Abstract
Acid sensing in the gastrointestinal tract is required for gut homeostasis and the detection of tissue acidosis caused by ischaemia, inflammation and infection. In the colorectum, activation of colonic afferents by low pH contributes to visceral hypersensitivity and abdominal pain in human disease including during inflammatory bowel disease. The naked mole-rat (
Introduction
The gastrointestinal (GI) tract coordinates the digestion of food, absorption of nutrients and evacuation of waste with acidification of the stomach contents a critical component of this process. Through compartmentalisation, sensory surveillance and specialised mucosal defence mechanisms are not only the breakdown of food and elimination of ingested pathogens achieved through acidification in the foregut but also the delicate gut microbiota-host symbiosis of the hindgut maintained. It is clear that when gastric acid regulation is lost then significant pathogenesis can occur, including acid-related diseases such as gastro-eosophageal reflux disease, gastroduodenal ulceration, dyspepsia and gastritis. 1
Sensory neurones innervating the GI tract are central to the feedback regulation of gastric acid secretion and can additionally detect tissue acidosis caused by inflammation, ischaemia and microbial activity,2,3 often resulting in visceral hypersensitivity and abdominal pain. 4 Whilst luminal pH varies along the length of the healthy human gut (with lower pH found in the stomach and colon5,6), both surgical intervention and disease (e.g. chronic pancreatitis and inflammatory bowel disease5,7,8) can also result in abnormal acidification of the gut.
The naked mole-rat (
Materials and methods
Animals
Experiments were performed in C57BL6/J mice (6–41 weeks; three females, six males) and subordinate NMR (25–198 weeks; two females, seven males). Mice were conventionally housed with nesting material and a red plastic shelter in temperature-controlled rooms (21°C) with a 12-h light/dark cycle and access to food and water ad libitum. NMRs were bred in-house and maintained in an inter-connected network of cages in a humidified (∼55%) temperature-controlled room (28°C) with red lighting (08:00–16:00) and had access to food ad libitum. In addition, a heat cable provided extra warmth under two to three cages/colony. NMRs used in this study came from two different colonies. Experiments were conducted under the Animals (Scientific Procedures) Act 1986 Amendment Regulations 2012 under Project Licences (70/7705 & P7EBFC1B1) granted to ESt.JS by the Home Office and approved by the University of Cambridge Animal Welfare Ethical Review Body.
Immunohistochemistry
The colorectum was dissected free before opening along the mesenteric border and pinning flat in a Sylgard lined dissection tray. After fixing in Zamboni’s fixative (2% paraformaldehyde/15% picric acid in 0.1M phosphate buffer; pH 7.4) overnight, the mesentery and mucosa were dissected free from the muscle layers. Small 1.5 cm × 1.5 cm sections were subsequently washed in 100% DMSO (3 × 10 min) and phosphate-buffered saline (PBS; 3 × 10 min). Tissues were blocked with antibody diluent (10% donkey serum, 1% bovine serum albumin in 0.2% Triton X-100) for 1 h, then primary antibodies were applied overnight at 4°C. The following day, tissues were washed (PBS, 3 × 10 min), donkey anti-rabbit IgG-AF488 (1:500, Life Technologies A21206) antibody applied for 2 h, washed (PBS; 3 × 10 min), mounted and coverslipped. Primary antibodies used were rabbit anti-calcitonin gene-related peptide (anti-CGRP; 1:5000, Sigma C8198) and rabbit anti-protein gene product 9.5 (anti-PGP9.5; 1:500, Abcam ab10404). No labelling was observed in control sections where primary antibody was excluded. Tissues were imaged using a Leica SP5 confocal microscope and z-stack reconstructions of nerve fibres within different layers of the NMR gut produced with ImageJ (v1.51a, National Institutes of Health).
