Abstract
Triptolide (T10), an active component of Tripterygium wilfordii Hook F, is reported to have potent anti-inflammatory and analgesic effects. Additionally, MK-801, a noncompetitive N-methyl-D-aspartate receptor antagonist, can reduce glutamate toxicity and has a significant analgesic effect on chronic pain. In this study, we tested the possible synergistic analgesic ability by intrathecal administration of T10 and MK-801 for the treatment of neuropathic pain. Single T10 (3, 10, or 30 µg/kg), MK-801 (10, 30, or 90 µg/kg), or a combination of them were intrathecally administrated in rats with spinal nerve ligation. We found that single administration of T10 caused a slow-acting but long-term analgesic effect, while single administration of MK-801 caused a fast-acting but short-term effect. Administration of their combination showed obviously synergic analgesia and the 1:3 ratio of T10 to MK-801 reached the peak effect. Furthermore, application of T10 and/or MK-801 significantly inhibited the activation of microglia and astrocyte and phosphorylation of STAT3 and NR2B in the spinal dorsal horn induced by chronic neuropathic pain. Our data suggest that the combination of T10 and MK-801 may be a potentially novel strategy for treatment of neuropathic pain.
Introduction
Neuropathic pain (NP) is a debilitating pain caused by peripheral nerve injury, such as traumatic injury, surgical intervention, diabetes, or infection.1–3 Patients who suffer from NP are essentially intractable to currently available antinociceptive therapies because the underlying cellular and molecular mechanisms are unclear yet. NP can cause both central and peripheral sensitization. Central sensitization is primarily caused by the enhancement of excitatory transmission in nociception.
Glial cells are a major cell type within the central nervous system and consist of diverse cell types, including microglia and astrocyte. Under NP condition, they are highly activated and involved in the nerve injury-induced central sensitization and mechanical allodynia. Microglia is activated within 24 h after peripheral nerve damage, which is commonly associated with the early establishment of NP. Astrocyte is activated as late as three days following microglia activation. 4–6
Tripterygium wilfordii Hook F. (TWHF) is a traditional Chinese herb. T10 is one of the major active components of TWHF and has potent anti-inflammatory and immunosuppressive properties for treatment of various diseases, such as rheumatoid arthritis, nephritic syndrome, lupus, Parkinson's disease, and Alzheimer's disease.7–9 Recent studies have suggested that T10 can inhibit tumor necrosis factor-alpha, interleukin (IL)-1beta, and nitric oxide production in spinal microglia.10,11 Our previous studies have demonstrated that T10 effectively relieves NP by inhibiting the activation of microglia and astrocyte in the spinal dorsal horn (SDH) after spinal cord injury. 8 T10 also inhibits the proliferation of reactive astrocyte and blocks the hypertrophy of astrocytic processes after spinal cord injury. 8 However, T10 has been investigated to have some side effects along with the increased dosage. Since the effect of T10 is mainly mediated by acting on the glia, 8 analgesic drugs that target on neurons may produce a synergistic effect with T10 and reduce its required dose.
MK-801, also named as dizocilpine, is a noncompetitive N-methyl-D-aspartate receptor (NMDAR) antagonist. Previous studies have indicated that MK-801 can block neuronal NMDAR-mediated glutamate toxicity during the induction phase of human optic neuritis and highlight a potential reagent for therapeutic neuroprotection. 12 However, other studies have reported that a large dose of MK-801 have unacceptable neurotoxic effects. 13
Synergistic, additive or antagonistic interactions can be observed when two analgesics are administered at the same time. When acting synergistically, lower doses of each drug can reach an equal or better analgesic effect, but with fewer overall side effects, than the individual compound.
14
Because both T10 and MK-801 are associated with an increased risk of side effects, combining the two at a lower dosage to produce a synergistic effect might be a better analgesic strategy. In the current study, by simultaneous intrathecal (
Methods
Experimental animals
Male Sprague-Dawley rats (220–250 g) were used in the present study and housed in cages that were maintained on a standard 12:12 h light/dark cycle. All experiments were conducted in accordance with approved experimental protocols of the Animal Use and Care Committee for Research and Education of the Fourth Military Medical University (Xi'an, China). All efforts were made to minimize animal suffering and reduce the number of animals used. 15
The i.t. Implantation
Rats were implanted with a single
Spinal nerve ligation
L5 SNL was performed according to Kim and Chung.
