Abstract
Mouse behavioral assays have proven useful for the study of thermosensation, helping to identify receptors and circuits responsible for the transduction of thermal stimuli and information relay to the brain. However, these methods typically rely on observation of behavioral responses to various temperature stimuli to infer sensory ability and are often unable to disambiguate innocuous thermosensation from thermal nociception or to study thermosensory circuitry which do not produce easily detectable innate behavioral responses. Here we demonstrate a new testing apparatus capable of delivering small, rapid temperature change stimuli to the mouse’s skin, permitting the use of operant conditioning to train mice to recognize and report temperature change. Using this assay, mice that were trained to detect a large temperature change were found to generalize this learning to distinguish much smaller temperature changes across the entire range of innocuous temperatures tested. Mice with ablated TRPV1 and TRPM8 neuronal populations had reduced ability to discriminate temperature differences in the warm (>35°C) and cool (<30°C) ranges, respectively. Furthermore, mice that were trained to recognize temperature changes in only the cool, TRPM8-mediated temperature range did not generalize this learning in the warm, TRPV1-mediated range (and vice versa), suggesting that thermosensory information from the TRPM8- and TRPV1-neuronal populations are perceptually distinct.
Introduction
The ability to perceive temperatures and temperature differences enables us to avoid painful cold or hot environments that are harmful.1–4 Additionally, the ability to sense temperature is vital to both produce appropriate autonomic but also behavioral responses to defend body temperature and protect against adverse environmental thermal conditions.5–8 Furthermore, certain context dependent thermal cues produce pleasurable and aversive responses (e.g. heat sources are pleasurable when one is cold but lose their appeal when one is hot). 9 Mammalian thermosensation relies on thermosensory neurons projecting from sensory ganglion to the skin which express receptors capable of transducing temperature changes into neuronal activity.10–14 A variety of temperature-sensitive ion channels have been identified, two of which have received the most attention: TRPV1, an ion channel activated by temperature increase and capsaicin and TRPM8, a channel activated by temperature below 25°C and menthol. TRPV1 has been implicated in the detection of warming and hot temperatures8,15–17 and TRPM8 in cold thermosensation.18–20 Much of our knowledge of the neurobiological mechanisms of thermosensation can be attributed to studies using murine models with specific ablation of neuron populations expressing these receptors or knockout of the genes encoding these receptors and coupling these transgenic animals with behavior thermosensory assays. However, there remains many unknowns and confusing results. For example, many unique TRP ion channels have been identified whose activity is modulated by temperature, but genetically knocking out many of these receptors in experimental animals produces little-to-no apparent deficit or change in thermosensory responses15,21–25 – only the TRPM8 gene knockout has produced a robust thermosensory deficit replicated across many studies.18–20 Alternatively, studies focusing on ablation of genetically defined cell types, such as those expressing TRPM8 or TRPV1, have had more success in producing phenotypes.26–29 However, where there is a warming-detection phenotype from TRPV1 cell ablation there is no detectable phenotype is observed from TRPV1 gene knockout suggesting that there is still much more to learn before we have a complete understanding of basic thermosensation.16,22,26–28,30–32
Our current knowledge of how individual cells and receptors contribute to thermosensation is limited at least in part by the types of behavioral assays available and their constraints. One common category of methods used to study thermosensation include the cold plantar, hot plate, tail flick, acetone evaporative, and Hargreaves tests which rely on measuring the animal’s robust escape and reflex behaviors to cold or painfully hot temperatures or learnt operant behavior to avoid noxious temperatures.33–36 However, thermosensation and nociception overlap at temperature extremes, making these assays which rely on aversive responses to thermal pain not ideal for disentangling neurobiological mechanisms of thermosensation and more general nociception. Other behavioral assays such as the two-plate preference and gradient tests circumvent this confound by instead relying on the animal’s innate preference between innocuous temperatures; this type of test has been utilized to identify the range of temperatures over which specific receptors and cell populations contribute to thermosensory discrimination.18,20,22,27,37 However, both temperature preference and aversive assays have other limitations, such as being restricted to study thermosensation only between temperatures large enough to reliably produce a detectable temperature preference, and unable to identify thermosensation responses between temperatures which produce similar or only mild levels of preference.
Recently, a conditioned behavioral assay for mice was developed that does not rely on the pain or preference caused by temperatures and which could produce reliable behavioral reactions to very small temperature increases. 38 While more technically challenging than other thermosensory tests, in that it requires both head and paw restraint, this type of operant assay allowed more precise measurements of innocuous thermosensory ability including the detection of temperature increases as small as 1°C. This same operant conditioning assay was also used to investigate warming temperature changes. 17 This assay offered a means to gain new insights into the effects of various TRP-channels on thermal perception, albeit over a limited set of temperature ranges. This type of assay, when utilized with more temperature change configurations and coupled with genetic tools for targeting specific thermosensory populations, might be able to provide more information about how individual receptors and cell populations contribute to thermal perception.
Here, we describe an operant assay to investigate thermosensation that does not require restraint and provides a way to measure responses of mice to range of temperature changes. This assay begins with conditioning mice to recognize and report a large thermal step. In particular, we wanted to examine behavioral responses in the innocuous temperature range and consequently limited our studies to examining perceptual ability of mice in the range of 15°C to 40°C.39–41 Mice trained in this fashion generalized for the detection of small temperatures changes across a wide range of baseline temperatures, including those associated with both warm-sensitive and cool-sensitive thermoreceptors. By varying the baseline and target temperature combinations used to train and test mice for operant conditioning of warming and cooling detection, we better delimited the contributions of individual cell populations to the perception of thermosensory stimuli and uncovered new insights about how conditioned thermosensory perception is generalized.
