Abstract
Trigeminal neuropathic pain has been modeled in rodents through the constriction of the infraorbital nerve (CCI-ION). Sensory alterations, including spontaneous pain, and thermal and mechanical hyperalgesia are well characterized, but there is a notable lack of evidence about the affective pain component in this model. Evaluation of the emotional component of pain in rats has been proposed as a way to optimize potential translational value of non-clinical studies. In rats, 22 and 50 kHz ultrasonic vocalizations (USVs) are considered well-established measures of negative and positive emotional states, respectively. Thus, this study tested the hypothesis that trigeminal neuropathic pain would result, in addition to the sensory alterations, in a decrease of 50 kHz USV, which may be related to altered function of brain areas involved in emotional pain processing. CCI-ION surgery was performed on 60-day-old male Wistar rats. 15 days after surgery, von Frey filaments were applied to detect mechanical hyperalgesia, and USV was recorded. At the same timepoint, systemic treatment with d,l-amphetamine (1 mg/kg) allowed investigation of the involvement of the dopaminergic system in USV emission. Finally, brain tissue was collected to assess the change in tyrosine hydroxylase (TH) expression in the nucleus accumbens (NAc) and c-Fos expression in brain areas involved in emotional pain processing, including the prefrontal cortex (PFC), amygdala, and NAc. The results showed that CCI-ION rats presented mechanical hyperalgesia and a significant reduction of environmental-induced 50 kHz USV. Amphetamine caused a marked increase in 50 kHz USV emission in CCI-ION rats. In addition, TH expression was lower in constricted animals and c-Fos analysis revealed an increase in neuronal activation. Taken together, these data indicate that CCI-ION causes a reduction in the emission of environmental-induced appetitive calls concomitantly with facial mechanical hyperalgesia and that both changes may be related to a reduction in the mesolimbic dopaminergic activity.
Keywords
Introduction
Trigeminal neuropathic pain is a consequence of a lesion or disease of the trigeminal nerve and may have multiple underlying causes and clinical presentations. Trigeminal neuralgia (TN) is the best-known form of trigeminal neuropathic pain, which is characterized by recurrent unilateral brief electric shock–like pain attacks, limited to the distribution of one or more divisions of the trigeminal nerve and triggered by innocuous stimuli.1–3 The trigeminal nerve consists of three main branches: ophthalmic (V1), maxillary (V2), and mandibular (V3), being the last two mainly affected in TN.2,3 Constriction of the infraorbital nerve (CCI-ION), the main branch of V2, is a widely used model to study TN pathophysiological mechanisms, consequences, and treatment effects in rodents.4–9 Previous studies of our group using this model have shown that rats subjected to CCI-ION developed late (i.e., after 10 days of surgery) but long-lasting facial mechanical hyperalgesia (i.e., more than 3 months), which is associated with the development of anxiety-like behavior.4,10 This finding is in agreement with clinical reports that show the impact of TN on emotional functioning of patients, an aspect that has gained attention but is still overlooked.11–13
Evaluation of the emotional component of pain in rats has been proposed as a way to optimize the potential translational value of pre-clinical studies. 14 In this regard, the analysis of ultrasonic vocalization (USV) is considered a well-established measure of positive and negative emotional states in rodents.15,16 It is well established that 22 kHz calls are emitted by rats in stressful and/or painful situations to express a negative state and/or to alert conspecifics of a potential danger. 17 In this regard, it has been shown that formalin injection into the hind paw or into the upper lip causes a significant increase in 22 kHz call emissions briefly after the injection.18–20 On the other hand, 50 kHz call emissions by rats are related to appetitive stimuli and social interaction. At least two main categories of appetitive calls have been described: (1) flat, which have a social-coordinating function and (2) frequency modulated (FM), which include step, mixed, and trill calls, that are mainly related to rewarding and hedonic situations.15,17 In fact, drugs with euphorigenic properties, such as amphetamine, are potent elicitors of 50 kHz USV, possibly by causing an increase in dopamine in the nucleus accumbens (NAc). 21
A previous study from our group reported that persistent inflammatory pain in the orofacial region is associated with significantly reduced 50 kHz call emissions from rats. Additionally, it was shown that this effect was accompanied by reduction of dopaminergic activity in the NAc. 19 These findings suggest that persistent orofacial pain may lead to a decrease in mesolimbic dopamine, which may be related to the reduction in the emission of 50 kHz calls. In the light of these considerations, the present study tested the hypothesis that trigeminal neuropathy would result in both sensory alterations and a decrease of 50 kHz USV, the latter of which may be related to altered function of brain areas involved in emotional pain processing.