16
Briefly, rats were anesthetized with 7% chloral hydrate (0.4 ml/100 g,
The i.t. Administration of T10, MK-801, or their combination and experimental design
T10 was obtained from Fujian Academy of Medical Sciences (Fujian, China) and MK-801 was obtained from Sigma-Aldrich (St. Louis, MO, USA). They were dissolved and diluted with preservative-free normal saline solution for administration. Normal saline (0.9%) was used as the negative control. The doses for
The experimental procedures are shown in Figure 1. A total of 272 animals were used in the study and randomly assigned to one of eight groups: (1) normal group, (2) sham saline group, (3) SNL saline group (a volume of 5–10 µl saline was injected), (4) SNL-T10 group (3, 10 or 30 µg/kg/daily), (5) SNL-MK-801 group (10, 30 or 90 µg/kg/daily), (6) SNL-T10/MK-801 group (1:1, 5 µl T10 + 5 µl MK-801), (7) SNL-T10/MK-801 group (1:3, 2.5 µl T10 + 7.5 µl ΜK-801), and (8) SNL-T10/MK-801 group (3:1, 7.5 µl T10 + 2.5 µl ΜK-801). Each group included 34 rats: 18 for behavioral testing (6 for low concentration, 6 for medium concentration, and 6 for high concentration), 8 for immunohistochemical staining (third day and seventh day), and eight for Western blot analysis (third day and seventh day). The T10 and MK-801 dosages, used singly and combined, for the immunohistochemical staining and Western blot analysis were the effective dose 50 (ED50) of each. Behavioral experiments were conducted by double-blinded technicians and occurred at a fixed time during testing days. The rats were always habituated to the testing room for 30 min before tests. At days 1, 3, 5, 7, 10, 14, 21, and 28, the paw withdrawal thresholds (PWTs) were tested using von Frey filaments. The rats used for immunohistochemical staining and Western blot analysis were sacrificed at day 3 or 7.
Schematic diagram of the experimental design (a) and the structural formulas of the drugs (b and c). This figure depicted the behavioral tests that were conducted for each group, the time course of i.t. implantation, the time course of the lidocaine test and SNL, the duration time for drugs administration, and the structural formulas of T10 and MK-801. PWT: paw withdrawal threshold, SNL: spinal nerve ligation.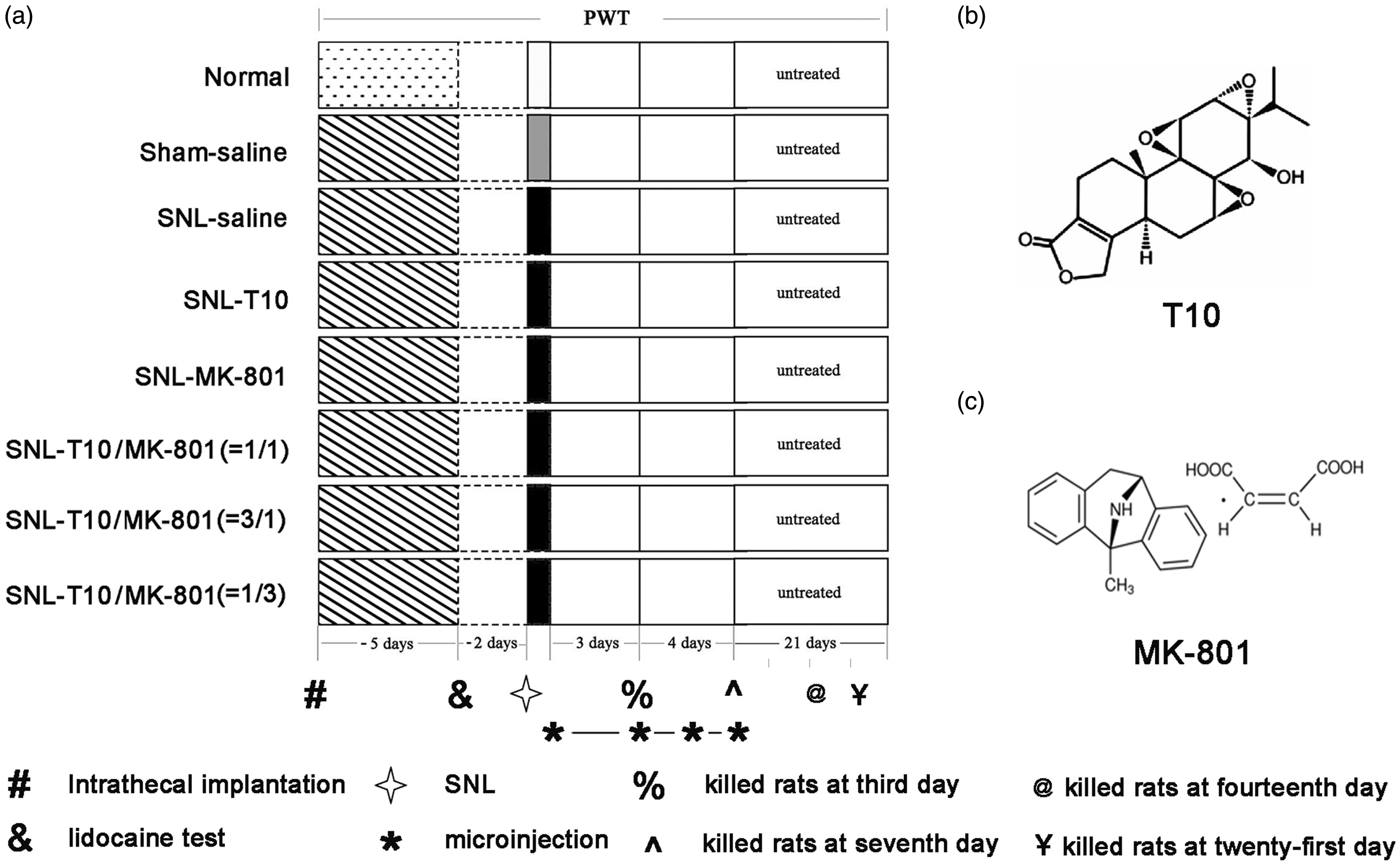
