Abstract
Oxytocin (OT) is recognized as a critical neuropeptide in pain-related disorders. Chronic pain caused by the comorbidity of temporomandibular disorder (TMD) and fibromyalgia syndrome (FMS) is common, but whether OT plays an analgesic role in the comorbidity of TMD and FMS is unknown. Female rats with masseter muscle inflammation combined with 3-day forced swim (FS) stress developed somatic hypersensitivity, which modeled the comorbidity of TMD and FMS. Using this model, the effects of spinal OT administration on mechanical allodynia and thermal hyperalgesia in hindpaws were examined. Furthermore, the protein levels of OT receptors and 5-HT2A receptors in the L4–L5 spinal dorsal horn were analyzed by Western blot. The OT receptor antagonist atosiban and 5-HT2A receptor antagonist ritanserin were intrathecally injected prior to OT injection in the separate groups. Intrathecal injection of 0.125 μg and 0.5 μg OT attenuated the hindpaw hyperalgesia. The expression of OT receptors and 5-HT2A receptors in the L4–L5 spinal dorsal horn significantly increased following intrathecal injection of 0.5 μg OT. Intrathecal administration of either the OT receptor antagonist atosiban or 5-HT2A receptor antagonist ritanserin blocked the analgesic effect of OT. These results suggest that OT may inhibit hindpaw hyperalgesia evoked by orofacial inflammation combined with stress through OT receptors and/or 5-HT2A receptors, thus providing a therapeutic prospect for drugs targeting the OT system and for patients with comorbidity of TMD and FMS.
Introduction
There exists a group of patients who experience a significant level of pain which cannot be classified as a symptom of a disease. These pain syndromes were newly termed chronic primary pain by the International Classification of Diseases-11 (ICD-11).1,2 Chronic primary pain is a chronic relapsing pain condition that has gained recognition in the recent years. The origin of the chronic primary pain is difficult to trace since there is no definitive physical pathology or available laboratory measures for chronic primary pain.1,3 Chronic headache, neck pain, back pain, pelvic pain, irritable bowel syndrome (IBS), fibromyalgia syndrome (FMS), and temporomandibular disorder (TMD) are examples of chronic primary pain. 3 More often than not, multiple chronic pain conditions co-occur in the same patient and are referred to as chronic overlapping pain conditions.
The features of TMD include pain in the temporomandibular joint and/or the masticatory muscles, limitations in mandibular motion, and clicking or crepitation produced by the temporomandibular joint during mandibular function. More than 50% of patients with TMD report pain conditions at sites beyond the masticatory system.4-6 For example, as many as 94% of patients with TMD are commonly diagnosed with FMS.7-9 However, despite its commonality, there are limited treatment strategies for the comorbidity of TMD and FMS in the clinic. 4
Chronic primary pain is related to psychological distress.1,3 For example, in patients with TMD and FMS, there is a close association between exposure to stressful stimuli and exacerbation of pain symptoms.10-12 In fact, both human and animal studies have reported a causative role of stress in the development of pain. A large proportion of FMS patients report experiencing physical or sexual abuse in their childhood. 13 In animals, stress such as forced swim (FS) or cold stress induces somatic hyperalgesia which can last for weeks.14-18 Thus, stress is a major triggering factor for both the generation of hyperalgesia and its aggravation.
One mechanism to account for the comorbidity between TMD and FMS is an imbalance of neurotransmitters causing augmented pain processing in the central nervous system (CNS), which is the chief pathogeny of FMS. 19 The oxytocinergic system is activated under different conditions such as inflammatory sensitization, 20 neuropathic pain,21, 22 or FS stress. Oxytocin (OT) is a nonapeptide synthesized by hypothalamic neurons. OT receptors play a vital role in pain modulation.23, 24 In the CNS, OT receptors can be coupled with both Gq and Gi proteins. 25 The inhibitory effect of OT on the firing properties of sensory wide dynamic range (WDR) neurons in the spinal cord is through OT receptor functionally binding a Gi protein. 20 FS significantly increased the release of OT within both the supraoptic nucleus and the paraventricular nucleus (PVN). 26 In a rat inflammatory pain model induced by carrageenan, spinal OT increased for at least 24 h, and OT receptor activation led to sustained GABAergic activity. 27 Intrathecal administration of OT affected the neuronal activity of Aδ and C fibers in rats, and the analgesic effect caused by spinal administration of OT was blocked by an OT receptor antagonist. 28 Similarly, the analgesic effect induced by endogenous OT was also blocked by an OT receptor antagonist at the spinal level in rats. 20
However, not all OT-provoked actions have been linked to its cognate receptors. OT receptors and 5-hydroxytryptamine 2A (5-HT2A) receptors are expressed in similar brain regions modulating central pathways related to nociceptive behaviors. 29 The participation of OT in modulating the serotonin (5-hydroxytryptamine, 5-HT) system has been demonstrated in multiple studies.30-33 Serotonergic projections innervate almost every brain region, and 5-HT is a key neurotransmitter involved in mood, stress, and social behaviors. 34 Interestingly, OT is known to affect the synthesis and release of 5-HT from 5-HT neurons in the median raphe nuclei, reducing anxiety-like behaviors in mice. 31 In addition, 5-HT receptors have been shown to mediate OT secretion from the PVN of the hypothalamus.35-37 Although OT and 5-HT2A receptors have been suggested to be involved in somatic hyperalgesia, the precise mechanism of OT and 5-HT2A receptor interaction has not yet been fully elucidated.
