Abstract
Background
Chronic postoperative pain affects approximately 20% of patients with knee osteoarthritis after total knee replacement. Circulating microRNAs can be found in serum and might act as biomarkers in a variety of diseases. The current study aimed to investigate the preoperative expression of circulating microRNAs as potential predictive biomarkers for the development of chronic postoperative pain in the year following total knee replacement.
Methods
Serum samples, collected preoperatively from 136 knee osteoarthritis patients, were analyzed for 21 circulatory microRNAs. Pain intensity was assessed using a visual analog scale before and one year after total knee replacement. Patients were divided into a low-pain relief group (pain relief percentage <30%) and a high-pain relief group (pain relief percentage >30%) based on their pain relief one year after total knee replacement, and differences in microRNAs expression were analyzed between the two groups.
Results
We found that three microRNAs were preoperatively dysregulated in serum in the low-pain relief group compared with the high-pain relief group. MicroRNAs hsa-miR-146a-5p, -145-5p, and -130 b-3p exhibited fold changes of 1.50, 1.55, and 1.61, respectively, between the groups (all P values < 0.05). Hsa-miR-146a-5p and preoperative pain intensity correlated positively with postoperative pain relief (respectively, R = 0.300, P = 0.006; R = 0.500, P < 0.001).
Discussion
This study showed that patients with a low postoperative pain relief present a dysregulation of circulating microRNAs. Altered circulatory microRNAs expression correlated with postoperative pain relief, indicating that microRNAs can serve as predictive biomarkers of pain outcome after surgery and hence may foster new strategies for preventing chronic postoperative pain after total knee replacement (TKR).
Introduction
Osteoarthritis (OA) is the most frequent painful musculoskeletal diagnosis in the elderly population and the most prominent cause of disability. 1 Total knee replacement (TKR) is the end-stage treatment of knee OA and provides pain relief for the majority of patients with severe OA. However, around 20% of knee OA patients will experience chronic postoperative pain after TKR surgery.2,3 Several studies have found that high preoperative pain intensities4,5 and sensitization of central pain pathways 6 act as predictors for chronic postoperative pain following TKR.
Recently, an exploratory study found that certain preoperative pro-inflammatory cytokines were associated with the development of chronic postoperative pain following TKR. 7 There is evidence that pro-inflammatory and anti-inflammatory cytokines are involved in pain. 8 Consequently, there is increasing interest in the evaluation of small non-coding RNAs, defined by Sommer et al. as “master switches,” acting in the development and maintenance of inflammation and pain. 9 The action of circulating non-coding RNAs, already verified in pathologies such as cancer and autoimmune disease, is receiving increasing attention in the pain field.10–12 In this context, studies have shown that small non-coding RNAs are directly involved in the production of cytokines.13,14
This indicates that the preoperative assessment of non-coding RNAs could be a potential prognostic biomarker venue in terms of assessing the risk for chronic postoperative pain following TKR and may represent a way to explore new therapeutic opportunities.
MicroRNA (miRNA) is a group of small non-coding RNAs (∼20–25 nucleotides) 15 involved in post-transcriptional gene expression regulation, with more than 7000 miRNAs recognized in human between precursor and mature structure. 16 Unlike the majority of miRNAs, which are usually detectable intracellularly, circulating or extracellular miRNAs have also been found in the interstitial environment, cell culture media, and in different biological fluids such as serum or plasma.17–20 Moreover, circulating miRNAs are known to be protected from the action of degradation enzymes, due to their inclusion in extracellular microvesicles 18 or formation of protein-miRNA complexes. 19 This makes the circulating miRNAs highly stable and their expression can be quantified in body fluids, making them suitable to act as potential biomarkers. 21 Circulating miRNAs have been found to be dysregulated in patients with various chronic pain conditions including complex regional pain syndrome, 22 migraine, 23 peripheral neuropathy, 24 and fibromyalgia when compared to healthy controls. 24 Recently, a study highlighted the different functions as well as potential diagnostic and predictive value of miRNAs in various pain states, 25 but no study thus far has assessed the potential use of circulating miRNA as predictive biomarkers for chronic postoperative pain. The current study aimed to investigate the preoperative expression of circulating miRNAs validated or predicted in previous study to be associated with OA pain and inflammation, as potential predictive biomarkers for the development of chronic postoperative pain one year after TKR.