Nociceptive behavioral tests
Rats were habituated to the testing environment for three days before baseline test and were then placed under inverted plastic boxes (30 × 30×50 cm) on an elevated mesh floor for 30 min before the threshold test was conducted to allow for habituation. Briefly, a logarithmic series of von Frey filaments (Stoelting, Kiel, WI, USA) was applied to the hind paw to determine the stimulus intensity threshold stiffness required to elicit a paw withdrawal response. The ipsilateral hind paw was pressed with one of a series of von Frey filaments with increasing stiffness (2, 4, 6, 8, 10, 15, and 26 g) applied to the plantar surface for 5 s for each filament. Rapid pulling back, biting, or shaking of the ipsilateral hind limb was taken as a positive sign of withdrawal responses. The interval between trials was at least 5 min. For each trial, the same hind limb was stimulated 10 times by a single von Frey filament before being stimulated by the next larger filament. The minimal value that resulted in at least six responses to 10 stimulations was recorded as the PWT.
Dose–effect curve and ED50 calculation
The dosages of
Isobolographic analysis
An isobolographic analysis was further performed to characterize the drug interactions according to the method originally described by Tallarida 14 and our previous reports. 17 Both drugs in this experiment achieved comparable levels of anti-nociception, such that ED50 values were used to obtain a theoretical dose-response curve for a fixed ratio combination of T10 and MK-801. We calculated a theoretical ED50 (ED50add) based upon the theoretical dose-response curve. Subsequently, an experimental dose-response curve was obtained by treating animals with one of the following combination doses: ED50add*0.3, ED50add*1 and ED50add*3 in ratios of 1:1 for T10 and MK-801. According to this dose-response curve, the ED50 of combinations could be calculated and presented as the ED50comb. An ED50comb that was lower than the ED50add suggested a synergistic effect of these two agents.
