Abstract
Background
Noncoding microRNAs have emerged as critical players of gene expression in the nervous system, where they contribute to regulating nervous disease. As stated in previous research, the miR-155-5p upregulation happens in the spinal cord at the nociceptive state. It was unclear if miR-155-5p is linked to bone cancer pain (BCP). Herein, we aimed at investigating the miR-155-5p functional regulatory function in BCP process and delineating the underlying mechanism.
Methods
The miRNA-155-5p levels and cellular distribution were determined by RNA sequencing, fluorescent in situ hybridization (FISH), and quantitative real-time PCR (qPCR). Immunoblotting, qPCR, dual-luciferase reporter gene assays, immunofluorescence, recombinant overexpression adeno-associated virus, small interfering RNA, intraspinal administration, and behavioral tests were utilized for exploring the downstream signaling pathway.
Results
The miR-155-5p high expression in spinal neurons contributes to BCP maintenance. The miR-155-5p blockage via the intrathecal injection of miR-155-5p antagomir alleviated the pain behavior; in contrast, upregulating miR-155-5p by agomir induced pain hypersensitivity. The miR-155-5p bounds directly to TCF4 mRNA’s 3′ UTR. BCP significantly reduced protein expression of TCF4 versus the Sham group. The miR-155-5p inhibition relieved the spinal TCF4 protein’s down-expression level, while miR-155-5p upregulation by miR-155-5p agomir intrathecal injection decreased TCF4 protein expression in naïve rats. Additionally, TCF4 overexpression in BCP rats could increase Kv1.1. Moreover, TCF4 knockdown inhibited Kv1.1 expression in BCP rats. Indeed, TCF4 and Kv1.1 were co-expressed in BCP spinal cord neurons.
Conclusion
The study findings stated the miR-155-5p pivotal role in regulating BCP by directly targeting TCF4 in spinal neurons and suggested that miR-155-5p could be a promising target in treating BCP.
Introduction
Bone cancer pain (BCP) brought considerable trouble to the quality of patients’ daily life. 1 Unfortunately, few patients obtain complete relief from the currently available analgesic treatment. It is critical to understand pathophysiology to propose a novel therapeutic measure in this field. The microRNAs (miRNAs) and nociceptive processing are strongly connected.2,3 miRNAs are a subset of non-coding functional RNAs that regulates eukaryotic gene expression via the direct target mRNA’s sequence, 3’-untranslated region (UTR), resulting in translational repression or mRNA destruction.4–6 Increasing evidence indicated that miRNAs were highly expressed in the nervous system 7 and had a pivotal function in neuroinflammation and neuropathic pain. 8 miR-155-5p was regarded as a master inflammation regulator, 9 playing a pivotal role in various diseases, like cancer formation 10 and myocardial infarction. 11 Additionally, it is indicated that miR-155-5p inhibition might decrease the neuropathic pain caused by chemotherapeutic bortezomib via neuroinflammation engagement. 12 Miao F et al. found that miR-155-5p was involved in exaggerating inflammation signals in neuropathic pain. 13 The results stated that miR-155-5p could be a crucial regulator in neuropathic pain. However, the miR-155-5p mechanism in pain pathogenesis in bone cancer pain remains unknown.
Gene TCF4 encodes a type I transcription factor with a basic helix-loop-helix (bHLH) structure located on chromosome 18. 14 It is well established that TCF4 identified and regulated gene expression by recognizing E-box (CANNTG) sequences within the target genes’ promoter and enhancer regions. 15 TCF4 is involved in the neurogenesis and specialization of cells regulated by many downstream target genes, such as Nav1.8 and KCNQ1.16,17 miR-155-5p could directly suppress TCF4 expression in colorectal cancer. 18 Indeed, TCF4 is essential in developing the brain and important leather. TCF4 also regulates DNA methylation, synaptic plasticity, and memory function in a rare autism spectrum disorder, Pitt Hopkins syndrome (PTHS). 19 Furthermore, it has been reported that upregulating transcription factor 4 could prevent rat inflammatory and neuropathic hypersensitivity development. 20 However, the TCF4’s major role in bone cancer-induced pain remains unknown.
It is also demonstrated that TCF4 controls intrinsic excitability, synaptic transmission, and synaptic plasticity. 21 TCF4 embryonic knockout led to Kv1.1 (Kcna1) downregulation, a voltage-gated potassium channel.22,23 Generally, Kv1.1 channels reduce neuronal excitability, and their blockade leads to low action potential production voltage thresholds. 24 It has been reported that Kv1.1 channel contributes to the mechano-sensitive K+ current regulating certain types of mechano-sensitive fibers’ threshold, like DRG and auditory neurons’ subsets. 25
Our work revealed that BCP induced miR-155-5p upregulation in spinal cord dorsal horn tissue using miRNA expression profiling and PCR. Then, we evaluated the miRNA-155-5p and its target genes’ expression, namely TCF4, in the rats’ spinal cord. We intrathecally administered the miR-155-5p agonist (miR-155-5p agomir) or antagonist (miR-155-5p antagomir) to investigate the miR-155-5p role in pain modulation. The study findings stated that miR-155-5p could aggravate bone cancer-induced mechanical hyperalgesia by inhibiting TCF4 and Kv1.1. This research could point to a new target for treating and alleviating bone cancer pain.
Materials and methods
Animals and groups
The Shanghai Leagene Biotech Co. Ltd (No.20200-0007) provided female adult Sprague-Dawley rats that weighed 180–200 g. They were put in plastic cages with sufficient water and food supply. The breeding room was always maintained with a light/dark cycle for 12 h. The JUMC Laboratory Animal Ethics Committee (JUMC2021-098) approved all experiments and protocols. For each experiment, the sample size (alpha: 0.05) for sufficient size was determined based on similar previous studies.
The rats were classified randomly into numerous groups: Naive, Naive + miR-155-5p Agomir (Naive +Agomir group), Naive + miR-155-5p Agomir control (Naive + Agomir control group), Sham, BCP, BCP + miR-155-5p Antagomir (BCP + Antagomir group), BCP + miR-155-5p Antagomir control (BCP + Antagomir control group) and BCP +TCF4 siRNA, BCP + scramble siRNA, BCP +TCF4 overexpression adeno-associated virus, AAV (BCP + AAV-TCF4 group), and BCP + TCF4 overexpression adeno-associated control (BCP + AAV control group).