Material and methods
Patients
One hundred thirty-six patients with a knee osteoarthritis (KOA) scheduled for TKR were recruited consecutively from the outpatient clinic at Hospital Vendsyssel, Frederikshavn, Denmark, and tested for preoperative miRNAs expression in serum. Patients with other diagnosed pain conditions (e.g., hip OA, rheumatoid arthritis, fibromyalgia, and neuropathic pain), sensory dysfunction, or mental impairment were excluded from the study. Radiological KOA progression was evaluated using the Kellgren and Lawrence (KL) score. 26 The KL score is a radiological assessment score of knee OA. The score ranges from 0 (no OA) to 4 (severe OA). 26
The patients were asked not to take any analgesic medication, such as non-steroidal anti-inflammatory drugs or paracetamol, 24 h before the pain scoring examination. The study was approved by The North Denmark Region Committee on Health Research Ethics (N-20120015) and conducted in accordance with the Helsinki Declaration. All patients read and signed an informed consent form prior to enrollment.
Pain assessment
Before surgery and one year after, the peak pain intensity within the last 24 h was collected (using the visual analog scale, hereafter VAS). The patients were asked to rate their pain intensity on the VAS scale from “0–10” where “0” represents “no pain” and “10” represents “worst pain imaginable.” The patients were divided into two groups based on the percentage of postoperative pain relief (the difference between pre- and postoperative VAS scores divided by preoperative VAS score). Patients achieving >30% pain relief after TKR were assigned to the “high-pain relief” group, whereas patients who achieved <30% pain relief were assigned to the “low-pain relief” group. This classification for the patients was based on the minimum clinical relevance observed in previous study. 27
Blood withdrawal and microRNA isolation
Venous blood was collected following standard procedures from patients before they underwent the surgery, between 07:30 and 09:00 in the morning. For the real-time PCR (qRT-PCR), 9 ml of whole blood was withdrawn in an untreated tube. After collection, the whole blood was left at room temperature for 15 min and allowed to clot; the serum was then separated from clotted cells by low-speed centrifugation (3000 rpm) for 15 min to allow the serum separation. The serum obtained was stored at −80°C until used. After thawing on ice, circulating miRNAs were isolated from 200 µl of serum using miRNeasy Serum/Plasma Advanced Kit (QIAGEN, Hilden, Germany) according to the manufacturer’s instructions. As a positive control, 3.5 µl (1.6 × 108 copies/µl) of Serum/Plasma Spike-in Control cel-miR-39-3p (QIAGEN, Hilden, Germany) was added to each reaction during the extraction protocol.
MicroRNA array expression
Isolated miRNAs were retrotranscribed to cDNA, using miScript II RT Kit (QIAGEN, Hilden, Germany) in a thermocycler (Applied Biosystems, Foster City, CA, USA) at 37°C for 1 h with heat inactivation of the retro-transcriptase at 95°C for 5 min. To perform qRT-PCR, miScript SYBR® Green qPCR Kit (QIAGEN, Germany) was added to each sample. Furthermore, this mixture was aliquoted into the wells of Custom miScript miRNA PCR Array (QIAGEN, Germany). The cDNA for every patient was analyzed for 21 putative miRNAs candidates known to be associated with inflammation, pain, and cartilage degeneration. Plates were pre-manufactured by QIAGEN and contained specific primers for miRNAs of interest based on previous evidence of their detection in human body fluids, and their involvement in pathological OA pathways, inflammation process, and findings of direct connections with pain sensation (see Table 1). Thermal cycling conditions consisted of a hot start at 95°C for 15 min followed by 40 cycles of each qPCR step: (denaturation) 94°C for 15 s, (annealing) 55°C for 30 s and (extension) 70°C for 30 s (AriaMx Agilent Technologies, Santa Clara, CA, USA). A melting curve analysis was carried out to ensure the specificity of the corresponding qRT-PCR reactions. The Cycle quantification (Cq) data obtained setting a single threshold of 12 between the assays using Agilent Aria software 5.1 (Agilent Technologies, Santa Clara, CA, USA), exported to a Microsoft Excel file (Microsoft 2016) and subsequently uploaded in Statistical Package for Social Sciences (SPSS, v. 25, IBM).