In our previous study, we established a rat model of TMD comorbid with FMS. Specifically, injection of complete Freund’s adjuvant (CFA) into masseter muscles induced orofacial inflammation in female rats. Then rats with preexisting masseter muscle inflammation that were exposed to 3-day FS stress developed somatic pain hypersensitivity that persisted for 2 weeks. In the model, masseter muscle inflammation was used to mimic the symptoms of TMD, while FS provided the physical and psychological stress.38-40
Using this model in female rats, through testing both mechanical withdrawal threshold and thermal withdrawal latency, the effects of OT on the mechanical allodynia and thermal hyperalgesia were evaluated. Western blot was used to investigate the expression of spinal OT receptors and 5-HT2A receptors post intrathecal injection of OT. The OT receptor antagonist atosiban or 5-HT2A receptor antagonist ritanserin was intrathecally applied before OT injection to confirm the functional role of these two receptors. Generally, this study provides evidence for the therapeutic prospect of drugs targeting the OT system for patients with comorbid TMD and FMS.
Materials and Methods
Animals
Female Sprague–Dawley rats weighing 200–230 g were obtained from Xi’an Jiaotong University Laboratory Animal Center (Xi’an, Shaanxi, China). All animals were housed in a temperature (22°C) controlled environment with a 12 h/12 h alternating light-dark cycle, with lights on at 7:00 a.m. Rats were housed in pairs, and food and water were available ad libitum. Experimental protocols were approved by the Institutional Animal Care and Use Committee of Xi’an Jiaotong University, China and adhered to the guidelines for experimental pain in animals published by the International Association for the Study of Pain.
Drugs
17-β-estradiol (E2; Sigma, St Louis, MO, USA) was dissolved in safflower oil to a concentration of 0.5 mg/mL. CFA (Sigma) was dissolved in saline at a 1:1 ratio. OT (Sigma) was dissolved in 0.9% sterilized saline to a concentration of 50 μg/mL. The OT receptor antagonist atosiban (Sigma) was dissolved in 0.9% sterilized saline to a final concentration of 0.1 μg/10 μL. The 5-HT2A receptor antagonist ritanserin (Sigma) was dissolved in 20% dimethyl sulfoxide (DMSO, MP Biomedicals, Irvine, CA, USA) to a final concentration of 20 μg/10 μL. Intrathecal administration of 20% DMSO has no significant effect on pain behaviors in rats according to our results 41 and other studies. 42
Rat model of TMD comorbid with FMS
Rats were anesthetized with isoflurane mixed with air (5% induction reduced to 3%) and ovariectomized (OVx) by a dorsolateral approach. Ten days after surgery, rats were subcutaneously injected with E2 (50 μg/100 μL), which was repeated at 4-days intervals to mimic the fluctuation of a normal estrous cycle. 39
CFA (150 μL, 1:1 in saline) was injected into the masseter muscles bilaterally. Sub-chronic stress was induced by a 3-day FS paradigm starting 1 day after CFA injection. 39 During the FS, rats were placed in a cylindrical container (30 cm diameter) filled with 20 cm water at 25–26°C for 10 min on the first day and 20 min on the following 2 days.39,43 After exposure to FS, rats were gently dried with a towel and a heater fan. The FS stress was performed at the same time from 9:00 a.m. to noon every day to avoid the influence of circadian rhythm.
Behavioral experiment protocol
The experimental procedures are shown in Figure 1. The behavioral tests were assessed before CFA injection (baseline), after FS (pre-OT), and after OT injection (post-OT application). The dose of OT (0.031, 0.125, and 0.5 μg) was chosen based on previous studies44,45 and our preliminary studies. Since our previous study found that the somatic hyperalgesia was similar from day 2 to day 15 after FS in the comorbidity group,
40
OT was intrathecally injected by lumbar puncture on the 4th, 8th, and 12th days after FS, that is, 4th day for 0.5 μg OT, 8th day for 0.125 μg OT, and 12th day for 0.031 μg OT. Separate cohorts were tested with mechanical and thermal stimuli. The mechanical withdrawal threshold was tested 30 min before injection of OT and 1, 2, 3, 4, 5, 6, and 7 h after the application of OT. The thermal withdrawal latency was tested before and 1, 3, and 5 h after injection of OT (testing took too long to retest hourly). Schematic experimental protocols. All rats were ovariectomized (OVx). After 10 days recovery, E2 was injected subcutaneously every 4 days. The baselines of mechanical withdrawal threshold (M) and thermal withdrawal latency (T) were tested prior to CFA injection. On the 4th, 8th, and 12th days post the last FS, the mechanical withdrawal threshold and thermal withdrawal latency were measured separately for each OT concentration group.
In order to investigate the effect of spinal OT receptors, rats were randomly assigned to the atosiban + OT group or saline + OT group. In the atosiban + OT group, the selective OT receptor antagonist atosiban (0.1 μg/10 μL) was intrathecally injected 30 min prior to injection of 0.5 μg OT.45-47 The control (saline + OT) group received an equal volume of 0.9% saline (10 μL, vehicle of atosiban) before OT injection.
