Abstract
Neuropathic pain (NP) is the cardinal symptom of neural injury, and its underlying molecular mechanism needs further investigation. Complements, especially complement 3 (C3), are involved in the pathophysiology of many neurological disorders, while the specific role of C3 in NP is still obscure. In this study, we found that both C3 and its receptor C3aR were upregulated in the spinal dorsal horn in a rat chronic constriction injury (CCI) model. In addition, C3 was mainly detected in astrocytes, while C3aR was expressed in microglia and neuron. Intrathecal injection of C3 antibody and C3aR antagonist alleviated NP in CCI model together with reduced M1 polarization of microglia. Our finding suggested that blockade of the C3/C3aR pathway might be a novel strategy for NP.
Introduction
Neuropathic pain (NP) is usually caused by injury or disease which damages the somatosensory nervous system. At present, opioid drugs are still the main management for NP. However, their effectiveness is not often unsatisfactory and they may induce unbearable side effects.1–3 Hence, many studies aimed to discover compensatory mechanisms of NP for more effective and applicable therapies. Glial cells are a group of special immune cells responsible for keeping the homeostasis of central nervous system (CNS). These glial cells could secret neuromodulatory substances activated in neuropathic conditions, such as cytokines and neuropeptides.4,5 Recent studies have revealed that input signals of pain from the primary sensory neuron are processed in the spinal dorsal horn (SDH) and transmitted to the brain, which is modulated by the glial cells.
Astrocyte, which accounts for approximately 20–40% of the glial cells in CNS, is critical for immunological homeostasis and neural metabolism.6,7 It provides nutrients to neurons, regulates fluid and iron homeostasis, and controls local blood flow.7–10 Under pathological conditions, such as infection, injury, or neurodegeneration, astrocyte transforms to reactive status in response to various molecular signals. Then, astrocyte secret mediators, such as CXCL1, CCL2, IL-1, growth factors, and complements, to modulate immune microenvironment in CNS. The activation of astrocytes after peripheral nerve injury maintains from the acute phase to the chronic phase of NP. 11 In addition, pharmacological inhibition of astrocyte proliferation in SDH attenuated NP. 12
Microglia, the second subpopulation of glia in CNS, acts as the first defense line and rapidly responds to external stimulus. Activated microglia could be classified into two phenotypes: M1 pro-inflammatory microglia and M2 anti-inflammatory microglia.13–17 The activation of microglia is earlier than the activation of astrocytes and the late-activated astrocytes could modify the activation and polarization of early-activated microglia due to the broad interaction between astrocyte and microglia.11,18 However, the details about the astrocyte-microglia crosstalk in NP are not well demonstrated.
The complement cascade was found in circulation as a key regulator of the innate immune system,19,20 while recent studies have highlighted its new role in the CNS. Among these complement elements, C3 functions as the center of the complement cascade. In the CNS, C3 has been thought to be predominantly produced by astroglia. and then it will combine with its receptors on astroglia, microcyte, and other cells. The receptors of C3 expressed on neurons and glias promote cellular interactions throughout CNS. This crosstalk regulates the pathological process of many neurological diseases, such as ischemia, epileptics, and Alzheimer’s.20–25 These studies indicated that aberrant microglia activation was attenuated together with alleviated neurological symptoms by blocking the C3-C3aR pathway. Meanwhile, previous studies have demonstrated that C3 was upregulated in both peripheral nervous systems and CNS under NP.26,27 However, the detailed effects of C3-C3aR activation in NP have not been explored. Therefore, this study aimed to discover the regulatory mechanism of the C3-C3aR pathway and its role in microglia polarization in NP.
Materials and methods
Animals
The adult male Sprague-Dawley rats (120–180 g) were purchased from HFK Bioscience Co., Ltd, Beijing, China. All rats were housed (4–5 rats per cage) in a controlled environment with a 12-h light/dark cycle. All experimental procedures were approved by the Animals Ethics Committee of our institution. Animals were randomly allocated into chronic constriction injury (CCI) or sham groups.
Animal model of NP
The CCI model in rats was carried out according to the study of Bennett and Xie. 28 All rats received intraperitoneal injection of sodium pentobarbital (40 mg/kg) for anesthesia. The sciatic nerve was exposed in the mid-thigh level on the right side and four snug ligatures of the chromic gut were sutured loosely around the nerve with 1 mm space between the knots. In the sham group, the sciatic nerve was exposed without ligation.