Material and methods
Animals
A total of 82 male 60-days old Wistar rats weighing 250–300 g and provided by the Federal University of Parana colony were kept in groups of three to four per cage (41 cm× 32 cm× 16.5 cm) in a climate-controlled room (22 ± 2°C) on a 12-h light/dark cycle with chow and water ad libitum. Fresh sawdust was provided every other day for bedding. Experimental procedures were performed during the light cycle, between 7 a.m. and 7 p.m., respecting at least 48 h for the acclimatization of the animals to the environment, in accordance with the ARRIVE guidelines, the guidelines from the International Association for the Study of Pain, the Federal University of Parana and Brazilian regulations, and the Canadian guidelines of the University of Calgary on animal welfare. The protocols were approved by both Universities’ Ethics Committees (CEUA/BIO-UFPR #1057 and AC18-0003). All efforts were made to optimize the number of animals used and to reduce their stress and suffering, respecting the 3R’s statement.
Drugs and reagents
Rats were anesthetized with an intraperitoneal injection of a solution of ketamine (90 mg/kg; Vetica Laboratories of Veterinary Products, São Paulo, Brazil) and xylazine (10 mg/kg; Rhobifarma Ind. Farmacêutica, São Paulo, Brazil), or via inhalation with 5% isoflurane (Abbott laboratories, USA). D,l-amphetamine sulfate (1 mg/kg; sc; Sigma-Aldrich, St Louis, Missouri, USA) was diluted in sterile saline. To quantify the expression of c-Fos and TH by immunohistochemistry (IHC), primary monoclonal antibodies rabbit anti-Fos (Abcam, AB190289) and mouse anti-TH (Sigma-Aldrich, T1299) antibodies were used. The secondary antibodies were goat anti-mouse Alexa Fluor 488 and Alexa Fluor 546 (Invitrogen, Thermo Fisher Scientific, CA, A21206 and A11030). For quantification of TH expression by western blot, mouse anti-TH primary monoclonal antibodies (Sigma-Aldrich, St Louis, MO, T1299) and secondary antibodies (mouse anti-IgG HRP conjugate; Jackson) were used. The loading control used was α-tubulin (Abcam, Cambridge, MA, 1: 5000). The doses of drugs and antibodies dilution were based on previous studies.5,22–25
Constriction of the infraorbital nerve
Induction of trigeminal neuropathic pain was performed through a constriction of the infraorbital nerve (CCI-ION) according to the method initially proposed by VOS et al. 26 and later modified by Chichorro et al. 5 First, animals were anesthetized with intraperitoneal ketamine and xylazine hydrochloride followed by local trichotomy and face asepsis with iodinated alcohol. After establishment of anesthesia, an incision was made in the skin below the right eye, approximately 3 mm posterior to the insertion of the vibrissae. The levator labii superioris muscle and the superficial layer of the anterior masseter muscle were retracted so that the rostral portion of the infraorbital nerve was exposed close to the infraorbital fissure. The infraorbital nerve was dissected from adjacent tissues, and then two loose ties were placed, approximately 2 mm apart, with 4.0 silk thread around the nerve bundle. The suture was made with the same type of needled thread used to make the ligatures. Sham animals underwent the same surgical procedure; however, the nervous bundle was not subjected to constriction. At the end of surgery, the animals were monitored in a warm room until complete recovery from anesthesia.
Assessment of facial mechanical hyperalgesia
Animals were individually habituated in acrylic observation boxes for 2 h (from 8 to 10 a.m.) prior to the beginning of the experiment. The start time of the experiments was defined based on previous studies, in which an improved behavioral response was observed. Von Frey filaments (Semmes–Weinstein monofilaments, Stoelting, USA, 0.04; 0.07; 0.16; 0.4; 1.0; 2.0; 4.0, and 8.0 g) were progressively applied in the vibrissa pad skin on the right side of the face, ipsilateral to the surgery. An experimenter blind to the animal’s condition applied each filament three consecutive times, with an interval of approximately 3 s between each application. The response threshold to mechanical stimulation corresponded to the filament that twice evoked rapid head withdrawal behaviors or attack/escape reactions. 4 Only rats that did not present nociceptive behavior in the pre-selection stage with the application of all filaments were included in the experiments, so that the mechanical threshold baseline was equal to or greater than 8.0 g.