Materials and methods
Animals
All procedures followed the NIH Guide for the Care and Use of Laboratory Animals and were approved by the National Institute of Dental and Craniofacial Research Animal Care and Use Committee. Mice with genetically targeted expression of the diphtheria toxin receptor in TRPV1 neurons (TRPV1-DTR) and TRPM8 neurons (TRPM8-DTR) were generated as described previously. 22 Animals were intercrossed to generate experimental and control animals (of either sex) as described in the text; the TRPV1-DTR and TRPM8-DTR mice were hemizygous for DTR. For diphtheria toxin (DT)-mediated cell ablation, DT administration began in adult mice (>=5 weeks old) with daily injection of 100 ng DT for 7 days for TRPM8-DTR animals and 200 ng DT for 3 weeks (5 days on, 2 days off) for TRPV1-DTR animals. Behavior training and testing began at least 1 week and no more than 2 months after DT administration using mice weighing 20–30 g (2–4 months of age). Control mice for each experimental series were DT-treated, non-DTR littermates cohoused with DTR siblings.
Temperature change apparatus
A small test clear acrylic chamber (4”L × 3”W × 3”H) was constructed. These dimensions confined a mouse to grasp on copper tubing (2 mm OD, 1 mm ID) bent to create a floor grating but allowed mice to turn within the chamber. A locking back door allowed mice to be placed inside the test chamber. Two choice ports (each with a 5 mm x 5 mm opening) were presented to the mouse through a narrow slot in the front of the test chamber. Access to the choice ports was controlled by a stepper motor which extended and retracted the ports toward or away from the test chamber. Each choice port was equipped with a photo interrupter (Digi-Key GP1A57HRJ00f) to detect nose pokes, a silicon tube (Cole-Parmer MasterFlex L/S 16) to deliver water rewards, and a pressurized air tube (10 psi) to deliver air puff punishments. The opening and closing of air and water outlet tubes was controlled by solenoid valves (Cole-Parmer Vaplock; Sci-Plus 40672P1). For the heating and cooling of the copper tubing, water was pumped through tubing connected to two recirculating heated/refrigerated water baths (Polyscience 1150 A, 7 L capacity, 1 L/min flow rate) with independent temperature control. Large solenoid valves (0.25” brass NPT) regulated which of the two water baths were connected to the tubing in the test chamber and which were set to an external recirculation loop (see Figure 1(a)). The surface temperature of the test chamber tubing was measured by a thin-wire exposed-junction type-K thermocouple fixed to the copper tubing with thermal paste and electrical tape and read by a digital thermocouple thermometer (Cole Parmer Digi-Sense EW-08505–86 and EW-91210–07). The entire apparatus was contained within a sound attenuating cubicle (Med Associates ENV-022MD). All electrical components and valves were controlled by Arduino Uno R3 which logged all training and testing data (e.g. nose pokes) to an on-board SD card.

A water-pumped test chamber and operant assay for learned innocuous temperature discrimination in mice. (a) A small acrylic test chamber for mice had a copper tubing floor and two nose poke detection ports, each equipped with a liquid reward spout. Water at different temperatures (a baseline reference temperature, Tref, and a test temperature, Ttest) maintained in separate circulating water baths were switched through sets of solenoid valves to rapidly change the temperature of the test chamber floor. (b) Schematic of the nose poke/water delivery ports used to train and test operant activity. (c) Description of a single trial for training a mouse to detect a temperature change and report a choice by nose poke. Water-deprived mice are given a water reward by correctly poking the left port during no-temperature-change trials and the right port during 15°C temperature-increase trials or given no reward for the incorrect choice. (d) Progression of success rates, defined as the average rates of correct choices between no temperature change and temperature increase trials, over 9 days of training sessions. The subset of mice which learned to correctly discriminate between the two trials/temperatures are advanced for further testing. The average daily success rate for each mouse group (those included or excluded from further testing as defined post-hoc after training) is shown as the mean of each group with error bars representing s.e.m.
Operant assay protocols
Mice were individually trained to recognize temperature changes through operant conditioning separated into stages of increasing complexity; the description of each training stage is briefly described below. Arduino programs defining and controlling each training and testing stage are available online (https://github.com/misaacson01/Operant_Temp_Assay). Mice were water-deprived prior to and all throughout the training and testing phases of the study to motivate drinking behavior. Mice were weighed daily to measure their weight relative to their baseline weight and given supplemental water in 1 mL increments to maintain a minimum of 75% baseline body weight. Mice were identified by ear-tags and assays were performed blinded to mouse identity which was only recorded after assays were completed.
Stage 1 (day 1): A mouse was placed alone in the test chamber to acclimate to the chamber and to learn to make nose pokes for rewards. Any nose poke made at this stage in either of the two choice ports resulted in a water reward (∼5 µL) delivered to that port. Pokes made within 2 seconds of a previous reward were ignored. Stage 2 (day 2): Temperature change trials introduced to begin the association of the temperatures stimulus to water reward (the choice-ports). Before a trial began, the choice ports were extended up to the test chamber (beginning from the retracted position where mice were unable to reach the ports) while the test chamber was held at the reference temperature. Once a mouse poked the choice port associated with the reference temperature (the left port), the mouse was given a reward at the port and a temperature change trial began. Trials were either reference or test trials, chosen in a random sequence. For test trials, valves were switched to deliver test temperature water to the tubing in the chamber. After a 4 second delay (allowing time for the temperature change to be most salient), nose pokes in the left (reference) port were punished with an air puff and nose pokes in the right are rewarded with water (volume 5 µl). Pokes were allowed for 10 seconds after the temperature change, allowing enough time for the mice to try each port multiple times. For reference trials, dummy valves were switched to mimic the sound of valves being switched while reference temperature water continued being delivered to the chamber. Pokes in the left port were rewarded and right-side pokes were punished. After the trial ended, the test chamber was switched back to the reference temperature and the choice ports were retracted for 10 seconds to allow time for the test chamber to return to the reference temperature. For the 1st 5 trials, punishments were disabled to prevent early discouragement. Stage 3 (day 3): Continuing association of temperatures with choice-ports, with more trials given and fewer rewards delivered per trial. Stage 3 was nearly the same as stage 2, except that the number of rewards given for correct nose poke choices during a trial was limited to 2, and incorrect pokes were corrected by delivering a reward to the opposite port. This encouraged mice to switch when they made incorrect choices. Punishments were still disabled for the 1st 5 trials. Stage 4 (days 4–7): More association, more trials, and more encouragement of correct choices. This stage was the same as stage 3, except that the mouse was only allowed a single incorrect poke per trial (allowing the mouse to switch sides after an incorrect choice; two incorrect pokes ended a trial early) and only the 1st 3 incorrect pokes resulted in delivery water to opposite side. Punishments were enabled the entire time of the trial. Stage 5 (days 8-11+): Introduce final version of the assay and continue operant conditioning. Test and reference trials were selected in a random sequence. The first poke was either rewarded if correct or punished if incorrect, after which the trial immediately ended. The “success rate” of the mouse for all trials completed in a session was quantified (see Data Analysis and Statistics for details).