Immunofluorescent histochemistry
Rats were deeply anesthetized with 7% chloral hydrate (0.4 ml/100 g,
For double immunofluorescence histochemical staining, the sections were incubated with a mixture of mouse monoclonal anti-glial fibrillary acidic protein (GFAP, a marker of astrocytes) IgG (1:4000; Chemicon, Temecula, CA, USA), rabbit polyclonal anti-phospho-signal transducer and activator of transcription 3 (pSTAT3) IgG (1:300; Cell Signaling, Danvers, MA, USA) overnight at 4℃. Then, all of the above sections were treated with a mixture of biotin-conjugated donkey anti-rabbit IgG antibody (1:500; Jackson Immuno-Research, PA, USA) for 6 h at room temperature. Finally, the sections were then treated by a mixture of 10 µg/ml Alexa488-conjugated donkey antibody to mouse IgG (1:500; Invitrogen, Carlsbad, CA, USA) and Alexa594-conjugated streptavidin (1:500; Jackson Immuno-Research, West Grove, PA, USA). The sections were observed under a confocal laser scanning microscope (FV1000; Olympus, Tokyo, Japan) with the appropriate laser beams and filter settings for Alexa488 (excitation, 488 nm; emission, 510-530 nm) and Alexa594 (excitation, 543 nm; emission, 590–615 nm).
Western blot analysis
Rats were deeply anesthetized by injection with 7% chloral hydrate (0.4 ml/100 g,
Statistical analyses
All data were collected and analyzed by researchers who were blinded to the surgery and reagents that were used. One- or two-way analysis of variance (ANOVA) followed by Bonferroni's
Results
Effects of i.t. alone administration of T10 or MK-801 on SNL-induced mechanical allodynia
Consistent with previous studies,8,17,18 SNL produces rapid and persistent mechanical allodynia as evidenced by significant decreases in ipsilateral PWTs, indicating successfully induced chronic NP by SNL.
In the present study, the PWTs of the ipsilateral hind limb decreased significantly at post-operational day (POD) 1 and remained decreased until POD 7 (Figure 2(a)). When compared with the SNL saline group, T10 dose dependently inhibited SNL-induced mechanical allodynia of the ipsilateral hind paw. (a) The analgesia effects of different doses of 
When compared with the SNL saline group, MK-801 dose dependently inhibited SNL-induced mechanical allodynia of the ipsilateral hind paw. (a) The analgesia effects of different doses of 
Taken together, these results demonstrate that
Effects of i.t. administration of T10 and MK-801 on SNL-induced mechanical allodynia
Due to the different analgesic profiles of Combination treatment of T10 or MK-801 with different ratios. (a, b, and c) The analgesia effect of different volumes of 
The
These results revealed that the 1:3 ratio of T10 and MK-801 synergistic analgesia enhanced analgesic intensity with significantly reduced doses of T10 and MK-801.
Effective drug duration of i.t. T10, MK-801 or their combination on SNL-induced mechanical allodynia after drug withdrawal
To determine the long-term effect after drug withdrawal, we continually to observe the possible analgesia effect for another three weeks after injecting drugs for one week (from POD 1 to POD 7). After drug withdrawal, the PWTs in the combination drug administration groups (Figure 5(b)) were significantly higher than those in the single drug administration groups (Figure 5(a)) and saline administration group. As summarized by the AUC values of the PWTs, the analgesic effects of Repeated administration of T10, MK-801, or their combination reversed SNL-induced mechanical allodynia. (a–b) The continual analgesia duration of 
Effect of i.t. administration of T10, MK-801, or their combination on SNL-induced glial activation
Our previous studies have suggested that microglia and astrocytic activation was involved in the development and maintenance of chronic pain. Thus, we questioned whether microglial or astrocytic inhibition contributed to synergistic analgesia following the co-administration of T10 and MK-801. To test this hypothesis, we used ionized calcium-binding adapter molecule 1 (Iba-1, a marker of microglia) to detect microglial activation on POD3 and GFAP (a marker of astrocytes) to observe astrocyte activation on POD 3, 7, 14, and 21.