Electrophysiology recordings of visceral afferent activity
Colonic nerves innervating the colorectum of mouse and NMR were isolated, and electrophysiological activity was recorded as previously described. 22 Mice were humanely killed by cervical dislocation of the neck and cessation of circulation. NMRs were humanely killed by CO2 exposure followed by decapitation. For both species, the colorectum with associated lumbar splanchnic nerve was dissected free from the animal and transferred to a recording chamber superfused with carbogenated Krebs buffer (in mM: 124 NaCl, 4.8 KCl, 1.3 NaH2PO4, 2.5 CaCl2, 1.2 MgSO4·7H2O, 11.1 glucose and 25 NaHCO3; 7 ml/min; 32°C–34°C). The colorectum was cannulated and perfused with Krebs buffer (100 µl/min) enabling distension of the colon by closure of the outflow. The Krebs buffer was supplemented with nifedipine (10 µM) and atropine (10 µM) to inhibit smooth muscle activity and with indomethacin (3 µM) to restrict endogenous prostanoid production. Multi-unit electrophysiological activity of the lumbar splanchnic nerve rostral to the inferior mesenteric ganglia was recorded using a borosilicate glass suction electrode. Signals were amplified and bandpass filtered (gain 5 K; 100–1300 Hz; Neurolog, Digitimer Ltd, UK) and digitised at 20 kHz (micro1401; Cambridge Electronic Design, UK) before display on a PC using Spike 2 software. The signal was digitally filtered online for 50 Hz noise (Humbug, Quest Scientific, Canada), and action potential firing counts were determined using a threshold of twice the background noise (typically 100 µV).
Electrophysiological protocols
Tissues were stabilised for 30 min before noxious intraluminal distension pressures were applied by blocking the luminal outflow of the cannulated mouse or NMR colorectum. The pressures reached are above threshold for all known visceral afferent mechanoreceptors 23 and evoke pain behaviours in rodents in vivo. 24 Mechanosensitivity and chemosensitivity were investigated using a combined sequential protocol. As such, a slow ramp distension (0–80 mmHg, 4–5 min) and set of six rapid phasic distensions (0–80 mmHg, 60 s at 9 min intervals) were applied as previously described 25 prior to bath superfusion of pH 4.0 Krebs buffer (50 ml volume) and a set of three phasic distensions (0–80 mmHg, 60 s at 9 min) to test for acid-induced acute mechanical hypersensitivity. After a 20-min wash-out period, 1 µM bradykinin was applied by bath superfusion (20 ml volume) and a further set of three phasic distensions were performed. Phasic distension protocols were automated using an Octaflow II perfusion system (ALA Scientific, USA) to standardise duration and intervals.
Data analysis
Peak changes in firing rates of electrophysiological nerve recordings were determined by subtracting baseline firing (3 min before distension or drug application) from increases in nerve activity following distension or chemical stimuli. Statistical analysis was performed using two-way analysis of variance (ANOVA) followed by Holm–Sidak’s post hoc test in Prism 6 (GraphPad Inc., USA). Statistical significance was set at
Drugs
Stock concentrations of bradykinin (10 mM; water), nifedipine (100 mM; DMSO), atropine (100 mM; ethanol) and indomethacin (30 mM; DMSO) were dissolved as described, diluted to working concentration in Krebs buffer on the day of experiment as described above and were all purchased from Sigma-Aldrich.
Results
GI neuroanatomy of the NMR
We first compared the gross anatomy of the NMR and mouse GI tract. As the NMR is greatly long lived compared to the mouse, with a life expectancy of >30 years, we chose animals from both species that could be deemed adults (see ‘Methods’ section). Compared to the mouse, NMR GI length (from pyloric sphincter to anus) was significantly shorter in length (mouse, 37.8 ± 0.4 mm; NMR, 25.8 ± 1.2 mm,
We next confirmed the presence of extrinsic sensory fibres innervating different layers of the NMR colorectum using immunohistochemistry. Equivalent staining in the mouse is widely available within the literature, and we did not seek to duplicate them here.
26
Using antibodies raised against CGRP and PGP9.5, we stained for neuronal fibres within flat-sheet whole-mount preparations of multiple layers of the NMR colon. Specifically, CGRP-positive extrinsic neuronal varicosities were identified encircling and tracking with blood vessels within the mesentery supplying the distal colon of NMR; such fibres likely contribute to the larger lumbar splanchnic nerve upon which these coalesce (Figure 1(b)). Although NMR lack CGRP in cutaneous afferent neurones, this finding is in line with the observation that mesenteric arteries in NMR and the common mole-rat (

Extrinsic sensory innervation of NMR colorectum. (a) Comparison of mouse and NMR gastrointestinal tracts from anus (
Colonic afferent mechanosensitivity does not differ in the NMR compared to mouse
In order to understand whether the peripheral terminals of sensory neurones innervating the GI tract of the NMR possessed altered acid and inflammatory sensitivity compared to mouse, we made ex vivo multi-unit electrophysiological recordings of lumbar splanchnic nerve activity using a suction electrode from the colorectum of both NMR and mouse. The lumbar splanchnic nerve innervates the colorectum and is a pathway through which pain is the predominant conscious sensation transduced. 28 The colorectum, once dissected free from the animal, was cannulated and both luminally perfused and bath superfused with Krebs buffer, thus allowing mechanical distension of the bowel or application of chemical stimuli, respectively.
