Abstract
The neurohypophysial hormone oxytocin (OXT) is synthesized in the hypothalamic paraventricular and supraoptic nuclei. Recently, some studies have considered OXT to be important in sensory modulation and that the OXT protein is upregulated by acute and chronic nociception. However, the mechanism by which OXT is upregulated in neurons is unknown. In this study, we examined the resting membrane potentials and excitatory postsynaptic currents in OXT-ergic neurons in the paraventricular nucleus in adjuvant arthritis rat model, a model of chronic inflammation, using whole-cell patch-clamping. Transgenic rats expressing OXT and monomeric red fluorescent protein 1 (mRFP1) fusion protein to visualize the OXT-ergic neurons were used, and the OXT-mRFP1 transgenic rat model of adjuvant arthritis was developed by injection of heat-killed
Keywords
Introduction
Oxytocin (OXT), a neurohypophysial hormone, is synthesized in the hypothalamic paraventricular nucleus (PVN) and supraoptic nucleus (SON) and enters the systemic circulation from the posterior pituitary. The functions of OXT are classically considered to be the contraction of the uterus and the milk reflex during lactation. Recently, it has been shown that OXT is also involved in maternal bonding, sexual behavior, and social affiliation.1,2 Furthermore, some studies have suggested that OXT is important in sensory modulation.
3
For example, OXT attenuates inflammation and myeloperoxidase activity induced by the subcutaneous injection of carrageenan
4
; subarachnoid administration of OXT lowers the threshold of pain,
5
and transient OXT administration abolishes hypersensitivity in the nerve injury rat model.
6
In addition, it is known that the activity of OXT-ergic neurons is upregulated by various stresses, such as conditioned fear (environmental stimuli previously paired with foot shocks), unconditioned fear (intermittently applied foot shocks), noxious stimuli,7–10 and morphine withdrawal.
11
A previous study reported that OXT was upregulated in the PVN, SON, posterior pituitary, and the dorsal horn of the spinal cord in rats with adjuvant arthritis (AA), which is a well-known chronic pain model.
12
However, the mechanism by which OXT-ergic neuron activity is upregulated is not known.
The hypothalamic PVN generates a homeostatic response. 14 It has been shown that neuroendocrine-autonomic integration is a homeostatic response in the PVN, 15 and that the activity-dependent dendritic release of OXT from the PVN acts diffusely to increase the activity of presympathetic neurons. 14 The dendrites release OXT, which then functions as a paracrine or autocrine signal at the site of release. 16 However, the mechanism of communication between OXT-ergic neurons is not well understood.
In this study, we investigated excitatory postsynaptic currents (EPSCs) in OXT-ergic neurons in the PVN in an AA rat model using whole-cell patch-clamping. A previous study demonstrated that
Materials and methods
Animals
We used OXT-mRFP1 transgenic rats (Wistar, male, aged six to eight weeks and weighing 200–300 g) that were bred and maintained as described previously.
18
All rats received food and water ad libitum and were maintained on a 12:12 h light/dark cycle (lights on at 07.00 h) at 22°C to 25°C, with three rats housed per plastic cage. PCR for genomic DNA was performed for all rats to confirm the presence of the
AA model
We intracutaneously (i.c.) injected heat-killed
Slice preparations
The rats were killed by decapitation on day 15 postinjection (vehicle or heat-killed
In vitro patch-clamp recordings
We placed the brain slices onto a glass-bottomed chamber and fixed the brain slice using a grid of parallel nylon threads supported by the weight of the C-shaped stainless steel. A low-pressure aspiration system kept the solution level constant. To identify the mRFP1-positive neurons, an upright microscope (BX-50, Olympus) and infrared differential interference contrast (DIC) optics with an mRFP1 filter (Olympus) were used. We acquired signals filtered at 3 kHz with a patch-clamp amplifier (Axopatch 200B; Axon Instruments, Sunnyvale, CA, USA) and digitized it at 1 kHz with an analog to digital converter (Digidata 1440 A; Axon Instruments). Spontaneous EPSCs (sEPSCs) were recorded from OXT-mRFP1 neurons in the magnocellular PVN (mPVN) of the hypothalamus. The neurons were voltage-clamped at −60 mV for sEPSCs and miniature EPSCs (mEPSC) or 0 mV for miniature inhibitory postsynaptic currents (mIPSCs). We filled the recording pipettes (3–6 MΩ) with a solution containing 145 mM K-gluconate, 1 mM MgCl2, 5 mM NaCl, 10 mM HEPES, 2 mM Mg-ATP, 0.2 mM EGTA, and 0.1 mM Na3-GTP (adjusted to pH 7.2 with KOH). The membrane potential was held at −60 mV, except during these experiments. We waited for 5 min after the formation of whole-cell recordings to allow the resting membrane potentials (RMPs) to stabilize, unless stated otherwise. Using the property that glutamic voluntary quantum release is insensitive to Na+ channel interception, we used tetrodotoxin (1 μM) to separate mEPSCs from other currents. mIPSCs were recorded with bath presence of cyanquixaline (20 µM), (2 R)-amino-5-phosphonopentanoate (50 µM), and holding voltage 0 mV. The patch electrode internal solution (112 mM Cs-gluconate, 5 mM TEA-Cl, 3.7 mM NaCl, 10 mM HEPES, 2 mM Mg-ATP, 0.2 mM EGTA, 0.3 mM Na3-GTP, and 5 mM QX-314, pH adjusted to 7.2 using CsOH) was used for recording mIPSCs.