Chemicals and reagents
Chemicals and reagents.

Tumor cells and BCP animal model’s preparation
The tumor cell preparation was done as reported before. 26 The cells were intraperitoneally inoculated into female rats, washed in PBS solution, and diluted to a 106 cells/mL concentration. The same amount of carcinoma cells was administered to Sham group, which was heat-killed for 30 min.
As reported earlier, Walker 256 tumor cells were injected into the intramedullary space of the left leg using the BCP model.26, 27 A small slit was done to the left tibia’s lower one-third while they were sedated using pentobarbital sodium (60 mg/kg, i.p.). Next, Walker 256 cells (106 cells/mL) or heat-killed cells (106 cells/mL for Sham group) were administered to the tibia intramedullary space. To prevent the cell’s leakage, a syringe was held for 60 s, and an instant blockage to the injection site by bone wax was done. The incision was sutured, and chlortetracycline eye ointment was applied. Animals were wrapped in warm blankets after surgery.
Intrathecal catheterization and intraspinal administration
As previously described, animals were first anesthetized with pentobarbital sodium. 28 PE-10 microtube was used for drug delivery after being inserted between L4 and L5 in the intervertebral space. The microtube was attached to the surrounding ligament using a 3.0 suture when PE-10 was placed into the lumbar enlargement’s subarachnoid space. Finally, the microtube’s free end was exposed for 2 cm and plugged. A lidocaine test was performed on the day following the surgery. After 15 s of intrathecal gavage of 10 μL lidocaine, both hind limbs were paralyzed, indicating normal catheter function.
AAV virus intraspinal injection was performed by exposing L4-L5 vertebrae and mounting the spinal column in a stereotaxic frame. After conducting, a minor laminotomy, an incision of the dura was used to reveal the spinal cord. The glass pipettes were inserted into the dorsal spinal cord at a tilting angle (45°). Micromanipulator was used to inject AAV into three sites (400 nL per site at a rate of 40 nL/min) of ipsilateral L3-5 dorsal horn. 10 minutes after the viral injection, the micropipette was withdrawn, and the incision was applied to close stitches.
Behavioral assessment
By employing von Frey monofilaments, the left hind paw’s mechanical allodynia was measured, as described before. 28 For 30 min, the rats were kept separately in translucent Plexiglas chambers (25 × 20 × 20 cm) on a metal-wired platform to adapt to their new surroundings. Each stimulus was repeated thrice with 5 min intervals. The maximum tolerance in grams was utilized to establish PWT, and each hind paw’s mean PWT was calculated as the three tests’ average. The procedures to test the behavior were executed by blinded experimenters to the animal groups to maintain the results’ accuracy and reliability.
CatWalk XT system has been suggested as an approach for assessing pain behaviors. The machine has a red ceiling light-emitting diode and a high-speed camera under a glass platform, allowing walking through the walkway. The rat was arranged in the open port of an enclosed walkway and allowed to move voluntarily, a high-speed camera positioned took pictures of each paw’s illuminated area, and the data were transferred to the gait analysis software.
Western blot analysis
Antibody and probe.

miRNA sequencing and data processing
In this experiment, total RNA samples were isolated from freshly harvested spinal cords of eight rats (4 BCP and 4 Sham). Then, agarose electrophoresis and quality control were performed using NanoDrop ND1000 (Nanodrop, USA). Fragments from PCR amplification (135-155 bp) were selected, repaired with 5′- and 3′-adaptors, and subsequently reverse transcribed into cDNA before amplification. miRNA libraries were built and sequenced by NEB Multiplex Small RNA Library Prep Set for Illumina (NEB, United States). EdgeR was utilized to analyze miRNAs differential expression between groups under cut-off values of p < 0.05, absolute values of fold-change change >1.5 and <0.7, and visualized with a heatmap of miRNA clusters.
Real-time quantitative PCR
L4-L6 spinal cord lumbar enlarged tissue was removed and stored at −80°C. Total RNA from the spinal cord was isolated by the commercially available trizol reagent, and its purity and concentration were determined by measuring the absorbance. For cDNA synthesis, isolated RNAS was reverse transcribed into cDNA and pre-heated for 30 s at 95°C, then 40 amplification cycles (95°C for 5 s and 60°C for 30 s). The Sangong Bioengineering (Shanghai) provided the primers. Their sequences are as follows: miR-155-5p:5’-3’: UUAAUGCUAAUUGUGAUAGGGGU. Experimental results were measured using the 2-ΔΔCt method for relative gene expression.
Immunofluorescence and FISH
Briefly, rats were anesthetized and exsanguinated in large doses. Following that, the lumbar enlargements underwent dehydration in a 10%–30% sucrose gradient in sterile water for 5–7 days, after storage for 4–6 h at 4°C in 4% phosphate-buffered paraformaldehyde. After treatment, the tissues were frozen in a cryostat at 25°C under zero using OCT compound and then cut into 20 μm thickness. First, the sections were blocked by 4% normal donkey serum and 0.1% Triton X-100 in PBS for 1 hour at room temperature. A primary antibody against TCF4 (Affinity, Santa Cruz), Kv1.1 (Alomone, Affinity), GFAP (Sigma), Iba-1 (Abcam), as well as NeuN (Abcam) were added to every section at 4°C (Table 2). The miR-155-5p hybrid probe was synthesized by GenePharm. Pre-hybrid drops (excluding probe) were added to the slice, then put the slice under the sealed box at 37°C. After 15 h, the incubated sections were washed with PBST (1× PBS with 0.1% Triton 100) and incubated for 1 hour with FITC- or Cy3-conjugated secondary antibodies (Abcam) at 23°C in the dark (Table 2). The nuclei were stained with VECTASHIELD Antifade Mounting Medium with DAPI (Vectashield hard-set mounting medium; Vector) (Table 2). At last, using LSM 880 with Airyscan, the stained sections were surveyed and analyzed.