We first investigated mechanosensitivity of visceral afferents in the NMR compared to mouse GI tract. There were no significant differences in the baseline spontaneous activity measured between NMR and mouse (3 min average: 7.2 ± 2.6 spikes/s vs. 8.3 ± 1.2 spikes/s, respectively,
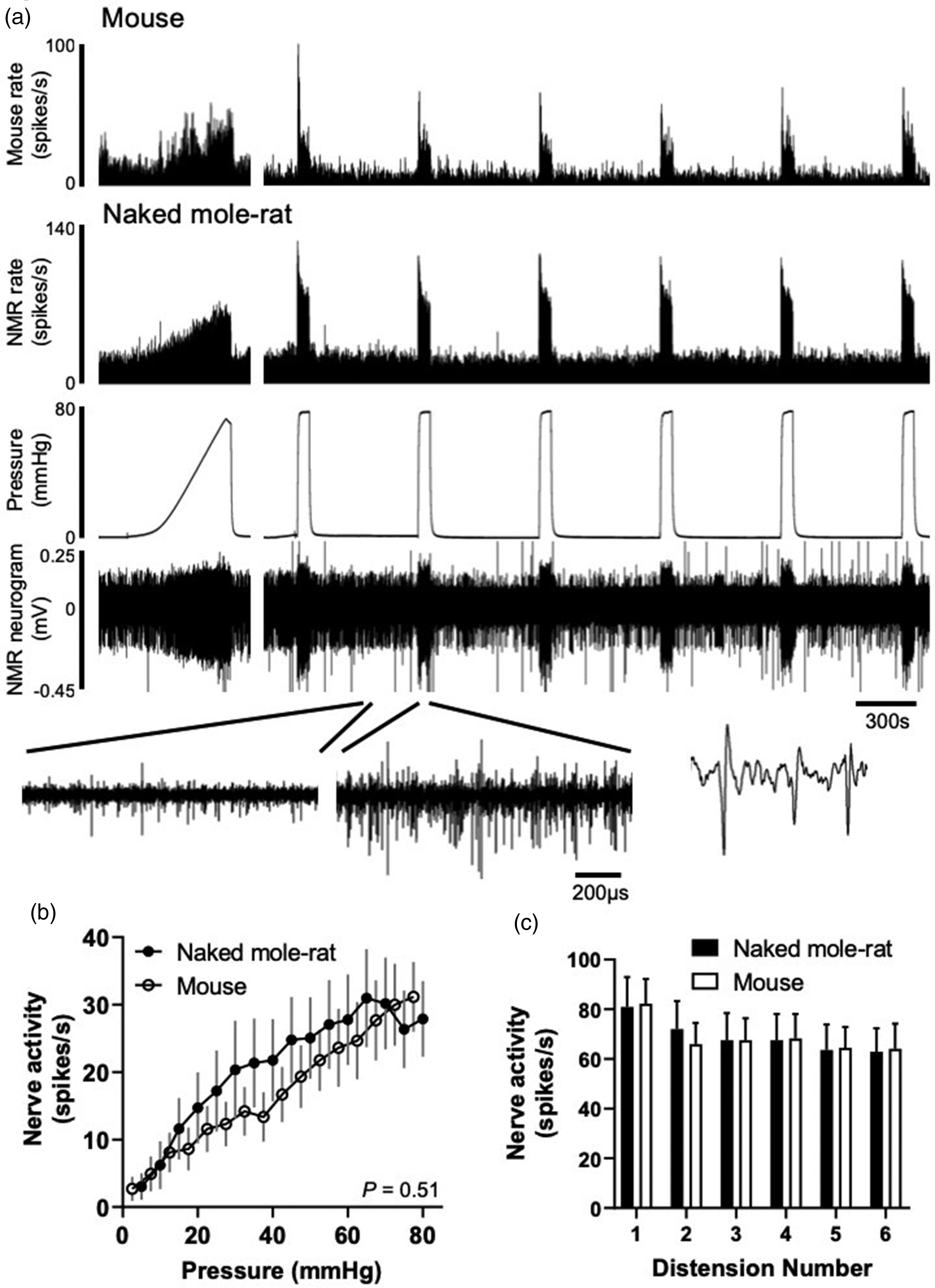
Colonic afferent responses to noxious ramp and repeat phasic distension in mouse and NMR. (a) Example rate histograms of colonic lumbar splanchnic nerve activity from mouse and NMR with intraluminal pressure trace and neurogram trace following ramp distension (0–80 mmHg) and repeat phasic distension (0–80 mmHg, 60 s, 9 min intervals). Below, expanded neurogram traces showing NMR before and after phasic distension and an example trace showing three action potentials. (b) Mean firing rates to ramp distension at 5 mmHg increments in mouse and NMR (
Extracellular acid evokes mechanical hypersensitivity in NMR but not mouse
Next, we investigated the effect of extracellular acid on visceral afferent firing and mechanical hypersensitivity to phasic distension (Figure 3(a)). We chose a pH 4.0 stimulus to broadly activate acid-sensitive ion channels15,31 and a stimulus that is capable of evoking pain both in humans and rodents when injected subcutaneously.11,32 The vast majority of colonic sensory neurones possess inward sustained currents in response to low pH.