In our present study, the results of immunofluorescence histochemistry for Iba-1 and Western blot on POD3 showed that SNL evoked significant microglial activation (Figure 6(A: c and c′)) and that the combination of T10 and MK-801 significantly inhibited the expression and activation of Iba-1 in the ipsilateral SDH (Figure 6(A: f, f′, g, g′, h, and h′), (B), and (C)) (## Administration of T10, MK-801, or their combination on SNL-induced microglial activation. Immunofluorescent results showed that SNL induced up-regulation of Iba-1 in the ipsilateral SDH (A; c and c′). Administration of individual or concomitant drugs down-regulated Iba-1 expression in the ipsilateral SDH on day 3 (d–h and d′-h′ ). The rectangle areas in a–h are enlarged in a′-h′, respectively. (B) Western blot of Iba-1 expression at day 3 in groups with saline or drug treatment. (C) The relative optic densities of Iba-1 shown in (B). ##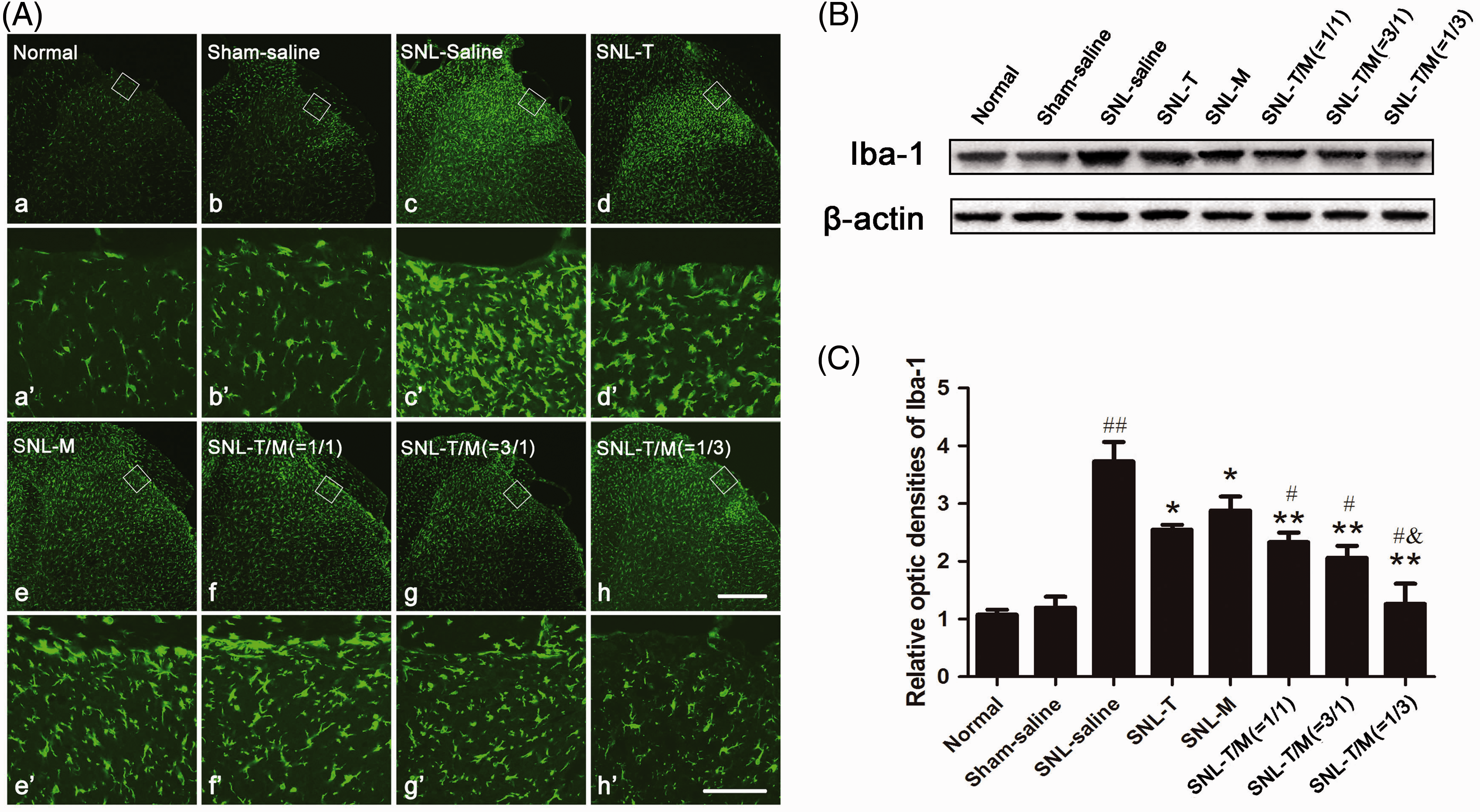
Our previous studies have also showed that SNL rats exhibited GFAP up-regulation, which is significant on day 3 and reaches the peak on day 7 after SNL. In the present study, we also found the activated astrocytes, presenting as hypertrophied cell bodies and thickened processes with enhanced GFAP-immunoreactivity (IR) (Figure 7(a) and (b)). Western blot analysis showed that the GFAP and p-STAT3 expression levels were significantly increased after SNL. However, treatment with T10 or MK-801 decreased the GFAP and p-STAT3 expression on POD 3 (Figure 7(e) and (g)) and POD 7 (Figure 7(f) and (h)), but not on POD 14 (Figure 7(i) and (k)) and POD 21 (Figure 7(j) and (l)), in comparison with the SNL saline group. The combined T10 and MK-801 treatment