MicroRNA candidates list and list of microRNA sequences contained in the RT-qPCR customized array.
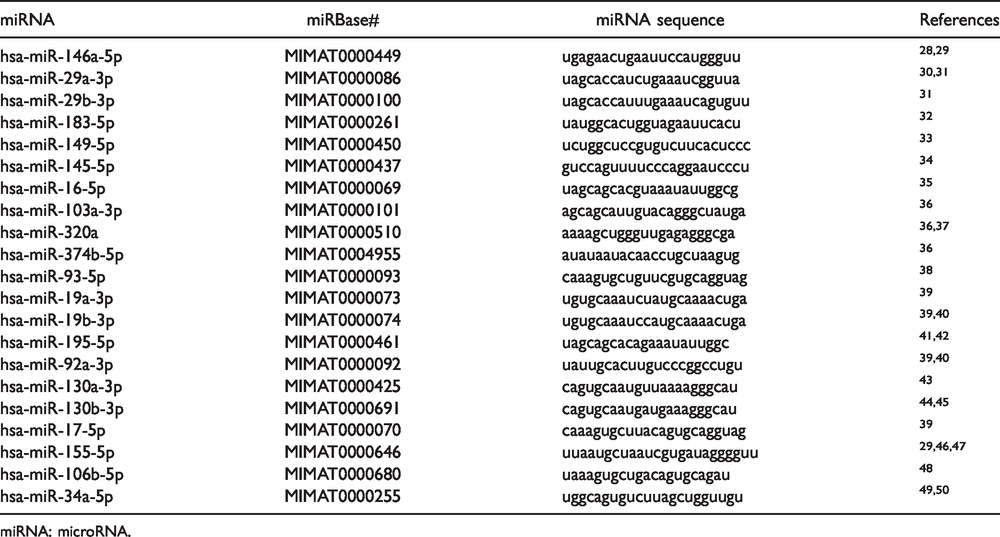
miRNA: microRNA.
Statistical analysis
Before the statistical analysis, the raw Cq values were normalized using the Global Mean normalization method on the expression of all miRNA assays. 51 Initially, patient demographics were calculated for the two groups with chi-square tests for categorical data and t-tests for continuous data. The qRT-PCR data were transformed into the fold change domain, defined as measure describing how much a quantity changes between an original and a subsequent measurement. Unpaired Student’s t-tests were used to compare the normalized Cq values for each miRNA between the two groups of patients. The data have been visualized in a volcano plot, combining P values (y-axis) with the fold changes (x-axis), thus highlighting miRNAs with higher or lower levels of expression. Correlations were conducted using Pearson’s correlation coefficients. Linear regression models were constructed to predict the pain relief following TKR, using the preoperative pain intensity and the miRNAs, which were significantly different between the two groups. Furthermore, linear regressions were utilized to identify independent preoperative predictive factors based on the miRNAs. Data analysis was performed in SPSS software V.25 (IBM, Armonk, NY, USA). P < 0.05 was considered significant.
In silico target prediction
A gene target analysis was conducted for the differentially expressed miRNAs to identify genes that represent putative targets. In silico prediction analyses were performed using DIANA-Tool TarBase v.8, 52 choosing the predicted targeted mRNA genes by their prediction score with a value between 0.5 and 1.0. To determine the biological relevance of the predicted mRNA genes targeted by the miRNAs, gene ontology (GO) analysis was subsequently explored using PANTHER classification system 53 and Reactome (www.reactome.org). Manual curation of targets associated with the immune system process, biological regulation, cellular process, and response to stimuli was performed.
