Abstract
Glutamate is a neurotransmitter present in most excitatory synapses in the nervous system. It also plays a key role in the spinal cord’s physiological excitatory circuit and is involved in pathological neurotransmissions such as those observed in inflammatory and neuropathic pain conditions. The actions of glutamate are mediated by different types of ionotropic glutamate receptors (iGluRs) and metabotropic glutamate receptors (mGluRs). Although expressions of iGluRs are well studied, those of mGluRs are not fully elucidated in the spinal cord. In this study, we examined the expressions of mGluRs (mGluR1-8) and investigated which mGluR subtypes can modulate pain transmission in the dorsal horn of the spinal cord using an inflammatory pain model. Reverse transcription-polymerase chain reaction revealed that mGluR mRNAs, except for mGluR2 and 6, were detected in the spinal cord. Double labeling analysis, in situ hybridization histochemistry with immunohistochemistry, was used to examine the distribution of each mGluR in neurons or glial cells in the lamina I–II of the spinal dorsal horn. mGluR1, 5, and 7 were generally, and 4 and 8 were frequently, expressed in neurons. mGluR3 was expressed not only in neurons but also in oligodendrocytes. We next examined the distribution of mGluR4 and 8 were expressed in excitatory or inhibitory neurons. Both mGluR4 and 8 were preferentially expressed in inhibitory neurons rather than in excitatory neurons. Furthermore, intrathecal delivery of CPPG((RS)-α-cyclopropyl-4-phosphonophenylglycine), an antagonist for mGluR 4 and 8, attenuated nocifensive behaviors and the increase in fos-positive-excitatory neurons of the dorsal horn induced by intraplantar injection of formalin. These findings suggest that mGluR4 and 8, which are preferentially expressed in inhibitory neurons, may play roles in the modulation of pain transmission in the spinal dorsal horn.
Introduction
Metabotropic glutamate receptors (mGluRs) belong to G protein-coupled receptors and mainly regulate glutamatergic transmission. To date, eight subtypes of mGluRs, mGluR1-8, have been identified and classified into three groups based on sequence similarities and pharmacological properties. Group I mGluRs includes mGluR1 and 5, Group II mGluRs includes mGluR2 and 3, and Group III includes mGluR4, 6, 7, and 8. Each group of mGluRs has specific cell signaling pathways; Group I mGluRs regulate neuronal excitability by coupling to Gαq/11, and Group II and Group III mGluRs are negatively coupled to Gαi/o to regulate neuronal inhibition. Therefore, it was thought that the responses of the neuronal network induced by mGluRs are dependent upon the expression balance, pattern, and levels of the Gαq/11- or Gαi/o-coupled mGluRs.
All mGluRs, except for mGluR6, are ubiquitously expressed throughout the whole nervous system, in both neurons and glial cells, with some subtype specificity in different regions. 1 Recently, using transgenic techniques, several studies have shown that mGluR6 expression is not restricted in the retina but is also found in some cortical areas in mice and other species.2,3
It is well known that mGluRs are involved in pain transmission in the spinal cord. The majority of studies indicated that Group I mGluRs, especially, are pronociceptive.1,4 For example, intrathecal administration of Group I mGluR antagonists suppresses the neuronal activity of dorsal horn associated with capsaicin-induced sensitization, repeated application of mustard oil, and knee joint inflammation.5–8 In addition, mGluR5 in the cytoplasmic membrane, as well as intracellular mGluR5, is actively involved in the relay of nociceptive information in the spinal cord. 9 Notably, intrathecal injection of Group I mGluRs agonist facilitates formalin-induced nociception, 10 produces persistent thermal and mechanical hypersensitivity, 11 and induces spontaneous nociceptive behaviors that persist for over 10 h. 12 It has been demonstrated that Group I mGluRs promote the activation of L-type calcium channels 13 or inhibit the Kv-channel in response to phosphorylation of mitogen-activated protein kinase, 14 a mechanism underlying the involvement of Group I mGluRs in pain modulation. Thus, Group I mGluRs are involved in the enhancement of pain neuraxis in the spinal cord. On the other hand, the roles of Group II and Group III mGluRs are controversial. Intrathecal injection of Group II and Group III mGluR agonists significantly suppresses mechanical and cold hypersensitivity induced by peripheral nerve injury, 15 and Group III mGluR agonists decrease the frequency of action potentials in dorsal horn neurons following von Frey stimulation to the receptive field in neuropathic pain model rats. 16 Furthermore, enhanced nocifensive behaviors induced by intraplantar injection of formalin has been observed in mGluR4 knock-out (KO) mice. 17 However, it was demonstrated that Group II mGluR agonists significantly enhanced formalin-induced nociception compared to the vehicle control group. 10 Intrathecal injection of a specific agonist for mGluR7 (Group III) does not relieve hyperalgesia in neuropathic conditions in rats. 18 Moreover, inhibition of Group III mGluRs tends to decrease the nocifensive behaviors in postoperative pain (Brennan model). 19 Therefore, the precise role of each mGluR in pain neuraxis in the spinal cord is controversial, and the possible reasons are as follows. First, gene recombination techniques are useful in understanding the function of one or two molecules; however, it is difficult to make a lot of lines of these mice and analyze all phenotypes simultaneously. Therefore, anatomical and functional studies of mGluRs are needed to focus on the comprehensive expression of each mGluR in situ using appropriate and reliable molecular tools. Second, the expression pattern of each mGluR subtype in different neurons (i.e., excitatory or inhibitory) remains unclear. Recently, several studies have shown that spinal neurons can be categorized according to the expression characteristics of specific proteins.20,21
Accordingly, the current study comprehensively analyzed the distribution of each mGluR in the spinal dorsal horn and investigated the effects of mGluR agonists or antagonists on nocifensive behaviors following peripheral inflammation.