33
Bath superfusion of pH 4.0 to mouse colon directly excited visceral afferents evoking a peak firing increase of 30.0 ± 5.1 spikes/s returning to baseline firing rates after 1735 ± 60 s. Direct excitation of NMR visceral afferents as a result of acid was significantly lower compared to mouse, but return to baseline did not differ between species (12.5 ± 4.3 spikes/s and duration of 1545 ± 197 s,
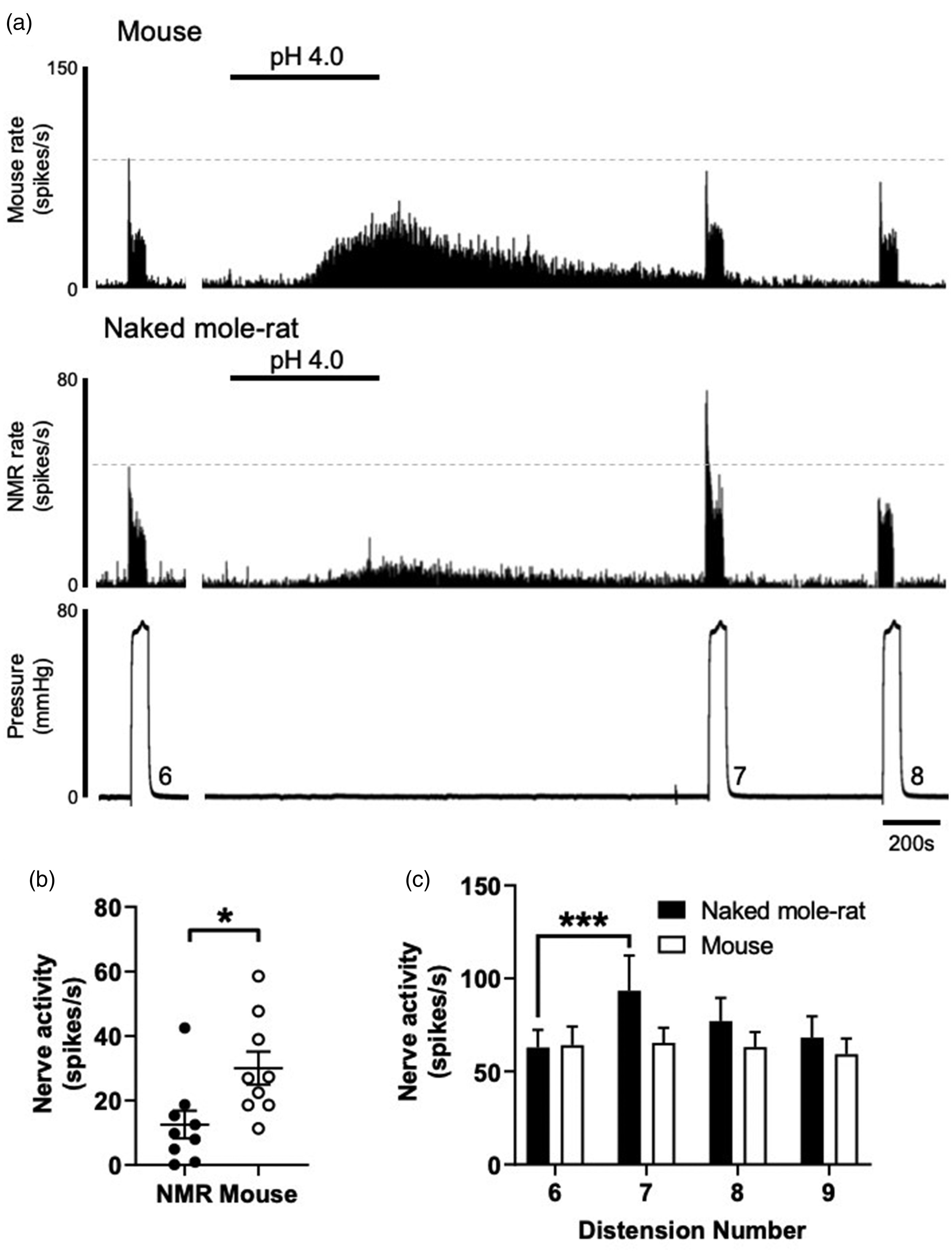
Extracellular acid evokes mechanical hypersensitivity in NMR, but not mouse. (a) Example rate histograms of colonic splanchnic nerve activity from mouse and NMR with accompanying pressure trace showing bath superfusion of pH 4.0 Krebs buffer (50 mL) and subsequent repeat (×3) phasic distension. (b) Mean increase in peak firing after application of pH 4.0 (*
Afferent excitation to bradykinin is blunted in NMR, but mechanical sensitisation is unaffected
Given that inflammatory pain responses in NMR are blunted to some inflammatory stimuli,
11
we investigated the ability for the prototypical inflammatory mediator, bradykinin, to not only activate but also evoke mechanical hypersensitivity in NMR visceral afferent fibres. Application of bradykinin (1 µM) by bath superfusion to mouse colonic afferents led to an increase in peak firing of 24.2 ± 3.9 spikes/s in agreement with previous studies in mouse and human colonic tissues (Figure 4(a)).35,36 In NMR this was not the case, with peak firing only increased by 4.0 ± 1.5 spikes/s following addition of bradykinin (