Mice were kept at stage 5 for 4–7 days, after which only mice which have successfully learned the task continued into the testing phase; the inclusion criteria for mice was set as a minimum success rate of 0.6 for 2 consecutive days during training stage 5. The testing phase protocol was identical to that of training stage 5, except that the reference and test temperatures are manipulated as described in the text. Each testing session lasted for 30 minutes or until the mouse completed at least 25 trials, as early testing showed that overall success rates did not change appreciably beyond 25 trials (see Supplemental Figure 1). If at least 25 trials were not completed by the mouse within 1 hour, the results from the entire test was discarded. Mice underwent testing for up to 3 sessions per day.
Data analysis and statistics
Success rates were calculated for each mouse, per experiment, using the formula
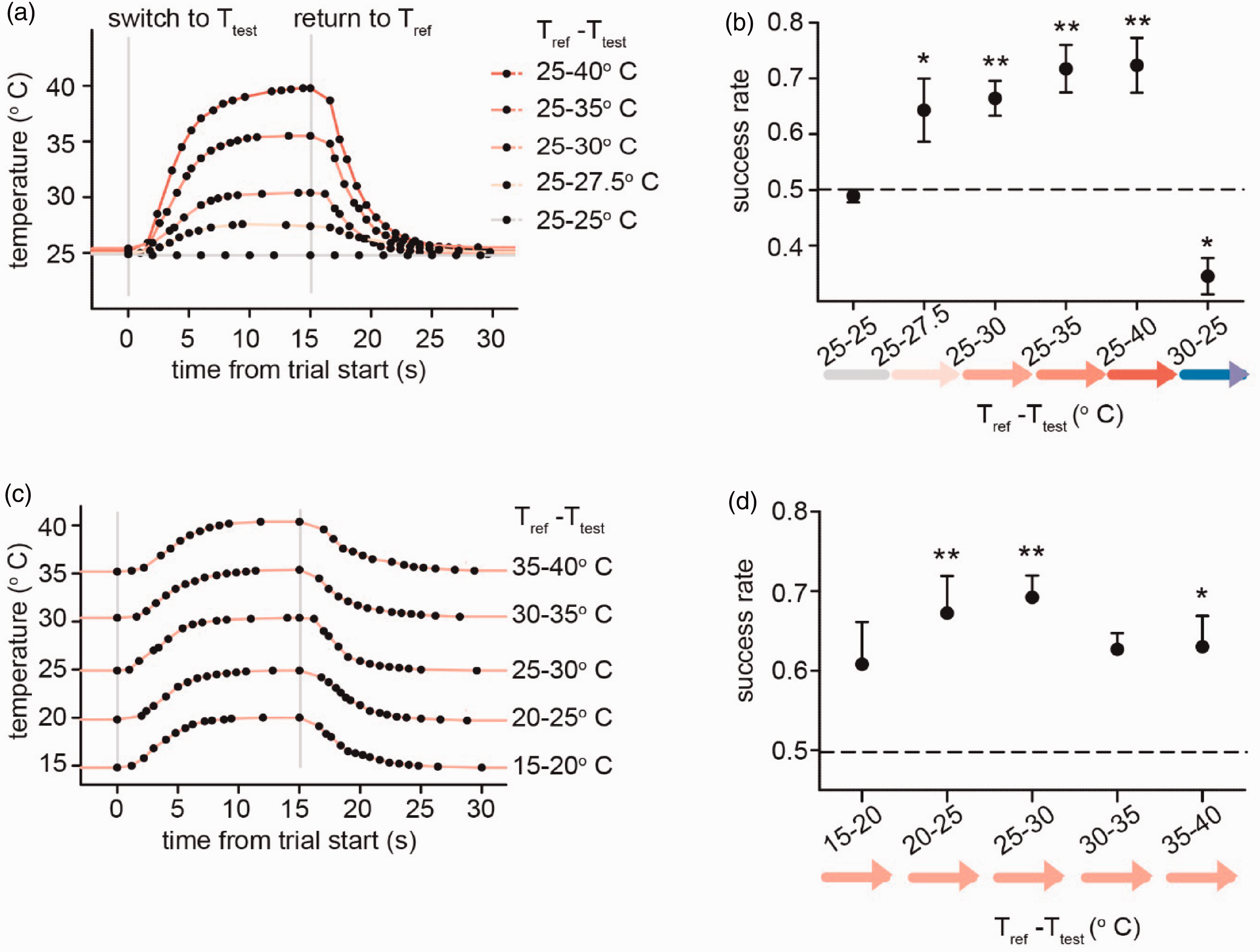
Learned temperature change detection demonstrates acuity of thermosensation and generalization of learning across a wide range of baseline temperatures. (a) Temperature profile of the test chamber floor during a no-temperature-change trial (25–25°C) and for trials of four magnitudes of temperature increase (2.5, 5, 10 and 15°C) from the 25°C baseline reference temperature. (b) Mean rates of successful temperature change detection of by magnitude and direction of temperature change, of mice trained to detect a 15°C temperature increase. Tested temperature changes and a no-temperature change control and four magnitudes of temperature increase (same as shown in [a]), as well as one magnitude of temperature decrease (Tref-Ttest = 30–25°C). (c) Temperature profile of the test chamber floor during trials of a 5°C temperature increase from five different baseline temperatures (15, 20, 25, 30 and 30°C). (d) Success rates of trials of 5°C temperature changes from five baseline temperatures, as shown in (c), of mice trained to detect a 15°C temperature increase from a baseline of 25°C. Data points in (b) and (d) are the mean success rate of all successfully trained C57BL6 mice (n = 5) with error bars representing s.e.m. Success rates which are significantly different from the no-temperature change control were determined by one-way ANOVA followed by Dunnett’s test for multiple comparisons (1 star = p < 0.05, 2 stars = p < 0.01; 1°C comparisons).