Analysis of ultrasonic vocalizations
Ultrasonic vocalization (USV) analysis was performed as described by Araya et al. 19 The experiment was carried out in an acoustic room with lighting at 40 lux. Each animal was placed individually in an acrylic box (30 cm3) with fresh bedding, an environment that was found to increase the 50 kHz USV emission. 27 An ultrasonic microphone (CM16 Avisoft Bioacoustics, Berlin, Germany), sensitive to frequencies between 15–180 kHz, was placed at a distance of 45 cm from the floor of the movement box allowing the capture of the USV. The Avisoft Recorder software (version 2.95; Avisoft Bioacoustics) allowed the recording and subsequent analysis of the spectrogram (Avisoft SAS Lab Pro version 4.34 Avisoft Bioacoustics), with a frequency of 488 kHz and 0.512 ms resolution, of the USV emissions by the animals. The call-to-call separation was defined by an inter-call interval of at least 190–320 ms, which is the inhalation time between two vocal calls. The duration of the USV recording test was 10 min, and the quantification analysis was performed manually by an experimenter blind to the animal’s condition and/or treatment, according to the frequency and shape of each USV.18,28 The 22 kHz USVs are calls in the frequency range 18–32 kHz, emitted in bouts or individually; 29 however, these emissions were not detected in our analysis. Moreover, all emissions above 33 kHz were considered as 50 kHz USV, and their classification into subtypes was based on their spectrographic shapes being termed: flat, when the peak frequency changes within a single call were equal to or less than 5 kHz; step, when a main flat call also had another short flat element shifted 5 kHz higher in frequency; trill, a single call element with peak frequency changes in opposite directions separated by at least 5 kHz forming a “zig-zag”; and mixed, which were calls that did not fit into other categories.28–30
Western blots
Rats were sacrificed by decapitation under deep anesthesia with inhalation of isoflurane on day 15 after CCI-ION or sham surgery. Brains were excised on dry ice and stored at −80°C according to a previous study. 24 The nucleus accumbens (NAc) was dissected bilaterally with a standard puncture biopsy (tissue extension of 0.15 cm in diameter). Tissue samples containing the NAc were lysed in modified radioimmunoprecipitation assay (RIPA) buffer: 50 mM Tris, 100 mM NaCl, 0.2% Triton X-100 (vol/vol), 0.2% NP-40 (vol/vol), and 10 mM diethylenediaminetetraacetic acid (EDTA) plus a protease inhibitor cocktail (Thermo Scientific™A32965) at pH 7.5. Lysates were prepared by shaking the samples for 30 min at 4°C and by centrifugation at 13,000 rpm for 20 min also at 4°C. Supernatants were transferred to new tubes, and protein quantification followed the Bradford method (Bio-Rad, Hercules, CA). Then the samples (60 mg) were boiled in Laemmli sample buffer for 10 min and separated in 10% SDS-polyacrylamide gel electrophoresis and transferred to a 0.45 mm polyvinylidene difluoride membrane (Merck-Millipore, Darmstadt, Germany). To prevent non-specific binding, membrane blocking was performed with 0.1% tris-tween 20 buffered saline (TBST, pH 7.6) containing 5% skim milk for 30 min. After washing 4 times for 10 min with 10 mL of TBST, the membranes were incubated with a mouse anti-tyrosine hydroxylase (TH) monoclonal antibody (Sigma-Aldrich, St Louis, MO, T1299) at a 1:2500 dilution overnight at 4°C. Horseradish peroxidase (HRP-Horseradish peroxidase)–conjugated secondary antibody anti-mouse IgG (Jackson ImmunoResearch, West Grove, PA) was diluted in 5% skim milk in TBST solution (1:5000) and incubated for 1 h under agitation. The loading control used was α-tubulin (Abcam, Cambridge, MA, 1: 5000). Western blot images were recorded with an LI-COR scanner (98–14,229, Biosciences, Canada), and the densitometry analysis was performed using the ImageJ software (National Institutes of Health).
