Abstract
Post-COVID syndrome (PCS) may manifest as chronic pain and fibromyalgia-like symptoms, but the specific risk factors contributing to these outcomes remain poorly understood. We conducted a prospective cohort study using data from the ongoing “Post-Acute Sequelae of SARS-CoV-2 Infection (PASC)” study to identify predictors of chronic pain and fibromyalgia-related phenotypes among previously hospitalized COVID-19 patients. Participants were adults (n = 756, mean age 55, 47% female) hospitalized between March and August 2020 at the University of São Paulo Medical School. Clinical and demographic data from hospitalization, along with psychological, functional, and cognitive assessments at 3–11 months post-discharge (phase 1), were used to predict chronic pain, chronic widespread pain (CWP), fibromyalgia-like symptoms (FLS), and probable fibromyalgia (FM) at a 2–3 year follow-up (phase 2). Multivariate logistic regression analyses revealed that higher anxiety/depression severity (OR 1.35; 95% CI 1.09–1.69; p = 0.008) and reduced left handgrip strength (OR 0.95; 95% CI 0.92–0.98; p < 0.001) at phase 1 significantly predicted chronic pain at phase 2. For fibromyalgia-related phenotypes, greater left-side pain, insomnia severity, and weaker left handgrip strength were consistently associated with increased risk across all outcomes (CWP, FLS, FM), with statistically significant odds ratios ranging from 1.07 to 1.44. These findings highlight key modifiable risk factors – particularly psychological distress, sleep disruption, and muscle weakness – that may contribute to the development of chronic pain and fibromyalgia symptoms following COVID-19 hospitalization. Early identification and targeted intervention on these domains could improve long-term outcomes in PCS populations.
Introduction
With COVID-19 now classified as an endemic disease, it is critical to understand the post-acute sequelae of SARS CoV-2 infection, referred to as post-COVID syndrome (PCS). PCS – which has been reported in 10%–35% of patients and up to 85% of hospitalized patients – is often characterized by an unexplained persistence or emergence of new symptoms lasting over 12 weeks after infection. 1 Cornerstone features of PCS include fatigue (28%–85% prevalence), memory or concentration difficulties (24%), joint or muscle pain (45%–75%), sleep disturbances (20%), and depression (18%),2–5 with higher rates of these symptoms observed in patients with more severe COVID-19. 6 These symptoms bear a strong resemblance to the defining characteristics of chronic pain and fibromyalgia syndrome (FM).
Chronic pain is defined as any pain lasting more than 3 months, and chronic widespread pain (CWP) is a known symptom of FM. 7 The overlap between PCS, chronic pain, and FM symptoms suggests some degree of interrelation between these disorders. In fact, patient surveys have revealed that 63.3% of post-acute COVID-19 patients experience chronic pain, and up to 30% have met modified 2011 American College of Rheumatology (ACR) criteria for FM.8–10 However, the exact nature of these relationships remains largely unknown.
Given that infections such as the Epstein-Barr virus have already been linked to FM onset, it is plausible that abnormal inflammatory pathways altered by COVID-19 infection may induce CNS hypersensitivity and, consequently, FM symptoms.11–13 Furthermore, the COVID-19 pandemic cultivated many negative environmental factors – such as mental stress and inactivity due to quarantine or hospital admission – that may trigger chronic pain and FM following COVID-19 infection.14,15 No existing studies have connected various facets of behavioral, psychological, and clinical stressors related to COVID-19 infection to the subsequent development of chronic pain or fibromyalgia-like symptoms (FLS).
A limited number of cross-sectional studies on probable post-COVID FM have yielded inconsistent results; Akel et al. 16 and Savin et al. 17 identified female gender as a strong predictor, whereas Ursini et al. 9 found male gender to be a significant predictor. Studies examining post-COVID chronic pain have revealed female sex, body mass index (BMI), COVID symptom load, pre-existing pain symptoms, and days hospitalized as risk factors.18–22 These studies were primarily conducted through web-based questionnaires or phone interviews, which may limit reliability of results and involve self-selection of study participants. Furthermore, longitudinal relationships between post-COVID recovery and FM/FLS onset are not well characterized.