Results
We set out to design and develop an operant assay that would allow us to directly probe the perceptual quality of thermal cues in mice. The design of the apparatus and methods for its use took into consideration several details. First, we found through preliminary testing that in order for mice to be trained to detect and report innocuous temperature changes a temperature change stimulus must be sufficiently rapid in order for learning to occur – we note that standard thermoelectric (Peltier) plates, in our hands, (for heating a floor of a compartment big enough for a mouse) were unable to achieve sufficiently fast warming and cooling rates. To overcome this challenge, we designed a different thermal stimulus delivery system made of copper tubing (with high thermal conductivity and diameter optimum for a mouse to grasp) which was in constant contact with the mouse’s paws (Figure 1(a)). Second, we wanted a device where the temperature stimulus could be flexibly adjusted and reliably delivered. For this reason, we used temperature-controlled water which was pumped through the floor tubing at a high rate. The temperature of the water was reliably maintained using two independent, temperature-controlled recirculating water baths, where solenoid valves provided rapid control over which of the two water sources were pumped through the tubing. While a mouse was inside the test chamber, one of the two water sources was held at a stable reference temperature (Tref) and water from it was pumped through the test chamber giving precise control over the baseline temperature. To deliver a rapid temperature change stimulus, water maintained at a different temperature (Ttest) in the 2nd water source was rapidly diverted from an external recirculation loop to the loop containing the tubing in the test chamber (while the reference temperature water was switched simultaneously to the external circulation loop). Third, we wanted an assay where mice were minimally stressed, and mice were not restrained. To allow mice to have relatively free movement while maintaining constant skin contact to the copper tubing, the tubing was looped across the entire test chamber so as to act as the chamber’s flooring, forcing the mice to have at least two paws gripped onto the tubing at all times (Figure 1(a) and see Video). Additionally, the behavioral assay was performed in a darkened and auditorily isolated chamber to further reduce animal stress levels. Lastly, we developed a standardized robust training procedure that permitted the training of mice in about 2 weeks (see Methods for details).
We began operant conditioning of mice using a relatively large, 15°C, temperature change between the reference and test temperatures (25°C and 40°C), see Methods for details. Water-deprived mice were conditioned with water rewards to nose poke into one of two ports in the test chamber during trials when tubing temperature was either maintained at the reference or switched to the test temperature (Figure 1(b) and (c)). After 9 days of training (see Methods for details of training method employed), a subset of mice (5/10) successfully learned to associate the two choice ports with either the reference (25°C) or test (40°C) temperature (Figure 1(d)) and met our criteria for inclusion and proceeded to the testing phase; preliminary results revealed that additional training days did not result in the recruitment of significantly more trained mice. The overall rate for inclusion of trained mice for all sets of conditioning stimuli (warming and cooling) was 58/105.
While keeping the reference temperature stable, mice trained with a 15°C change were found to associate different test temperatures with the test choice port even when the test temperature was only slightly warmer than the reference temperature, with temperature change magnitudes as small as 2.5°C (Tref–Ttest = 25–27.5°C) (Figure 2(a) and (b)). Importantly, in control trials when the test temperature was set as colder than the reference temperature (instead of the warming cue they received during training), mice were more likely to make the incorrect choice (Figure 2(b)). This suggests that trained mice were incapable of extending their training to novel stimuli outside of their experience as far as direction of temperature change, a result similar to that previously reported in operant thermal tests. 17 However, mice consistently associated the reference and test choice ports when the relatively colder and warmer temperatures respected the learned paradigm (i.e. the test temperature was higher than the reference temperature) even when they were challenged to discriminate over temperature configurations they were not trained to learn. Indeed, this generalization of associating the two choice ports to the relatively colder (reference) and warmer (test) temperatures rather than their absolute temperatures even included temperatures outside the range of their training (e.g. <25°C) (Figure 2(c) and (d)). Together, these results suggest that in the format we used, mice can reliably generalize from a training task suggesting that temperature change stimuli can be readily associated, and importantly successful responses are not dependent on absolute temperatures.
Since the TRPM8 sensory neuron population and the TRPM8 ion-channel have been consistently associated with cold detection,18–20,22,27 we asked whether the specific ablation of this population using diphtheria toxin and genetic targeting of the diphtheria toxin receptor (DTR) or the genetic knockout (KO) of the TRPM8 receptor would affect the mouse’s ability to generalize this learned temperature change detection. When trained mice in the same temperature increase protocol used in our initial studies (Tref -Ttest = 25–40°C), both TRPM8-ablated and TRPM8-KO mice had responses similar to wild-type (WT) animals in the 30–40°C range. As expected, TRPM8-ablated and TRPM8 null mice lost the ability to recognize temperature changes in the cool range (15–25°C) (Figure 3(a) and (b)) which contrasted with wild-type (WT) littermates that had undergone the same training protocol (Figure 3(a) and (b)). These results show that the recognition of a difference between two cool temperatures (<= 30°C) requires the TRMP8 receptor and the TRPM8-neuronal population.