Behavioral assessments
Paw withdrawal mechanical threshold (PWT) was used to evaluate mechanical pain determined by electric von Frey. And paw withdrawal thermal latency (PWL) was used to evaluate the thermal pain measured by the radiant heat. PWT and PWL values were obtained at 10 a.m. for each behavioral assay. The rats underwent 30 min to acclimate to the environment before each test. Three repeat measures were acquired in each rat with a 5-min interval, and the average was applied.
Intrathecal injection and drug delivery
C3 antibody (Abcam, ab200999) was mixed with Phosphate-Buffered Saline (PBS) to the concentration of 0.25ug/ul and 0.10 ug/ul. Trifluoroacetate (Selleck, SB290157), a specific antagonist of C3aR, was mixed with 10% DMSO to a concentration of 1.0ug/ul and 2.0ug/ul. Drugs (10ul antibody or Trifluoroacetate) were given into the subarachnoid cavity at L5-L6 intervertebral disc by insulin needles from POD7 to POD12. The spinal tissue was harvested at POD12 for immunostaining and RT-qPCR. The L5-L6 intervertebral disc was determined by the iliac crest. Drugs were injected when typical rat tail shaking appeared.
Fluorescent immunostaining
Sodium pentobarbital (40 mg/kg) was intraperitoneally injected into rats for anesthesia. Rats were perfused transcardially with cold PBS followed by 4% paraformaldehyde (Sigma, USA). After perfusion, L4–L6 SDH were collected and post-fixed in 4% paraformaldehyde overnight at 4°C. Then the tissue was gradient dehydrated with 20% and 30% sucrose until tissue sink down to the bottom of the EP pipe at 4°C. The tissue was then embedded into OCT (Tissue-Tek, Japan) and sectioned into 15-μm-thickness slices. The tissue sections were permeabilized with 0.3% Triton X-100 at room temperature and blocked with 5% donkey serum for 1 hour. The tissue sections were then bound with the target primary antibodies at 4°C overnight in a wet box. The next day, corresponding secondary antibodies were incubated for 1 hour at room temperature. The slides were then washed in PBS and covered by DAPI Mounting Medium (ZSJB-Bio, Beijing, China). Images were captured by laser confocal microscopic imaging system (FV1000 and Olympus FluoView software, Olympus, Japan). The method of quantification for immunofluorescence staining referred to previous studies.29,30 Briefly, two non-adjacent spinal cord sections were randomly selected from the spinal cord segment (L3–L5) of each rat, and three rats were selected for each group. The intensity of GFAP and IBA1 staining was measured with Image J. The used antibodies were listed in Table S1.
Quantitative real-time PCR
The total RNA of rat spinal dorsal horn was extracted by TRIzol reagent (Invitrogen, Grand Island, NY, USA) and reverse transcribed using PrimeScript™ RT Master Mix (Takara, Japan) according to the manufacturer’s instructions. Real-time PCR was performed using TB Green qPCR Master Mix (Takara, Japan) and analyzed with Bio-Rad system. Real-Time PCR primer sequences are shown in Table S2.
Statistical analysis
Data were expressed as means and standard errors (Mean±SEMs). Shapiro-Wilk test was used to determine the normality of the parametric test. The student’s t-test was applied to examine differences between the two groups. For the behavioral test, two-way analysis of variance (ANOVA) followed by the Bonferroni post hoc test was used to test differences among multiple groups. In the two-way ANOVA tests, one factor was set as time point, and the other was the treatment of the rats. All statistical analyses were performed in GraphPad Prism for Windows version 7.0 (GraphPad Software, Inc., San Diego, CA, USA). A significant difference was defined as a two-sided p-value < 0.05.