To address these existing gaps, this prospective cohort analysis aims to (1) characterize long-term chronic pain, CWP, FLS, and probable FM in a sample of post-hospitalized COVID-19 patients at two time points and (2) analyze potential sociodemographic, clinical, behavioral, and psychological risk/protective factors on the no-pain to probable FM continuum. This study will contribute to ongoing investigations of COVID-19 as a potential trigger for chronic pain and FM, providing new insights for post-COVID symptom management and risk-stratified treatment approaches.
Methods
Study design
This analysis consists of post-hospitalized COVID-19 survivors from the ongoing “Post-acute sequelae of SARS-CoV-2 infection (PASC)” study conducted at the General Hospital of the University of Sao Paulo Medical School. Eligible participants were 18 or older and hospitalized with confirmed COVID-19 infection for over 24 h between March and August 2020. All patients discharged during this period were invited to participate in this study. Phase 1 evaluation occurred 3–11 months after discharge, and phase 2 follow-up assessments took place 2–3 years after phase 1. Data collection was performed by a multidisciplinary team of clinical professionals via in-person assessments and teleconsultation questionnaires. Sociodemographic and clinical information, as well as a wide range of psychological, functional, physiological, and cognitive measures, were collected to provide a comprehensive patient health assessment. In this prospective cohort analysis, these measures were used to identify risk factors at phase 1 that are associated with the presence of post-COVID chronic pain, CWP, FLS, or probable FM in phase 2. The institutional review board of the Hospital das Clínicas, Faculdade de Medicina, Universidade de São Paulo gave ethics approval for all protocol components for this study (approval numbers: 4.270.242, 4.502.334, 4.524.031, 4.302.745, and 4.391.560). Informed written consent were obtained from participants (or their legal guardians) prior to study procedures.
Experimental measures
Exposure measures included the Insomnia Severity Index, handgrip strength, WHO Disability Assessment Schedule (WHODAS – functioning across six domains), EQ-5D questionnaire (mobility, self-care, usual activities, pain/discomfort, and anxiety/depression, self-reported health status VAS), and the Post-COVID Functional Status scale (post-COVID symptoms and functional limitations).23,24 PCFS and EQ-5D scores, as ordinal categorizations, were treated as continuous variables to align with other measures of patient function.
Select measures were used to classify chronic pain and FM/FLS, as explained below and in eTable 1.
Classification criteria
Chronic pain
Chronic pain was classified based on the presence of any pain sites and associated visual analogue scale (VAS) scores (mild to severe) throughout the body, with the exception of pain sites associated with a previous injury unrelated to COVID-19 infection.
Chronic widespread pain
CWP was defined as pain present in at least four of the five Widespread Pain Regions – left upper, right upper, left lower, right lower, or axial – as described by the International Association for the Study of Pain (IASP). 14
Fibromyalgia
To capture the wide range of FM diagnostic standards, two distinct sets of classification criteria were used to sort patients into having either no FM/FLS or probable FM/FLS in Phase 2. The presence of FM/FLS was stored separately as a binary variable for each definition.
Patient characteristics at phase 1.

Numbers may not sum to totals due to the exclusion of missing data.
n (%); Mean (SD).
Wilcoxon rank sum test for numerical variables; Fisher’s exact test for categorical variables.
We assumed that the duration of symptoms reported by patients, as measured in assessments, reflects a sustained period of discomfort. Additionally, comorbid disorders at phase 2 were disregarded as a diagnosis of FM is valid irrespective of other diagnoses.