Drug application
GABAA receptor antagonist (picrotoxin), OXT, and Nω-Nitro-L-arginine methyl ester hydrochloride (L-NAME) were obtained from Sigma Aldrich (St. Louis, MO, USA). The OXT receptor antagonist (L-368,899) and cannabinoid receptor 1 antagonist (AM 251) were obtained from Tocris Cookson (Bristol, UK). Immediately before use, all drugs used were diluted from stock solutions to the final desired concentration in ACSF.
Data analysis
The data were collected and analyzed with Clampex 10.7 and Clampfit 10.7 (Molecular Devices, San Jose, CA, USA). An unpaired student’s
Results
In vitro whole-cell patch-clamp recording of the hypothalamic mPVN in the OXT-mRFP1 transgenic rat
Whole-cell patch-clamp recordings of OXT-mRFP1 neurons were performed in the mPVN of OXT-mRFP1 transgenic rats (Figure 1(A), left). The mRFP1 fluorescence was observed in the PVN by fluorescence microscopy (Figure 1(A), right). The OXT-mRFP1 neurons were distinguishable from other neurons by DIC fluorescence in the PVN (Figure 1(B), a to f).
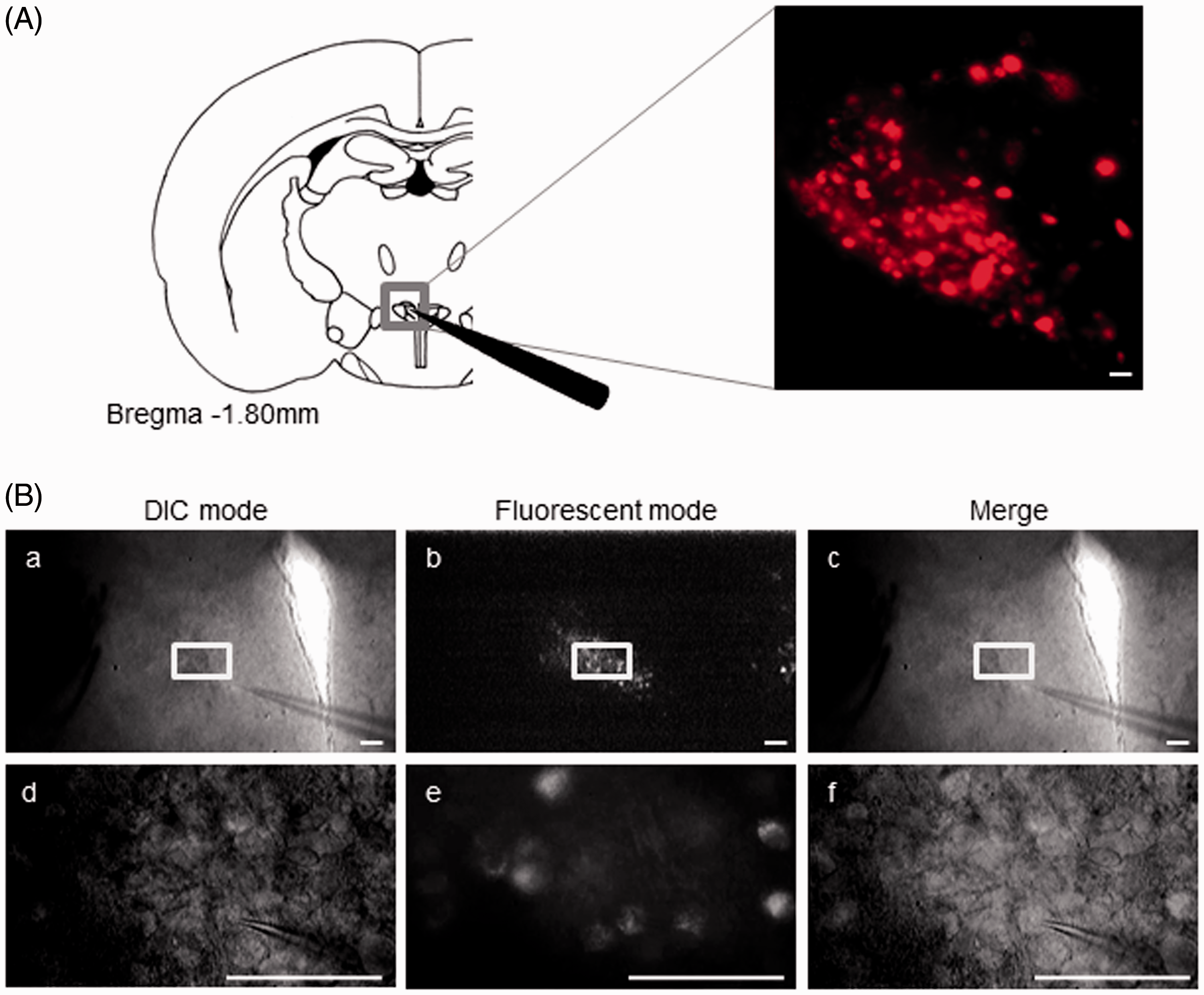
Electrophysiological response of OXT-mRFP1 neurons in the PVN using whole-cell patch-clamp recordings