Results
C3 and C3aR were elevated in spinal dorsal horn after CCI
Both PWT and PWL values on CCI rats were significantly lower than those in Sham group (p < .0001, Figure 1A and B). PCR analysis showed that mRNA of complement 3 (C3) in the dorsal horn was upregulated after CCI, which was maintained to POD21 (Figure 1C). The immunofluorescence (IF) showed that protein level of C3 was increased when compared to the sham group (Figures 1D and E). The mRNA level of C3aR in the CCI group was also elevated from POD14 to POD21 (Figure 1F). On POD14, IF showed that dorsal C3aR was upregulated in the CCI group as well (Figures 1G and H). To identify the cellular distribution of C3 and C3aR, we performed double staining among three major subpopulations at POD14, including NeuN (for neurons), GFAP (for reactive astrocytes), and IBA1 (for reactive microglia). As is shown in Figure 2A–C, C3 signal mainly co-localized with GFAP, while no obvious overlap was detected on NeuN and IBA1 (Figure 2D). In addition, C3aR signal is mainly co-localized with IBA1 and NeuN (Figure 3). C3 and C3aR are overexpressed after CCI injury. Double staining of C3 with different cellular markers. Double staining of C3aR with different cellular markers. 


Neutralizing C3 alleviated neuropathic pain and reduced M1 polarization of microglia
To investigate biological function of C3, we neutralized C3 by intrathecal injection of C3 antibody from POD7 to POD12. The IF experiment showed that intrathecal injection of C3 significantly decreased the content of C3 protein in the SDH of CCI rats (Figures 4A and B). The behavioral tests showed that intrathecal injection of 0.1ug/ul and 0.25ug/ul C3 antibody could alleviate both mechanical and thermal pain compared with isotype IgG (Figures 4C–D). Further, we observed that higher concentration of C3 antibody presented more potent effects, suggesting that neutralization of C3 could alleviate NP. Nonetheless, neutralization of C3 did not change the pain relative behavior of sham rats. Furtherly, compared to isotype IgG injection, injection of C3 antibody decreased the expression of GFAP and IBA1, which indicated decreased activation of astrocyte and microglia (Figure 4E–H). Considering the location of C3aR in microglia and the effect of C3-C3aR on the polarization of microglia, we performed PCR to examine the microglial