Materials and methods
Animal procedures
Male Sprague Dawley rats weighing 200–250 g were anesthetized with sevoflurane (2.5%–5.0% inhalation administration, in the air) and were used in all experiments in this study. The animals were group-housed in polycarbonate cages (three animals per cage) with a deep layer of sawdust, in a thermostatically controlled room at 25.0 ± 1.0°C. The room was artificially illuminated from 8:00 a.m. to 8:00 p.m., and the animals received commercial pelleted rat food (CE-2; CLEA Japan Inc.) and water ad libitum. All animal experimental procedures were approved by the Hyogo College of Medicine Committee on Animal Research (approval number, 17–026) and were performed in accordance with the National Institutes of Health guidelines on animal care. Every effort was made to minimize animal suffering and reduce the number of animals used.
Reverse transcription-polymerase chain reaction and in situ hybridization histochemistry
Rats were sacrificed by decapitation under deep ether anesthesia. Half of their spinal cords (L4–L5) were removed and rapidly frozen with powdered dry ice and stored at −80°C until use. Extraction of total RNA was performed using a single step extraction method with ISOGEN (Nippon Gene, Tokyo, Japan), as described in a previous study. 22 PCR primers for mGluR1-8 and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) cDNA are shown in Table 1. PCR products were used to generate the cRNA probes for in situ hybridization histochemistry (ISHH). The L4–L5 spinal cords were dissected out, rapidly frozen in powdered dry ice, and cut on a cryostat at a thickness of 16 µm. The protocol for ISHH was described in detail previously. 23 The clone (pCRII-TOPO; Promega, Madison, WI, USA) containing a partial sequence corresponding to the coding regions of mGluR1-mGluR8 was prepared. Using the enzyme-digested clones, 35 S UTP-labeled antisense and sense cRNA probes were synthesized using enzyme-digested clones. The 35 S-labeled probes in hybridization buffer were placed on the section and then incubated at 55°C overnight (>18 h). Sections were then washed and treated with 1 µg/ml RNase A. Subsequently, sections were dehydrated and air-dried. After the hybridization reaction, the slides were coated with NTB emulsion (Kodak, Rochester, NY, USA) and exposed for 4–8 weeks. Once developed in D-19 (Kodak), the sections were stained with hematoxylin-eosin and were then coverslipped.
Sequence location of primers.
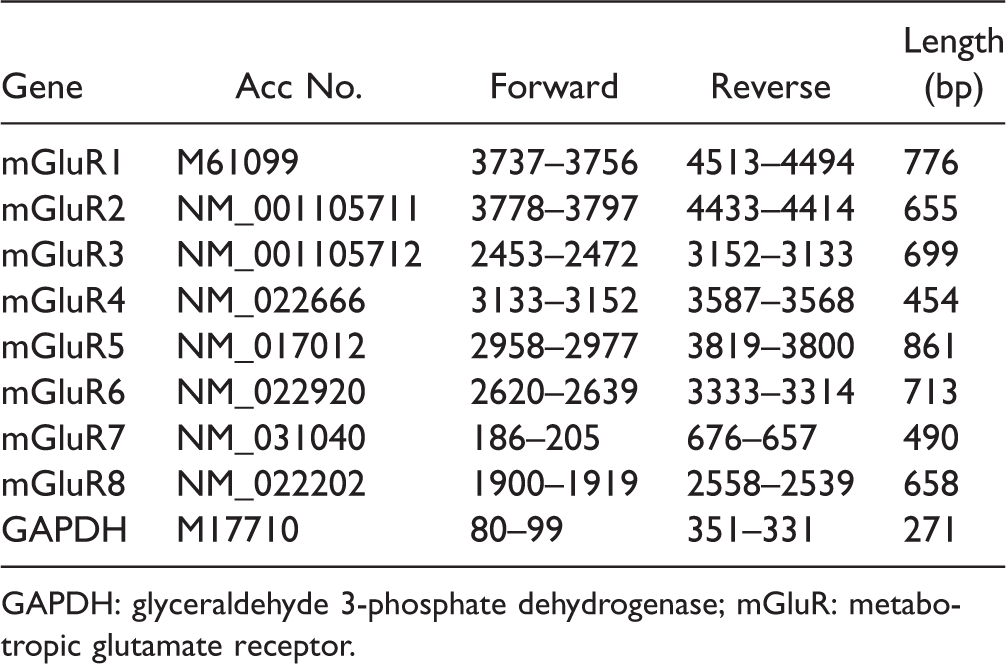
GAPDH: glyceraldehyde 3-phosphate dehydrogenase; mGluR: metabotropic glutamate receptor.