groups showed stronger reversal effects of the GFAP expression than T10 or MK-801 single treatment groups, except on POD 21 (Figure 7(g), (h) and (k), (l)). When the ratio of T10:MK-801 was 3:1, the expression levels of GFAP and p-STAT3 were inhibited maximally (Figure 7(g), (h), and (k)).
Administration of T10, MK-801, or their combination on SNL-induced astrocytic activation and STAT3 phosphorylation. (a–d) Immunohistochemistry indicated that activated astrocytes presented as hypertrophied cell bodies and thickened processes with enhanced GFAP immunoreactivity (IR) and many of the p-STAT3-IRs was co-labeled with GFAP-IR. Western blot analysis showed that the GFAP and p-STAT3 expression levels were significantly decreased on days 3 (e and g) and 7 (f and h) but not on days 14 (i and k) and 21 (j and l), in all intervention groups compared with the SNL saline group. However, the combined treatment groups experienced better outcomes than the single treatment groups except on day 21 (g, h and k, l); When the ratio of T10 and MK-801 was 1/3, the expression levels of GFAP and p-STAT3 were significantly reduced (g, h, and k). ##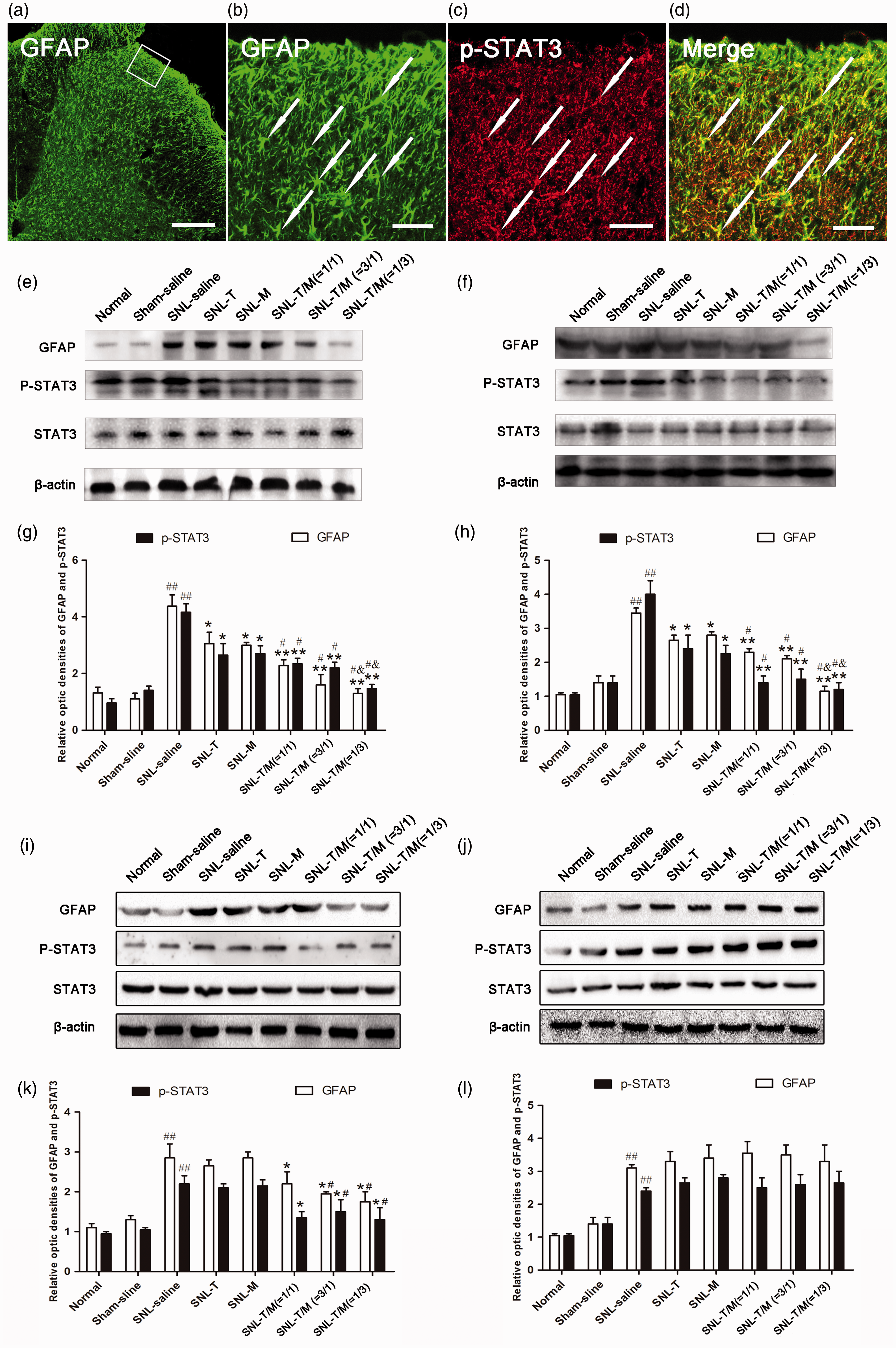
Effect of i.t. T10, MK-801, or their combination on SNL-induced activation of NMDAR
Activation of NMDARs, especially the activated subtype 2B of NMDAR (NR2B), is widely reported in rodents' SDH in chronic pain condition, we thus wanted to check whether Administration of T10, MK-801, or their combination on SNL-induced expression of pNR2B. SNL injury resulted in increased levels of pNR2B in the ipsilateral SDH seven days after nerve ligation. Administration of T10, MK-801, or their combination could prevent SNL-evoked pNR2B accumulation. Combination groups have better effects than single drug groups. ##