Immunohistochemistry
Rats underwent transcardiac perfusion with 4% paraformaldehyde under deep anesthesia with inhalation of isoflurane on day 15 after CCI-ION or sham. Brains were submerged in 4% paraformaldehyde overnight and then transferred to 30% sucrose solution for 3 days. Subsequently, a tissue block was made with Tissue-Tek on dry ice and stored at −80°C. 30 mm NAc slices were obtained with cryostat cut, then washed three times with phosphate-buffered saline followed by blocking for 2 h with a solution containing 0.5% bovine serum albumin, 10% bovine serum normal goat, and 0.3% Triton X-100 in phosphate-buffered saline. Sections were then incubated with anti-TH or anti-c-Fos primary antibody overnight at 4°C and washed three times (same solution as above) before incubation with secondary antibody for 2 h at room temperature. After washing three times in phosphate-buffered saline, the sections were mounted on slides. All images were captured digitally with an 8-bit camera, thus giving color level (intensity) values from 0 to 255. Immunostaining was visualized using a 40 × 0.4 numerical aperture objective lens on a Zeiss LSM510META confocal system, run speed 6. ImageJ (National Institutes of Health) was used to quantify fluorescence intensity. This protocol was adapted from Huang et al. 23
Experimental procedures
Facial mechanical threshold (baseline) was assessed randomly in all animals at 10 a.m. after 2 h of individual acclimatization in an acrylic box (according to protocol 2.4) prior to surgery (according to protocol 2.3). Likewise, on day 15 after CCI-ION or sham surgery, the development of mechanical hyperalgesia was confirmed in constricted (n = 8) compared to sham animals (n = 7). At the same time point, after a 4-h interval, the same animals were exposed to a USV test (according to protocol 2.5) to detect CCI-ION–induced changes related to hyperalgesia (Figure 1A). The evaluation was performed randomly by an experienced experimenter blind to the condition of the animals. Pain-related responses induced by CCI-ION in rats. (A) Timeline of the experimental procedures. Constriction of the infraorbital nerve (CCI-ION) or sham surgery was performed on day zero (DO) after evaluation of the baseline mechanical threshold (BL). (B) On day 15 (D15) after CCI-ION, constricted rats (n = 8) developed mechanical hyperalgesia compared to sham (n = 7). (C) At the same time point, CCI-ION rats showed a reduction in the emission of flat calls and in the total number of 50 kHz USV compared to sham. (D) Illustrates the reduction in the total number of 50 kHz calls, but with no change in the percentage of subtypes emission between groups. Data are expressed as mean ± SEM *p < 0.05 when compared with sham. Two-way ANOVA (C andD) with repeated measures (B) followed by Bonferroni post-hoc test.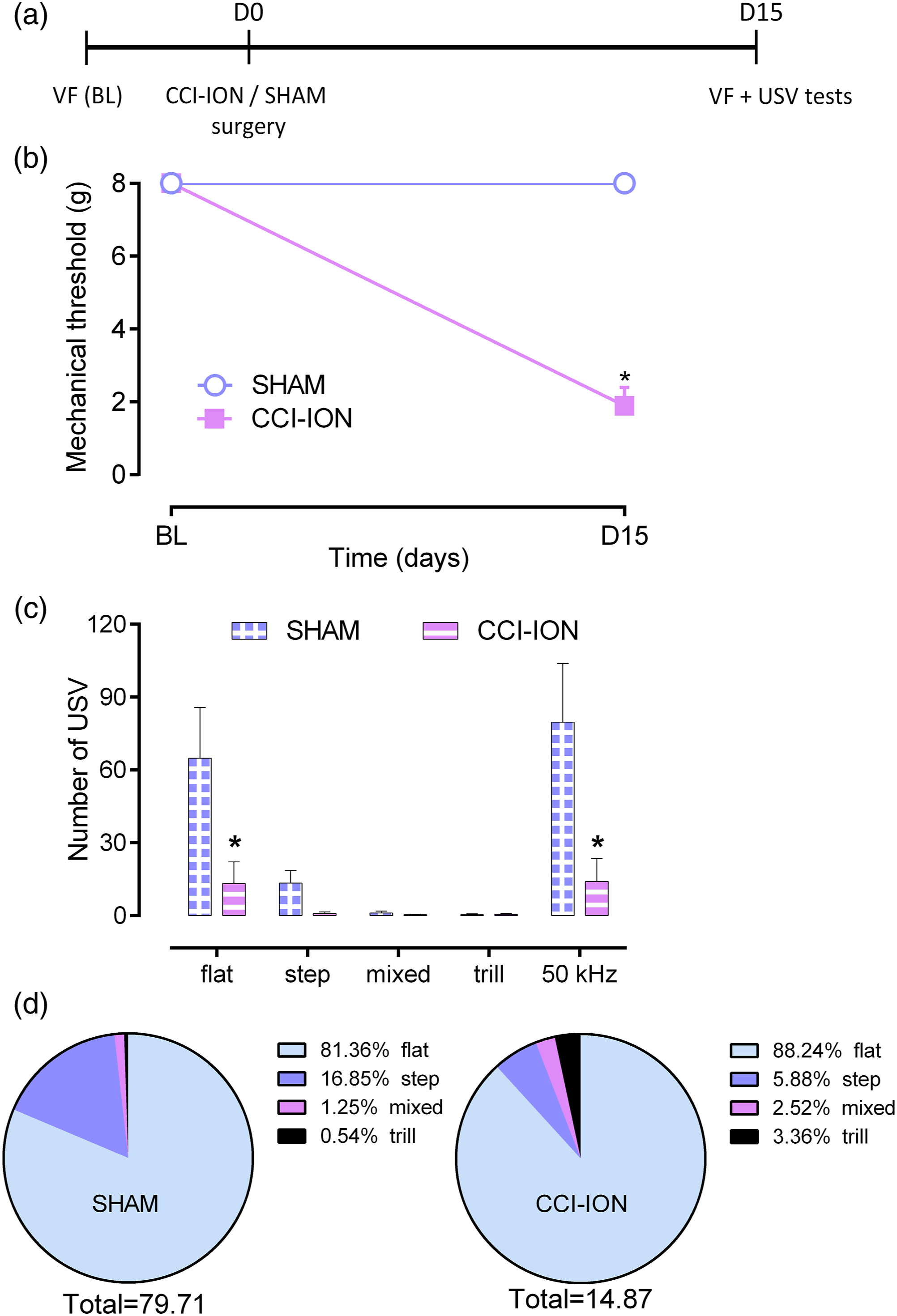