We confirmed AI to evaluate the degree of inflammation on the day of the experiment. AI of AA rats was significantly higher than in the control rats (control group: 0,
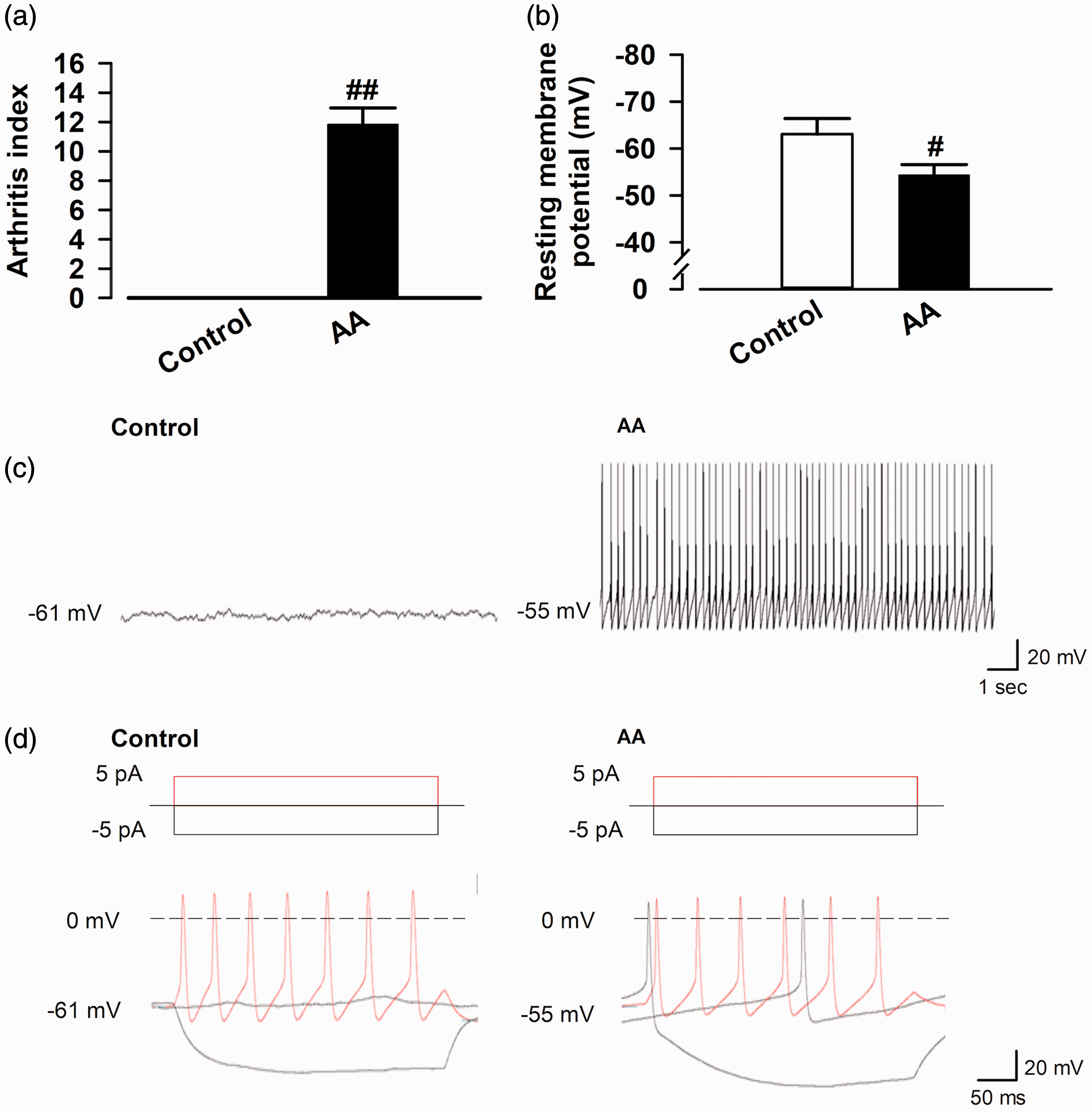
Electrophysiological response of OXT-mRFP1 neurons in the magnocellular PVN using whole-cell patch-clamp recordings. (a) The arthritis index in control and AA rats (control group:
To examine whether the synthesized endogenous OXT affects the RMPs, we recorded the RMPs following the bath application of 1 µM OXT (5 min) to the OXT-mRFP1 neurons in the mPVN of control and AA rats. We show representative RMPs recorded in the OXT-mRFP1 neurons in slices from the control and AA rats (Figure 3(a), top and bottom); 1 µM OXT application increased the RMPs in slices from the control rats (ΔRMPs control group: 3.47 ± 1.80 Hz,

Effect of bath application of OXT on RMPs in OXT-mRFP1 neurons in slices from control and AA rats. (a) Example RMPs traces in the OXT-mRFP1 neurons in slices from control (top) and AA (bottom) rats. (b) Summary RMPs change data of OXT (1 μM) in control and AA rats (control group,
Next, to examine whether excitatory presynaptic transmitter release changed in the OXT-ergic neurons after chronic inflammation, we recorded sEPSCs and mEPSCs in the OXT-mRFP1 neurons of the mPVN in control and AA rats. We show representative sEPSCs and mEPSCs recorded in the OXT-mRFP1 neurons in slices from control and AA rats at a holding potential of −60 mV, and a cumulative histogram of the interevent interval and amplitude (Figure 4(a), (b), (d), and (e)). The frequency of mEPSCs and sEPSCs in OXT-mRFP1 neurons in the mPVN increased significantly in the AA group compared with the control group (sEPSC control group: 1.28 ± 0.16 Hz,

sEPSCs and mEPSCs recorded in the OXT-mRFP1 neurons in control and AA rats. (a, d) Representative sEPSCs and mEPSCs recorded in the OXT-mRFP1 neuron in slices from a control rat (left) and AA rat (right) at a holding potential of −60 mV. Cumulative interevent interval (left) and amplitude (right) histograms of sEPSCs (b) and mEPSCs (e) recorded in slices from control rats (sEPSCs;
We investigated whether synthesized OXT from AA rats affects the EPSPs in OXT-ergic neurons. We used a bath application of the OXT receptor antagonist L-368,899.