Immunohistochemistry
Rats were deeply anesthetized with sodium pentobarbital and perfused transcardially with 250 ml of 1% formaldehyde in 0.1 M phosphate buffer (pH 7.4), followed by 500 ml of 4% formaldehyde in 0.1 M phosphate buffer. The spinal cords were dissected out and postfixed in the same fixative at 4°C overnight, followed by immersion in 20% sucrose in 0.1 M phosphate buffer at 4°C for two days. The spinal cord tissue was frozen in powdered dry ice and cut on a cryostat at 16 µm thickness. The sections were processed for immunohistochemistry (IHC) using ABC method. 24 The following antibodies were used: guinea pig anti-LIM homeobox transcription factor 1-beta (Lmx1b) polyclonal antiserum (1: 10,000; kindly gifted from Drs T. Müller and C. Birchmeier), rabbit anti-c-fos polyclonal antiserum (1: 20,000; Ab-5; Oncogene), rabbit anti-oligodendrocyte transcription factor 2 (Olig2) polyclonal antiserum (1:20,000; Millipore), rabbit anti-pax2 polyclonal antiserum (1:20,000; Abcam), rabbit antiionized calcium-binding adapter molecule 1 (Iba1) polyclonal antiserum (1:2000; Wako Chemicals), mouse antineuronal neuclei (NeuN) monoclonal antiserum (1:1000; Chemicon), and rabbit antiglial fibrillary acidic protein (GFAP) polyclonal antiserum (1:1000; Dako Cytomation). In brief, spinal cord sections were incubated with primary antibodies overnight at 4°C, followed by goat, donkey, or horse biotinylated secondary antibodies (1: 500; Vector Laboratories, Burlingame, CA, USA) overnight at 4°C. Immunoreactivities were visualized using 0.05% 3,3-diaminobenzidine tetrahydrochloride (Wako Chemicals). Double-immunofluorescent staining was performed by incubation with two different primary antibodies followed by incubation with antiguinea pig Alexa Fluor 633 IgG (1:1000; Invitrogen) and antirabbit Alexa Fluor 488 IgG (1:1000). Detailed methods of double labeling of IHC and ISHH have been described previously. 25
Photomicrographs
All 3,3'-diaminobenzidine-stained and emulsion-coated slides were digitized with a Nikon Eclipse 80i microscope connected to a Nikon DXM-1200F digital camera (Nikon Corporation, Tokyo, Japan). Immunofluorescence images were obtained using an Olympus FLUOVIEW FV1200 confocal microscope (Olympus Corporation, Tokyo, Japan). A maximum of 15 images of confocal z stacks were sectioned at a thickness of 0.5 µm. Z projections of stack series were averaged using ImageJ (1.46r). Adobe Photoshop CS4 (Adobe Systems, Mountain View, CA, USA) was used to optimize the images and compose all figures.
Drug treatments
The L5 vertebra of rats was laminectomized under adequate anesthesia with sevoflurane (2.5%–5.0% inhalation administration, in air), and a 7-cm soft tube (SILASTIC laboratory tubing, Dow Corning Corporation, Auburn, MI, USA; outer diameter, 0.64 mm) filled with saline was inserted into the subarachnoid space at a length of ∼0.5 cm. After the muscle incision was closed, the tube was laid under the skin and the cut end was ligated. Then, the incision was closed. Ten microliters of group III mGluR agonist L-(+)-2-amino-4-phosphonobutyric acid (L-AP4) (Tocris Bioscience) and group III mGluR antagonist (RS)-α-cyclopropyl-4-phosphonophenylglycine (CPPG) (Tocris Bioscience) were carefully administered, followed by 5 µl of saline. According to previous studies,19,26 the doses of L-AP4 and CPPG were 10 and 50 nmol/µl diluted in phosphate-buffered saline (PBS), respectively. One dose of each drug was given 5 min prior to formalin injection. Rats were anesthetized, and drugs were administered using a Hamilton syringe.
Behavioral testing
Nocifensive behaviors were examined using a formalin test. First, rats were placed in a wire mesh cage for adaptation before testing. Subsequently, rats received intraplantar injection of formalin (1.5% in saline) into the left hindpaw. Immediately, the rats were placed in a cage again. The number of flinching at a 5-min interval during the 60 min after injection of formalin was measured.