Luciferase assay
Wild-type (5′-AGCATTA-3′) or mutated (5′-TCGTAAT-3′) miR-155-5p binding sites were made using RIBOBIO and cloned into the psi-CHECK-2 luciferase plasmid’s KpnI and BglII sites, respectively. The 293 T cells underwent co-transfection with reporter 200 ng pMIR-Report Luciferase vector and miRNA mimic using Lipofectamine 2000 (Invitrogen) at a final 100 nM concentration. Finally, the dual-luciferase reporter assay kit assessed the luciferase activity of cells.
Bone histological analysis
The rats were administered pentobarbital in large doses (80 mg/kg: i.p.), then following a 12-days tumor implant, and they were sacrificed. The left tibia tissues around the injection site were taken (1 cm). For a whole day, the tibia bones underwent decalcification using a 10% EDTA solution. The bone tissues were then dried and cut using a rotary microtome into 8 μm-thick sections stained by hematoxylin and eosin (H&E) to determine the tumor cell infiltration’s extent. All pictures were taken using a microscope with a 10 × or ×20 objective.
Computed tomography reconstruction
The bone destruction was observed using 3D CT bone reconstruction technology. The following CT scan parameter settings were based on a previous report 27 : helical scanning, 120 vvkVp; 1 mm layer thickness; care dose 4D technique; 1 mm layer interval; kemel: U30u medium smooth, with SD rats tibia’s CT high-resolution imaging (100 mm view field). At last, for reconstructing the images, the Siemens Syngo MMWP post-processing workstation was utilized.
Open Field Testing
An open field test was conducted on the rats to evaluate their spontaneous exploration of the environment. The open field comprises a 1x1 m cube devoid of wooden box covers. ANY-maze V6.14, a software-based system for acquiring and analyzing behavioral data, laid out the area into 16 equal parts, including four centrally located areas and 12 peripherally located areas. Typically, a camera was placed over the field to record the rats' behavior clearly and smoothly. 30 minutes before the test, rats were introduced to a testing room to being familiar with a new environment (acclimatization) and lowering stress. Following an adaptation period of 30 s, we used ANY-maze to record and analyze the rats' behavior during the 5-min test.
Statistical analysis
The SPSS version 25.0 was utilized for the statistical significance analysis. The results are expressed as mean ± SD. The sample size calculations were done following past behavioral and molecular investigations. The Student t-test was adopted for two group comparisons. The multiple group comparisons were analyzed by the one-way ANOVA, two- or three-way repeated-measures ANOVA, and Bonferroni multiple comparison post hoc test analyzed the differences between groups. To assess data distribution, we employed Shapiro-Wilk test. Statistical significance was defined as p < 0.05.
Results
Intratibial inoculation with walker 256 breast carcinoma cells induces bone destruction and nociceptive hypersensitivity
The flowchart includes the study design to assure that BCP model was established successfully (Figure 1(a)). To identify the bone cancer pain animal model’s establishment, we detected the mechanical allodynia and bone destruction after tumor inoculation. After intramedullary inoculation, BCP rats had bone deterioration and sustained mechanical allodynia. The tumor-bearing rats’ PWT was decreased 6 days following inoculation and decreased gradually during the observation period (***p < 0.001 vs. Sham group; n = 10, two-way ANOVA, Figure 1(b)). Besides, following tumor inoculation, 3D reconstruction revealed a considerable cortical bone loss (Figure 1(c)). In addition, histopathological analysis was used to detect osteolysis. The medullary cavity was filled with large morphologically altered cells’ amount, characterized by a large nucleus, a high nucleoplasm proportion, and nuclear pyknosis (Figure 1(d)). As depicted in Figures 1(e) and (a) Catwalk analysis was done in the bone cancer pain studies context, which was considered a reliable method to evaluate the pain behavior in neuropathic and inflammatory pain models.29,30 We chose two parameters that declined in a significant way in BCP rats in comparison to the Sham group: Max Contact Area and Max Contact Mean Intensity, which were considered the hind paw print area at maximum contact, and the hind paw print strengths of the intensity at maximum contact, respectively (**p < 0.01, ***p < 0.001; n = 10 in each group, unpaired t-test, Figures 1(e) to (g)). 12 days after surgery, the rat received heat-killed cells, and BCP rats were treated with open field testing. Compared with heat-killed cells treated rats, bone cancer-induced pain significantly increased anxiety disorders. As revealed in Figure 1(h), time spent and distance in the center area of BCP rats were reduced in the open field test (***p < 0.001 vs. Sham group; n = 10, unpaired t-test). BCP establishment model was successfully proved by behavioral tests and histochemical analysis. (a) The flowchart includes the study design to assure that BCP model was established successfully. (b) The left hind paw’s PWT in BCP group started decreasing after 6 days. The sham group’s pain sensitivity did not alter significantly (F(2,27) =349.94, ***p < 0.001; BCP versus Sham group; two-way ANOVA, then the Bonferroni test; n = 10). (c–d) CT scanning and HE staining created after 12 days following the tumor inoculation in sham and BCP groups. Active cancer cell invasion in bone medullary cavity destroyed normal structure. (e–g) Representative CatWalk gait, including print and timing view, and print intensity, in Sham and BCP groups. Tumor invasion significantly declined. (e) Max Contact Area and (f) Max Contact Mean Intensity on days 12 following tumor inoculation treatment. (*p < 0.05, **p < 0.01, unpaired t-test). The detected animals’ number within each column is determined. (LH, left hind, RH, right hind. (h) Representative animal tracks from Sham and BCP group rats in the open field test (***p < 0.001, versus Sham group; unpaired t test; n = 10, Fig. 1(h)).
Increased miR-155-5p is located in the neuron in BCP models
Recently, miR-155-5p has been identified as involved in intense neuropathic pain and neuronal inflammation.
31
Subsequently, we verified whether miR-155-5p is a key molecule in nociceptive hypersensitivity behaviors (Figure 2(a) for the timeline of drug administration and testing). To investigate the potential spinal miR-155-5p modulatory role in the bone cancer-induced nociceptive response, we first collected Sham and BCP group spinal dorsal horn tissues and detected microRNA expression. The microRNA profiling database analysis disclosed that 45 microRNAs were significantly changed between Sham and BCP groups from 569 candidates (Figure 2(b)).