Results
Demographics
Of 136 patients with KOA (82 female), 114 patients (84%) had pain relief percentage >30% (high-pain relief group) and 22 patients (16%) had pain relief percentage <30% (low-pain relief group). No significant differences were found between the two groups regarding preoperative patient demographics or KL (Table 2). Moreover, no statistical difference was found for preoperative pain intensity in patients when subdivided for gender (P = 0.944). From the 82 females enrolled in the study, 82% were in the high-pain relief group and 18% in the low-pain relief group. From the 54 men enrolled in the study, 87% were in the high-pain relief group and 13% in the low-pain relief group.
Demographic characteristics of sub grouped patients with severe knee osteoarthritis before total knee replacement.
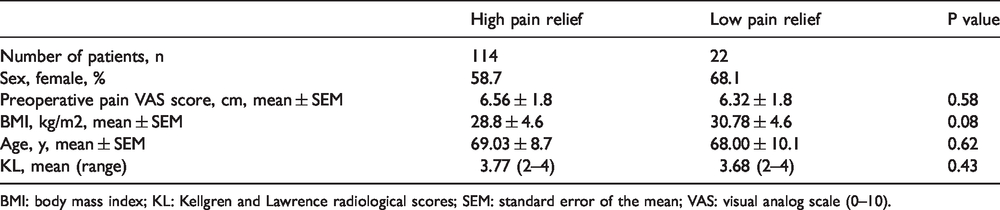
BMI: body mass index; KL: Kellgren and Lawrence radiological scores; SEM: standard error of the mean; VAS: visual analog scale (0–10).
MiRNA serum assays quality control
MiRNAs amplification cut-off was set at 35 cycles (Figure S1). Moreover, to avoid “false positive” results, a melt curve analysis was carried out to ensure the specificity of the corresponding RT-qPCR reactions. Figure S2 shows that the assessed miRNAs peak at the same time as the positive control cel-miR-39-3p (both peak at temperatures of 76.5°C). Conversely, the PPC (internal PCR positive control) peaked at higher temperatures (81.5°C). The single aligned peak for assessed miRNA and cel-miR-39-3p suggests that no unspecific amplicons were produced.
Differential expression of miRNAs in patients with low-pain relief following TKR
Three miRNAs were differentially expressed between low-pain relief and high-pain relief group (a volcano plot is displayed in Figure 1). Hsa-miR-146a-5p (fold change = 1.50 ± 0.86 SD, P value = 0.021), hsa-miR-145-5p (fold change = 1.55 ± 1.24 SD, P value = 0.037), and hsa-miR-130b-3p (fold change = 1.61 ± 1.4 SD, P value = 0.039) had a significantly higher expression in the low-pain relief group relative to the high-pain group.

Volcano plot of the differential serum miRNAs expression. Statistical significance versus fold change was showed on the y- and x-axes, respectively. Volcano plot of data shows the fold change of the 21 assessed miRNAs in patients with less than 30% pain relief (low-pain relief group) compared with patients with more than 30% pain relief (high-pain relief group) following total knee replacement. Three miRNAs exhibited higher significantly (black dots) expression (P < 0.05) between the two groups.
Prediction of postoperative pain relief
Pooling all patient data showed significant Pearson correlations between pain relief and preoperative pain intensity (R = 0.500, P < 0.001) and hsa-miR-146a-5p (R = 0.300, P = 0.006). Furthermore, linear regression models were established to investigate the predictive value using the significant miRNAs hsa-miR-146a-5p, hsa-miR-130b-3p, hsa-miR-145-5p, and preoperative pain intensity. Model 1 consisted of all the parameters with a predictive value (R2) of 30% and identified preoperative pain intensity (P < 0.001) as significant factors (Table 3). Model 2 was constructed using a backward selection of the parameters included in model 1 and identified preoperative pain intensity (P < 0.001) as a significantly independent parameter for postoperative pain relief prediction with a value of R2 of 30% and showed a trend of hsa-miR-146a-5p (P = 0.06) (Table 3, model 2).