The FACIT-F scale, which ranges from 0 to 52, was used to characterize fatigue level, with lower scores indicating more severe fatigue. Scores under 43 are considered clinically relevant, and existing literature supports using scores below 30 to define a severe fatigue population. 26 To account for missing data, cognitive status was determined by both the Patient Memory Complaints Scale and total score from the Mini-Mental State Examination, with the more severe of the two used in the final diagnostic criteria. The EPWORTH Sleepiness Scale was used as a proxy for waking unrefreshed, a common symptom of FM. Depression and anxiety were determined using the Hospital Anxiety and Depression Scale. Headache and abdominal pain were determined through recorded pain site locations. Fatigue, cognitive function, and daytime sleepiness scores were re-coded into a four-point scale, ranging from 0 (no symptoms) to 3 (severe symptoms) based on standard assessment interpretations. Depression, anxiety, headache, and abdominal pain were converted to binary variables for the SSS calculation.
Study sample
A flow diagram of study participants is shown in Figure 1. Participants with no pain-related assessments at phase 2 (n = 415) were excluded from the sample of 756 COVID-19 survivors. Chronic pain status at phase 2 was classified for the remaining 341 participants, representing both pain persistence and development.

Flow diagram of study sample.
For the CWP/FM/FLS portion of the study, patients with any rheumatological disease at Phase 1 were excluded from both chronic pain and non-chronic pain groups at phase 2 (n = 10 and n = 5, respectively) due to the high likelihood that they were already taking measures to manage their condition. Patients with CWP at phase 1 were removed in CWP comparisons, and patients with FLS at phase 1 were removed from FM/FLS comparisons because many measures relevant to FM classification were not available at phase 1. Classifications of CWP, FLS, and FM were not mutually exclusive. These classifications formed a continuum between patients with chronic pain, CWP, FLS, and finally probable FM.
Statistical analysis
Phase 1 demographics, as well as a range of clinical and functional characteristics, were characterized for the sample of participants. Four comparisons were made based on phase 2 outcome classifications: (1) chronic pain versus no chronic pain (2) CWP versus no CWP (n = 308) (3) FM versus no FM (n = 318) and (4) FLS versus no FLS (n = 318). Comparisons of relative frequencies or means between these groups were conducted using Fisher’s exact test and Wilcoxon rank sum test, respectively. The specific test used is mentioned in the footnote of Table 1.
Bivariate and adjusted multivariate logistic regressions were used to assess predictive relationships between various measures and post-COVID outcome at phase 2. Sex, age at hospital admission, and resting heart rate – an indicator of general patient health – were established as fixed covariates in these analyses. Missing data were removed in individual analyses. Results are presented as odds ratios with 95% confidence intervals, and p values <0.05 were considered statistically significant. All statistical analyses were conducted using Python and R.
Results
Baseline results
Clinical characteristics of this sample were detailed in a prior analysis by Battistella et al. 27 The mean age at hospital admission was 55 years and the average length of hospital stay was 18 days. Of the 801 post-hospitalized COVID-19 patients initially included in the sample, 62% (497) of the patients were admitted to intensive care. During their hospitalization, 41.6% (333) were intubated. 27 Linear regression models indicated that intubation status – which can be regarded as a measure of COVID-19 severity – had no significant impact on VAS pain scores in phase 1. Furthermore, participants’ pain VAS severity classifications did not show a significant difference when grouped by the time between hospital discharge and phase 1 assessment.
Chronic pain
Chronic pain was identified in 70.1% of (530 of 756) patients at phase 1, an average of 6.8 months post-hospitalization. Females made up 51% of chronic pain patients, as compared to 38% of the patients with no chronic pain (p = 0.002). Patients with chronic pain at phase 1 had more moderate to severe problems across all five dimensions of the EQ-5D assessment (mobility, self-care, usual activities, pain/discomfort; p < 0.001; Table 1). They also had more problems with sleepiness and insomnia (EPWORTH: 9.7 vs 8.2, p = 0.001; ISI: 8 vs 5, p < 0.001), weaker handgrip strength (R: 19 vs 24, p < 0.001; L: 17 vs 22, p < 0.001), worse WHODAS performance (p < 0.001), and moderate to severe post-COVID functional limitations (p < 0.001). Similar associations were found in patients with chronic pain at phase 2 (an average of 2.7 years follow up from phase 1), with the addition of higher BMI (33 vs 31, p = 0.029) and higher systolic blood pressure (136 vs 136, p = 0.012; eTable 4).