Similarly, to identify the involvement of the motivational system, the effect of amphetamine on USV emission was investigated once nociception was established, on day 15 after CCI-ION. Independent groups of animals were treated subcutaneously with saline or d,l-amphetamine (sham-SAL n = 6, CCI-ION-SAL n = 7, sham-AMPH n = 6, CCI-ION-AMPH n = 8) 15 min before the USV recording test with a duration of 10 min (Figure 2A). First, saline-treated animals were exposed to the USV test, followed by amphetamine-treated animals to reduce experimental bias in control animals. In addition, the animals that underwent the test were kept in a separate experimental room from the animals that had not undergone USV recording to avoid communication between them. Quantification was performed manually in a condition- and treatment-blind manner (according to protocol 2.5). Influence of amphetamine in USV emission after CCI-ION. (A) Timeline of the experimental procedures. Constriction of the infraorbital nerve (CCI-ION) or sham surgery was performed on day zero (DO). On day 15 after (D15) CCI-ION, rats were treated with d,l-amphetamine (1 mg/kg; sc; AMPH) or saline (1 mL/Kg; sc.; SAL) 15 min before USV recording during 10 min. (B) Shows the number of call emissions and (C) Illustrates the total number of 50 kHz calls of each group and the percentage of each call subtype within the group (sham+SAL n = 6; CION+SAL n = 7; sham+AMPH n = 6; CION+AMPH n = 8). Data are expressed as mean ± SEM *p < 0.05 when compared with sham+SAL and #p < 0.05 when compared with CION+SAL. Two-way ANOVA followed by Bonferroni post-hoc test.
At the same time point, on day 15 after surgery, brains of the CCI-ION and sham rats were collected to quantify the expression of TH in the NAc, and thus identify possible changes in the mesolimbic dopaminergic function in animals with trigeminal neuropathic pain (Figure 3A). Independent groups of animals were used for western blots (sham n = 8, CCI-ION n = 8) and immunohistochemistry assays for TH immunostaining (sham n = 8, CCI-ION n = 8) in the NAc. In addition, quantification of c-Fos expression by immunohistochemistry (sham n = 4, CCI-ION n = 4) was performed in several central areas implicated in pain processing including the NAc, medial prefrontal cortex (mPFC), central amygdala (CeA), and basolateral amygdala (BLA). It is important to emphasize that all animals were mechanically stimulated in the vibrissa pad skin between 1 and 2 h before euthanasia for harvest tissue (according to the protocol 2.7). Quantitative analysis of tyrosine hydroxylase in the nucleus accumbens after CCI-ION. (A) Timeline of the experimental procedures. Constriction of the infraorbital nerve (CCI-ION) or sham surgery was performed on day zero (DO). Samples of the nucleus accumbens (NAc) were extracted from sham and CCI-ION rats on day 15 (D15) after surgery for quantification of tyrosine hydroxylase (TH). (A) Representative image for western blot of TH; (B) Protein expression levels of TH by western blot (sham n = 8, CION n = 8; (C) Immunofluorescence for quantification of tyrosine hydroxylase (TH) expression observed in the dopaminergic fibers reaching the nucleus accumbens shell (NAc) by fluorescence microscopy (sham n = 8; CCI-ION n = 8). (D) Representative images of ipsilateral NAc shell expressing TH-positive fibers from sham and CCI-ION rats. Data are expressed as mean ± SEM *p < 0.05 when compared with sham. Unpaired two-tailed t-test.