32
We then assessed the miR-155-5p temporal expression pattern in the spinal cord. The qPCR analysis was done to examine the variances in miR-155-5p levels in Sham and BCP groups; the miR-155-5p levels were markedly increased on day 12 post-BCP surgery in comparison with those after Sham surgery in the rat’s dorsal horn (Figure 2(c), ***p < 0.001 vs. Sham group; n = 4, one-way ANOVA). Additionally, to demonstrate the miR-155-5p expression in spinal cell types, we created a miR-155-5p mRNA antisense probe and then performed fluorescence in situ hybridization. FISH results reveal that increased miRNA-155-5p was distributed in the spinal cord. Immunofluorescence staining analysis further illustrated an 18.3% overlap between miR-155-5p signal and NeuN (a neuron marker) in the Sham group and a 32.1% overlap in BCP group (Figures 2(d) and (e), ***p < 0.001 vs. Sham group; n = 4, unpaired t-test). The miR-155-5p upregulation mediates neuropathic pain. (a) This part experiments’ timeline (b) heatmap of 45 microRNAs was significantly changed on day 12 post-operation. (c) The miR-155-5p relative expression level in the spinal dorsal horn at different time points following tumor inoculation treatment (F(3,12) = 73.913,***p< 0.001 vs. the Sham group, one-way ANOVA, n=4). As the mean ± SD, the data are presented. (d) FISH analysis revealed that miR-155-5p was co-expressed with spinal dorsal horn neurons (arrow). Scale bar= 100 μm; (n=4). (e) Immunofluorescence showed miR-155-5p immunoactivities in different groups. (f–g) Effects of suppression and enhancement miR-155-5p on mechanical sensitivity. Intrathecal injection of miR-155-5p agomir (1 nmol each day over five consecutive days) and miR-155-5p antagomir (1 nmol every day for 5 days consecutively) were performed. Compared to controls, miR-155 agomir intrathecal injection significantly decreased PWT in naïve rats. (F(2,27) = 122.16,**p< 0.01, ***p< 0.001, control rats versus rats with agomir, two-way ANOVA, then the Bonferroni test; n = 10). Conversely, miR-155 antagomir intrathecal injection significantly increased PWT compared with BCP +control group (F(2,27) = 14.31,*p<0.05, **p<0.01, ***p<0.001 control rats versus rats with antagomir, two-way ANOVA, then the Bonferroni test; n = 10). (h) Representative animal tracks from rats treated with miR-155 antagomir/control at BCP for 12 days in the open field test. (*p<0.001, unpaired t-test, vs. the corresponding antagomir control group, n = 10).
The miR-155-5p antagomir microinjection effect on nociceptive hypersensitivity maintenance
For assessing the increased miR-155-5p role in the bone cancer-induced nociceptive hypersensitivity’s maintenance, we intrathecally injected miR-155-5p antagomir starting on day 6 after surgery when mechanical hypersensitivities were initially developed at this time point. After antagomir intrathecal injection, the nociceptive behavior of rats with bone cancer pain was specifically attenuated compared to the antagomir control group. This research finding stated that miR-155-5p antagomir intrathecal injection (for five consecutive days) significantly reduced mechanical allodynia compared to Antagomir control rats (Figure 2(f), **p < 0.01 vs. control group; n = 10, two-way ANOVA). To understand whether miR-155-5p is related to BCP and pain-related anxiety, the total distance traveled and the time spent in the antagomir group’s central area was significantly reduced compared with control rats in the open field test (*p < 0.05 vs. control group, n = 10, unpaired t-test, Figure 2(h)). Based on this evidence, miR-155-5p has been implicated in bone cancer-induced nociceptive hypersensitivity maintenance.
Effect of microinjection of miR-155-5p agomir increases hypersensitivity to nociceptive stimuli
Furthermore, our research investigated if increased miR-155-5p in the spinal cord contributes to nociceptive hypersensitivity. Intrathecal injection of miRNA agomir upregulated the spinal miR-155-5p function. In the naïve group, miR-155-5p upregulation via miR-155-5p agomir intrathecal injection, but not agomir control, for five consecutive days, produced pain-like behavior. As illustrated in Figure 2(g), the naive animal with agomir showed significantly reduced PWT in response to von Frey on day 6 after intrathecal injection (***p < 0.001 vs. control group; n = 10, two-way ANOVA, Figure 2(g)). This result further stated that increased miR-155-5p in the dorsal horn might result in nociceptive hypersensitivity.
TCF4 was a miR-155-5p target
To investigate how miR-155-5p regulates bone cancer pain in the spinal cord on a molecular level, we conducted the following experiments. miRNA in mammals exerts a transcriptional regulation effect by combining the 3'UTR zone of transcription sequence. To predict miR-155-5p candidate gene, targetscan databases were utilized. The potential miR-155-5p and TCF4 interaction by their complementary binding sites are indicated in Figure 3(a). To provide direct functional evidence of the target gene on miR-155-5p, a luciferase reporter vector containing the 3′UTR region of TCF4 recognized by miR-155-5p was constructed. As depicted in Figure 3(b), the miR-155-5p co-transfection mimics with the wild-type reporter CHK-wt-TCF4 reduced the luciferase activity compared to the mutation-type reporter CHK-mut-TCF4. The results show that miR-155-5p could suppress TCF4 expression by binding to its 3′-UTR. TCF4 is a miR-155-5p target. (a) A potential binding site was predicted between miR-155-5p and the TCF4 mRNA’s 3’UTR. (b) miR-155-5p administration mimics significantly decreased the luciferase activity (**p<0.01 vs. miR-155-5p control group, unpaired t-test, n=3). (c) miR-155-5p regulates TCF4 expression in spinal in bone cancer pain. (**p < 0.01, ***p < 0.001, unpaired t-test.) (d) Western blot analysis revealed the expression of TCF4 dynamic changes in the spinal cord. (F(3,12) = 30.07, **p< 0.01, ***p < 0.001, versus Sham group; n = 4, one-way ANOVA). (e) Immunofluorescence showed immunoactivities of TCF4 in different groups. Scale bar: 100 μm. (f) The TCF4 relative immunofluorescence expression level in the spinal dorsal horn at different time points following the tumor inoculation treatment (F(3,12) = 30.77,**p< 0.01, ***p < 0.001, versus Sham group; n = 4, one-way ANOVA). (g–h) Expression of spinal TCF4 protein in the ipsilateral and contralateral on day 12 after BCP surgery (**p< 0.01, unpaired t-test, compared to the contralateral side).