Of the 530 patients with chronic pain at phase 1, 47% (117) had chronic pain at phase 2, compared to 33% of 226 in the no pain group (p = 0.029; Table 1). Chronic pain was classified in 43% (147 of 341) of patients at phase 2. Pain sites at phase 2 appeared to be more diffuse compared to phase 1, although most commonly reported pain sites remained to be the right knee (118 phase 1 counts, 51 phase 2 counts), left knee (106 phase 1 counts, 48 phase 2 counts), and lower back (90 phase 1 counts, 33 phase 2 counts; eFigure 1).
Both bivariate and adjusted multivariate analyses revealed many associations between exposure measures and chronic pain development (Table 2, eTable 3). To further examine these relationships, adjusted multivariate logistic regression models including a combination of exposure measures showed that severity of anxiety/depression (Odds Ratio (OR) 1.35; 95% Confidence Interval (CI) 1.09, 1.69; p = 0.008) and weaker left handgrip strength (OR 0.95; 95% CI 0.92, .98; p = 0.00) at phase 1 are statistically significant predictors for the presence of chronic pain at phase 2 (Table 3). It is important to mention that both weaker right and left handgrip strength were associated with the presence of chronic pain at phase 2, as presented in Table 2. However, we included only left handgrip strength because it improved the goodness-of-fit of the final model. Therefore, handedness dominance was not an important factor in these results. Pain severity did not appear to have a significant effect.
Bivariate analyses by chronic pain, CWP, FLS, and FM at phase 2.

CI: confidence interval; OR: odds ratio. Boldface values represent statistically significant results.
Adjusted multivariate logistic regression models for the presence of chronic pain at phase 2.
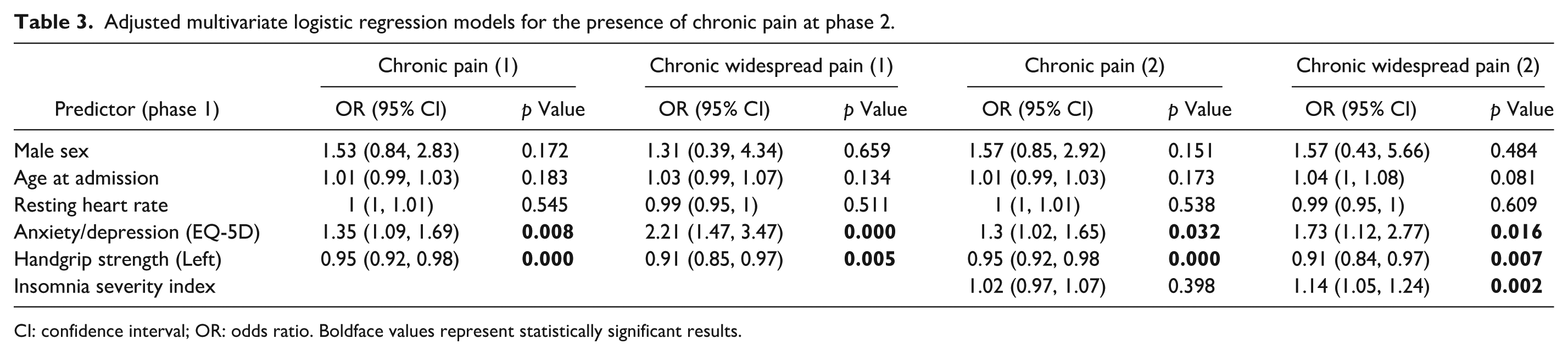
CI: confidence interval; OR: odds ratio. Boldface values represent statistically significant results.
Chronic widespread pain, fibromyalgia-like symptoms, and fibromyalgia
CWP was found in 5% (42 of 756) of patients at phase 1 and 7.7% (24 of 308) of patients in phase 2. FLS were found in 12% (38 of 318) of patients in phase 2. Further, 4.4% (14 of 318) of patients in phase 2 were classified as having probable FM. Phase 1 characteristic comparisons based on the separate classifications at phase 2 are summarized in eTable 2. Results of bivariate and multivariate analyses are displayed in Table 2 and eTable 3.