21
The results show representative sEPSCs and mEPSCs recorded in the OXT-mRFP1 neurons in slices from control and AA rats at a holding potential of −60 mV (Figure 5(a) and (d)). Bath application of L-368,899 dose-dependently increased the frequency of mEPSCs and sEPSCs in OXT-mRFP1 neurons in AA rats (sEPSC; 10 nM: 111.30 ± 11.68% of baseline,

Effect of bath application of L368,899 on sEPSCs and mEPSCs in OXT-mRFP1 neurons in slices from control (left) and AA (right) rats. Example sEPSC (a) and mEPSC (d) traces are shown. Summary frequency data of the different doses of L368,899 (10 and 100 nM and 1 µM) on sEPSCs (b) and mEPSCs (e). Summary amplitude data of the different doses of L368,899 (10 and 100 nM and 1 µM) on sEPSCs (c) and mEPSCs (f) (each group,
We also recorded mIPSCs in OXT-mRFP1 neurons in the mPVN of control and AA rats to examine whether inhibitory presynaptic transmitter release changed in the OXT-ergic neurons after chronic inflammation. Representative mIPSCs recorded in the OXT-mRFP1 neurons in slices from control and AA rats at a holding potential of 0 mV, and a cumulative histogram of interevent intervals and amplitudes is shown in Figure 6(a) and (b). The frequency of mIPSCs in OXT-mRFP1 neurons in the mPVN significantly increased in the AA group compared with that in the control group (mIPSC control group: 3.88 ± 0.33 Hz,
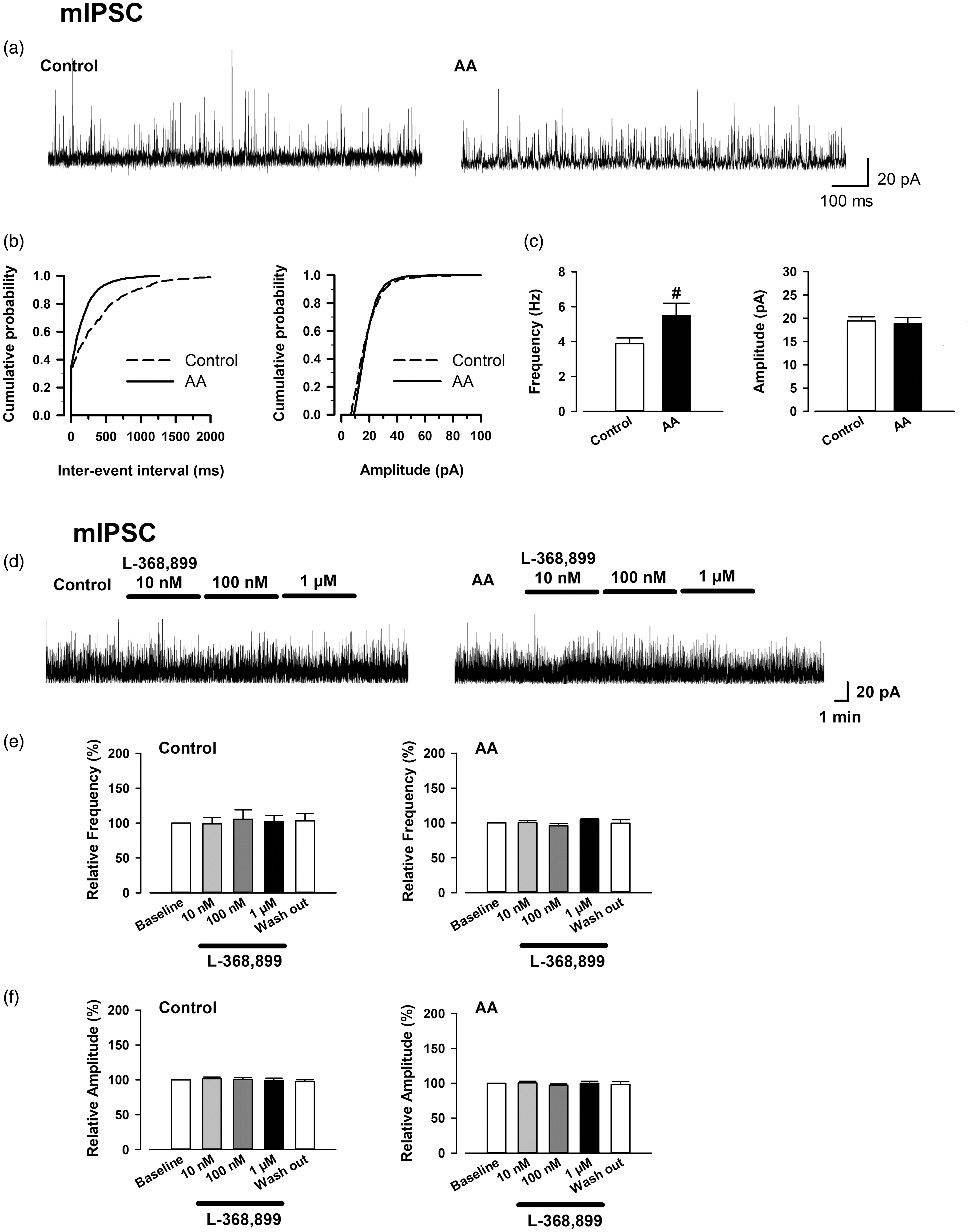
mIPSCs recorded in the OXT-mRFP1 neurons in control and AA rats, and effect of bath application of L368,899 on mIPSCs in OXT-mRFP1 neurons in slices from control (left) and AA (right) rats. (a) Representative mIPSCs recorded in the OXT-mRFP1 neurons in slices from a control rat (left) and AA rat (right) at a holding potential of 0 mV. (b) Cumulative interevent interval (left) and amplitude (right) histograms of mIPSCs recorded in slices from control rats (mIPSCs;
In addition, we investigated whether synthesized OXT from AA rats affects the IPSCs in OXT-ergic neurons. The results show representative mIPSCs recorded in the OXT-mRFP1 neurons in slices from control and AA rats at a holding potential of 0 mV (Figure 6(d)). Bath application of L-368,899 did not change the frequency and amplitude of mIPSCs in OXT-mRFP1 neurons of the control and AA rats (each group:
Therefore, the results suggest that feedback of mEPSCs and sEPSCs from synthesized OXT occurs in OXT-ergic neurons in AA rats.
Examination of retrograde transmitters in the feedback system in OXT-mRFP1 neurons in AA rats