To test the potential role of spinal 5-HT2A receptors in OT-induced analgesia, the effect of intrathecal application of the 5-HT2A receptor antagonist ritanserin was examined. 48 Rats were randomly assigned to the ritanserin + OT group or DMSO + OT group. Rats in the ritanserin + OT group were given 20 μg/10 μL ritanserin 30 min before injection of 0.5 μg OT.49,50 The control (DMSO + OT) group received an equal volume of 20% DMSO (10 μL, vehicle of ritanserin) before OT injection.
Behavioral measurements
Mechanical withdrawal threshold
All behavioral tests were performed in the laboratory at stable temperatures (22 ± 1°C). The mechanical sensitivity was measured via a series of calibrated von Frey filaments according to the up and down method.47,51 In brief, each rat was placed in an individual Plexiglas chamber (20 × 20 × 25 cm) with a metal mesh floor and allowed to acclimate for at least 30 min before testing. Calibrated von Frey filaments with a range of force from 0.41 to 26 g (4–255 mN) were pressed perpendicularly against the left hindpaw plantar surface. The 26 g filament was the cut-off force to avoid excessive tissue injury. 52 If there was no response to the initially selected filament, the next stronger stimulus was selected. If there was a withdrawal response, the next weaker stimulus was tested. After the first positive response, five more stimuli were tested. Including the first positive stimulus, the six responses were used to calculate the withdrawal threshold using the threshold calculation software (JFlashDixon Calcultor, University of Arizona, USA).
Thermal withdrawal latency
Rats were placed in separate clear Plexiglas chambers on an elevated glass surface and allowed to acclimate for 30 min before testing. An infrared (IR) beam was emitted and focused on the plantar surface of the right hindpaw, and the paw withdrawal latency was recorded by the equipment automatically. A 20 s cut-off was set to avoid excessive tissue injury. Three records were collected per animal with an inter-stimulus interval of no less than 5 min. The average of the three trials was determined as the thermal withdrawal latency.
Intrathecal administration
Rats were anesthetized with isoflurane as described above. After shaving the injection area, rats were placed in a prone position with a round tube underneath the abdomen. A disposable 30-gauge needle connected to a 25 μL Luer tip Hamilton syringe was inserted at the intervertebral space between the L4–L5 vertebrae to penetrate the dura. A quick flick of the tail indicated the needle entering the intrathecal space. The drug or vehicle was injected over 1 min and then the needle was withdrawn.
Western blot
Rats were injected with either OT (0.5 μg) or sterile saline on the 4th day after FS stress. Three hours later (the time of maximum efficacy of OT), rats were anesthetized with isoflurane (5%) and decapitated. The spinal cord was removed by pressure ejection with ice-cold saline. 41 The L4–L5 section of the dorsal spinal cord was isolated and stored at −80°C until use.
Tissues were homogenized in RIPA buffer (1% NP-40, 1% sodium deoxycholate, and 0.1% SDS) and protease inhibitor cocktail (Boster, AR1182, Wuhan, China). The homogenates were centrifuged at 10,000 g for 10 min at 4°C, and the supernatants were obtained. Protein concentration in supernatants was measured via the bicinchoninic acid (BCA, Boster, AR0146) method. Protein samples (20 μg) were boiled for denaturing, fractionated 18 μg per lane on 4–12% SDS-PAGE gel (Boster, AR0138) and blotted to PVDF membranes (Millipore, IPVH00010, Darmstadt, Germany). The membranes were immunoblotted using 5% nonfat milk for 2 h and then were incubated with primary antibody directed against OT receptor (1:5000, Abcam, EPR12789, Cambridge, MA, USA), 5-HT2A receptor (1:4000, Bioss, bs-12,049R, Beijing, China), and GAPDH (1:4000, Boster, BA2913) at 4°C overnight, respectively. The membranes were further incubated for 1 h in TBST (10 m
Data analysis
All data are presented as mean ± SEM. Statistical analyses were performed using GraphPad Prism 6 software (GraphPad Software, San Diego, CA, USA). The paired Student’s t-test was used to identify the rats’ hyperalgesia condition, and the unpaired Student’s t-test was used for testing the protein levels of the OT and 5-HT2A receptors. Two-way ANOVA followed by Sidak’s post hoc test was used to examine the differences in the pain behaviors among different OT concentration groups, and for intra-group comparisons. One-way ANOVA followed by Dunnett post hoc test was used for comparing post-application of OT receptor antagonist atosiban/5-HT2A receptor antagonist ritanserin and the analgesic effect of OT on the mechanical withdrawal threshold and thermal withdrawal latency. P < 0.05 was considered statistically significant.
Results
Intrathecal injection of OT has an analgesic effect
Administration of OT reverses mechanical allodynia
Injection of CFA into the masseter muscles combined with 3-day repeated FS stress lowered the hindpaw mechanical withdrawal threshold on the 4th day after the last FS compared with baseline (paired Student’s t-test, p < 0.0001, Figure 2(A)). OT at 0.125 and 0.5 μg inhibited mechanical allodynia (two-way ANOVA, F14,126 = 4.309, p < 0.0001 for interaction; F7, 126 = 12.67, p < 0.0001 for time factor; F2, 18 = 87.09, p < 0.0001 for group factor, Figure 2(B)). The analgesic effect of OT on mechanical stimuli lasted over the period from 2 to 4 h after 0.5 μg OT injection. After application of 0.125 μg OT, the mechanical withdrawal threshold peaked at 4 h. There was no significant difference in the mechanical withdrawal threshold after injection of 0.031 μg OT compared with pre-OT injection (p > 0.05). OT inhibited mechanical allodynia. (A) The mechanical withdrawal threshold significantly decreased following orofacial inflammation combined with FS stress (E2 + CFA + FS) on the 4th day after FS compared with baseline. **** p < 0.0001 vs. baseline. N = 21. (B) OT blocked the decrease in the mechanical withdrawal threshold. Intrathecal administration of OT at 0.125 μg and 0.5 μg, but not 0.031 μg reversed the mechanical allodynia induced by orofacial inflammation combined with FS stress. #, #### p < 0.05, 0.0001 vs. pre-OT with the same dose, respectively. Pre-OT represents the time point 30 min before OT injection. N = 7 in each group.