Imaging analysis
A box measuring 1.35 × 105 µm2 was placed onto areas of lamina I–II of the dorsal horn under 20× magnification using a microscope. Subsequently, the number of profiles positive for the aggregated-grain cells, and each marker was counted in this area. The assessments were performed on 13–15 spinal sections of each animal.
Statistics
Data are expressed as mean ± standard error of the mean. One-way analysis of variance (ANOVA) followed by individual post hoc comparisons (Fisher’s exact test) or pairwise comparisons (t test) was used to assess differences between control groups and each drug-treated group. Two-way repeated ANOVA followed by Fisher’s Protected Least Significant Difference (PLSD) was applied to analyze the behavioral data. A difference was accepted as significant if p < 0.05.
Results
Reevaluation of mGluR mRNA expression in the spinal cord of rats
We first examined mRNA expressions of eight subtypes of mGluRs mRNAs in the spinal cord, which have been investigated in the previous studies.27–30 We used the L4–L5 spinal cords, which were obtained from naive rats, were used to detect mGluR mRNAs with the conventional reverse transcription-polymerase chain reaction (RT-PCR) method (Figure 1(a)). mGluR1, 5 (Group I); 3 (Group II); and 4, 7, and 8 (Group III) mRNAs were detected in the spinal cord, but mGluR2 and 6 mRNAs could not be detected in the spinal cord. These results are consistent with those in the previous studies.27,28 The PCR results led us to examine whether the primers for mGluR2 and 6 were right primers. We found that primers for mGluR2 and 6 could generate the precise size of PCR products using rat cortex RNA as positive controls (Figure 1(b)), and DNA sequencing confirmed that the PCR fragments were correct (data not shown). Information of each primer is shown in Table 1. Subsequently, we performed ISHH using radioisotope-labeled cRNA probes to elucidate the expression pattern of mGluR mRNAs in the spinal cord. Consistent with the results of RT-PCR, we detected mRNA expressions of mGluR1, 3, 4, 5, 7, and 8 (Figure 1(d) to (f)), but not mGluR2 and 6 (data not shown), in the spinal cord. Figure 1(d) to (f) shows the lower magnification dark field images (upper lane) of the half of the spinal cord (see Figure 1(c)) and the higher magnification bright field images of lamina I–II of ISHH for each mGluR (lower lane). Lower magnification dark field images showed that mGluR1 and 4 mRNAs were expressed in the gray matter of the spinal cord (upper lane in Figure 1(d) and f). Because mGluR3 was uniformly expressed throughout the spinal cord, it might be localized not only in the neurons but also in the glial cells (upper one in Figure 1(e)). mGluR5 and 7 mRNAs showed a similar expression pattern, and they were unevenly distributed in the dorsal horn of the spinal cord; aggregated-grains were especially located in lamina I–III (upper lane in Figure 1(d) and (f)). mGluR8 mRNA was relatively expressed in the gray matter of the spinal cord (upper lane in Figure 1(f)). Higher magnification bright field images of lamina I–II of the spinal dorsal horn revealed that expression of mGluRs, except for mGluR3, was frequently localized in the cells whose nuclei were lightly stained by hematoxylin, indicating that they are expressed in neurons. Expression of mGluR3 mRNA was occasionally observed in densely hematoxylin-stained cells, indicating that it could be expressed in glial cells (under one in Figure 1(e)). Because a single labeling study was not enough to identify the expression pattern of each mGluR in the spinal dorsal horn, we conducted a double-labeling analysis using ISHH and IHC of NeuN, which is a reliable antibody for neuronal cell markers (Figure 2). No specific immunostaining of the antibody