status with M1 marker (CD86, inos) and M2 marker (CD206, Arg1). Intrathecal injection of C3 antibody significantly reduced the mRNA levels of CD86 and inos, whereas it increased the mRNA levels of CD206 and Arg1 compared with IgG group (Figure 4I). These data suggested that C3 mediated M1 polarization of microglia, which may contribute to the maintenance of NP. Intrathecal administration of C3 antibody alleviated neuropathic pain and decreased M1 microglia polarization. 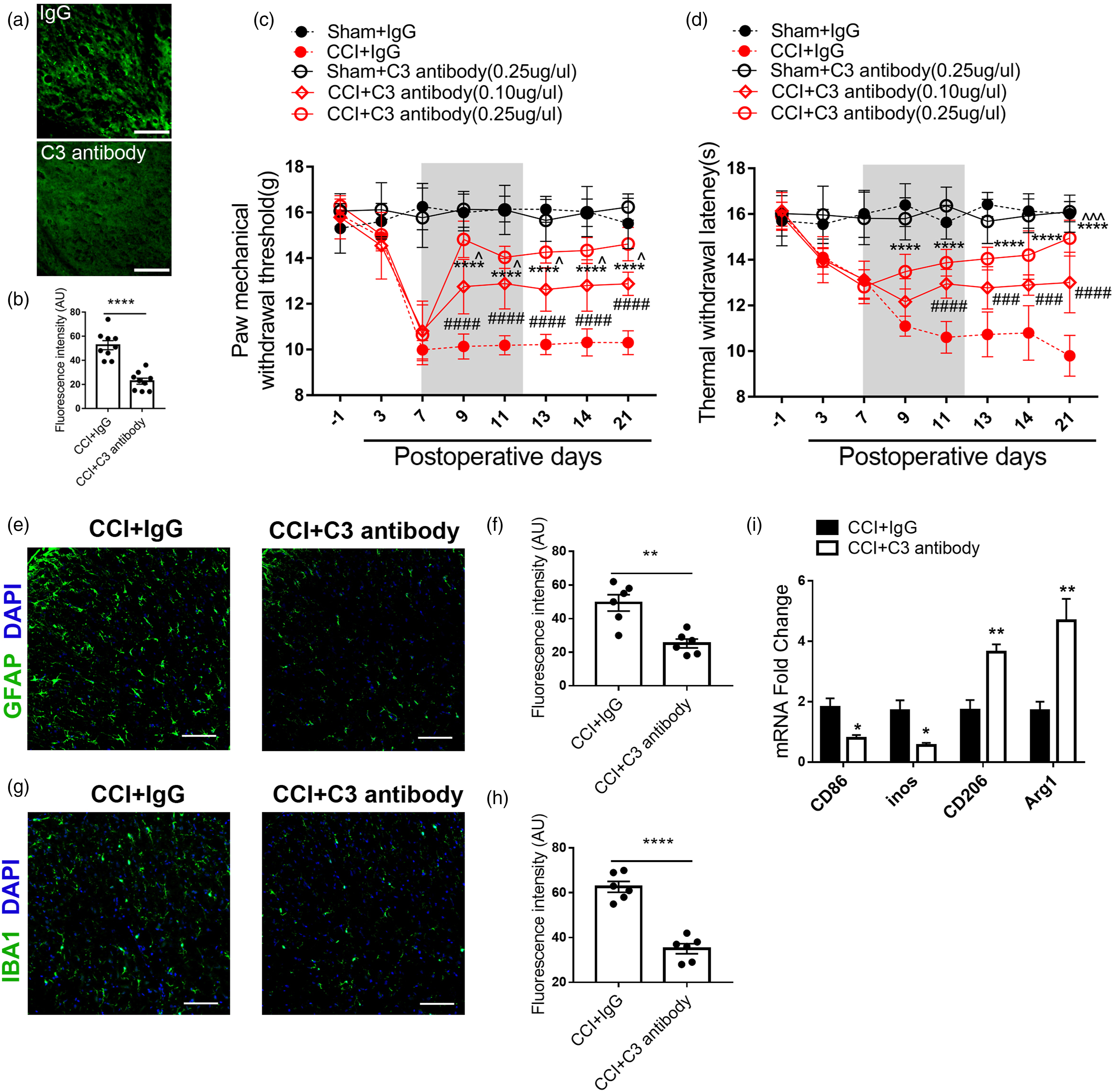
Blocking C3aR alleviated neuropathic pain and reduced M1 polarization of microglia
Then we explored the effects of the receptor of C3 (C3aR) on microglial polarization by application of C3aR specific antagonist. Compared with vehicle groups, the values of PWT and PWL were markedly increased in the C3aR antagonist group. The behavioral results also showed that higher concentration of C3aR antagonist Trifluoroacetate (10ug versus 20ug) presented stronger effects, suggesting that blocking C3aR could alleviate NP. The behavioral results of sham group showed that C3aR specific antagonist did not change the pain behavior baseline (Figures 5A and B). Immunofluorescent analysis showed that the injection of trifluoroacetate downregulated both IBA1 and GFAP compared with vehicle (Figure 5C–F). Upregulated CD206 and Arg1 and decreased CD86 and inos were also detected after the application of trifluoroacetate, which suggested the antagonization of C3aR induced the transformation of microglial to M2 phenotype (Figure 5G). The above data indicated that C3aR mediated C3-induced NP and microglial pro-inflammatory polarization. Intrathecal administration C3aR antagonist alleviated neuropathic pain and decreased M1 polarization of microglia. 
Discussion
In this study, we revealed that in SDH 1) C3 and its receptor C3aR elevated after peripheral nerve injury with chronic NP; 2) C3 was expressed on astrocytes while C3aR was mainly expressed on microglia and neuron; 3) suppressing C3-C3aR pathway by intrathecal injection of C3 antibody and C3aR antagonist could alleviate NP; 4) the activation and polarization of microglia could be regulated by C3-C3aR pathway. Taken together, our results demonstrated the critical roles of C3-C3aR pathway in astrocyte-driven microglia activation and polarization during the development of NP.