Administration of OT reverses thermal hyperalgesia
Following orofacial inflammation combined with FS stress, rats displayed thermal hyperalgesia as represented by the significant decrease in thermal withdrawal latency (paired Student’s t-test, p < 0.0001, Figure 3(A)). Both 0.125 μg and 0.5 μg dose, but not of 0.031 μg of OT inhibited the thermal hyperalgesia (two-way ANOVA, F6, 63 = 7.829, p < 0.0001 for interaction; F3, 63 = 53.48, p < 0.0001 for time factor; F2, 21 = 72.69, p < 0.0001 for group factor, Figure 3(B)). After injection of 0.5 μg OT, thermal withdrawal latency significantly increased at 1, 3, and 5 h (p < 0.0001 for 1 and 3 h and p = 0.0212 for 5 h). Administration of 0.125 μg OT induced an analgesic effect at 1 and 3 h compared with pre-OT injection (p = 0.001 and p < 0.0001, respectively). Taken together, these results suggest that OT inhibits both mechanical allodynia and thermal hyperalgesia. OT inhibited thermal hyperalgesia. (A) The thermal withdrawal latency significantly decreased following orofacial inflammation combined with FS stress (E2 + CFA + FS) on the 4th day after FS compared with baseline. **** p < 0.0001 vs. baseline. N = 24. (B) OT blocked the decrease in the thermal withdrawal latency. Intrathecal administration of OT at 0.125 μg and 0.5 μg, but not 0.031 μg reversed the thermal hyperalgesia induced by orofacial inflammation combined with FS stress. #, ###, #### p < 0.05, 0.001, 0.0001 vs. pre-OT with the same dose. Pre-OT represents the time point 30 min before OT injection. N = 8 in each group.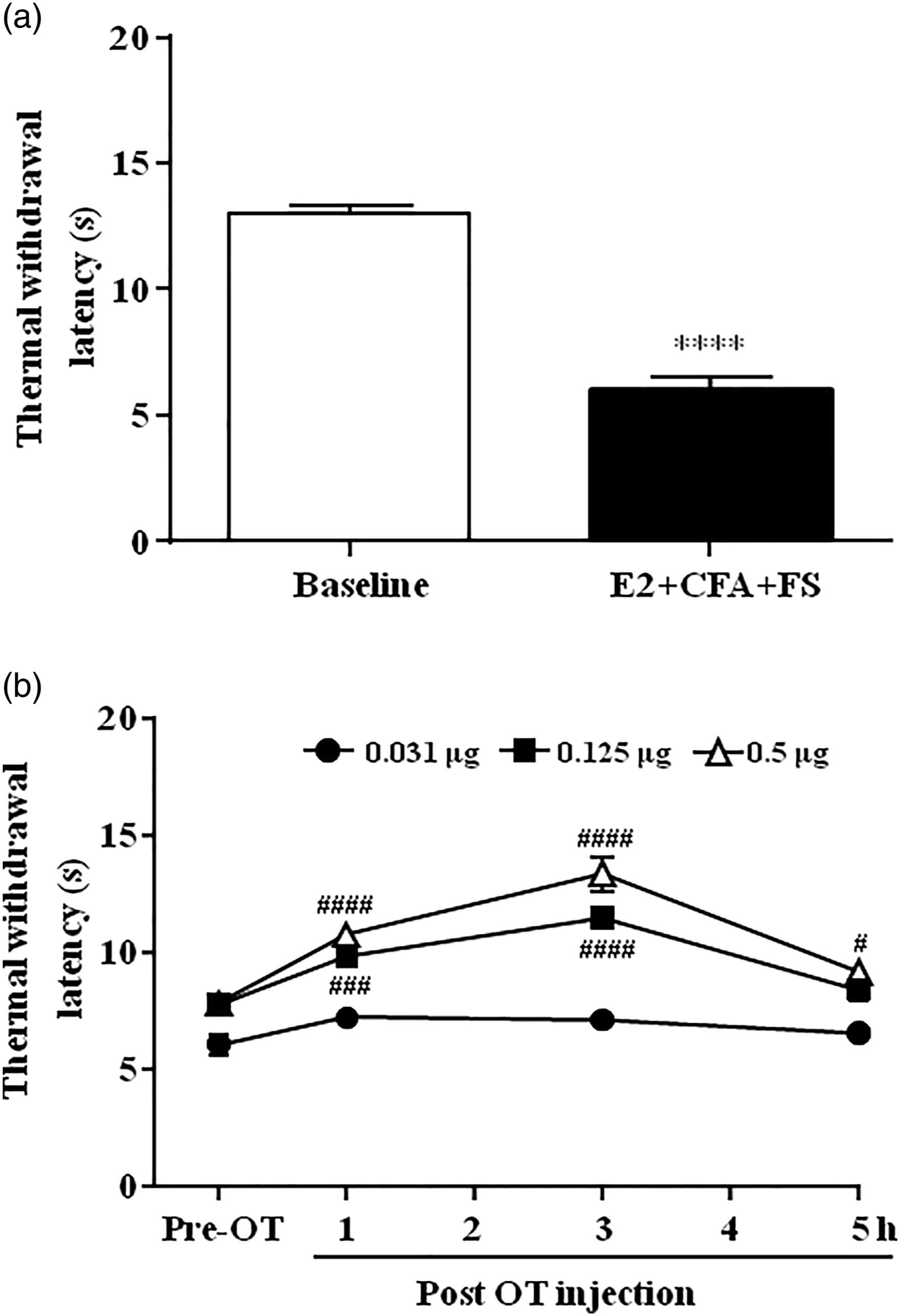
OT increases the expression of OT receptors in the spinal cord