was observed in the absence of the primary antibody (data not shown). Representative images of double labeling experiments (ISHH aggregation of grains with IHC brown staining) are shown in Figure 2(a) to (f). mGluR1, 5, and 7 mRNAs were heavily colocalized with NeuN-positive cells (Figure 2(a), (b), and (e)), and the percentages of colocalization of mGluR1, 5, and 7 mRNAs with NeuN (denominator; NeuN, numerator; mGluRs) were 93.6 ± 0.6%, 96.6 ± 1.0%, and 95.7 ± 0.6%, respectively. Conversely, colocalization of NeuN with mGluR1, 5, and 7 mRNAs (denominator; mGluRs, numerator; NeuN) were 90.2 ± 1.0%, 92.1 ± 0.6%, and 96.9 ± 0.4%, respectively (Figure 2(g) and (h)). mGluR3 mRNA was occasionally detected in NeuN-positive cells (Figure 2(c)), and the percentage of colocalization of mGluR3 mRNA with NeuN was 57.1 ± 2.7%. Conversely, colocalization of NeuN with mGluR3 mRNA was 59.2 ± 0.8% (Figure 2(g) and (h)), indicating that it is expressed in a subset of neurons and glial cells. Expression of mGluR4 and 8 mRNAs were very similar in lamina I–II, their mRNAs were frequently colocalized with NeuN-positive cells (Figure 2(d) and (f)), and the percentages of colocalization of their mRNA with NeuN were 94.0 ± 0.9% and 94.7 ± 1.1%. Conversely, colocalization of NeuN with their mRNAs were 54.4 ± 2.2% and 54.6 ± 2.6% (Figure 2(g) and (h)), suggesting that mGluR4 and 8 are expressed in a subset of neurons in the spinal dorsal horn. Our findings indicate that almost all mGluRs are normally expressed in neurons in the dorsal horn, and only mGluR3 is expressed in glial cells in the spinal dorsal horn. Hence, we next used antibodies for several glial markers, that is, GFAP for astrocytes, ionized calcium-binding adapter molecule 1 (Iba1) for microglia, and oligodendrocyte transcription factor 2 (Olig2) for oligodendrocytes to identify the mGluR3 mRNA-expressing cells (Figure 3). We found that mGluR3 mRNA was occasionally expressed in oligodendrocytes (Figure 3(c)), but not in astrocytes (Figure 3(a)), or microglia (Figure 3(b)), in the spinal dorsal horn.
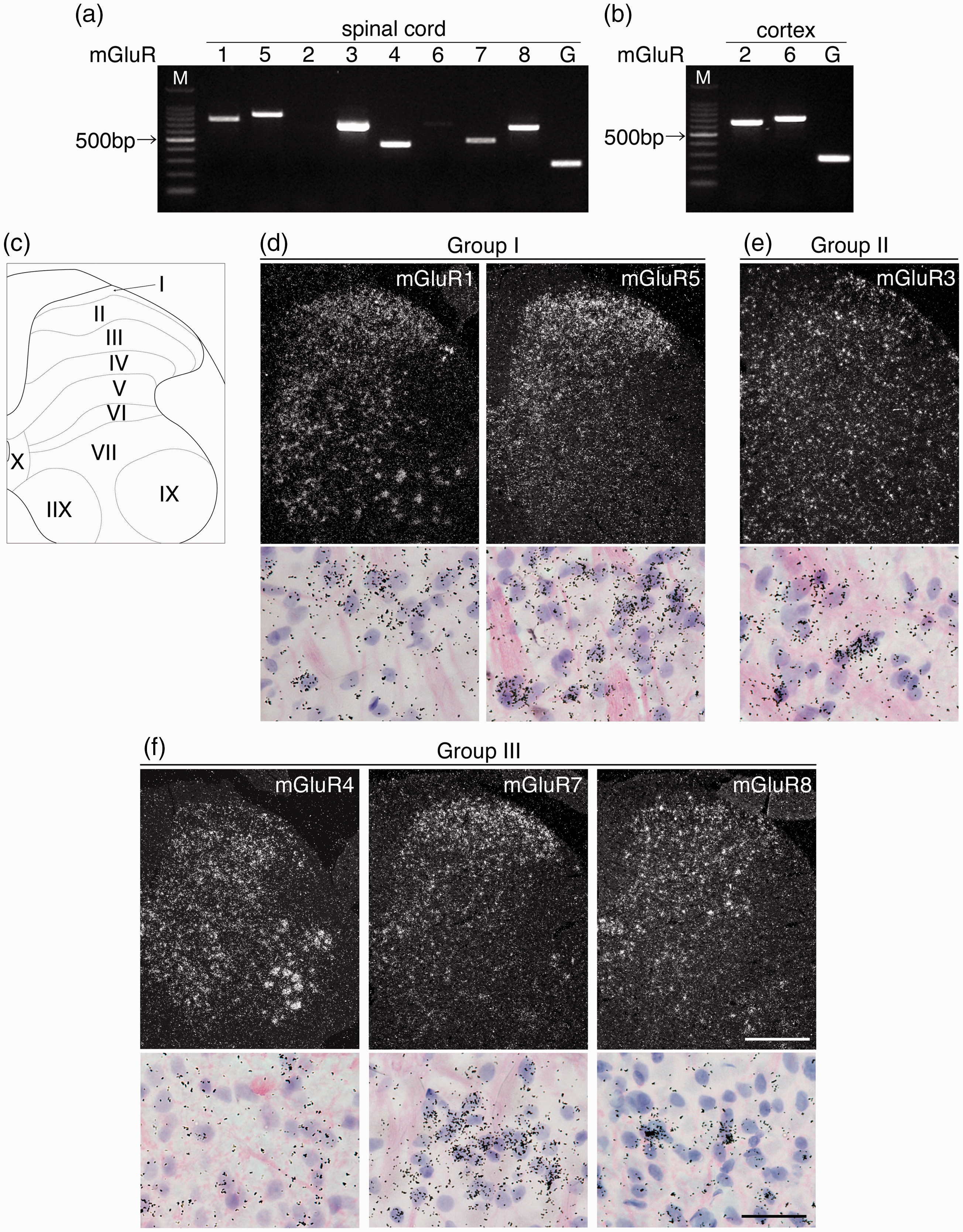
Expression patterns of a series of mGluRs in the spinal cord. Electrophoresis images show mRNA expressions of mGluRs in L4–L5 of the spinal cord (a) and mGluR2 and 6 in the cortex for positive control (b). Different gene primers produced different lengths of PCR products: 776 bp (mGluR1), 655 bp (mGluR2), 699 bp (mGluR3), 454 bp (mGluR4), 861 bp (mGluR5), 713 bp (mGluR6), 490 bp (mGluR7), 658 bp (mGluR8), and 271 bp (GAPDH). (c) The drawing indicates the lamina structure of the half of spinal cord for dark field ISHH images. Dark field images (upper panels) show the expression pattern of Group I (d), Group II (e), and Group III (f) mGluRs in the spinal cord. Lower panels show higher magnification bright field images of the dorsal horn (lamina I–II) at the upper panels. Sections were counterstained by hematoxylin-eosin. Aggregation of grains indicates positive cells in ISHH. Scale bar = 500 μm in upper panels and 12.5 μm in lower panels (d–f).
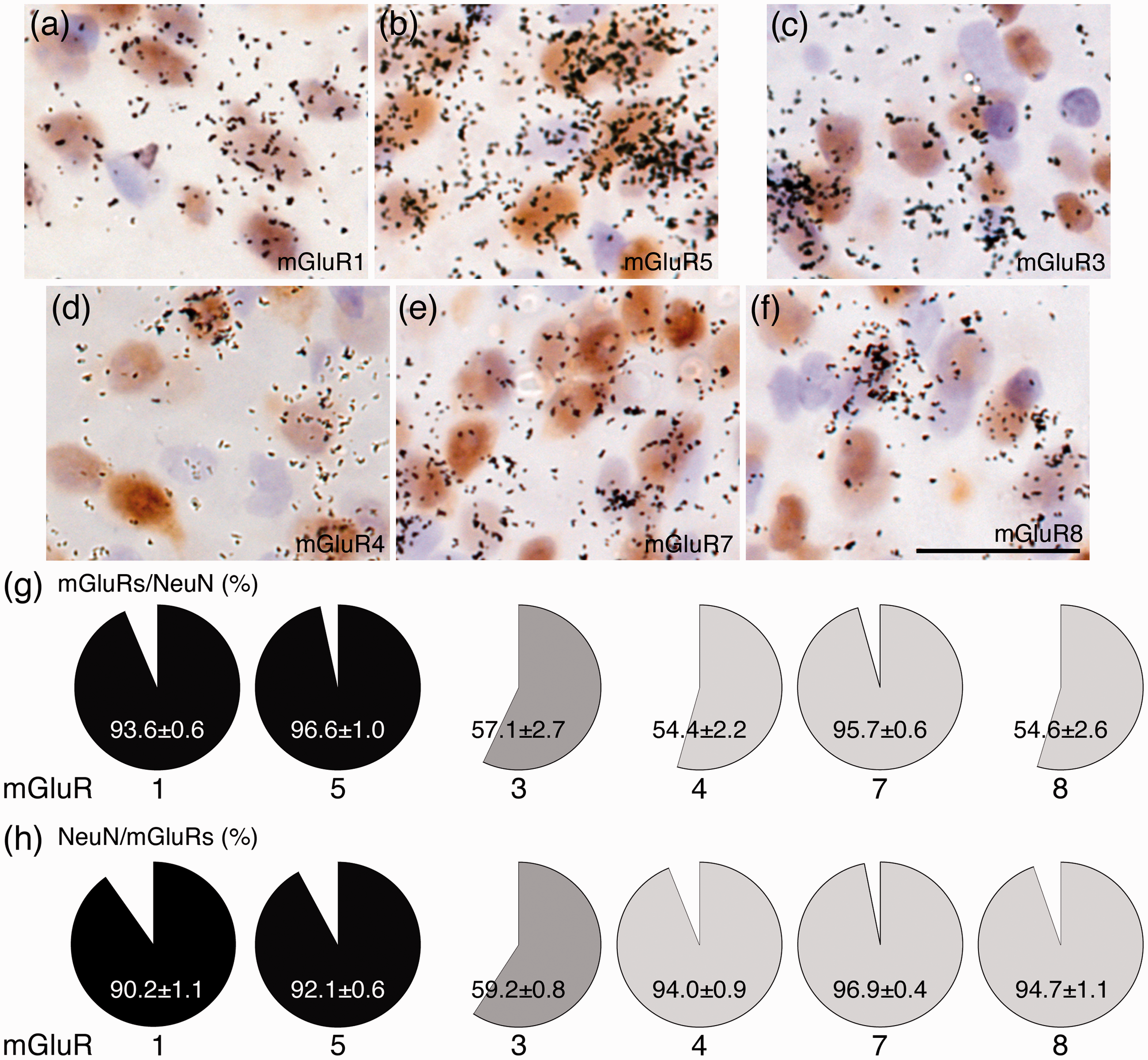
Percentages of each mGluR colocalization with neurons in the lamina I–II of the spinal dorsal horn. (a–f) Double labeling ISHH study of each mGluR mRNA with immunohistochemistry of NeuN in the lamina I–II. Panels show colocalization of Group I (a and b), Group II (c), and Group III (d–f) mGluR mRNAs (aggregation of grains) with NeuN (brown staining). Aggregation of grains indicates the positive cells in ISHH. Brown-stained cells indicate IHC-positive cells. The percentages of colocalization of each mGluR mRNA-expressing cells in NeuN-positive cells (g). NeuN-positive cells in each mGluR mRNA-expressing cells (h). Sections were counterstained with hematoxylin. Scale bar = 10 μm in (a) to (f). Data are presented as mean ± standard error of the mean (n = 4 in (g) and (h)).