Linear regression models of preoperative pain intensity, hsa-miR-146a-5p, hsa-miR-130b-3p, and hsa-miR-145-5p aiming to predict postoperative pain relief in patients with knee osteoarthritis following total knee replacement.

Note: Model 1 consists of all the parameters and model 2 is constructed using backward selection. R2 indicates the combined predictive value. miRNA: microRNA.
Potential target mRNA genes of differentially expressed circulating miRNAs
In silico target prediction analyses, through DIANA-TarBase v.8, were performed to highlight the potential mRNA targets of the dysregulated miRNAs. An mRNA was considered a potential target if it has been validated with high/low throughput experiment and if its prediction score was between 0.5 and 1.0. Using these criteria, the potential number of mRNA targets for the hsa-miR-146a-5p was 103, for hsa-miR-145-5p was 35, and for hsa-miR-130b-3p was 337. All the targeted genes were analyzed for the GO analysis of the biological processes (Figure 2). The majority of the identified gene targets were involved in various cellular and metabolic processes. Our interest in qualitative exploration were genes related to sensory transduction, inflammatory response, and neuronal sensitization. The interleukin-1 receptor-associated kinase 1 (IRAK1) gene was found to be targeted by hsa-miR-146a-5p and is involved broadly in cytokine-mediated signaling as well as the toll-like receptor signaling pathways.54,55 Moreover, the hsa-miR-145-5p, which was expressed at higher levels in the low-pain relief group, had transcription-factor JunB (JUNB) as a target. This transcription factor is part of the family of the Jun-protein normally identified as a transcriptional repressor; however, there is evidence that shows its involvement in the positive transcription regulation of IL-2, IL-4, IL-6, and TNF-α56–58 and its activation by external or endogenous stimuli leads to a cellular control, regulating pathway of differentiation or cell death (osteoclast and osteoblast genesis and proliferation).59–61

Gene ontology analysis. (a) Bar chart of biological process for genes regulated by hsa-miR-146a-5p. (b) Bar chart of biological process for genes regulated by hsa-miR-145-5p. (c) Bar chart of biological process for genes regulated by hsa-miR-130b-3p.
Discussion
The current study is the first to assess preoperative circulating miRNAs as serological preoperative predictors for postoperative pain relief one year after TKR in painful knee OA patients. Higher levels of hsa-miR-146a-5p, hsa-miR-145-5p and hsa-miR-130b-3p were demonstrated in patients with low-pain relief as compared to patients with high postoperative pain relief. Furthermore, preoperative pain intensity was found to be an independent predictor of postoperative pain relief with a trend for hsa-miR-146a-5p.