Statistical analysis
Two-way analysis of variance (ANOVA) with repeated measures followed by Bonferroni post-hoc test was used to analyze the time course of mechanical hyperalgesia, according to condition (i.e., sham/CCI-ION) and time as independent factors. Two-way ANOVA followed by Bonferroni post-hoc test was performed to analyze the USV emission after d,l-amphetamine treatment. Analyzes of western blot and immunofluorescence intensity were performed using the unpaired two-tailed Student’s t-test. Sample size was determined based on the GPower 3.1 software 31 defining a large standardized effect size of F = 0.5; power of 0.8; and α = 0.05, and estimated six to eight rats per group in the different experimental designs. For c-Fos detection, the sample size was based on previous studies32,33 to recruit the smallest number of rats. All data were expressed as mean ± SEM, and the results were considered statistically significant if p < 0.05. GraphPad Prism version 6 for Windows was used for statistical analysis of data (GraphPad Software, San Diego, CA).
Results
Changes in the facial mechanical threshold and ultrasonic vocalization emission after constriction of the infraorbital nerve
Baseline (BL) threshold to mechanical stimuli was similar in all selected rats (22/32) before surgery. It is important to mention that the surgery did not affect the weight of the animals, their coat, or the brightness of their eyes, showing signs of well-being. On day 15 after surgery, 8/15 CCI-ION rats developed mechanical hyperalgesia (time factor F (1,13)=111.8, p < 0.01; condition factor F (1,13)=111.8, p < 0.01; and interaction factor F (1,13)=111.8, p < 0.01; Figure 1B). Moreover, hyperalgesic rats subjected to nerve constriction showed a significant reduction in the total number of 50 kHz call emissions (condition factor F (1,65)=15.41, p < 0.01; treatment factor F (4,65)=9.020, p < 0.01; and interaction factor F (4,65) = 4.232; p < 0.01; Figure 1C). More specifically, the reduction was detected in the flat calls subtype (p < 0.01; Figure 1C). Figure 1D illustrates the total number of calls in each group and the percentage of each subtype within the group. Despite the marked reduction in call emissions by CCI-ION rats, the percentage of 50 kHz subtypes did show statistical differences between groups (p > 0.1; Figure 1D). Non-hyperalgesic CCI-ION rats were excluded from the experiment.
Influence of amphetamine in ultrasonic vocalization emission by rats subjected to constriction of the infraorbital nerve
Sham animals (12/12) and CCI-ION rats (15/15) were randomly treated with amphetamine or vehicle on day 15 after surgery. D,l-amphetamine (1 mg/kg; sc; AMPH) administered on sham rats did not change the emission of flat calls (p > 0.1; Figure 2B) or the total number of 50 kHz USV emissions (p > 0.1; Figure 2B) compared to sham rats treated with vehicle (saline, 1 mL/kg; sc). CCI-ION promoted a considerable reduction in the total number of 50 kHz USVs and specifically in the flat subtype (condition factor F (1,55)=43.05; p < 0.01; treatment factor F (4,55)=14.92; p < 0.01; and interaction factor F (4,55)=12.66; p < 0.01; Figure 2B) compared to sham rats treated with vehicle. On the other hand, d,l-amphetamine treatment in CCI-ION rats resulted in a significant increase in both the total number of USV emissions (condition factor F (4,115)=8.629; p < 0.01; treatment factor F (3,115)=6.674; p < 0.01; and interaction factor F (12,115)=1.941; p < 0.05 Figure 2B) and flat calls (p < 0.01; Figure 2B) when compared to CCI-ION group treated with vehicle. Figure 2B illustrates the total number and percentage of 50 kHz subtypes within each group. There was no difference in the number or subtype of emissions between sham groups (p > 0.1; Figure 2C). However, in CCI-ION rats, d,l-amphetamine caused a marked increase in the number of 50 kHz call emissions (p < 0.01 compared to sham-SAL and p<0.05 compared to CCI-ION-SAL; Figure 2C), but without changing the percentage of each emission subtype (p > 0.1, flat; p > 0.1, step; p > 0.1, mixed; and p > 0.1, trill; compared to CCI-ION-SAL; Figure 2C).
Analysis of tyrosine hydroxylase expression in the nucleus accumbens after constriction of the infraorbital nerve
TH expression was determined on day 15 after surgery by western blot and immunohistochemistry. It first revealed a reduction in TH protein expression in the NAc of constricted compared to sham rats (t (14)=3.487; p < 0.01; Figure 3B). Figure 3C provides a representative image for western blot of TH. Moreover, the immunohistochemistry technique demonstrated a reduction in the TH-positive fibers expressed in the NAc shell of the CCI-ION compared to the sham group (t (14)=5.437; p < 0.01; Figure 3D). Figure 3E provides a representative image of TH-positive fibers expression in sham and CCI-ION rats.