Colonic afferent excitation to bradykinin is blunted in NMR, but mechanical sensitisation is unaffected. (a) Example rate histograms of colonic splanchnic nerve activity from mouse and NMR with accompanying pressure trace showing addition of 1 µM bradykinin (20 mL) and subsequent repeat (×3) phasic distension. (b) Mean increase in peak firing after application of 1 µM bradykinin (***
Discussion
Acid sensing in the GI tract is necessary to maintain gut homeostasis by providing feedback for gastric and intestinal acid regulation and secondly for detecting tissue acidosis caused by inflammation, infection and ischaemia during disease. Here, we assessed the mechanical and chemical sensitivity of NMR and mouse colonic afferents in response to differing noxious stimuli.
In NMRs, the absence of thermal hypersensitivity induced by capsaicin and lack of histamine-induced scratching are thought to be due to a lack of cutaneous neuropeptides, such that both behaviours can be ‘rescued’ by intrathecal administration of substance P.11,12 We show here by immunohistochemistry that CGRP is expressed within nerve fibres found encapsulating both blood vessels of the NMR colonic mesentery and myenteric plexi within the smooth muscle layers of the colon wall, which aligns with previous findings of CGRP-positive fibres innervating NMR mesenteric arteries. 27 By contrast, PGP9.5 staining identified nerve fibres within the submucosa that did not express CGRP, highlighting potential restricted penetration of extrinsic sensory fibres innervating the NMR colorectum. Such differences in sensory innervation termini did not manifest as altered mechanosensitivity to distension during baseline conditions of NMR colonic afferents compared to mouse. This suggests that visceral mechanotransduction is not significantly altered in the NMR.