Nevertheless, the miRNA’s ability to regulate BCP via targeting TCF4 remains unclear. Then, we found that miR-155-5p agomir intrathecal injection inhibit expression of TCF4 in naïve rat, but not by agomir control (*p < 0.05, **p < 0.01 ***p < 0.001 vs. naive group; n = 4, one-way ANOVA). By contrast, spinal TCF4 expression was increased by miR-155-5p inhibition (with miR-155-5p antagomir intrathecal injection) in BCP rats (*p < 0.05, ***p < 0.001 vs. control group; n = 4, unpaired t-test, Figure 3(c)). These findings reveal that the spinal miR-155-5p regulation after surgery, which exhibited its TCF4 inhibitory effect, may be required for BCP maintenance.
Neuronal TCF4 participated in the bone cancer pain regulation
This research also explored the TCF4 role in pain modulation. For the first step in determining TCF4’s role in bone cancer pain, the TCF4 protein expression level decreased considerably in comparison to the Sham group 12 days after the bone cancer model’s establishment (*p < 0.05, ***p < 0.001 vs. Sham group; n = 4, one-way ANOVA, Figure 3(d)). To examine TCF4 expression in spinal cord tissue, we collected the lumbar spinal tissues and further determined the expression by double immunofluorescence staining and immunofluorescence staining. Subsequently, we analyzed whether TCF4 expression was changed in the spinal cord following carcinoma inoculation. The statistical assessment of TCF4-immunoreactive intensity further verified the decrease of TCF4 expression after BCP surgery (**p < 0.01, ***p < 0.001 vs. the Sham group, one-way ANOVA, Figures 3(e) and (f)). On day 12 after surgery, it was found that tumor cell inoculation dramatically diminished TCF4 expression in the ipsilateral dorsal horn compared to the contralateral side (*p < 0.05 vs. the Sham group, unpaired t-test, Figures 3(g) and (h)).
The current study also examined whether TCF4 displays distinct cellular locations in the spinal cord. As demonstrated in Figure 4, TCF4 was found in the spinal dorsal horn tissue and co-localized with spinal dorsal horn neurons but not astrocytes or microglia. Additionally, to identify whether TCF4 is linked to bone cancer pain regulation, PWT was utilized to assess the intrathecally given over-expression virus impact on behavioral pain. Reagent injection and behavioral assessments were performed, as displayed in Figure 5(a). TCF4 expression and localization in BCP. TCF4 (green), NeuN (red), Iba-1 (red), and GFAP (red) representative double-immunofluorescence images suggesting that TCF4 is co-localized with a neuron in the spinal cord. Arrowheads show double-labeled cells. Scale bar: 100 μm. TCF4 regulates the nociceptive behaviors of BCP rats. (a) The experiment schedule. (b) Western blot analysis of TCF4 dynamic changes following a TCF4 siRNA’s specific inhibitor’s intrathecal injection (F(2,9) = 49.37, ***p< 0.001, versus BCP + scramble siRNA group; by one-way ANOVA, then