Activation of pain-processing brain areas after constriction of the infraorbital nerve
The expression of c-Fos by immunohistochemistry allowed us to identify changes in neuronal activation in brain areas associated with pain processing. CCI-ION rats presented an increase in c-Fos expression in the ipsi- and contralateral sides in the nucleus accumbens shell compared to the sham group (Condition factor F (3,12)=7.498, p < 0.01; NAc; Figure 4A), in the medial prefrontal cortex (Condition factor F (3,12)=8.259, p < 0.01; mPFC; Figure 4C), in the central amygdala (Condition factor F (3,12)=22.47, p < 0.01; CeA; Figure 4E), and in the basolateral amygdala (Condition factor F (3,12)=11.73, p < 0.01; BLA; Figure 4G) of CCI-ION compared to sham rats. Figures 4B,D,F,H show representative images of c-Fos immunofluorescence in the NAc, mPFC, CeA, and BLA, respectively, of sham and CCI-ION rats. Quantification of c-Fos expression levels in pain-processing brain areas after CCI-ION. Euthanasia for tissue harvesting was performed on day 15 after infraorbital nerve constriction (CCI-ION) or sham surgery. (A) Quantification of c-Fos (sham n = 4; CCI-ION n = 4) expression in neuronal nuclei in the nucleus accumbens shell (NAc); (C) in the medial prefrontal cortex (mPFC); (E) central amygdala (CeA); and (G) basolateral amygdala (BLA) by intensity of fluorescence. (B, D, F, and H) Representative images for c-Fos immunofluorescence from sham and CCI-ION rats. Data are expressed as mean ± SEM *p < 0.05 when compared with sham. One-way ANOVA followed by Bonferroni post-hoc test.
Discussion
The main finding of the present study is that trigeminal neuropathic pain causes a reduction in the emission of appetitive calls, concomitantly to facial mechanical hyperalgesia, which may be related to a reduction in the mesolimbic dopaminergic activity. These data corroborate our previous observations 19 and reinforce the idea that analysis of USV can contribute to a better understanding of pain-related phenotypes associated with different rat models.
Sensory alterations after CCI-ION are well characterized in the literature. Long-lasting facial mechanical hyperalgesia4–6,8,26,34 has been associated with the development of anxiety-like behavior in this model, possibly due to persistence of hypersensitivity.10,35 It has been proposed that analysis of USV may be useful in the study of the affective component of pain.18,20 Previous studies of our group showed that inflammatory orofacial pain that lasts for a few days causes a decrease in the emission of 50 kHz USV emission, which is accompanied by the development of anxiety-like behavior, suggesting that persistence of pain can induce mood changes and affect social interaction. 19 Accordingly, the current study shows that animals subjected to CCI-ION present a significant reduction in environmental-induced emission of 50 kHz USV compared to sham, more specifically in the flat calls subtype. The full implication of this finding remains to be elucidated, but it is possible to speculate that the chronic pain states cause impairment in social interaction, an idea that is corroborated by previous clinical and non-clinical observations.18,36–38 It is noteworthy that we failed to detect the emission of aversive (i.e., 22 kHz) USV in the protocol used herein. Previous studies that aimed to analyze 22 kHz USV emission in chronic pain models have similarly failed to see such emissions, suggesting that this call subtype may serve to signal danger, potential danger, or an aversive situation.39,40 In this regard, it has been extensively shown that acute pain in rats is associated with increased emission of 22 kHz USV.18–20,41–44 On the other hand, it was recently demonstrated in a different rat model of neuropathic pain that when USV was recorded in a 24 h-period (in 15 min bins), rats showed both a decrease in the emission of 50 kHz USV, as well as an increase in the emission of 22 kHz USV. 45 However, other studies have observed mixed results. 46 As observed in the present study, when the rat was monitored alone in a test arena (or in the cage), 22 kHz USV was not detected or detected at a very low rate.46,47 Thus, it is possible that some methodologies (e.g., USV recording for longer periods or at different time points, during social interaction or group cage maintenance, different chronic pain model, etc.) could influence changes in 22 kHz calls in chronic pain states 39,45,48,46
After CCI of the sciatic nerve, rats also showed reduced rates of 50 kHz USV during the tickling test, which is a hedonic stimulus. 45 Here, we also used a hedonic stimulus to try to revert the influence of CCI-ION on the emission of appetitive calls. Dopaminergic psychostimulants, including amphetamine, are considered the most potent non-social elicitors of 50 kHz USV. 21 According to our data, systemic administration of amphetamine caused a marked increase in the emission of 50 kHz USV in CCI-ION rats, without affecting the emission of USV by sham rats. This finding contrasts with previous observations that acute administration of amphetamine in rats stimulates the emission of total 50 kHz USV in naïve rats.21,28,49–51 Interestingly, we did not observe an increase in the emission of FM calls, which are mainly related to hedonic situations, but we observed a drastic enhancement (well above sham levels) in the emission of flat calls by amphetamine treatment. Although most studies have shown that amphetamine influences mainly trill-type USVs, there are reports of increase in flat calls after its acute administration.49,51 The reason for this discrepancy requires further investigation, but some factors had already been shown to influence the effect of amphetamine on USV, such as dose, site of injection, the cathecolaminergic effect of amphetamine, and the dopamine content in the present condition.21,52 For instance, administration of propranolol in rats previously challenged with amphetamine caused a change in the call profile, resulting in suppression of trill calls and significant enhancement of flat calls. 22 Altogether, these findings suggest that the chronic pain states may be associated with the reduction of both hedonic and non-hedonic 50 kHz USV and reinforce the idea that USV analysis can be used to capture pain-induced changes in the positive emotional arousal expressed by 50 kHz USV emission.