Similar associations were observed when using CWP as the outcome variable in the chronic pain model. However, including insomnia as an exposure measure for CWP enhanced the model’s performance but slightly attenuated the effect size of anxiety/depression (Anxiety/Depression: OR 1.73; 95% CI 1.12, 2.77; p = 0.016. Handgrip Strength (Left): OR 0.91; 0.84, 0.97; Insomnia Severity Index: OR 1.14; 1.05, 1.24; p = 0.002; Table 3). On the other hand, insomnia was not a significant predictor for chronic pain in general.
In adjusted multivariate models for the presence of CWP/FLS/FM at phase 2 with a combination of exposures, more severe left side pain (CWP: OR 1.21; 95% CI 1.01, 1.5; p = 0.049. FLS: OR 1.16; 1.01, 1.33; p = 0.037. FM: OR 1.44; 1.11, 2.04; p = 0.016), insomnia (CWP: OR 1.17; 1.08, 1.27; p = 0.000. FLS: OR 1.07; 1.01, 1.15; p = 0.026. FM: OR 1.12; 1.02, 1.24; p = 0.022), and weaker left side handgrip strength (CWP: OR 0.91; 0.84, 0.97; p = 0.010. FLS: OR 0.93; 0.88, 0.98; p = 0.005. FM: OR 0.92; 0.83, 0.99; p = 0.042) at phase 1 were statistically significant predictors (Table 4). Similarly to chronic pain, both weaker right and left handgrip strength were associated with the presence of CWP/FLS/FM at phase 2, as presented in Table 2. However, we included only left handgrip strength because it improved the goodness-of-fit of the final model. Therefore, handedness dominance was not an important factor in these results. Neither the EQ-5D measurement of pain/discomfort nor the post-COVID functional status presented a significant effect.
Adjusted multivariate logistic regression model for CWP/FM/FLS at phase 2.
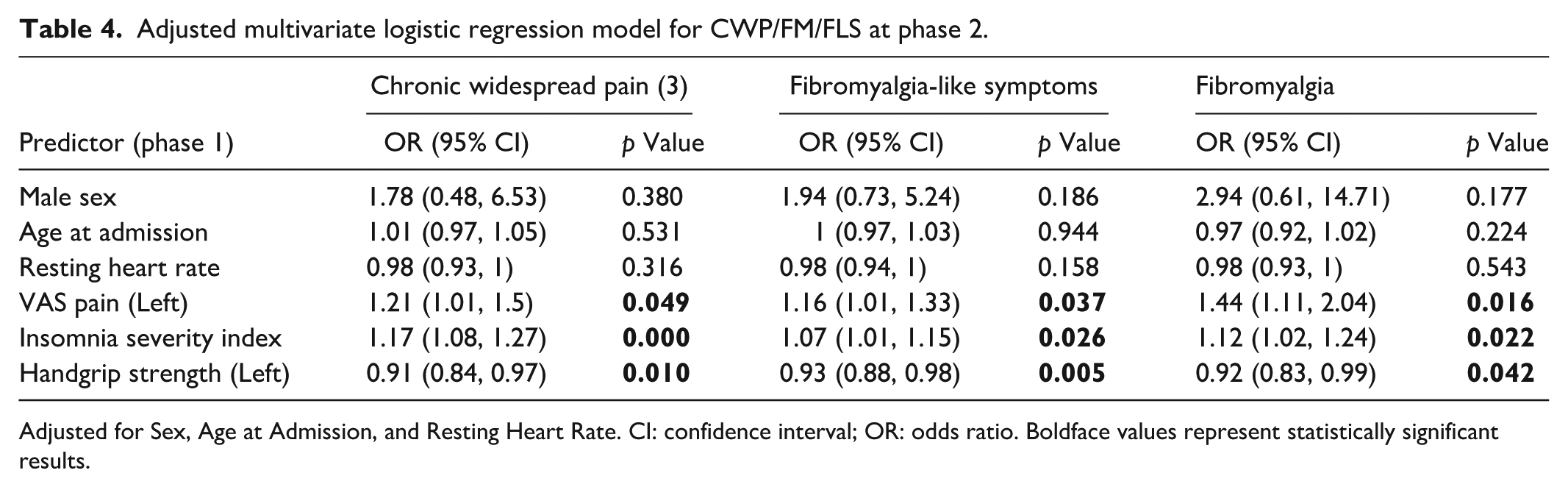
Adjusted for Sex, Age at Admission, and Resting Heart Rate. CI: confidence interval; OR: odds ratio. Boldface values represent statistically significant results.