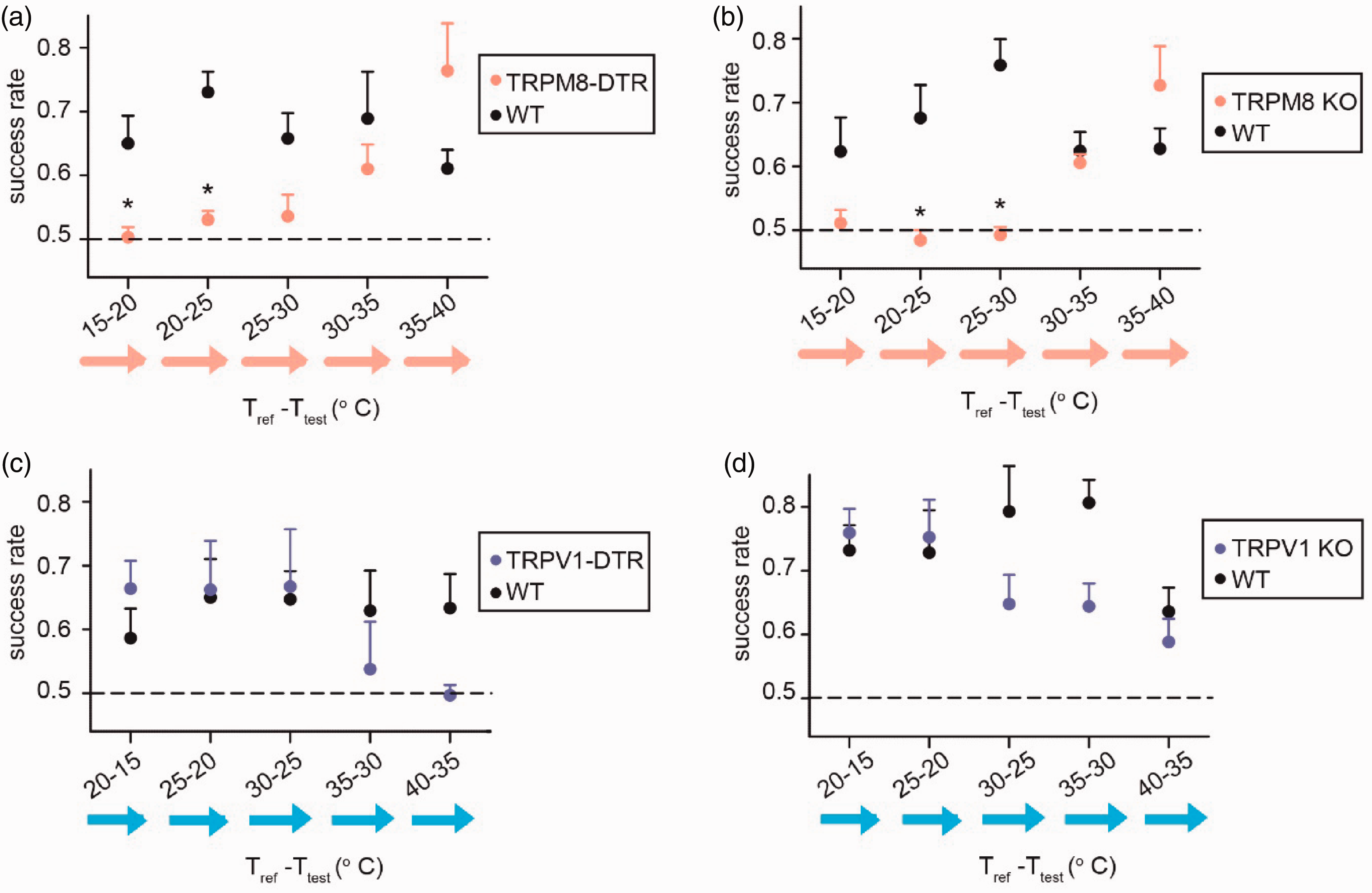
Effects of genetically targeted ablation of TRPM8 and TRPV1 populations and knockout of their receptors on the generalizability of learned temperature change detection. (a) Success rates by baseline reference temperature of trained mice with diphtheria-toxin-ablated TRPM8 cells (TRPM8-DTR, n = 4, in red) as compared to similarly trained WT littermates (WT, n = 4, in black). Both groups were trained to detect a 15°C increase from a baseline of 25°C (trained: 25-40°C) and tested in their ability to detect 5°C increases across the entire test range (15-40°C). (b) Similar to (a), success rates by reference temperature of trained mice with genetic knockout of the TRPM8 receptor (TRPM8-KO, n = 4, in red) compared to WT littermates (n = 4, in black). (c) Success rates by reference temperature of trained mice with diphtheria-toxin-ablated TRPV1 cells (TRPV1-DTR, n = 4, in blue) as compared to similarly trained WT littermates (WT, n = 4, in black). Both groups were trained to detect a 15°C decrease from a baseline of 40°C (“trained: 40–25°C”) to avoid a potential inability to train mice lacking TRPV1 cells to detect a temperature increase, and tested in their ability to detect 5°C decreases across the entire test range (15-40°C). (d) Similar to (c), success rates by reference temperature of trained mice with genetic knockout of the TRPV1 receptor (TRPV1-KO, n = 4, in blue) compared to wild-type littermates (WT, n = 4, in black). All data is shown as the mean success rate of all mice in that group +/− s.e.m. Significant differences between mouse groups were determined by Mann-Whitney U-Test (1 star = p < 0.05, 2 stars = p < 0.01; alpha = 0.05).