Double labeling analyses of mGluR3 mRNA with glial cell markers. ISHH of mGluR3 mRNA and IHC of GFAP (a), Iba1 (b), and Olig2 (c). Solid arrowheads indicate single-labeled cells using ISHH, open arrowheads indicate single-immunostained cells, and arrows indicate double-labeled cells. Sections were counterstained by hematoxylin. Scale bar = 12.5 μm.
Group III mGluRs were more frequently expressed in inhibitory than excitatory neurons of the spinal dorsal horn
Our results indicate that mGluR1, 5, and 7 are expressed in almost all neurons (>90%), but mGluR3, 4, and 8 are expressed in approximately 60% of neurons, indicating that mGluR3, 4, and 8 may be expressed in specific subsets of neurons in the spinal dorsal horn. Therefore, we examined whether mGluR3, 4, and 8 show different expression patterns in excitatory and inhibitory neurons using lmx1b and pax2 antibodies (Figure 4). mGluR3 mRNA was expressed in 20.9 ± 1.8% of lmx1b-positive and 25.6 ± 1.7% of pax2-positive neurons, indicating that mGluR3 mRNA is almost equally expressed in a subset of excitatory and inhibitory neurons (left column in Figure 4(a) to (d)). Approximately 20% of lmx1b-positive neurons expressed mGluR4 (21.5 ± 1.7%) and 8 (19.1 ± 1.6%) mRNAs, but approximately 60% of pax2-positive neurons expressed mGluR4 (59.7 ± 3.3%) and 8 (60.9 ± 2.6%) mRNAs (middle and right columns in Figure 4(a) to (d)). These results indicate that mGluR4 and 8 are preferentially expressed in inhibitory than excitatory neurons in lamina I-II of the spinal dorsal horn.

Percentages of colocalization of mGluR3, 4, and 8 with lmx1b- or pax2-positive cells in lamina I–II. Representative images show colocalization of mGluR3 (left column), 4 (middle column), and 8 (right column) mRNAs with lmx1b (a) or pax2 (c) immunoreactive cells. Percentage of colocalization of each mGluR-expressing cells with lmx1b (b) or pax2 immunoreactive cells (d). Solid arrowheads indicate single-labeled cells using ISHH, open arrowheads indicate single-immunostained cells, and arrows indicate double-labeled cells. Sections were counterstained with hematoxylin. Scale bar = 12.5 μm. Data are presented as mean ± standard error of the mean (n = 4).
Group III mGluR antagonist reduced nocifensive behaviors in phase II of the formalin test
Given that mGluR4 and 8 are preferentially expressed in inhibitory neurons, we wondered whether they are involved in the modulation of pain transmission in the spinal dorsal horn. Therefore, we conducted the formalin test that is widely used as an observation method for nocifensive behaviors. First, we performed an intraplantar injection of formalin (1.5% in 50 μl saline) and observed nocifensive behaviors in rats. As demonstrated in the previous studies,31,32 formalin injection induced a typical biphasic response as shown in Figure 5(a). An increased number of flinching was noted between 0 and 5 min and between 20 and 50 min, whereas such behaviors were not observed in vehicle (PBS)-injected rats (data not shown). Second, we examined whether Group III mGluRs, including mGluR4 and 8, agonist (L-AP4), or antagonist (CPPG) influenced nocifensive behavioral responses following a formalin injection. These drugs were injected into the spinal subarachinoid space (L4–L5). Five minutes after intrathecal pretreatment with CPPG, the number of flinching in phase II of the formalin test, but not in phase I, was significantly reduced compared with that of vehicle-treated rats. Intrathecal pretreatment of L-AP4 did not change the flinching behavior in either phase. The cumulative number of flinching during phase I and phase II clearly differed among the three groups (Figure 5(b)). No significant change in phase I was observed among the three groups (left in Figure 5(b)); however, in phase II, pretreatment with CPPG but not L-AP4 reduced the number of formalin-induced flinching (right in Figure 5(b)).