The effects of OT on OT receptor expression were examined 4 days following the end of the stress paradigm. Three hours following OT injection (0.5 μg), the protein level of OT receptors in the L4–L5 spinal dorsal horn was significantly up-regulated post OT application compared with the control (saline) group (unpaired Student’s t-test, p = 0.0016, Figure 4(A)). These data suggest that OT receptors in the spinal cord are involved in the modulation of OT in alleviating hindpaw hyperalgesia induced by orofacial inflammation combined with FS stress. OT receptors contributed to the inhibitory effect of OT on hindpaw hyperalgesia. (A) Intrathecal injection of OT increased the protein level of OT receptors in the L4–L5 spinal dorsal horn. * p < 0.05 vs. saline group. N = 6 for each group. (B and C) Intrathecal pretreatment with the OT receptor antagonist atosiban blocked the anti-nociceptive effect of OT on mechanical allodynia (B) and thermal hyperalgesia (C). ##, ### p < 0.01, 0.001 vs. the Saline + OT group, respectively. N = 8 in each group.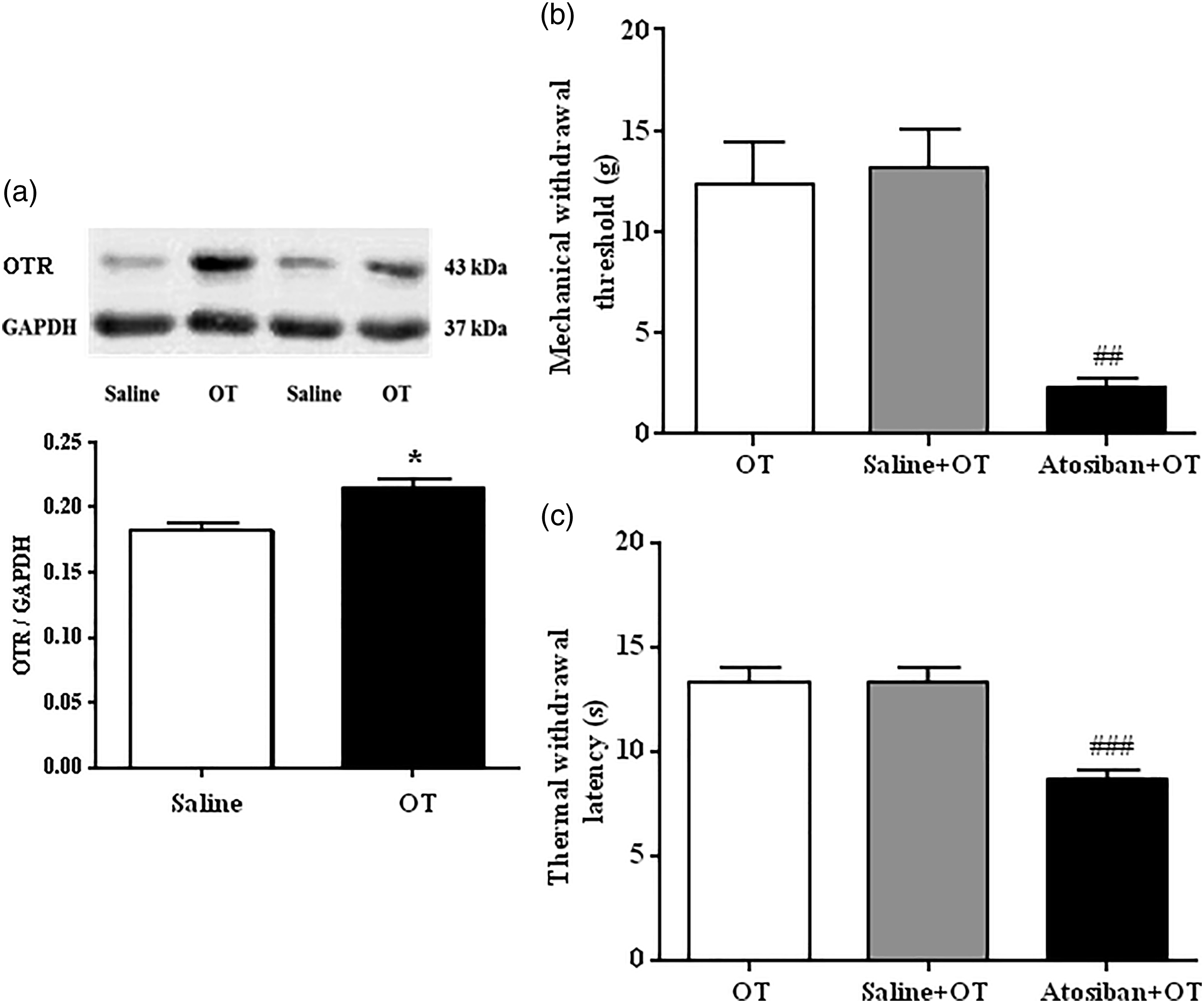
Pretreatment with OT receptor antagonist atosiban blocks the analgesic effect of OT
To further confirm the role of OT receptors in OT-mediated inhibition of hindpaw hyperalgesia induced by orofacial inflammation combined with FS stress, the behavioral responses of rats to mechanical and thermal stimuli were investigated after intrathecal injection of OT receptor antagonist atosiban on the 4th day after FS stress. Intrathecal injection of atosiban (0.1 μg) blocked OT evoked analgesia on the mechanical withdrawal threshold (F2, 21 = 12.45, p = 0.0009, Figure 4(B)) and on the thermal withdrawal latency (F2, 21 = 17.18, p = 0.0001, Figure 4(C)). Saline had no effect. These results indicate that OT alleviates mechanical allodynia and thermal hyperalgesia through activation of OT receptors.