Discussion
To our knowledge, this is the first study that explores the synergistic analgesia of T10 and MK-801 in chronic NP rat. Our data demonstrate that
Administration of T10 and MK-801 had synergistic analgesic effect but with weak side effects
The interest in exploiting traditional Chinese medicine for the prevention or treatment of chronic pain has been increased greatly.8,18,19 T10 has an important role in modulating immune function both through and independent of the nervous system. It is reported that T10 can inhibit neurotoxic and proinflammatory cytokine, such as inducible nitric oxide synthase and cyclooxygenase-2 in microglia.10,20 Furthermore, T10 also exerts protective effects on neurons suffering from inflammation-mediated damage. 21 A recent study has demonstrated that T10 suppresses the development of NP and T-cell activation.22,23
NMDARs are involved in the development and regulation of chronic pain.24,25 MK-801, also called dizocilpine, displays a wide array of biological properties, including anticonvulsant and anesthetic properties, due to its activity as a noncompetitive NMDAR antagonist. It is reported that
In our present study, we found that the alone administration of T10 or MK-801 can inhibit SNL-induced mechanical allodynia. Based upon previous studies and our present experimental data, the anti-nociceptive effect of T10 or MK-801 is dose dependent. However, high dose of T10 or MK-801 will induce accompanying toxic side effects. For example, T10 accumulates in the testis and liver will damage the normal functions of these organs. A large dose of MK-801 impairs locomotion.