Previous studies have demonstrated that regarding feedback in central neurons, retrograde synaptic transmission is known to occur via transmitters such as the nitric oxide (NO), cannabinoid receptor 1 (CB1), and γ-aminobutyric acid (GABA).22–24 Thus, we investigated whether these transmitters were involved in the feedback from synthesized OXT in AA rats. Following bath application of the GABAA receptor antagonist (100 µM picrotoxin) (Figure 7(a) and (b)) and CB1 antagonist (2 µM AM 251) (Figure 7(c) and (d)), bath application of L-368,899 still increased the frequency of mEPSCs (picrotoxin; 100 µM picrotoxin: 125.41 ± 10.7% of baseline,

Effect of bath application of picrotoxin, AM 251, and L-NAME on induced mEPSCs increased by L-368,899 in OXT-mRFP1 neurons in slices from AA rats. Example trace of mEPSCs following the bath application of 100 µM picrotoxin (a), 2 µM AM 251 (c), and 100 µM L-NAME (e) after the bath application of L-368,899 in OXT-mRFP1 neurons in AA rats are shown. Summary frequency (left) and amplitude (right) data of the effect of picrotoxin (b), AM 251 (d), and L-NAME (f) after the bath application of L-368,899 in OXT-mRFP1 neurons in AA rats (each group,
Confirmation of the effect of OXT and the OXT receptor antagonist L-368,899 in OXT-mRFP1 neurons in slices from control and AA rats
We confirmed the effects of OXT and L-368,899 in the OXT-mRFP1 neurons. After recording a stable baseline at a holding potential of −60 mV, we recorded mEPSCs following the application of 1 µM OXT and different doses of L368,899 (10 and 100 nM and 1 µM) on mEPSCs in the OXT-mRFP1 neurons in control rats and the application of 1 µM OXT on mEPSCs in the OXT-mRFP1 neurons in AA rats. Following the bath application of OXT, the frequency of mEPSCs in OXT-mRFP1 neurons was significantly increased (OXT; 1 µM OXT: 233.45 ± 25.99% of baseline,

Effect of bath application of OXT and L-368,899 on mEPSCs in OXT-mRFP1 neurons in slices from control and AA rats. (a) Example trace of mEPSCs following the application of 1 µM OXT and different doses of L368,899 (10 and 100 nM and 1 µM) on mEPSCs in OXT-mRFP1 neurons in control rats are shown. (b) Summary frequency (left) and amplitude (right) data of 1 µM OXT and different doses of L368,899 (10 and 100 nM and 1 µM) on mEPSCs in OXT-mRFP1 neurons in control rats (each drug group,
We confirmed that the bath application OXT affects the presynaptic current in OXT-mRFP1 neurons in the control rats, and L-368,899 inhibited the effect of OXT. However, the bath application OXT did not affect the presynaptic current in OXT-mRFP1 neurons in the AA rats.