the Bonferroni test; n=4 rat in every group). (c) Mechanical allodynia for rats receiving A TCF4 overexpression AAV was measured with von Frey tests (F(2,27) = 457.41, *p < 0.05, **p < 0.01 versus BCP + AAV control group using one-way ANOVA, then the Bonferroni test; n=10 rat in every group). (d) Schematic showing experimental strategy for AAV injection. AAV transfection was confirmed by the enhanced green fluorescent protein (EGFP) expression in the spinal dorsal horn. Double-immunofluorescence images (e) and Western blots (f) show efficient TCF4 overexpression in the spinal cord. (f) Following the recombinant AAV-TCF4 intraspinal injection, TCF4 expression was significantly upregulated. (F(2,9) = 10.48,*p< 0.05, **p< 0.01 versus BCP + AAV control group; by one-way ANOVA, then the Bonferroni test; n=4 rat in every group). (g–h) Quantification of behavioral parameters in OF after AAV injection. (*p<0.05 unpaired t-test, vs. the corresponding AAV control group, n = 10).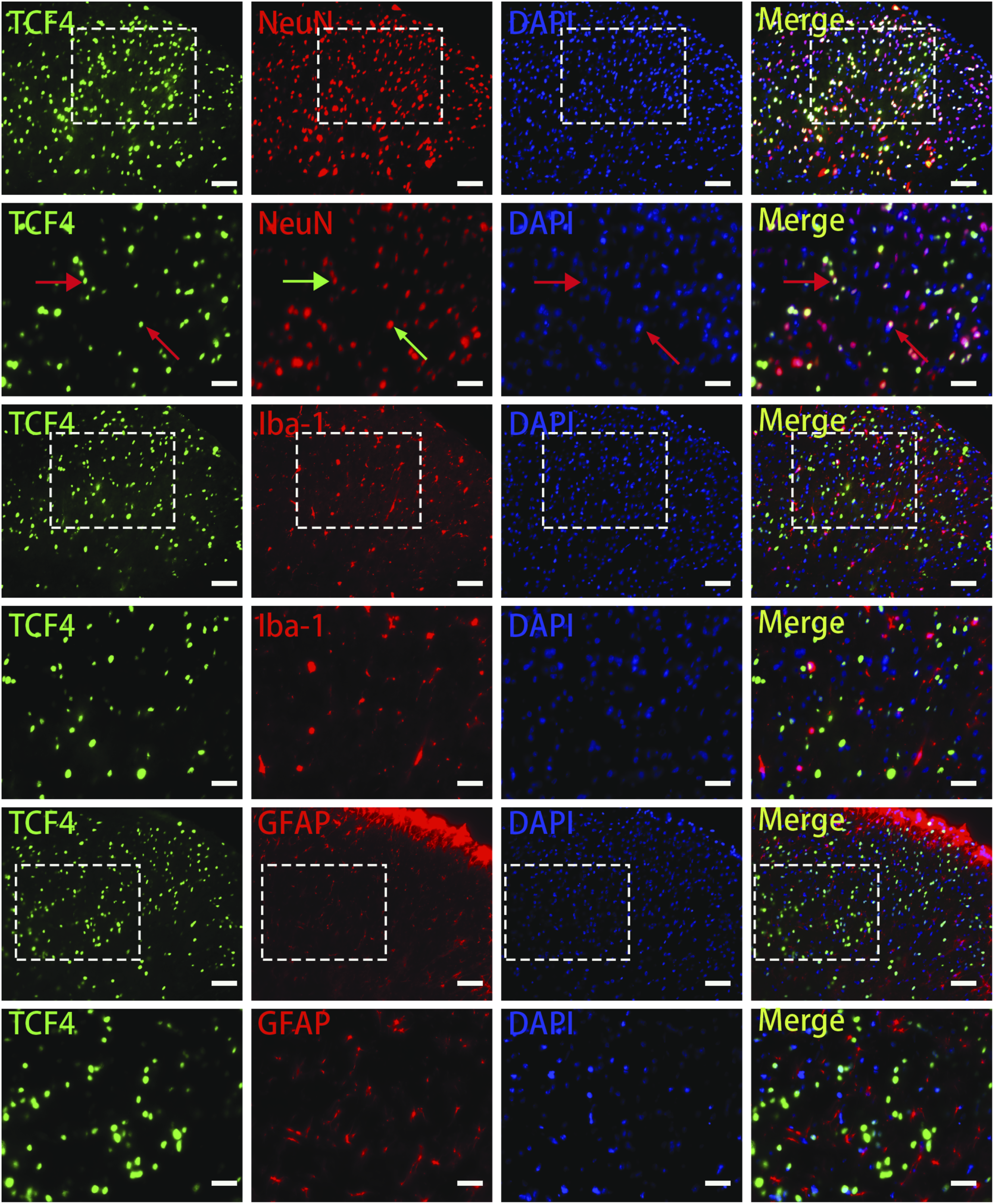
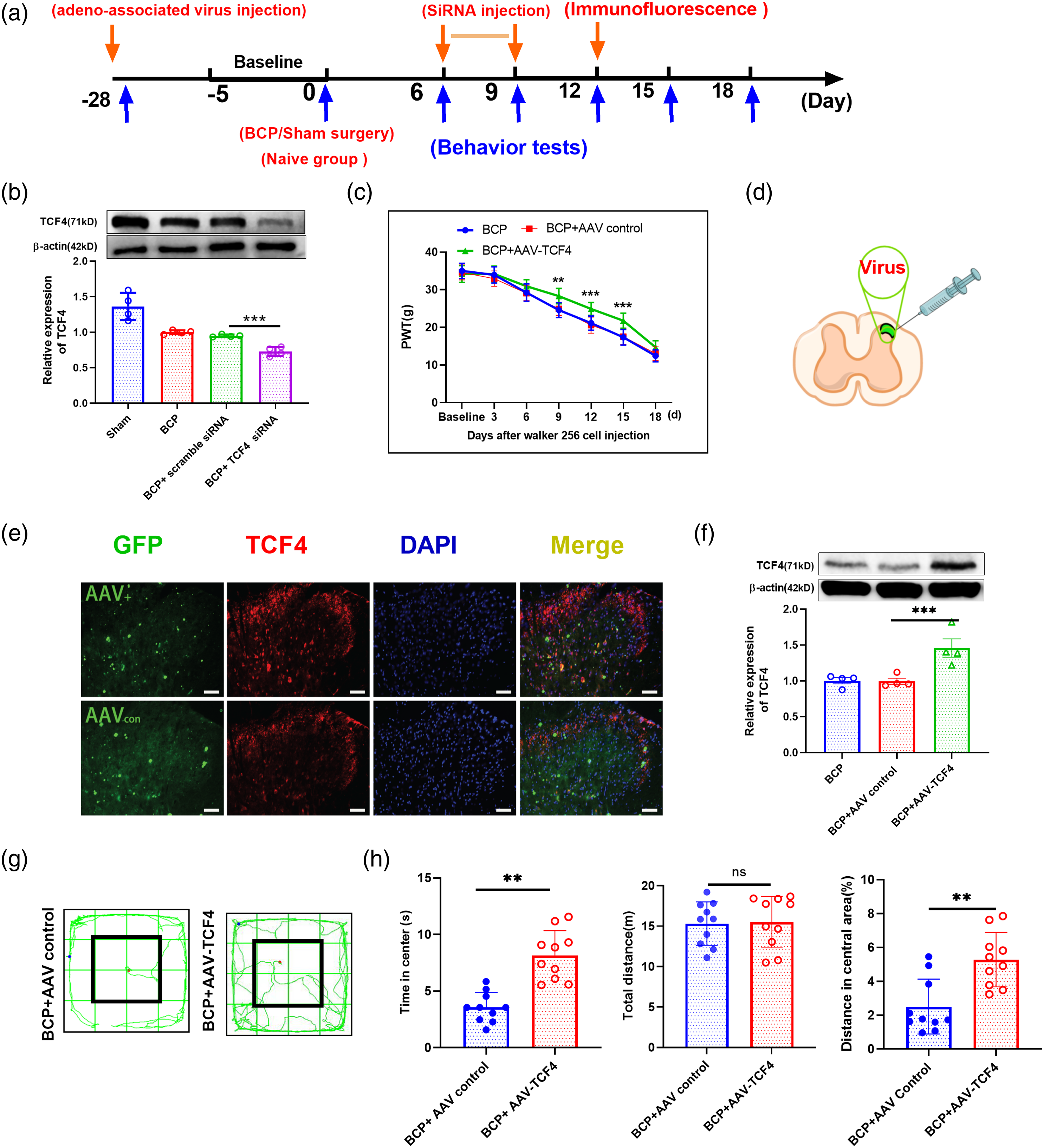
We first determined the TCF4 role in bone cancer pain by TCF4 inhibition by siRNA, a TCF4-specific antagonist. The siRNA treatment brought a lower TCF4 expression level in BCP + TCF4 siRNA group than in the counterpart group (***p < 0.001; BCP + TCF4 siRNA group vs. BCP +scramble siRNA group; one-way ANOVA; n = 4, Figure 5(b)). To address whether spinal neuron-derived TCF4 influences the mechanical allodynia in the current study, we performed adeno-associated virus (AAV) overexpression to mediate the target gene’s expression. With overexpression adeno-associated virus-mediated expression, overexpression of TCF4 with AAV-TCF4 partly reversed the hypersensitivity for mechanical stimulus, after intraspinal injection in comparison to that in AAV control group (*p < 0.05, **p < 0.01, ***p < 0.001; BCP + AAV-TCF4 group vs. BCP + AAV control group; one-way ANOVA; n=10, Figure 5(c)). Following a previous study, the virus works well in many cells like neurons. 33 A schematic demonstration shows the experimental strategy for AAV injection (Figure 5(d)). The efficiency of TCF4 overexpression confined in the spinal cord was confirmed accordingly (Figures 5(e) and (f)). TCF4 was upregulated after the administration of TCF4 overexpression virus in comparison to after AAV control administration. (**p < 0.01, ***p < 0.001; BCP + AAV-TCF4 group vs. BCP + AAV control group; one-way ANOVA; n=4, Figure 5(f)). After AAV injection, anxiety-like behaviors, including in the Open filed test, were reduced (decreased distance traveled in the central area’s percentage and the time spent, Figures 5(g) and (h)). These findings revealed that the spinal TCF4 attenuated bone cancer pain modulation. Overall, these findings stated that decreased spinal neuron TCF4 had a pivotal function in bone cancer pain’s pathogenesis and may regulate spinal neuronal sensitization.