Inhibition of Group III mGluRs suppressed fos expressions in lmx1b-positive cells and nocifensive behaviors. Rats were pretreated with CPPG (500 nmol), L-AP4 (100 nmol), or PBS intrathecally 5 min before formalin (1.5% in 50 μl PBS) injection. Time course (a) and cumulative data (b) of the number of flinching after formalin injection with CPPG, L-AP4, or PBS pretreatment were measured. The number counted per 5-min interval in the initial hour postinjection period in (a). (b) The cumulative number of flinching during phase I (0–5 min) and phase II (5–60 min) after formalin injection. (c) Representative images of fos (upper lane, green) and lmx1b (middle lane, red) immunoreactive cells in the dorsal horn of controls (left column), vehicle-treated (second column), CPPG-treated (third column), and L-AP4-treated (right column) animals. (d) Bar graphs show the quantitative number of total fos-expressing cells (left), and that of fos-expressing cells in lmx1b-positive (middle) or lmx1b-negative cells (right) in the dorsal horn. Solid arrowheads indicate the merged cells. ctr: control; veh: vehicle. #<0.05 versus ctr, § <0.05 versus veh. Scale bar = 12.5 μm. Data are presented as mean ± standard error of the mean (n = 5–6). CPPG: (RS)-α-cyclopropyl-4-phosphonophenylglycine; L-AP4: L-(+)-2-amino-4-phosphonobutyric acid; n.s.: not significant.
Pretreatment with CPPG suppressed the formalin-induced fos expression in Lmx1b-positive cells
Behavioral tests showed that pretreatment with CPPG attenuated nocifensive behaviors induced by a formalin injection. Therefore, we examined whether fos expressions are affected in the dorsal horn lamina I–II neurons 2 h after formalin injection (Figure 5(c) and (d)). IHC revealed that intraplantar injection of formalin dramatically induced fos expressions in dorsal horn neurons, and the findings are consistent with those in the previous studies (first vs. second column in Figure 5(c)). We quantified the number of fos-positive cells in the lamina I–II of the dorsal horn (Figure 5(d)). Pretreatment with CPPG and L-AP4 had no effects on the total number of fos-positive cells induced by formalin injection (left column in Figure 5(d)). Interestingly, CPPG decreased the number of fos-positive cells in Lmx1b-positive neurons (third column in Figure 5(c), middle in Figure 5(d)), but not in Lmx1b-negative neurons (right in Figure 5(d)), in the lamina I–II of the dorsal horn. L-AP4 did not affect the fos-positive cells in Lmx1b-positive or Lmx1b-negative neurons (right column in Figure 5(c) and (d)). These results suggested that the antagonist of Group III mGluRs triggers the disinhibition of inhibitory neurons. Therefore, inhibition of Group III mGluRs suppresses the excitability of excitatory neurons in the lamina I–II of the spinal cord, and formalin-induced nocifensive behaviors may be ameliorated by the antagonism of Group III mGluRs.
Discussion
Expressions of mGluRs in the spinal cord
The present study precisely investigated the expression pattern of all mGluRs mRNAs in the lamina I–II of the spinal dorsal horn. We found that mRNAs of all mGluRs, except mGluR2 and 6 mRNAs, were expressed in the spinal cord of rats. A previous ISHH study indicates that mGluR2 mRNA is frequently expressed throughout the central nervous system, but no significant hybridization signals are detected in the spinal cord. 28 In addition, previous studies indicate that the expression of mGluR6 is restricted in the retina; however, recent studies using transgenic techniques have identified its expression in some brain regions, such as the superior colliculus, hypothalamus, and olfactory bulb, in mice or other species.2,3 Considering these findings, the conventional RT-PCR findings are likely regarding the expression of each mGluR mRNA, as shown in Figure 1(a) and (b). We next attempted to assess the expression patterns of all mGluR mRNAs in the spinal cord using ISHH. Expressions of mGluR1 and 5 (Group I) had similar patterns and levels of distribution. Notably, the signals of mGluR1 were relatively lower and broader throughout the gray matter, especially around motoneurons, but the mGluR5 expression was very strong in the superficial dorsal horn, mainly in the lamina I–III. These data were supported by previous studies. The expressions of mGluR1 mRNA and protein have been demonstrated in the lamina I–III of the dorsal horn and to a lower extent in the ventral horn.27,33 Several studies have shown robust expressions of mGluR5 mRNA and protein in the dorsal horn.34,35
Our results showed that mGluR3 mRNA was expressed throughout the spinal cord but not significantly in motoneurons. Furthermore, double labeling analysis (Figures 2(c) and 3) demonstrated that mGluR3 mRNA was expressed in a subset of neurons and glial cells in the spinal dorsal horn. These findings are in agreement with those in the previous studies.27,29 In the central nervous system, especially in the brain, mGluR3 is known to be expressed in glial cells and neurons.29,36–38 Notably, in vitro studies indicate mGluR3 is abundantly expressed in astrocytes, microglia, and oligodendrocytes.39,40 However, it is unclear which types of glial cells express mGluR3 in the nervous system in vivo. Our double labeling analysis found that mGluR3 was expressed in oligodendrocytes in the spinal dorsal horn in rats. In vitro studies have indicated that mGluR3 plays a role in differentiation and myelination in oligodendrocytes. 41 However, further studies are needed to investigate the predominant function of mGluR3 in oligodendrocytes in adult rats.