A hypothetical scheme for the excitatory system, the inhibitory system, and the feedback mechanism of OXT-ergic neurons in the hypothalamus of the chronic inflammation rat model is shown in Figure 9.

A hypothetical scheme for the excitatory system, the inhibitory system, and the feedback mechanism of OXT-ergic neurons in the hypothalamus in the rat chronic inflammation model. (Left) Basal transmission: OTR is present in both excitatory pre- and postsynaptic neurons in OXT-ergic neurons. (Right) Chronic inflammation pain: Chronic inflammation pain enhances synaptic excitatory and inhibitory transmissions. Activated OXT works as negative feedback excitatory system around itself via the OTR. The signaling pathway of the negative feedback system contributes to the nNOS to promote NO production. NO and GABA act as retrograde neurotransmitters causing negative feedback at the pre-or postsynaptic OXT-ergic neurons.
Discussion
In this study, we investigated RMPs, mEPSCs, and sEPSCs in the OXT-ergic neurons of the mPVN by using OXT-mRFP1 transgenic rats with AA. We have demonstrated that the RMPs and the frequency of mEPSCs and sEPSCs in the absence of picrotoxin, or mIPSCs were increased in OXT-mRFP1 neurons in the mPVN of AA rats. Furthermore, the feedback system of synthesized endogenous OXT was also investigated using L-368,899. L-368,899 dose-dependently further increased the frequency of mEPSCs and sEPSCs in the neurons of AA rats. Following the bath application of picrotoxin and AM 251, L-368,899 still increased the frequency of mEPSCs in the AA rats. However, following the bath application of L-NAME, L-368,899 did not change the frequency of mEPSCs in AA rats. Our results indicate that the activity of OXT-ergic neurons is upregulated by increasing the glutamate release in AA rats, and upregulated OXT neurons have a feedback system with the released OXT. It is possible that NO and not GABA may contribute to the feedback system in OXT neurons in AA rats.
A previous study demonstrated that
In this study, we focused on the increase of the release probability for the glutamate and surrounding synthesized OXT. Glutamate is the main excitatory neurotransmitter, and the glutamate receptor is critical for synaptic plasticity. In animals of chronic pain model, the glutamate is increased in various neurons such as the substantia gelatinosa neuron of the spinal cord dorsal horn and the pyramidal neuron of the anterior cingulate cortex, which play an important role in the pain perception.25–27 The increased glutamate induces a long-term potentiation, which is proposed to be the cellular model for learning and memory.
28
OXT also enhances the synaptic transmission in the hippocampus
Interestingly, AA rats produced an increase of RMPs compared to the control rats. Furthermore, the bath application of OXT produced an increase of RMPs in the OXT-mRFP1 neurons of the control rats; however, the bath application of OXT did not change the RMPs in the AA rats. The results demonstrate that surrounding synthesized endogenous OXT may have already produced an increase of the RMPs in the OXT-ergic neurons in the AA rats. Increased APs have been reported in the spinal cord dorsal horn in rats of a chronic pain model without any change in the RMPs. 25 In the hypothalamic OXT-ergic neuron, a chronic inflammation model such as the AA rats induces the increase of APs; RMPs are also increased by the surrounding synthesized endogenous OXT. Although the mechanisms to produce APs and increase of RMPs are not yet fully understood, endogenous OXT may depolarize the OXT-ergic neurons, and this depolarization may be mediated by L-type Ca channel. 31 Taken together, these results suggest that the presynaptic glutamate release and the synthesized endogenous OXT are the factors that upregulate activity in the OXT-ergic neurons.