It has been reported that the TRPV1 neuron population is associated with detection of warmth16,17,22 and therefore we also set out to investigate the effect their ablation and knockout would have on thermal discrimination. To avoid the possibility of TRPV1-DTR or TRPV1 KO mice are unable to learn the initial temperature change detection protocol which uses a warming temperature change stimulus, we instead used a large cooling temperature change for training (Tref -Ttest = 40–25°C). After being trained with this protocol, WT animals learned to generalize their temperature change detection training similar to WT mice that had been trained using the warming protocol, except now these mice associated the reference and test choice ports to the relatively warmer and colder temperatures, respectively. While the differences between experimental mice and their WT littermates did not reach the same statistical significance as observed between TRPM8-DTR/KO and WT mice, the only experimental condition in which successful temperature change detection rates of TRPV1-DTR mice dropped to the chance level was between the warmest temperatures tested (Tref -Ttest = 40–35°C). This is noteworthy since this was our expectation, and temperature change detection rate of all WT control animals never dropped to the chance level at any tested temperature change (other than the no-temperature-change control condition). TRPV1-KO mice also showed relatively smaller reductions in the higher temperature ranges, though still well above the chance level and more similar to the WT littermates (Figure 3(c) and (d)). Due to the relatively low statistical power of these experiments, we cannot confidently say that TRPV1-ablation produced a stronger deficit in warm-range (40–35°C) temperature change detection than TRPV1-knockout, though this would be consistent with previous studies that showed the TRPV1 population, but not the TRPV1 receptor, is necessary for warm temperature discrimination and that input from TRPM8 contributes to warm sensation.17,22
Among the innocuous temperatures tested in this study, the apparent temperature ranges where TRPM8 and TRPV1 populations appear to be most relevant are the cooler range for TRPM8 (15–30°C) and the warmer range for TRPV1 (35–40°C). Notably, the large temperature change initially chosen in our training protocol (25–40°C) overlaps with both the cool and warm ranges. This suggests that the thermosensory information which WT mice are provided during the training phase may be derived from afferent input from both the TRPV1 and TRPM8 expressing neurons, which may explain the ability of mice trained with a large temperature conditioning stimulus to generalize their training across the entire range of temperatures tested. We wondered whether mice would still be able to generalize across this broad temperature range if the training phase only contained thermosensory information relevant for one of these two thermosensory neuron populations. Accordingly, we trained mice for temperature changes which we expected to be selective for either TRPM8- or for TRPV1-afferent input. Specifically, a training range of 15–30°C was chosen to transmit information through the TRPM8-neuron population, and a range of 35–45°C was chosen for the TRPV1-population. The later temperature change was chosen to be smaller in order to keep the temperature range in the non-noxious range (<=45°C). 41
Interestingly, unlike mice trained over the broad 25–40°C range, WT mice trained over the warm (35–45°C) range did not generalize their training to the cooler range. These mice did not appear to perform any differently than TRMP8-DTR mice trained over the same range (Figure 4(a)). Similarly, WT mice trained in the cool range did not generalize their training to the warmest range where the TRPV1 population was expected to be required and did not perform any better than their TRPV1-DTR littermates (Figure 4(b)). These results suggest, for mice, that temperature perception is separated into at least two domains – a cool range and a warm range – where a learned temperature discrimination behavior within one of these ranges does not provide generalized discrimination into the other (Figure 4(c)).
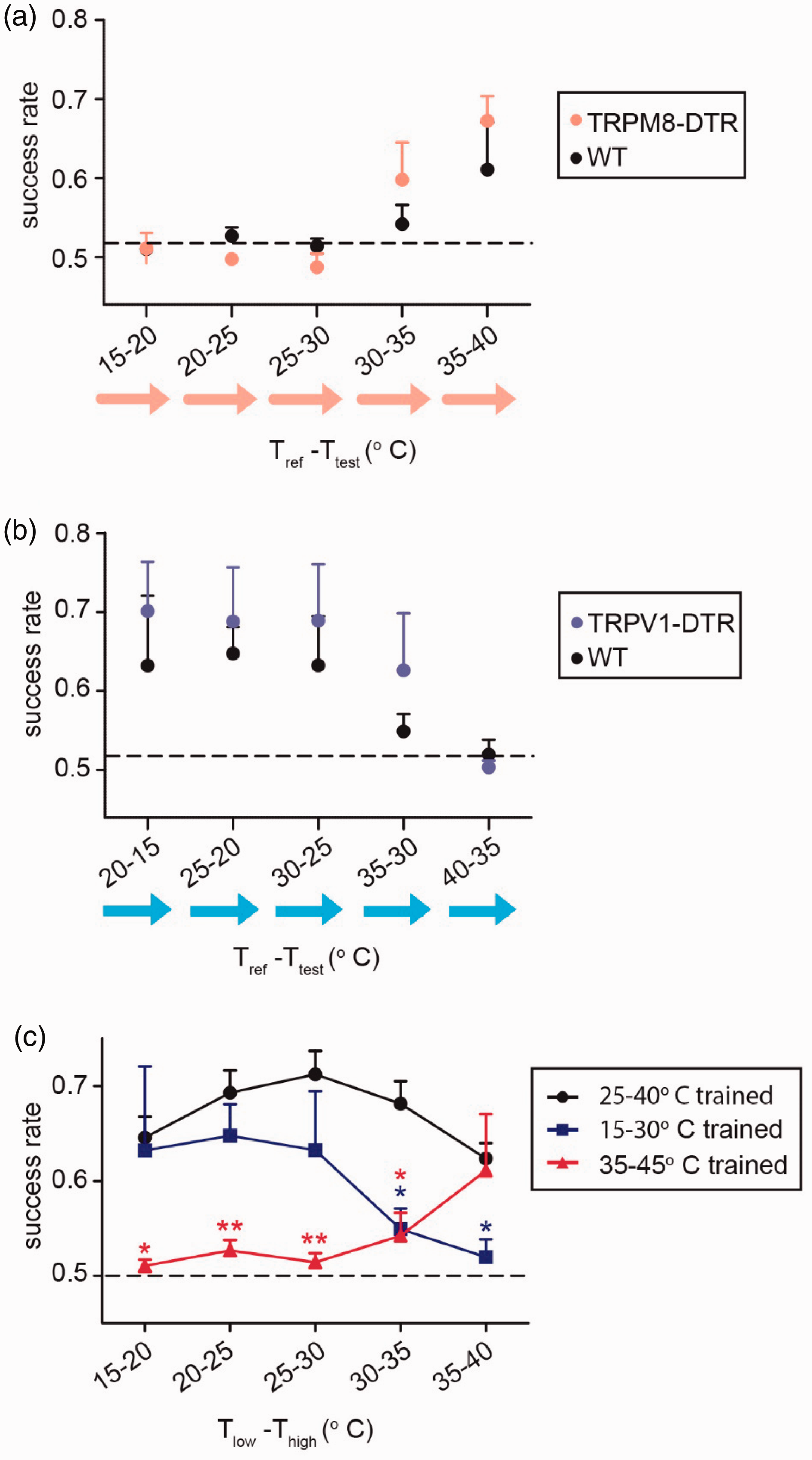
Temperature change detection learned within the warm temperature range does not generalize into learning in the cool range, and vice versa. (a) Success rates of mice with diphtheria toxin-ablated TRPM8 cells (M8DTR; n = 4, in red) and wild-type littermates (WT; n = 4, in black) trained over the 35–45°C “warm” temperature range tested in their ability to detect 5°C increases across the entire test range (15–40°C). (b) Success rates of mice with ablated TRPV1 cells (TRPV1-DTR; n = 4, in blue) and wild-type littermates (WT; n = 5, in black) mice trained over a 30–15°C “cool” temperature range tested in their ability to detect 5°C decreases across the entire test range (15–40°C). (c) WT mice trained over either a warm 35–45°C temperature range (warm trained; n = 4, in red), a cool 30–15°C temperature range (cool trained; n = 4, in blue), or all mice trained over a broad 25–40°C temperature range (broadly trained; n = 26, in black), tested in their ability to detect temperature changes across the entire testing range (15–40°C). Data of warm trained and cool trained mice are the same as those shown as WT in (a) and (b) respectively, while data for broadly trained mice includes all WT mice used throughout this paper that were trained over 25–40 or 40–25°C ranges (see Supplemental Figure 4 for all groups plotted separately). Significant differences are noted between the warm/cool trained mouse groups, as compared to the broadly trained mouse group. All success rates are calculated as the mean of each group +/− s.e.m. Significant differences between the warm/cool trained groups and the broadly trained group were determined by Mann-Whitney U-Test (1 star = p < 0.05, 2 stars = p < 0.01).