13
Therefore, we checked whether
Administration of T10 and MK-801 inhibited activation of glial cells and NR2B in the spinal cord
Our previous study indicated that
Modification of synaptic NMDAR expression will influence NMDAR-mediated synaptic function and associated physiological and pathological effects. Membrane NMDARs is dynamic, especially the NR2B subunit. NR2B binds to the membrane-associated guanylate kinases that regulate surface and synaptic NMDAR trafficking
29
. It is reported that NR2B plays role in the transmission of nociceptive information and development of chronic pain.30,31 The expression of NR2B in SDH increases 48 h after SNL in rats. This increase reaches its peak at 3 days, lasts for 14 days, and returns to pre-operational levels 28 days after SNL.
32
The activation of astrocytes leads to a consequent release of neuro-excitatory substances, including prostaglandins, IL-1, IL-6, and subsequently induces the phosphorylation of NR2B subunit in spinal neurons. In this study, Western blot analysis revealed that
Based upon these previous studies and our experimental data, we hypothesize that the synergistic analgesic mechanisms of
Administration of T10 as an adjuvant could be a new analgesic strategy
Clinical cases have shown that drug combinations can provide better pain relief and minimize side effects compared with singly groups. However, most analgesic methods focus on neuronal participation during chronic pain progression. In consideration of the pivotal role of glial cells during the initiation and maintenance of chronic pain, the combination of a neuronal inhibitor and a glial inhibitor may represent a new strategy for a more effective treatment of chronic pain.
Our present results showed that the co-administration of T10 and MK-801 can enhance the synergistic analgesic effects on NP, compared with the single administration of either drug. The experimental ED50 (ED50comb) of the drug combination was lower than that of the theoretical ED50 (ED50add). Moreover, the present study showed that the combination of MK-801 with T10 could induce a long-term effect even at 7–14 days after drug injection, particularly in the group with MK801:T10 equals to 3:1. Although the lifetime of MK-801 is short (approximately three days), the sedative and analgesic properties of MK-801 might contribute to this long-lasting and accumulative analgesic effect. In contrast, the lifetime of T10 is long (approximately seven days). Therefore, a combination of T10 and MK-801 could compensate the short lifetime of MK-801. More importantly, in the combination groups, the dosages of ED50comb of T10 and MK801 are all decreased but the analgesic effect are all potentiated, compared with the single application groups. Therefore, these combined groups reduce T10 and MK801 administration, indicating less potential neurotoxicity and a possible reduction in additional side effects.
Conclusions
These results demonstrate that the
Footnotes
Author contributions
J Wang, Y Qiao, and R-S Yang contributed equally to this work. Y-Q Li, Y-L Dong, and J-L Li corresponded equally to this work in designing and supervising the project. J Wang, Y Qiao, R-S Yang, and C-K Zhang performed the animal surgery, carried out the immunofluorescence and Western blot study, and drafted the manuscript. J-J Lin and T Zhang performed the behavioral test. H-H Wu participated in producing graphics and performed the statistical analysis. J Wang, Y Qiao, R-S Yang, and C-K Zhang conceived the study and participated in its design and coordination. T Chen, Y-Q Li, Y-L Dong, and J-L Li designed the experiment. All authors read and approved the final manuscript.
Declarations of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (81571074, 31671087, 31371126 and 81671095), the National Key Scientific Instrument & Equipment Development Program of China (2012YQ03026009 to Y-Q Li), and an intramural grant of the Fourth Military Medical University (4139C4IAA1).
Supplemental Material
Supplementary material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