OT increases the expression of 5-HT2A receptors in the spinal cord
Both OT receptors and 5-HT2A receptors are involved in pain and stress related behaviors within brain regions. Therefore, an interaction between OT and 5-HT2A receptors in the L4–L5 spinal dorsal horn was examined. Intrathecal injection of OT (0.5 μg) increased 5-HT2A receptor expression compared with injection of saline (unpaired Student’s t-test, p = 0.0495; Figure 5(A)). Therefore, aside from OT receptors in the spinal cord, 5-HT2A receptors may also be affected by OT, alleviating hindpaw hyperalgesia induced by orofacial inflammation combined with FS stress. 5-HT2A receptors contributed to the inhibitory effect of OT on hindpaw hyperalgesia. (A) Intrathecal injection of OT significantly increased the protein level of 5-HT2A receptors in the L4–L5 spinal dorsal horn. * p < 0.05 vs. saline group. N = 6 for each group. (B and C) Intrathecal pretreatment with the 5-HT2A receptor antagonist ritanserin blocked the anti-nociceptive effect of OT on mechanical allodynia (B) and thermal hyperalgesia (C). ##, ### p < 0.01, 0.001 vs. the DMSO + OT group, respectively. N = 8 in each group.
Pretreatment with 5-HT2A receptor antagonist ritanserin blocks the analgesic effect of OT
Since Western blot data showed that 5-HT2A receptor expression significantly increased in the OT group compared with the control group, the changes of mechanical withdrawal threshold and thermal withdrawal latency were further investigated after intrathecal injection of 5-HT2A receptor antagonist ritanserin. Intrathecal administration of ritanserin (20 μg) reversed the analgesic effect of OT on both the mechanical withdrawal threshold (one-way ANOVA, F2, 21 = 9.313, p = 0.0037, Figure 5(B)) and thermal withdrawal latency (F2, 21 = 18.39, p < 0.0001, Figure 5(C)). Pre-injection of the vehicle (DMSO) had no effect. Both the behavioral and Western blot results suggest that administration of OT inhibits hindpaw hyperalgesia through an up-regulation of 5-HT2A receptors in the spinal cord.
Discussion
In the present study, we found that intrathecal injection of OT blocked hindpaw hyperalgesia induced by orofacial inflammation combined with repeated FS stress in female rats. This extends our previous observations that orofacial inflammation plus stress increases pain in other areas of the body away from the head.39, 40, 54 In the current study, OT increased the expression of both OT receptors and 5-HT2A receptors (but not 5-HT2C receptors, data not shown) in the spinal cord. Moreover, the analgesic function of OT was blocked by intrathecal administration of either the OT receptor antagonist atosiban or the 5-HT2A/2C receptor antagonist ritanserin, which is selective for 5-HT2A and 5-HT2C receptors over other 5-HT receptors. The results are consistent with the findings that intraperitoneal OT significantly increases cardiac OT receptor mRNA expression at 2 h after OT treatment 55 and intranasal OT induces higher OT receptor expression in affected subcortical brain regions than unaffected regions. 56 OT locates in about half of 5-HT neurons in the median raphe nucleus and OT affects the release of 5-HT from 5-HT neurons. 31 Therefore, it is speculated that OT may facilitate the synthesis and accumulation of OT receptors and 5-HT2A receptors in the spinal cord after OT is intrathecally injected. These results indicate that the inhibition of OT in the spinal cord on hindpaw hyperalgesia induced by orofacial inflammation combined with FS stress is not only through the activation of OT receptors, but also through the involvement of 5-HT2A receptors. In the previous study, we found that CFA alone, 3-day FS stress alone and estrogen alone did not induce hindpaw hyperalgesia; therefore, we did not examine the effects of OT on these animals. The estrogen treated rats with 3-day FS stress were also not tested in the present study because the lasting time of hindpaw hyperalgesia occurred in these rats was shorter than that in the estrogen treated rats with orofacial inflammation plus 3-day FS stress. 40
The analgesic effects of OT have been investigated in both the central and peripheral nervous systems in numerous animal pain models.57,58 At the supraspinal level, OT has anti-nociceptive effects if OT is injected into the periaqueductal gray (PAG), 59 raphe magnus, 60 lateral ventricle, 61 or the lateral habenular nucleus. 62 Intracerebroventricular injection of OT significantly attenuated hindpaw incision-induced hyperalgesia; a dose-dependent inhibition of mechanical hypersensitivity was detected 30 min post administration of OT which lasted for 3 h. 63 Administration of OT into the trigeminal ganglion decreased activities in sensitized neurons and inhibited orofacial mechanical allodynia triggered by partial ligation of the infraorbital nerve. 64 The evidence of OT-induced analgesia at both peripheral and supraspinal levels indicates that OT may have a therapeutic effect on neuropathic pain.