The complement system was found to initiate or amplify pain signals from the periphery to the central nerve system, including skin, primary sensory neurons, spinal cord, and brain.25,26,31–34 C3 is the point of convergence gating complement system activation. Scholars previously identified excessive C3 in the spinal cord of the CCI model. 26 However, the observation of the previous study only focused on a relatively acute phase of the disease. In fact, NP last for a long time and it was widely recognized as a long-term health issue clinically. Therefore, we extended the observation to the chronic phase, indicating that regulated roles of C3 are far beyond the acute phase. Intriguingly, we found that elevation of C3aR was behind C3, which started at POD14 (chronic phase). Consistent with our finding, a previous study found C3aR knockout barely changes basal and acute burn pain. 35 Thus, the role of C3aR might be different in the acute and chronic phases of pain and need to be further investigated.
The spatial information of C3 and C3aR expression identified in the present study. We found that C3 was mainly expressed on astrocytes, while its receptor C3aR is expressed on microglia and neuron in SDH, which is consistent with findings in previous studies.36–38 Temporally, we found both C3 and C3aR get elevated between POD7-14, which is also an overlapped period of astrocyte and microglia activation.39–42 Therefore, it is necessary for expanding our knowledge regarding the bridge role of C3-C3aR in crosstalk between astrocytes and microglia. It has been found that microglia can induce astrocytes to synthesize C3 by secreting cytokines, such as Il-1α, TNF, and C1q.24,43 Our finding replenished the other side of this feedback loop, which is astroglia’s regulation of microglia via the C3/C3aR pathway. We found the underlying molecular mechanism that by blocking the C3-C3aR pathway, pro-inflammatory M1 microglia reduced while the anti-inflammatory M2 microglia was increased. According to Zheng et al. and Chen et al., the inhibition or deletion of STAT3 promote microglia polarization to M2.44,45 And it is known that C3-C3aR mediates microglia activation via increased STAT3 level and its phosphorylation,46,47 therefore in the process of NP, the polarization to M2 after blocking C3-C3aR pathway might be via STAT3. Neutralizing C3 and blocking C3aR would inhibit STAT3 activation with decreased release of inflammatory factors such as IL-6, Il-1β and TNF-α. Therefore, the microglia transformed from pro-inflammatory M1 subtype to M2 subtype and the neuropathic pain was alleviated. Moreover, one vitro study showed that inflammation-induced elevation of NF-κB mainly in astroglia and then promote the C3 release, 48 which could be regarded as the upstream of C3-C3aR pathway. Accumulating vivo studies have provided evidence that impressing NF-κB alleviates NP,49–51 it is our belief that the analgetic function might be through C3-C3aR.
Admittedly, the present study has certain limitation. Since we found that C3aR not only express on macroglia but also expressed on other cells such other neurons and astrocytes, which might also contribute to NP. There are also previous studies reported that C3aR on neurons can regulate synaptic plasticity and maintenance of normal dendritic extensions 48 or astroglia can be activated by C3/C3aR pathway secreting cytokines. Therefore, the crosstalk among glial cells and neurons is also conceivable mechanism linking the network with C3/C3aR pathway and NP. Those underline mechanisms should be further well investigated.
Conclusion
In summary, we found that polarization of microglia could be regulated by C3-C3aR pathway and cutting off the C3-C3aR pathway between astrocyte and microglia reversed NP.
Supplemental Material
Supplemental material - Astrocyte-microglia interaction through C3/C3aR pathway modulates neuropathic pain in rats model of chronic constriction injury
Supplemental material for Astrocyte-microglia interaction through C3/C3aR pathway modulates neuropathic pain in rats model of chronic constriction injury by Wanying Mou, Lulu Ma, Afang Zhu, Huan Cui, and Yuguang Huang in Molecular Pain
Footnotes
Acknowledgements
We thank Dr Wenliang Su in Department of Anesthesiology and Critical Care Medicine, Peking University First Hospital for his technical assistance in establishing animal models.
Author contributions
Wanying Mou drafted the manuscript, performed the immunofluorescence staining and behavioral test. Wanying Mou and Huan Cui performed the CCI model and behavioral assessment. Huan Cui and Lulu Ma performed the data analysis. Yuguang Huang, Lulu Ma and Afang Zhu conceived of the study, participated in its design, and helped to draft the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (grant no. 82071252).
Ethics approval and consent to participate
All animal procedures performed in this study were reviewed and approved by the Institutional Animal Care and Use Committee of the Institute of Peking Union Medical College Hospital and were conducted in accordance with the guidelines of the International Association for the Study of Pain.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