We also found that L-368,899 dose-dependently increased the frequency of mEPSCs and sEPSCs in OXT-mRFP1 neurons in AA rats, suggesting that the activity of presynaptic excitatory neurons was suppressed by increasing OXT in the AA model. In this study, it is unclear which OXT-ergic neurons secrete OXT. It is possible that one of the major pathways involved in the secretion of OXT is the paracrine, 32 which is thought to enter the ventricle and act on the OXT receptor after release from the PVN. 33 OXT is also synthesized in the parvocellular PVN neurons, and some of these neurons are thought to project to the magnocellular PVN OXT neurons. 3 Consequently, it is assumed that the synthesized endogenous OXT suppressed the excitatory neurotransmission in the surrounding or its own OXT neurons in AA rats as a feedback system. In previous studies, it was unclear how feedback was provided to OXT neurons in the central nervous system. At the spinal level, OXT activates the presynaptic OXT receptor at the glutamatergic interneuron ends and promotes the synaptic release of glutamate and inhibitory GABA-ergic interneurons. 34 Therefore, OXT may activate the excitatory and inhibitory neurons by stimulating the presynaptic OXT receptor in the central nervous system.
Meanwhile, there is a mechanism that uses a retrograde transmitter as a mechanism for feedback through the postsynaptic OXT receptor. NO has been shown to be present in the central nervous system 35 and has long been considered a retrograde neurotransmitter because it is a fat-soluble gas that diffuses across cell membranes. 36 Neuronal NOS (nNOS) is abundant in the PVN, and nNOS in the PVN has been shown to be increased in several rat models of stress.37–39 It has been suggested that NO plays a role as an important messenger in the PVN-spinal pathway and may act in concert with OXT. 40
NO synthesis is competitively inhibited by L-NAME, which has a similar structure to the L-arginine. 41 L-NAME suppresses the hippocampal long-term potentiation, and it has been reported that NO may be a hippocampal retrograde transmitter. 42 On the other hand, it has been suggested that NO is also associated with signaling via the OXT receptor. Exogenous OXT hyperpolarizes the dorsal root ganglia neurons that induce the feeling of pain in rats, and that this hyperpolarization is mediated by the Ca2+/nNOS/NO/KATP pathway. 43 NO is also associated with other neuronal signal pathways, such as the NO/sGC/cGMP pathway, and downregulation of N-methyl-D-aspartic acid (NMDA)-receptor activity by reaction with thiol group(s) of the NMDA receptor’s redox modulatory site.44,45 Thus, released OXT may cause hyperpolarization and downregulation of NMDA-receptor activity in several putative pathways in the AA rats. As a result, OXT may modulate the glutamine release though the OXT receptor in OXT-mRFP1 neurons. Pretreatment with the OXT receptor antagonist Atociban or the selective NOS inhibitor N-propyl-L-arginine significantly attenuates the hyperpolarization caused by OXT. 43
We found that OXT-ergic neurons generate negative feedback with OXT itself in the PVN in AA rats. In addition, NO is a contributor to the feedback mechanism.
Although we investigated the contribution of GABAA to the feedback system through the OXT receptors, increased mEPSCs induced by the OXT receptor antagonist did not change following the bath application of the GABAA antagonist. It is thought that GABAA is not related to the feedback system through OXT receptors. Instead, GABAA may work as a retrograde transmitter in other feedback systems that are not related to the OXT receptor.
A limitation of the study is that the evoked EPSC was not investigated. Thus, we did not examine the postsynaptic changes of the OXT-ergic neurons in AA rats. Moreover, the relationship between OXT-ergic neurons was not described. In the future, the plasticity of OXT-mRFP1 neurons should be examined, and paired recordings with OXT-mRFP1 neurons should be conducted.
In conclusion, the activity of OXT-ergic neurons is upregulated by increasing the glutamate release in AA rats, and OXT neurons have a feedback system with released OXT. It is possible that NOS may contribute to the feedback system of the OXT-ergic neurons.
Footnotes
Acknowledgment
The authors thank Ms. Yuki Nonaka (University of Occupational and Environmental Health, Kitakyushu, Japan) for her technical assistance.
Author Contributions
TM designed the project. TF and TM performed the electrophysiological experiments. TF, TM, and MK wrote the initial draft and finished the final version of the manuscript. HS, HN, KB, YY, HO, YU, and AS contributed to the analysis and interpretation of data and assisted in the preparation of the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from JSPS KAKENHI Grant Number JP 19K21360 and 20K18079 to TM and (C) 19K09564 to MK.