We did observe greatly differing responses to both, the direct exposure of extracellular acid and to induced mechanical hypersensitivity, implicating altered integration of acid sensors, mechanotransducers and modulators of spontaneous afferent firing. NMR acid sensing differs significantly to other mammals. For example, in hippocampal and cortex neurones, the peak current density of NMR ASIC-like responses is reduced compared to mouse brain neurones. 37 In the peripheral nervous system, subcutaneous injection of acid (pH 3.5), capsaicin or histamine does not cause the nocifensive or pruriceptive behaviours in NMR that such stimuli characteristically induce in mice.11,12 This acid insensitivity is a function of altered ASIC responses compared to mouse 19 and a variation in NMR NaV1.7, which renders the channel hypersensitive to proton-mediated block and therefore prevents acid-driven action potential initiation from the skin. 18 Such intrinsic differences in the sensitivity of NMR to acid may explain our observation of significantly lower firing rates in response to application of acid to NMR colonic afferents compared mouse. We have shown previously that pharmacological inhibition or genetic ablation of NaV1.7 in mouse does not impair colonic afferent firing or alter pain behaviours. 25 Therefore, if NaV1.7 is redundant in colonic afferents compared to those innervating the hindpaw, then it would be predicted that NMR colonic afferents would not be as insensitive to acid as their somatic equivalents. Whilst this hypothesis does not hold true for the direct action of acid on NMR colonic afferent firing, that is, it is diminished compared to the mouse, it does fit with the lack of mechanical hypersensitivity observed following acid application in the mouse, which has been reported previously. 34 In contrast, the robust sensitisation observed in NMR colonic afferents to acid suggests differential coupling of molecular acid sensors to mechanotransducers compared to the mouse. Further studies would be required to elucidate the intracellular signalling cascades involved.
Responses to the inflammatory mediator bradykinin failed to activate NMR colonic afferents but could induce a robust mechanical hypersensitisation comparable to the effects observed in mouse. Although we do not confirm bradykinin B2 receptor expression in NMR colonic afferents in this study, such mechanical hypersensitivity suggests that the B2 receptor activity is unimpaired. The bradykinin B2 receptor is a predominantly Gαq protein-coupled receptor which upon activation modulates the function of a number of molecular transducers (including KV7, 38 TRPV1, 39 TRP ankyrin 1, 40 Ca2+-activated Cl− channels38,41 and KCa42,43) via downstream signalling cascades including increased intracellular Ca2+ and protein kinase C-dependent phosphorylation. Differences in the activity or coupling to of these molecular transducers in the NMR may explain the altered response profiles compared to mouse.
Given our experimental paradigm, we believe our observations are most likely the result of species differences in molecular sensors and transducers responsible for action potential firing at the level of the primary afferent terminal; however, we cannot exclude a number of confounding factors that may influence colonic afferent sensitivity. Firstly, whilst every effort was made to match the relative age range and sex of the NMR and mice used in this study, we acknowledge that age may have an impact on afferent sensitivity. For example, reduced responses of afferent firing to some stimuli are observed in tissues from older mice 44 and humans.45,46 We accept that this may have introduced variability into our study, but given the relatively comparable spread in age of the individuals used from both species, we do not believe that this is responsible for the dramatic differences observed between species. Secondly, all NMR used in this study were subordinates and therefore their sexual maturity will have been suppressed. Sex steroid hormones including oestrogen can modulate visceral sensory function. 47 Without a direct comparison to breeding NMRs, which is a challenge due to the eusociality of the species, we are unable to exclude that the effects observed are not due to sexual immaturity. Thirdly, all electrophysiological recordings were made at 32°C to 34°C and as such represents a 2°C to 3°C difference from optimum body temperature for both species, albeit in opposing directions. Temperature affects afferent firing; however, the equivalence of responses to ramp and phasic distension acts as a positive control and suggests that NMR colonic afferents are capable of producing significant action potential firing in these conditions.
In summary, understanding how noxious pH is sensed and GI homeostasis is maintained in the NMR may help to inform our understanding of other model species and ultimately, GI acid sensing during human disease.
Footnotes
Authors’ Contributions
JRFH designed the research studies, conducted the experiments, acquired and analysed the data and wrote the manuscript. KHB, TST, GC and ZMH acquired and analysed the data. DCB wrote the manuscript. ESt.JS designed the research studies and wrote the manuscript. All authors approve the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Rosetrees Postdoctoral Grant (A1296; JRFH and ESt.JS), Biotechnology and Biological Sciences Research Council grant (BB/R006210/1; JRFH and ESt.JS), Versus Arthritis Pain Challenge Grant (RG21973; GC and ESt.JS), AstraZeneca PhD studentship (KHB), European Molecular Biology Organization Long-Term Fellowship (ALTF1565-2015; ZMH) and University of Cambridge Vice Chancellor’s Award (TST).
