Abstract
Metabotropic glutamate receptor 5 (mGluR5) and transient receptor potential vanilloid subtype 1 (TRPV1) have been shown to play critical roles in the transduction and modulation of cutaneous nociception in the central nervous system. However, little is known regarding the possible involvement of mGluR5 and TRPV1 in regulating visceral nociception from the uterine cervix. In this study, we used a rat model of uterine cervical distension to examine the effects of noxious stimuli to the uterine cervix on expression of spinal mGluR5 and TRPV1. Our findings included the following: (1) uterine cervical distension resulted in a stimulus-dependent increase in electromyographic, spinal c-Fos signal, and expression of mGluR5 and TRPV1 in the spinal cord; (2) intrathecal administration of the mGluR5 antagonist 2-methyl-6-(phenylethynyl)-pyri-dine significantly reduced the increased TRPV1 and c-Fos expression induced by uterine cervical distension; (3) the TRPV1 inhibitor SB-366791 inhibited increased spinal c-Fos expression but had no effect on the expression of mGluR5 in response to uterine cervical distension. Our findings indicate that the spinal mGluR5-TRPV1 pathway modulates nociceptive transmission in uterine cervical distension-induced pathological visceral pain.
Keywords
Introduction
Metabotropic glutamate receptor 5 (mGluR5) and transient receptor potential vanilloid 1 (TRPV1) channel both play important roles in the transmission of nociceptive stimuli. Studies in mice and rats have demonstrated the role of mGluR5 in the transmission of inflammatory1–3 and neuropathic 4 pain and the role of TRPV1 in the transmission of painful chemical and thermal stimuli. 5 The transmission of nociceptive signals can be inhibited by the use of antagonists selective against mGluR5 and TRPV1. 2-methyl-6-(phenylethynyl)-pyridine (MPEP) is a potent, selective mGluR5 antagonist, 2 while N-(3-methoxyphenyl)-4-chlorocinnamide (SB-366791) is a highly potent and selective TRPV1 antagonist. 6
The role of mGluR5 and TRPV1 in the transmission of visceral pain from the gastrointestinal and urinary systems has been studied in rodent models. MPEP was found to decrease colorectal distension-evoked visceromotor responses in rats, indicating that mGluR5 plays a role in the transmission of visceral nociceptive signals from the gastrointestinal tract. 7 Similarly, in vitro experiments using mouse bladders demonstrated that TRPV1 is critical for transmission of stretch-evoked signals, 8 and TRPV1 has also been found to be involved in nociception in the mouse jejunum 9 and colon. 10 It appears that a relationship between TRPV1 and mGluR5 may exist, in which sensitization of TRPV1 through mGluR5 signaling enhances nociceptive stimuli. 11
While the role of mGluR5 and TRPV1 in transmitting nociceptive signals in the gastrointestinal and urinary systems has been well characterized, their roles in uterine cervical pain is less well understood. It is known that afferents in the hypogastric nerve originating from the rat uterine cervix express TRPV1, 12 and that estrogen may play a role in enhancing the uterine cervical nociceptive signals transmitted by TRPV1. 13 However, the interplay between mGluR5 and TRPV1 during the transmission of painful stimuli during uterine cervical distension (UCD) is unclear.
The aims of this study were to investigate the effects of UCD in a rat model on mGluR5 and TRPV1 expression and to investigate the effects of MPEP and SB-366791 on the expression of TRPV1 and mGluR5. Our hypothesis was that the expression of mGluR5 and TRPV1 would be increased with UCD, and that MPEP and SB-366791 may decrease TRPV1 and mGluR5 expression.
Materials and methods
All experimental procedures were carried out in accordance with the National Institute of Health Guide for the Care and Use of Laboratory Animals (NIH Publications No. 86-23). The Animal Ethnics and Welfare Committee of the Zhejiang University Department of Medicine approved all experimental procedures. Every effort was made to minimize the number of animals used and their suffering.
Animals
A total of 160 Sprague-Dawley-derived adult virgin female rats (Zhejiang Chinese Medical University, Hangzhou, China, 180–220 g) were used. Rats were divided into the following groups: Rats subjected to varying degrees of distension forces (5 groups of 18 rats each, n=90), rats treated with antagonists or vehicle (4 groups of 12 rats each, n=48), and control rats (n=16). All rats were allowed one week to acclimate to the house facilities. Rats in whom catheter implantation surgery was planned were housed one per cage; all other rats were housed five per cage. Rats were kept in a temperature-controlled room (22°C) with free access to food and water and a 12:12-h light-dark cycle. At the end of the experiments, all rats were euthanized.
UCD model
A UCD model previously described was modified for the purposes of this study. 14 Rats were anesthetized with inhalation of isoflurane (Hebei Yipin, China) in oxygen via an isoflurane vaporizer (Matrix, USA) and were maintained with spontaneous ventilation. The concentration of isoflurane was adjusted from 0.7% to 0.9% so as to allow for detection of rectus abdominis visceromotor response but to prevent spontaneous motor movement. Catheters were inserted into the right carotid artery and vein for blood pressure monitoring and intravenous fluid administration, respectively. A tracheotomy was performed for artificial ventilation. Body temperature was maintained at 37°C to 38°C by a circulating water heating pad throughout the experiment.