Interestingly, between phase 1 and phase 2, pain severity improved in patients with no previous pain, which was not observed in patients with chronic pain at phase 2 (eTable 5). Handgrip strength improved in both no pain and chronic pain groups, whereas ISI scores increased in severity in both groups.
Discussion
In this study, we tracked relevant exposure measures extending many years post-hospitalization to predict symptoms resembling chronic pain, CWP, FLS, and FM in a sample of patients from Brazil – a country significantly impacted by COVID-19. A key strength of our analysis is the 2.7-year follow-up, which allows for sufficient time for chronic pain and fibromyalgia-related networks to develop. We identified weaker handgrip strength and more severe anxiety/depression to be risk factors for long-term chronic pain as well as chronic widespread pain. However, moving toward the FM state, sleep-related factors became more significant than anxiety or depression.
Pain severity and insomnia scores did not improve from phase 1 in patients with chronic pain at phase 2, suggesting potential chronification rather than recovery. Accordingly, 80% of the patients with chronic pain at phase 2 had chronic pain at phase 1. The prevalence of chronic pain at phase 1 (70.1%) and phase 2 (43%) is comparable to findings in similar studies of hospitalized patients with PCS and approximately double that of the general population (20.9%), potentially pointing to the lasting effects of COVID-19 infection. 28
Unlike other studies, FLS was evaluated on a continuum ranging from chronic pain to probable FM. Comparisons revealed the differential roles of anxiety/depression and insomnia for general chronic pain and chronic pain resembling FM. Our study expands on previous research by highlighting anxiety, depression, and weaker handgrip strength as significant long-term predictors for general chronic pain. These results are supported in non-COVID samples, where anxiety/depression have been associated with pain chronification.28–30
While the intensity of pain at phase 1 was not linked to general chronic pain at phase 2, it was significantly associated with CWP/FM/FLS at phase 2. This suggests that factors underlying pain intensity in the acute phase of COVID-19 recovery may play a role in triggering characteristic symptoms of a fibromyalgia-like state. The significant effect of more intense pain, weaker handgrip strength, and insomnia as predictors of FM supports results found in non-COVID samples.31–33 The lack of an effect of anxiety/depression for FLS may indicate that psychological effects may be more of consequence of experiencing FLS rather than a causative variable. Surprisingly, potential markers of inflammation such as BMI did not survive in our final models for chronic pain/FLS, despite being implicated as risk factors in other studies. 18 An explanation for this result could be that pain-related PCS could arise from an interaction between inflammation and the identified risk factors of insomnia, and muscle weakness. Inflammation may lead to a more vulnerable neurological state and exacerbate the impact of these factors on triggering fibromyalgia-like symptoms. Other potential physiological pathways to explain the lack of association are changes in neuropeptides expression (e.g. CGRP blood levels), immune dysregulation (unbalanced lymphocyte populations and autoimmune reactions), or central sensitization (altered conditioned pain modulation test), which could account for these differential changes in pain sensitivity following COVID-19 hospitalization. Future studies should explore these biomarkers as potential mechanistic variables for post-COVID pain syndromes. Moreover, our findings further underscore the importance of controllable physical factors – such as strength training and sleep improvement interventions – in a post-COVID context.