Discussion
The assay for temperature change detection used in this study is relatively unique in the field of thermosensation in that it does not rely on innate temperature preferences or to reflexive escape behaviors produced by uncomfortable or painful temperatures. Instead, the assay makes use of a very rapid temperature change stimulus to maximize the salience of innocuous thermal stimuli which by their nature may seem to be subtle and inconspicuous. Approximately 2 weeks of training sessions was required for approximately 50% of mice to successfully learn the assay. Mice were motivated to learn this task using water rewards while under a water-restriction protocol; recent research has found that different reward strategies (e.g. nutritive rewards) can result in different levels of motivation in task learning.42,43 Therefore, it is possible that a different reward scheme might have helped motivate more mice, resulting in larger groups recruited for testing. With only approximately half of mice meeting our criterion for testing in this study, some of our results must be interpreted carefully to not assume our findings are representative of all mice. For example, the mice in this study could detect temperature changes as small as 2.5°C, but this may be only be true of a fraction of mice and may not be typical of all. Indeed, when measuring the success rate versus temperature change magnitude in additional mouse groups used in this study, the success rate for 2.5°C temperature change - while nearly always greater than chance - did not reach statistical significance (Supplemental Figure 2). Notably, all WT and transgenic mouse groups trained in this study had similar fractions of recruited mice, suggesting that any selection biases is not dependent on the genotype of animals we used in our studies.
The assay we developed was designed to train mice to detect a specific conditioning stimulus; a temperature change with either an increase or decrease from the baseline “reference” temperature to the “test” temperature, as compared to a no temperature change control. While the specific values chosen for the reference and test temperatures had a significant effect on how mice generalized their training, the arrangement of those temperatures – which of the two is the reference and which is the test – did not appear to make any difference. Mice trained with a cooler reference and warmer test temperature performed in the same way to those trained with a warmer reference and cooler test temperature, with the only difference being which choice ports the mice associated with the relatively colder or warmer temperatures (Supplemental Figure 3). Additionally, mice successfully trained with a test temperature warmer than the reference temperature performed similarly when the two temperatures were switched, maintaining the association of the choice ports to whichever temperature was relatively colder or warmer (Figure 2(b)). This result provides support for our ability to compare the results from mice across groups that were trained with opposite direction temperature changes, such as the TRPM8-DTR and TRPV1-DTR mice, rather than being limited to comparing each group to their WT littermate control. However, one downside to this feature is that this assay is not capable of dissociating the detection of temperature increases or decreases; a mouse trained to detect increases (i.e. test > reference) are also apparently trained to detect decreases (reference<test), and vice versa. This may be due to the design of the training and testing trials, since all temperature increases (and decreases) during the test condition are paired with a similarly fast temperature change in the opposite direction as the test chamber returns to the reference temperature in preparation for the next trial. In terms of measuring thermal acuity, another limitation of our assay is that it does not provide information as to whether it is the magnitude of temperature change or the speed of temperature change that is most important for mice to recognize a temperature change as has been reported previously33,44 – larger magnitudes of temperature change will also cause the temperature of the test chamber to change more rapidly (Figure 2(a)). Due to these limitations, we may only be confident in this assay’s capability of determining whether mice can detect a difference between two temperatures delivered, regardless of whether it is the warming and/or cooling aspects of the switch between these, the magnitude of the difference, and/or the rate of the change between them.