Kv1.1 is regulated by the miR-155-5p/TCF4 pathway in BCP spinal neurons
The mechanisms by which decreased TCF4 facilitates bone cancer-induced nociceptive hypersensitivity was also examined. Consistent with previous reports, TCF4 can regulate Kv1.1 expression,22–24 which works as a mechanical brake in the touch and pain senses. The changes in Kv1.1 were detected in this study. As revealed in Figure 6(a), BCP reduced Kv1.1 expression (***p < 0.001, BCP vs. Sham, n = 4, unpaired t-test). Additionally, we measured the levels of TCF4 in the group infected with overexpression virus. Compared with the control group, BCP-induced reduction was partly reversed (**p < 0.01; BCP + AAV-TCF4 group vs. BCP + AAV control group; one-way ANOVA; n = 4, Figure 6(b)). Further immunofluorescence staining detection showed that the located cell types of TCF4 and Kv1.1 were collocated in the spinal dorsal horn neurons (Figure 6(c)). Kv1.1 expression in the rats’ spinal cord having bone cancer pain. (a) Kv1.1 expression in the rats’ spinal cord with BCP significantly decreased on day 12. (***p<0.001 unpaired t-test, vs. the Sham group, n = 4). (b) Kv1.1 proteins in the spinal cord after the overexpression of AAV-TCF4 microinjection or control in BCP rats. Representative Western immunoblots and the densitometric analysis statistical summary are shown. Per group (F(2,9) = 73.17,**p<0.01; by one-way ANOVA, then the Bonferroni test; n=4 rat in every group, versus the corresponding AAV control group). (c) Immunofluorescence showed that Kv1.1 was localized to the spinal dorsal horn. Immunostaining revealed that Kv1.1 and TCF4 were colocalized with a neuronal marker. Original magnification: ×200; scale bar:100 μm.
Discussion
Pain is the main symptom of osteosarcoma, a global major public health problem.34,35 Although bone cancer pain has been extensively studied for several decades, it remains unclear how hypersensitivities under this condition are caused. Moreover, there are few effective clinical measures to manage pain symptoms due to drug resistance and dependencies. 36 It is necessary to find new targets for the effective treatment development for bone cancer pain.
This research successfully established the bone cancer pain model, as described previously. 28 With the popularity of transcriptome sequencing technology, accumulating research indicated that non-coding RNAs are involved in essential physiological and pathological processes and are functional during diseases. 37 RNA sequencing results showed that miR-155-5p expression performed differential changes between Sham and BCP groups. We tested the pain behavior changes after manipulating spinal miR-155-5p. The miR-155-5p suppression increased the withdrawal threshold and upregulated TCF4 expression in spinal dorsal horn neurons, while miR-155-5p enhancement reduced the withdrawal threshold and downregulated TCF4 expression in naïve rodents. Next, we collected spinal cord tissue and detected miR-155-5p and TCF4 co-location. miR-155-5p and TCF4 are both co-located with neurons. Next, BCP rats were treated with intrathecal injection of TCF4 overexpression virus. We found that TCF4 upregulation prevented bone cancer-induced hypersensitivity. The study findings were the first to reveal that the novel miR-155-5p/TCF4 pathway has a pivotal role in bone cancer pain.