When OT was injected into the intrathecal space, neuropathic pain induced by spinal nerve ligation was inhibited in a dose-dependent manner. 65 Moreover, a single intrathecal injection of OT produced significant analgesia for 1 h in rats with neuropathic pain. 44 When OT was continually administered intrathecally for the first 3 days after peripheral nerve injury, the prevention of neuropathic pain lasted for 2 days after OT administration. 44 Intrathecal injection of OT also inhibited inflammatory hyperalgesia induced by carrageenan in rats. 46 These findings indicate an analgesic effect of OT in attenuating neuropathic and inflammatory hyperalgesia.
An important additional consideration is the frequent co-occurrence of overlapping pain conditions and psychological stress. In our animal model, psychological stress in the form of FS is a trigger factor for pain, while orofacial inflammation is critical for the generation and aggravation of TMD symptoms. FMS is characterized by somatic hyperalgesia, a clinical symptom that can be translated into animal models by measuring mechanical withdrawal threshold and thermal withdrawal latency. Therefore, the hindpaw hyperalgesia model induced by orofacial inflammation combined with FS stress utilized in the present study may mimic the comorbidity of TMD and FMS. Both the thermal withdrawal latency and mechanical withdrawal threshold decreased in the hindpaws in these animals. We demonstrated that spinal administration of OT reversed both mechanical allodynia and thermal hyperalgesia. The peak value and duration of analgesia in 0.5 μg OT group were higher and longer than 0.125 μg OT group. But there was no significant difference in the mechanical and thermal thresholds after injection of 0.031 μg OT compared with pre-OT injection. Thus, these data suggest that OT may provide a therapeutic prospect for treating patients with comorbid TMD and FMS. To examine how long the analgesic effects of OT maintain, the mechanical withdrawal threshold was tested hourly and the thermal withdrawal latency was tested at 1, 3, and 5 h after injection of OT. These repetitive stimuli might have adversely impacted on the mechanical/thermal sensitivity and make rats more sensitive to mechanical and thermal tests.
The evidence of OT modulating pain perception in humans is less substantive than that in animals. However, several studies have found that exogenous OT administration reliably reduces pain sensitivity. For example, in a placebo-controlled study, intrathecal injection of OT relieved low back pain. 66 A clinical study on IBS showed that intravenous infusion of OT significantly increased visceral sensitivity thresholds and inhibited colonic pain induced by distention. 67 Furthermore, intranasal OT administration relieved headache, which partly circumvented the poor blood-brain-barrier penetration of OT and increased OT levels in the cerebrospinal fluid.68, 69 Therefore, OT has a potential therapeutic effect on pain perception in humans and may be a strong candidate drug in the treatment of TMD comorbid with FMS.
Further investigation is necessary to ascertain the mechanisms by which spinal OT suppresses somatic hyperalgesia in the TMD/FMS model. In the brain, OT contributes to synaptic plasticity and neuronal integration in response to physiological and environmental stimuli. 70 Axonal projections from OT neurons and OT receptor expression have been shown in several regions which are important for pain processing, such as the spinal cord, amygdala, PAG, and the rostral ventromedial medulla (RVM). 71 The PVN is the primary source of OT in the CNS, and the PVN has an important projection to the spinal cord, which has been demonstrated by retrograde and anterograde tracers. 72 Electrical stimulation of the PVN enhanced OT concentration in both the cerebrospinal fluid and the spinal cord. 21 Thus, the spinal cord, which is exceedingly important in the transmission of nociceptive information, 71 may play a vital role in the regulation of OT on hindpaw hyperalgesia induced by orofacial inflammation combined with FS stress. Previous data showed that OT innervation was particularly robust in the spinal cord around lumbar levels L2 and L6 in rats, monkeys, and humans.27,73,74 Moreover, the density of OT binding sites was higher between L4 and L574. These data provide anatomical evidence for the modulatory effects of OT on the spinal cord neurons.
OT receptors are strongly implicated in pain perception in the spinal cord. In our study, the expression of OT receptors in the OT-treated group significantly increased compared with the vehicle group. This was further confirmed by the behavioral results, with the inhibitory effects of OT on mechanical allodynia and thermal hyperalgesia reversed by intrathecal administration of atosiban. Therefore, the up-regulation of OT receptors may partly account for the inhibitory effect of OT on hindpaw hyperalgesia induced by orofacial inflammation combined with FS stress. Consistent with our findings, a recent study also indicated an analgesic effect of OT in modulating visceral hypersensitivity in an IBS model. Administration of atosiban augmented responses to colorectal distension and reversed the effect of OT. 75 The nociceptive-related neuronal activities of Aδ and C fibers are reduced by spinal application of OT, an effect blocked by atosiban. 28 Exogenous or endogenous OT released through the PVN also leads to the inhibition of WDR neurons in response to Aδ and C fiber stimulation. 28 Therefore, activation of OT receptors can inhibit nociceptive processing evoked by Aδ and C fibers.