Several mechanisms could explain the relationship between these post-COVID predictive variables and associated long-term pain syndromes. Handgrip strength is often considered a marker of muscle weakness and poor overall health status. 34 Performance may reflect the extent of COVID-19 infections’ impact in deteriorating muscle and joint strength throughout the body. Anxiety or depression, along with poor sleep quality, may lower patient’s pain threshold and lead to FLS.35,36 The severity of these measures may be triggered or exacerbated by both COVID-19 and hospitalization. Given that FM may also lead to more severe anxiety and muscle weakness, further understanding is needed on the temporal interplay between these measures and symptom chronification.30,37 Ultimately, aligning with existing theories, these syndromes may arise due to a persistent cycle of pain, functional limitations, and psychological stressors. 15
Across all the models, the PCFS score – which measures the direct impact of COVID-19 on functional status – did not appear to be a significant predictor in any of the models despite being significant in bivariate analyses. Thus, it appears that CWP/FLS/FM may be affected by manageable risk factors after COVID-19 infection – such as handgrip strength – rather than the impacts of the infection themselves.
Clinical implications
Our findings have important public health implications for the management and/or prevention of chronic pain and FLS/FM upon a potential trigger such as COVID-19 infection. The strongest predictive variables – poor sleep quality and handgrip strength – can be targeted early on in addition to existing pain management techniques. Muscle strength and mass, as indicated by handgrip strength, can also be improved through home-based training and stretching exercises. Additionally, poor sleep quality, which is often overlooked in rehabilitation programs, may be critical to address to prevent further development of post-COVID symptoms. Early identification and management of these measures by healthcare providers or the patients themselves may be key to slowing the cycle of post-COVID related symptom chronification.
Limitations
Limitations of this analysis include the absence of a direct set of questions specific to the ACR FM diagnostic criteria. We aimed to control for this through establishing the continuum of PCS from chronic pain to probable FM. Additionally, most of the measures assessed at Phase 1 and Phase 2 could not be evaluated during hospital admission or immediately afterward due to logistical complications arising from the severity of the pandemic at the time. Additionally, the present study have potential risk of selection bias due to sample attrition, 415 subjects were excluded from consideration because of no pain assessment in phase 2 follow-up. However, it is unlikely, this subset of sample would modify the results, since the sample characteristics at phase 1 were homogeneous (Table 1). Further, this study was conducted on a sample of patients at one hospital in Brazil, which may limit the generalizability of our findings to moderate to severe COVID-19 patients. Future analyses could expand the sample size to patients affected by different severities of COVID-19 and employ more specific measures of FM at multiple timepoints after infection to validate our findings.
Conclusions
Our findings indicate that PCS has long-term impacts that resemble chronic pain and fibromyalgia symptoms at a higher rate than the general population. In our sample, significant risk factors for both regional and widespread chronic pain over an average of 2.7 years were identified to be anxiety/depression and weaker handgrip strength. Risk factors for FLS/FM include high baseline pain, insomnia, and weaker handgrip strength. Understanding these risks inform prevention strategies to reduce the risk of chronic pain associated with PCS.
Supplemental Material
sj-docx-1-mpx-10.1177_17448069251369069 – Supplemental material for Risk factors associated with chronic pain and fibromyalgia-like symptoms post-COVID hospitalization
Supplemental material, sj-docx-1-mpx-10.1177_17448069251369069 for Risk factors associated with chronic pain and fibromyalgia-like symptoms post-COVID hospitalization by Linamara Rizzo Battistella, Marta Imamura, Xinyi Christine Zhang, Kevin Pacheco-Barrios and Felipe Fregni in Molecular Pain
Footnotes
Acknowledgements
We are thankful for the support received from members of the HCFMUSP COVID-19 Study Group.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was partially supported by donations from the general public under the HC-COMVIDA crowdfunding scheme (![]() ) with funds managed by the Fundação Faculdade de Medicina – USP.
) with funds managed by the Fundação Faculdade de Medicina – USP.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