The current analysis highlights that hsa-miR-146a-5p, hsa-miR-145-5p, and hsa-miR-130b-3p showed higher preoperatively levels in serum of patients with low postoperative pain relief one year after TKR. All of these miRNAs have been assessed in previous study, where their action to different pathological conditions, for example, cancer, has been proved through high-throughput analysis.62–64 As previously stated, these miRNAs are involved in numerous processes that can include metabolic, cellular, or pathological processes. In this study, their involvement in inflammatory processes regulation seemed to be of considerable interest since inflammation is one of the main actors for the sensitization of peripheral nerve endings that leads to pain. 8
Hsa-miR-146a-5p is codified by a gene located on chromosome 5 and its mature product differs only by 2–6 nucleotides in the 3′ region from the other products of miRNA-146’s family. 65 It has been shown that hsa-miR-146a-5p expression is regulated by nuclear factor kappa B (NFκB), induced by the action of proinflammatory mediators and in response to the activation of the innate immune response in monocytes and macrophages. 66 Hsa-miR-146a-5p has been shown to be upregulated in synovial tissues of patients with rheumatoid arthritis when compared to healthy controls, and its level of expression is stimulated by inflammatory cytokines such as tumor necrosis factor α (TNF-α) and Interleukin-1β (IL-1β). 67 A previous study has suggested that suggest that hsa-miR-146a-5p is expressed in OA cartilage at higher levels than in normal cartilage. 68 In addition, a study found higher levels of miR-146-5p in peripheral human blood mononuclear cells in early-stage OA patients when compared with healthy controls, indicating that the dysregulation of miR-146-5p is important for the initial stage of OA. 69 In a mouse model, Lu et al. showed higher levels of miR-146-5p may partially attenuate neuropathic pain conditions in rats by decreasing the expression of TNF receptor-associated factor 6 (TRAF6). 70 Hsa-miR-146a-5p also governs feedback of cytokine expression, and consequently the production of cartilage–degrading enzymes, such as matrix metalloproteinase (MMPs) and a disintegrin and metalloproteinase with thrombospondin motifs (ADAMTS). 28 The current study shows that hsa-miR-146a-5p is present at higher levels circulating in serum of patients with low chronic postoperative pain relief and shows a trend for this miRNA as an independent predictor of postoperative pain relief.
Recent studies have highlighted that high levels of TNF-α and IL-1β induce the expression of hsa-miR-145-5p, a pathway involved in the pathogenesis of OA.34,71 Previously, hsa-miR-145-5p has also been identified as a regulator of chondrogenic differentiation, and miRNA microarray data show that hsa-miR-145-5p is significantly upregulated in human chondrocytes isolated from KOA patients at the last stage (K&L scale III and IV) of the pathology. 71 The expression of hsa-miR-145-5p is sensitive to the level of TNF-α and is implicated in modulating the expression of TNF-α-induced enzymes such as MMPs and ADAMTS, which are involved in the cartilage disruption and OA progression. 34 Furthermore, the hsa-miR-145-5p expression is significantly upregulated in OA chondrocytes in response to IL-1β stimulation, which is important for cartilage degradation.71–73Has-miR-145-5p have also been found to be involved in another painful condition wherein cerebrospinal fluid of patients with fibromyalgia was strongly downregulated compared with healthy controls, but higher levels of it were found to correlate to higher levels of pain and fatigue. 74 The present results show preoperative higher expression levels of hsa-miR-145-5p in the serum of patients with post-operative low-pain relief, suggesting that hsa-miR-145-5p might increase and sustain the inflammation in low postoperative pain relief patients causing their painful condition. However, further studies to confirm this hypothesis are needed.
Hsa-miR-130b-3p is a well-known marker, studied in cellular and animal models, involved at different level in the progression and maintenance of different kinds of cancer,75,76 but it has also been shown to be involved in chondrogenesis and osteogenesis, 77 associated with obesity, insulin resistance, and other pathophysiological processes.45,78 A previous study has shown how inflammatory signals, such as TNFα and IL-1β, induce its action in adipose inflamed tissue. 44 So far, the presented results show a higher expression of hsa-miR-130b-3p in patients with low chronic postoperative pain relief. Future studies are needed to demonstrate whether the involvement of this miRNA can be directly related to the postoperative pain condition in patients with KOA and then serve as a new biomarker for it.
In this study, before the qRT-PCR, a per sample quantification of the extracted total RNA was not performed; however, by employing a standardized kit and several internal control assays (PPC, miRTC, cel-miR-39-3p), the quality of the qRT-PCR was validated. Data normalization, a considerable challenge for circulating miRNA data analysis given the lack of robust house-keeping genes,79,80 was performed using the common global mean normalization approach. 51 Moreover, adjustments for multiple comparisons using the Benjamini–Hochberg method (false discovery rate, FDR) was not performed due to the exploratory nature of this study. Many cell types are capable of secreting and receiving circulating free and exosomal miRNAs,18,19,21 which makes it impossible to determine the tissue origin as well as the recipient cells of the presently observed dysregulated miRNAs. Future research on the transporting mechanisms of circulating miRNAs is needed. With this in mind, in silico target predictions should be interpreted with caution until luciferase assays have been conducted on suspected miRNA-mRNA interactions of interest. In addition, interactions between miRNA and circulating metabolites released in the blood stream by any pharmaceutical treatment was not evaluated.