OT neurons are modulated by serotonergic neurons. Central administration of serotonin receptor agonists stimulates OT release from the pituitary.37, 76 OT neurons in the hypothalamus receive projections containing serotonin transporters. 77 The present study demonstrated that the expression of spinal 5-HT2A receptors increased after intrathecal administration of OT, and the analgesic effect of OT was blocked by a 5-HT2A receptor antagonist. Collectively, these findings reveal a novel functional relationship between OT and the serotonergic system in nociceptive processing at the spinal level. The formation of an OT-5-HT2A receptor heterocomplex has been observed in cortical and subcortical limbic regions, including the hippocampus, cingulate cortex, and nucleus accumbens. This heterocomplex formation may have a special role in distinct limbic circuits related to social salience and memory. 29 The question as to whether such a heterocomplex formation exists in the spinal cord and is involved in an analgesic effect warrants further investigation.
Several studies have shown that 5-HT2A receptors exert inhibitory effects on pain processing. 78 5-HT2A receptor agonists inhibit inflammatory pain 79 and neuropathic pain, 80 suggesting that 5-HT2A receptors are involved in 5-HT descending pain modulation system. The pathway involving 5-HT2A receptors and the potassium-chloride cotransporter (KCC2) in the spinal cord attenuates pain. KCC2 is responsible for maintaining a low Cl− concentration and decreasing the activity of neurons in the CNS. Both the physiological and pathophysiological impacts of KCC2 have emerged as potential key underlying mechanisms in chronic pain. 81 In the rat incision pain model, intrathecal injection of the 5-HT2A receptor agonist, methylamine hydrobromide (TCB-2), increased pain thresholds and inhibited the incision-induced decrease in transcription and translation of KCC2, while the delivery of the 5-HT2A receptor antagonist ketanserin aggravated the hyperalgesia and decreased transcription and translation of KCC2. 78 Similarly, administration of TCB-2 increased membrane KCC2 expression in the spinal dorsal horn and reduced the mechanical and thermal hyperalgesia in rats with spinal cord injury. 82
Parvocellular OT neurons located in the PVN synthesize OT and project extensively toward the brain stem and the spinal cord. 83 These projections are strongly related to pain perception. 20 In fact, OT administered by different methods has been shown to have an anti-nociceptive effect which is mediated by the activation of descending anti-nociceptive pathways to the spinal cord.21,84,85 The descending pain inhibitory system is an endogenous analgesic system mainly including the PAG, RVM, and the dorsal horn of the spinal cord. 86 Electrophysiological, pharmacological, and behavioral data have shown that the paraventricular hypothalamic descending OT pathway participates in anti-nociceptive processes.84,85
In the hypothalamo-spinal projection, parvocellular OT neurons in the PVN directly project to the WDR neurons in the deep layers of the spinal cord in rats. 20 Both endogenous OT released from the PVN and exogenous OT administrated into the spinal level prevented and reduced long-term potentiation in spinal WDR neurons. 87 In addition, after lesioning of the PVN, 5-HT2A receptors were activated, which mediated the release of OT. 88 Selective stimulation of 5-HT2A receptors in the PVN also increased OT concentrations. 89 Therefore, the activation of 5-HT2A receptors in the PVN may increase the endogenous secretion of OT. Moreover, when OT is released into the spinal cord, the 5-HT2A receptor-KCC2 pathway may also participate in analgesic processes.
The PAG and the RVM are closely associated areas noted for their function in the descending pain regulatory pathways. 90 Injection of OT into the PAG had an analgesic function and administration of an OT receptor antagonist blocked this effect. 91 In addition, pain stimulation elevated endogenous OT levels in the PAG, indicating that the PAG is a promising target for pain regulation through OT. 91 OT may have anti-nociceptive role on the spinal cord neurons through PAG-RVM-spinal cord pathway. 53 Thus, besides the direct analgesic effect of OT on the spinal cord circuitry, orofacial inflammation combined with FS stress may activate the PVN and increase the endogenous release of OT, which in turn inhibits pain perception through the PAG-RVM–spinal cord descending pathway and the direct hypothalamo-spinal projection. Further studies are needed to confirm the effects of endogenous OT.
Conclusion
The results of the present study indicate a therapeutic effect of OT in hindpaw hyperalgesia induced by orofacial inflammation combined with FS stress at the spinal level. Both OT receptors and 5-HT2A receptors are involved in OT’s alleviation of hindpaw hyperalgesia in female rats. Our findings provide a key insight that OT may be used as a novel therapy for the treatment of comorbid TMD and FMS.
Footnotes
Authorship Contribution
DYC designed the research; YXL, JHL, YG, ZYT, and SHQ performed the experiments; YXL, RJT, HA, and DYC analyzed the data and wrote the paper. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (81971049, 81671097) and the Social Development Program of Shaanxi Province, China (2020SF-018) to DYC, and partially by the National Institutes of Health, USA (R01 DE029074) to RJT.