Previous research shows that thermosensation can be divided into innocuous thermosensation as well as regions of painfully cold or noxious heat. 1 Here, we observed that innocuous thermosensation can be further divided into cool and warm regions associated with the TRPM8 and TRPV1 neuronal populations respectively. The TRPM8 receptor appears to be required for the TRPM8 neuron-associated range of thermosensation, as TRPM8-KO mice were equally unable to recognize temperature changes in the cooler region as mice with DT-ablated TRPM8 cells. The same may not be true for the TRPV1 receptor, in that previous studies suggest (and our study weakly supports) that while the ablation of TRPV1-expressing cells can cause a thermosensory deficit over warm temperatures, the TRPV1 receptor is not entirely necessary for 32–42°C warm perception.16,17,22 This suggests that in addition to TRPV1, there are other receptors and perhaps additional cellular mechanisms responsible for warm temperature change detection. One potential confound of our TRPM8/TRPV1 ablation strategy is that a small subset of TRPM8 trigeminal neurons are known to co-express TRPV1, which could also be feature of some thermosensitive DRG neurons which were the focus of our study since thermal stimulation was isolated to the mouse’s paws. However, since there were no clear differences in temperature change recognition between the TRPM8-DTR (which might ablate a small number of TRPV1 DRG neurons as well) and TRPM8-KO mice (which may be more selective), we are more confident that this potential confound would not have affected our results. Though it is important to note that our study likely would not have the statistical power to identify phenotypes caused by only ablating small subsets of these thermosensory populations, so we cannot rule out that neurons co-expressing TRPV1 and TRPM8 could potentially have an effect on innocuous thermosensation.
Interestingly, in this study we find that there are at least two separable domains of innocuous thermosensation – the TRPM8-mediated cool range and TRPV1-mediated warm range which appear in mice to be perceptually distinct. Wild-type mice trained in the cool range show normal cool-range temperature change detection but are unable to discriminate temperature changes in the warm range. The same appeared to be true for mice trained in the warm range in that they were incapable of generalizing their training to the cool range. Mice had to be trained across mid-range temperatures (25–40°C) which overlapped with both the warm and cool ranges to be able to detect temperature changes across the entire tested range. This suggests that temperature change in the cool and warm ranges result in qualitatively different perceptions, at least with respect to the learning paradigm used in our studies. These results also further highlight that the mammalian thermosensory system does not discriminate absolute temperature but instead functions to report relative temperature. Our results do not, however, mean that TRPM8 and TRPV1 circuits never interact with each other. Previous work has identified tonic cross-inhibition between these two circuits at the spinal level and found this to have an effect on sensitivity to painful and uncomfortable (e.g. <15°C, >45°C) temperatures and even on innocuous (20–40°C) temperature preference and thermoregulation. 45 This is not in conflict with the results of our study, since for temperature changes entirely within the cold range (for example), the tonic inhibition from the warm range’s circuit is expected to stay constant and only affect the absolute activity levels of the cold circuit, whereas temperature change detection requires some relative activity change as the temperature changes. But unlike innocuous temperature change detection in which we found that TRPM8 and TRPV1 have separate domains of influence, innocuous temperature preference appears to depend on the interaction between TRPM8 and TRPV1 circuits.
The terms “warming” and “cooling” are often used to refer to either increases or decreases in temperature, respectively, however this may oversimply and thereby miss an important component of thermosensation. Our results point to a different and perhaps more perceptually meaningful distinction that relates to what is known about the physiological responses of thermoreceptors. It has been shown that “cold sensory fibers” in mammals have maximal activity between 25–30°C46–49 and therefore, depending on the starting temperature, these fibers can be either activated or inhibited, in a similar way, “warm fibers” have been described to also have bell-shaped activation curves to temperature but at higher temperatures (approximately 40°C) and are oppositely affected by of temperature change.50–52 Therefore, both increases and decreases in temperature have opposite effects on cold-fibers to warm-fibers and these effects are temperature dependent and likely to some extent these inputs are compared with each other.17,22 This may be the reason why mice that are trained with a broad conditioning temperature range of 25–40°C were capable of distinguishing warming and cooling across the full range while, mice trained on restricted 15–30°C (cool) and 35–45°C (warm) were unable to generalize beyond their trained temperature ranges. This model of how sensory input at the periphery can shape behavioral responses suggests that the cold and warm signals are transmitted through pathways that remain distinct from each other at the level of what is recognized by the mouse in our operant assay.
We developed an operant behavioral method and used it to investigate the contributions of molecularly defined thermoreceptors. Additionally, controlling the stimuli used during operant conditioning shed light on how different sensory channels are perceptually organized. This type of assay may aid in understanding the contribution of other receptors, spinal, and supraspinal neuronal networks in thermosensation as it permits a more direct examination of sensory perception.
Footnotes
Author Contributions
MI designed and built the testing apparatus; designed, performed, and analyzed all behavioral experiments. MAH and MI conceived the project and wrote the manuscript.
Declaration of Conflicting Interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the intramural research program of the National Institute of Dental and Craniofacial Research, National Institutes of Health, project ZIADE000721-20 (MAH).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