There is accumulating evidence for a role of the mesolimbic dopaminergic system in the affective pain component, and particularly the NAc has been implicated, since it is considered a master regulator of motivational drive and has several connections to other pain modulating brain areas. Ongoing pain produces decreases in dopaminergic tone in the NAc, while pain relief is associated with increase in dopamine release in NAc shell (for review see 14,53). Moreover, in TN patients, a decrease in NAc gray matter volume has been reported, but also that there is an increase in the neuronal activity in this structure.54,55 In line with these observations, results of the current study demonstrated significant neuronal activation of NAc neurons after CCI-ION, which was accompanied by a reduction in the expression of TH. Protein levels of TH were shown to be diminished in CCI-ION rats compared to sham, indicating reduced content levels of dopamine in NAc. A lower dopaminergic activity in the mesolimbic system can contribute to the maintenance of the chronic pain state. Also, it may account for the reduction in the emission of flat calls observed in CCI-ION rats, since 50 kHz calls, to both social and non-social contexts, are associated with an augmentation in dopaminergic activity in the NAc.17,52 Thus, the present data corroborate previous observations that pain states cause a significant reduction in the emission of 50 kHz calls, which may be related to decreased dopaminergic activity in the mesolimbic system.19,56–58
Human imaging studies have helped the identification of changes in brain circuits associated with chronic pain. There are several reports of alterations of structural and functional connections in TN patients.12,55,59–61 Relevant to the current study, we noted the clinical observation that TN patients presented reduced gray matter volumes in the bilateral amygdala, but increased functional circuits between the amygdala and the mPFC, compared with healthy control subjects. Interestingly, this circuit was associated with emotional state ratings. 12 Also, it has been shown that increased functional connectivity of the NAc with the PFC predicts pain persistence, suggesting that this circuit contributes to the transition to chronic pain (for review see 53 ). Here, we showed increased neuronal activation in the NAc, amygdala, and the mPFC in CCI-ION rats compared to sham animals. Although these findings are preliminary, they are in agreement with several clinical observations that, although a decrease in the gray matter volume of these structures may occur in chronic pain states, the neuronal activity in these areas collectively increases.53–55,62 Taken together, these findings indicate that CCI-ION alters the function of the mesolimbic system, which includes a reduction in the dopaminergic activity in the NAc. These changes may have a role in the maintenance of the chronic pain state and affect its emotional perception. Considering that dysfunctions of this system can contribute to chronic pain comorbidities, a better comprehension of its functioning in chronic pain states may also provide new insights on pain-related disorders, such as depression or drug addiction.
Footnotes
Author Contributions
EA and JC conceived and designed the study. EA, RA, GW, and JC established the experimental protocols. EA and EC performed the experiments for data acquisition. EA and JC. performed the statistical analysis and interpretation. RA, GW, and JC reviewed the article critically for important intellectual content. All authors discussed the results, commented on the final version of the manuscript, and approved its submission.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001 and by National Council for Scientific and Technological Development—Brasil (CNPq). The CAPES also grants fellowship support for EA and the CNPq provides fellowship support for EC, JC, and RA are recipients of CNPq research productivity fellowship. GZ is supported by the Canadian Institutes of Health Research, the Canada-Israel Health Research Initiative, jointly funded by the CIHR, the Israel Science Foundation, the International Development Research Centre, and the Azrieli Foundation, and he holds a Canada Research Chair (https://www.chairs-chaires.gc.ca/home-accueil-eng.aspx).