We further detected mRNA expressions of Group III mGluRs, that is, mGluR4, 7, and 8, in the spinal cord. mGluR4 mRNA was expressed in a dispersed manner over the gray matter including motor neurons in the ventral horn. In contrast, mGluR7 mRNA was localized within the dorsal horn and to a somewhat lesser extent in the deep dorsal and ventral horn. The expression pattern of mGluR7 mRNA was similar to that of mGluR5 mRNA. These data are consistent with those in the previous studies. 27 mGluR8 has been known to be absent in the adult spinal cord,27,30,42 but recently pharmacological studies indicated that mGluR8 modulates pain signaling in the dorsal horn, suggesting that it might be expressed in the spinal cord.16,26,43 However, no studies have investigated whether mGluR8 is expressed in the spinal cord, in vivo. In this study, we found that mGluR8 was relatively expressed in the dorsal horn and to a somewhat lesser extent in the deep dorsal and ventral horn.
Taken together, the findings suggest that excitatory output from neuraxis of spinal dorsal horn is the summation determined by the complicated combination of Gαq/11 and Gαi/o mGluR expression.
Different expression patterns of mGluRs in spinal excitatory and inhibitory neurons of lamina I–II
This study is the first to investigate the percentage of colocalization of each mGluR with neurons in the spinal cord. According to this analysis, >90% of neurons expressed mGluR1, 5, and 7 mRNAs; conversely, >90% of these mGluRs-positive cells were colocalized with neurons, indicating that almost all neurons express these mGluRs. We thought that mGluR1, 5, and 7 might have limited roles in the regulation of the excitatory output of the neuronal network in lamina I–II under normal conditions. On the other hand, double labeling analysis showed that approximately 60% of neurons expressed mGluR3, 4, and 8 mRNAs, and more than 90% of mGluR4 and 8-positive cells were colocalized with neurons. These findings indicate that mGluR4 and 8 have some specific expression patterns in the neurons and may modulate the excitatory output of the neuronal network in the spinal cord. It has been suggested that Group II and Group III mGluRs are located in both excitatory and inhibitory neurons4,44; however, the precise distribution and percentage of these mGluRs in both types of neurons are still unclear. Therefore, we next examined whether mGluRs are specifically expressed in excitatory and inhibitory neurons in the lamina I–II of the dorsal horn. mGluR3 was evenly expressed in both excitatory and inhibitory neurons. These results may indicate that mGluR3 has limited roles in the balance of excitatory and inhibitory signals in the lamina I–II of the spinal cord. Interestingly, mGluR4 and 8 mRNAs were preferentially expressed in inhibitory neurons (pax2 positive) rather than in excitatory ones (lmx1b positive). Given these results and intracellular signaling (Gi/o) of these receptors under normal conditions, it is likely that mGluR4 and 8 mainly suppress inhibitory neurons, resulting in increased responsiveness in excitatory neurons.
Inhibitory neurons-expressing-mGluR4 and 8 might play a key role in the determination of excitatory output of pain transmission in spinal dorsal horn
Studies have demonstrated the roles of mGluRs in pain transmission in the spinal cord. Accumulating evidence indicated that the effective control of pain hypersensitivity can be achieved by either inhibiting Group I mGluRs or stimulating group II and III mGluRs. Group I mGluRs, especially mGluR1 and 5, are known to be pronociceptive. For example, it has been reported that inhibition of Group I mGluRs significantly suppressed formalin-induced pain behaviors. 45 Our data in this study mostly agreed with many of these previous reports. Although pharmacological inhibition of mGluR1 and 5 in spinal neurons suppresses pain transmission, inhibition of these receptors has an impact on the function of all neurons, which might lead to side effects. It may be possible to develop a more efficient method for pain relief based on the differential expression pattern of mGluRs in the spinal cord. Indeed, intrathecal injection of CPPG, an antagonist for Group III mGluRs, significantly suppressed the second phase of the formalin test. Increased number of fos-positive cells in excitatory neurons induced by formalin were attenuated by CPPG injection, although the behavioral findings are poorly supported by those in the previous studies.16,43,46 Furthermore, L-AP4, a Group III mGluRs agonist, could not reverse the pain behaviors. The findings are also inconsistent with those in the previous studies.10,15,16,43 Presently, it is difficult to clarify the reason for this discrepancy; however, our behavioral results are corroborated by the findings that the agonist of Group III mGluRs had no effect on the increased number of c-fos-positive cells in the spinal cord. We hypothesized that suppression of inhibitory neurons induced by the agonist of Group III mGluRs has limited roles in the nociceptive networks in the dorsal horn. Further studies are needed to evaluate this issue. In addition, injection of CPPG and L-AP4 might have some effects on motor neurons-expressing mGluR4. However, mGluR4 KO mice do not show any behavioral changes in normal condition. 17 Therefore, it seems that doses of CPPG and L-AP4 in this study have no effects on the motor function.
In summary, this study illustrated the precise expression patterns of each mGluR in the lamina I–II of the spinal cord, and the findings indicated that mGluR4 and 8 are preferentially expressed in inhibitory neurons rather than in excitatory neurons. Suppression of Group III mGluRs, including mGluR4 and 8, could attenuate formalin-induced nocifensive behaviors and the increase in fos expression in excitatory neurons.
It is known that the functions of glutamate are mediated by ionotropic glutamate receptors (iGluRs) and mGluRs. Because iGluRs are cation channel, they always lead the neurons to ionic activation as action potentials. Given the combination of the expression and function of iGluRs and mGluR, the final output of pain transmission from the spinal cord is mediated in an extremely complicated manner in the dorsal horn. Further studies using different techniques are needed to clarify the mechanisms by which iGluRs and mGluRs induce pain relief. It is not necessarily that the expression levels of mRNAs match with those of proteins, but we believe that our results may contribute to a fundamental understanding of the function of mGluRs in the spinal cord and their role in pain modulation.
Footnotes
Acknowledgments
Authors’ Contributions
MO and HY designed the study; MO and KK conducted experiments; MO prepared figures; MO drafted the paper; HY supervised experiments; and KN edited the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Japan Society for the Promotion of Science KAKENHI (Grant number: 17H04113).
