKCC1 activity in vivo increases mechanical and thermal nociceptive thresholds in SCI animals. Numerous reports illustrate that the alteration of Cl− homeostasis and GABAergic function are involved in nociceptive input hypersensitivity following nerve injury. 34 The NKCC1 protein accumulates chloride intracellularly in DRGs, which is the primary mechanism that sets the reversal potential for chloride conductance through GABAAR. 35 Unlike most CNS neurons, DRGs maintain depolarizing responses to GABAAR agonists throughout postnatal development, which is dependent on NKCC1 expression. 36 Furthermore, spinal interneurons release GABA, which activates GABAAR on primary afferent terminals and produces PAD. 37 PAD is known to underlie presynaptic inhibition in the spinal cord, which could shunt the magnitude of incoming action potentials and decrease excitatory amino release at the primary afferent central terminals.37–39 In several pathological conditions, such as after spinal cord contusion, PAD may be enhanced via a GABAergic mechanism causing dorsal root reflexes and orthodromic firing of nociceptors.40–42 Intrathecal delivery of the NKCC1 antagonist BU decreased the concentration of chloride in the DRGs and inhibited the hyperalgesia caused by capsaicin. 43 An in vitro study found that inflammatory mediators increase the NKCC1 channel expression of DRGs within three hours. 44 Taken together, these findings indicate that the NKCC1 of DRGs might be an important player in pain. However, the detailed upstream mechanisms regulating NKCC1 expression in DRGs remain largely unknown. A recent report shows that activation of TRPV1 by exogenous agonists can increase the expression and function of the NKCC1 protein in DRGs, which is mediated by the activation of the PKC/pERK signaling pathway. 43 This needs to be confirmed in future studies. Although more experiments are needed, our data have provided evidence to demonstrate that changes in the peripheral nervous system also play a major role in promoting pain sensitization after SCI.
Likewise, we further investigated the expression of NKCC1 in spinal cords. On days 35 post-SCI, the mean fluorescence intensity of NKCC1 significantly increased in the contusion epicenter of spinal cords. In addition, the NKCC1 protein was up-regulated on days 35 post-SCI. It was reported previously that adult male SD rats showed increased NKCC1 channel expression 2–14 days post-SCI at the injury epicenter. 15 These results suggested that alteration of Cl− homeostasis by changes in NKCC1 function may contribute to hyperalgesia development. BU rats displayed a decrease in NKCC1 intensity, with the expression of NKCC1 observed to be 67% of SCI rats in lesion epicenter tissues at days 35 post-SCI. These data indicated that NKCC1 in the dorsal horn of the lesion epicenter may be involved in NP following SCI. Moreover, inhibition of NKCC1 activity in spinal cords attenuated allodynia and hyperalgesia in nerve-injured animals. Earlier studies have also confirmed that GABAergic interneurons in laminae I–IV in the dorsal horn are essential for primary afferent nociceptive attenuation.38,39,45,46 The generation of GABAAR responses is critically dependent on the activity of NKCC1. Nerve injury disrupts anion homeostasis in lamina I neurons and shifts the normally inhibitory synaptic currents to excitatory, thereby increasing lamina I neuronal excitability. 47 Therefore, we speculated that spinal cord contusion might induce modification of this inhibitory action, which had the potential to alter the processing of nociceptive information in the dorsal horn and result in NP. Consistent with GABAergic mechanisms, reduction of intracellular chloride by BU, a diuretic NKCC1 chloride co-transporter antagonist, mimicked its effects on GABA responses in nociceptive neurons. 48 More importantly, nociception is strongly regulated not only by the amount of the GABAAR mediated anionic conductance, but also by its reversal potential (EGABA), as the depolarization shift of EGABA in the nociceptive and dorsal horn neurons is related to the elevation of pain. 49 Pain is alleviated by pharmacological blockade or the gene knockout of NKCC1, which is the dominant cause of chloride elevation and GABA depolarization in nociceptive neurons.50–52 Inhibition of NKCC1 causes a negative shift in EGABA and reduces pathological pain.51,53,54 We found that BU exerted an analgesic action in thoracic spinal cord contusion. Therefore, blockade of the NKCC1 chloride co-transporter in rats with spinal cord horn, which is the main mediator of chloride accumulation in primary nociceptive neurons, attenuates SCI-induced allodynia and hyperalgesia.
This study is a preliminary study to examine the effect of bumetanide treatment in the contusion SCI rat. One limitation of this study is that it is unknown whether the bumetanide affects the sham-treated rats. To provide more scientific evaluation, further research on sham-treated rats is crucial.
In summary, the present study illustrates that inhibiting the expression of NKCC1 in dorsal horn and DRGs alleviates NP and promotes locomotor behavior in SCI rats. The anti-hyperalgesic effect of NKCC1 inhibition suggests that normal or elevated NKCC1 function plays a critical role in the development and maintenance of SCI-induced hyperalgesia and allodynia. These findings may provide new evidence that NKCC1 in DRGs mediate SCI-induced pain sensitization.
Footnotes
Acknowledgements
Thanks to the members of Department of Spine Surgery, Beijing Bo’ai Hospital, China Rehabilitation Research Center for the generous help they offered.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key R&D Program of China (Project No. 2021YFF0501600 and Subject No. 2021YFF0501604), the Capital’s Funds for Health Improvement and Research (No. CFH 2022-2-6013), and the Projects of China Rehabilitation Research Center (No. 2021ZX-09).
Author contributions
Wang FY conceived the study, analyzed the data, and revised the manuscript; Wu Y acquired the data, interpreted the results, and drafted the article. All authors have read and approved the final version of the manuscript.
Data availability
The datasets used during the current study are available from the corresponding author on reasonable request.