The study results revealed that BCP increased the miR-155-5p expression in the spinal cord neuron is required for bone cancer-induced nociceptive hypersensitivity. TCF4 was suggested to regulate the occurrence of Kv1.1 in spinal cord horn neurons, which has an important role in hyperalgesia regulation in BCP rats. Schematic shows that increased miR-155-5p regulated BCP via interaction with TCF4 and the subsequent Kv1.1 pathway in the spinal neuron (Figure 7). Schematic shows that increased miR-155-5p regulated BCP via interaction with TCF4 and the subsequent Kv1.1 pathway in the spinal neuron.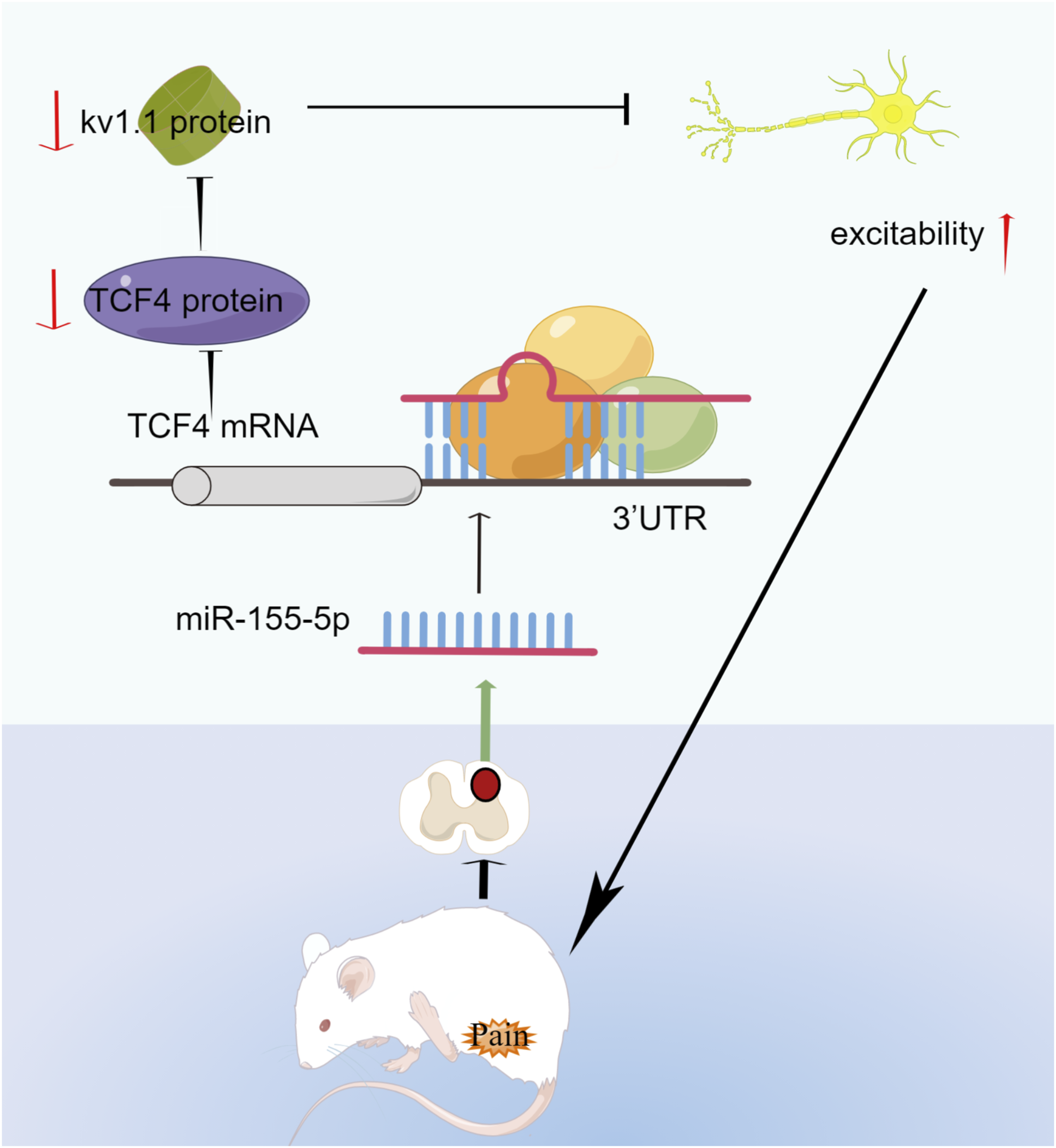
TCF4 affects synaptic plasticity, DNA methylation, and memory. 19 Neurogenesis in adults is facilitated by inhibiting the latent inflammatory potential of neural stem cells through TCF4. 38 TCF4 has been found to regulate the prefrontal neurons' excitability and participate in nociceptive behaviors by regulating Nav1.8 in a recent study.17,20
Considering hyperexcitability is a hallmark of pain, epilepsy, and anxiety disorders, potassium channels may provide a molecular target to influence neuronal excitability. 39,40 Various plasma membrane and intracellular potassium channels could be pharmacologically targeted to treat pain diseases,41–44 such as Piezo2, Nav1.8 and TACAN.45,46 Kv1.1 channel controls the excitability of neuronal cells. The voltage-gated potassium channel Kv1.1 belongs to the Shaker subfamily and regulates mouse hippocampal excitatory neurons to dampen excitability in CNS and PNS. 47 A previous study has demonstrated that potassium channel KCNA1 modulates oncogene-induced senescence and transformation, which restricts tumor development. Moreover, KCNA1 expression declined in human cancers. This decreased expression was related to breast cancer aggressiveness. 48 Taken together, we speculate that Kv1.1 may be a novel pathway for BCP treatment caused by bone metastasis of breast cancer. Nonetheless, because Kv1.1 is expressed in other tissues, potential unwanted side effects should be evaluated.
In conclusion, these findings revealed that increased spinal neuron miR-155-5p has a crucial role in the BCP model’s mechanical allodynia. The major findings are as follows: (1) the persistently increased miR-155-5p expression was induced in bone cancer pain; (2) the spinal miR-155-5p modulation significantly regulates nociceptive pain behaviors; (3) TCF4 was the miR-155-5p target, and (4) miR-155-5p regulated neuropathic pain via modulating TCF4/Kv1.1 expression. These results may suggest a new mechanism for controlling bone cancer pain in the spinal cord.
Footnotes
Acknowledgements
The authors thank Home for Researchers (www.home-for-researchers.com). Drawing of rat image and pathway diagram was performed by Figdraw (![]() ).
).
Authors’ contributions
HDN and XWL conceived and designed the study. QLH performed the experiments. YHW, CFX and MX coordinated and supervised the experiments. LL, JF and BXZ participated in part of the behavioral testing and immunofluorescence staining experiments. CBN performed the data analysis. MY, LL, and JJZ wrote the article. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported, in part, by grants from the National Science Foundation for Young Scientists of China (81901124), the Natural Science Foundation of Zhejiang Province (LQ19H090007, Y20H090134, LGD22H090002, and LQ22H090009), the Medical and Health General Research Program of Zhejiang Province (2020RC122, 2021RC130, and 2022RC076), the Key Medical Subjects Established by Zhejiang Province and Jiaxing City Jointly Pain Medicine (2019-ssttyx), Key Discipline of Anesthesiology of Jiaxing City (2019-zc-06), Science and Technology Project of Jiaxing City (2022AY10017), the Construction Project of Key Laboratory of Nerve and Pain Medicine in Jiaxing City, Key disciplines of pain construction in the city and Zhejiang Province and Interdisciplinary Innovation team for Integrated traditional Chinese and Western Medicine in diagnosis and treatment of senile headache and Vertigo of Zhejiang Province.
Data availability
All data generated or analyzed during this study are included in this published article.