The current study utilized a 30% decrease in pain after surgery as a cut-off for chronic postoperative pain. This yielded 16% of patients with chronic postoperative pain, which is in line with the overall risk for chronic postoperative pain following TKR, 3 but it is important to note that different cut-offs have been utilized previously.4,5
Results obtained from this study and the biological pathways potentially involved in the action of miRNAs highlighted need to be better investigated and validated in further studies, and a second independent cohort of patients. Females were frequent in the current study and further research is encouraged to address gender differences in expressions of miRNA in relation to chronic postoperative pain.
In conclusion, this is the first study to show that postoperative pain is associated with specific preoperative serum circulating miRNA signatures. In the subset of patients who developed chronic pain one year after TKR, hsa-miR-146a-5p, hsa-miR-145-5p, and hsa-miR-130b-3p showed higher levels of expression. Furthermore, a prediction model shows that preoperative pain intensity is an independent predictive factor for postoperative pain relief and highlights a trend for hsa-miR-146a-5p, although further validation analysis is needed in this regard. This exploratory study gives the first insight into preoperative circulating miRNAs dysregulation and how they can serve as potential biomarkers for postoperative pain condition.
Supplemental Material
sj-pdf-1-mpx-10.1177_1744806920962925 - Supplemental material for Preoperative serum circulating microRNAs as potential biomarkers for chronic postoperative pain after total knee replacement
Supplemental material, sj-pdf-1-mpx-10.1177_1744806920962925 for Preoperative serum circulating microRNAs as potential biomarkers for chronic postoperative pain after total knee replacement by Rocco Giordano, Kristian Kjær Petersen, Hjalte Holm Andersen, Jacek Lichota, Massimiliano Valeriani, Ole Simonsen and Lars Arendt-Nielsen in Molecular Pain
Supplemental Material
sj-pdf-2-mpx-10.1177_1744806920962925 - Supplemental material for Preoperative serum circulating microRNAs as potential biomarkers for chronic postoperative pain after total knee replacement
Supplemental material, sj-pdf-2-mpx-10.1177_1744806920962925 for Preoperative serum circulating microRNAs as potential biomarkers for chronic postoperative pain after total knee replacement by Rocco Giordano, Kristian Kjær Petersen, Hjalte Holm Andersen, Jacek Lichota, Massimiliano Valeriani, Ole Simonsen and Lars Arendt-Nielsen in Molecular Pain
Supplemental Material
sj-pdf-3-mpx-10.1177_1744806920962925 - Supplemental material for Preoperative serum circulating microRNAs as potential biomarkers for chronic postoperative pain after total knee replacement
Supplemental material, sj-pdf-3-mpx-10.1177_1744806920962925 for Preoperative serum circulating microRNAs as potential biomarkers for chronic postoperative pain after total knee replacement by Rocco Giordano, Kristian Kjær Petersen, Hjalte Holm Andersen, Jacek Lichota, Massimiliano Valeriani, Ole Simonsen and Lars Arendt-Nielsen in Molecular Pain
Footnotes
Acknowledgments
The Danish Rheumatism Association and The Innovation Fund Denmark (j.no. 136–2014-5) are acknowledged for providing the opportunity to conduct the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Danish National Research Foundation (DNRF121) supports the center for Neuroplasticity and Pain. HHA received support from the EliteForsk Travel Stipend (2016) awarded by the Danish Ministry of Science and Higher Education as well as the Spar Nord Foundation’s Research Award 2018. KKP received support from the Aalborg University Talent Management Programme (2018). LAN received support for Shionogi Science Program and TaNeDS Program.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