Once the rats were anesthetized, a 1.5-cm midline lower abdominal incision was made to expose the uterus. All incisions were infiltrated with lidocaine (Hebei Yipin, China). Two sterilized fine metal rods (23G needles, RWD Life Science, China) were inserted through the cervical os, with one end entering the uterus through the abdominal incision and the other end exiting through the upper vaginal wall. Each rod was connected to a 1-0 silk suture, with one end attached to a metal stand and the other end connected to a pulley system for application of manual weighted traction. Each rat was then subjected to UCD with a distension force of 0, 25, 50, 75, or 100 g. To simulate contractions occurring at regular intervals during labor, distension forces were applied for 20 s every 3 min for a total duration of 1 h. Following UCD, a 30-min recovery period was allowed prior to subsequent procedures. The submaximal distension force of 75 g was the only force applied in rats that had drug or vehicle administration.
Visceromotor responses
The visceromotor responses to UCD were quantified using electromyographic (EMG) recordings from the rat rectus abdominis muscle. Two concentric monitoring needle electrodes (Mei Yi, Nanjing, China) were inserted into the rectus abdominis muscle immediately after placement of the UCD rods. The baseline EMG amplitude was measured for 10 min, after which visceromotor responses were recorded throughout the entire hour of UCD. Physiological measurements, including electrocardiography and blood pressure, were also recorded by a MedLab biological signal acquisition system (Mei Yi, Nanjing, China). The EMG signal was amplified and filtered at 3000 Hz, and raw data were then integrated and digitized by calculating the area under the curve. Following measurement of the raw EMG amplitude during the 20 s of each distension cycle, the net EMG amplitude was calculated by subtracting the mean EMG amplitude during the first 20 s after each distention ceased from the raw EMG amplitude.
Intrathecal catheter placement
A previously described method for insertion of an intrathecal catheter was used 15 for those rats that underwent intrathecal catheter placement. After anesthetizing the rats with an intraperitoneal injection of 60 mg/kg sodium pentobarbital (Hebei Yipin, China), a midline incision at the atlanto-occipital junction was made. A fine bore polythylene-10 catheter (Smiths Medical, USA) filled with 0.9% sterile saline was passed through the dura and into the subarachnoid space. The catheter was advanced 7.5 cm caudally such that the tip was placed at the level of the lumbar enlargement, while the cephalic portion of the catheter was secured at the neck. Three days after intrathecal catheter placement, catheter position was verified by injecting 2 mg/kg of 2% lidocaine (Hebei Yipin, China). Rats were given five to seven days to recover after intrathecal catheter placement. If signs of motor impairment were present, rats were immediately euthanized.
Drugs
MPEP and SB-366791 (Selleck Chemicals, USA) were dissolved in 5% dimethyl sulfoxide (DMSO) (Sigma-Aldrich, USA) to a concentration of 20 nmol/µl. Thirty minutes prior to UCD, MPEP (30 µg in 25 µl) or SB-366791 (30 µg in 25 µl) was administered intrathecally via micro syringe (Shanghai High Pigeon, China) in those rats that were randomized to receive the mGluR5 and TRPV1 inhibitors. Rats randomized to vehicle treatment received 25 µl DMSO in the same manner, while rats randomized to the control group received no drug via intrathecal catheter. Following administration of the drug or vehicle, 5 µl saline was injected to ensure complete delivery. MPEP and SB-366791 dosing was based on previous reports.16–18 The dose of DMSO was based on previous studies that demonstrated a lack of noxious effect on spinal cord neurons at this dose. 2 Unless otherwise specified, chemical reagents were HPLC grade and purchased from Sinopharm Chemical Reagent.
Tissue preparation
After the experiments, rats were euthanized and incisions were made on both sides of the vertebral column. The tissues were dissected, and the area of the lumbar enlargement of the spinal cord encompassing the T12-L2 segments was removed under aseptic condition. All samples were then fast-frozen in liquid nitrogen and stored at –80°C.
Western blot experiments
The tissues were homogenized using mortars and pestles in an ice-cold lysis buffer (1% NP-40, 0.5% sodium deoxycholate, 0.1% sodium dodecyl sulfate, Boster, Wuhan, China) with freshly added protease inhibitor cocktail tablets (Roche Diagnostics, USA). Following 30 min of incubation, the liquid was centrifuged at 12,000 r/min at 4°C for 20 min. The supernatant was collected, and the protein concentration was calculated using the Bicinchoninic acid method according to the manufacturer’s instructions (KeyGen Biotech, Nanjing, China). An equivalent amount of protein (30 µg) and prestained protein ladder (ThermoFisher Scientific, USA) was loaded on a 12% sodium dodecyl sulfate polyacrylamide gel (Invitrogen, ThermoFisher Scientific, USA). Electrophoresis was run for 50 min at 200 V, after which the protein was transferred to polyvinylidene fluoride membranes (Millipore, USA), and the blots were blocked with 5% non-fat milk in tris-buffered saline for 2 h at room temperature. The blots were then incubated with primary antibodies at 4°C overnight. The antibodies used included the following: anti-β-actin antibody (rabbit affinity purified polyclonal antibody; 1:3000, Abcam, Hong Kong, China), anti-mGluR5 antibody (rabbit affinity purified monoclonal antibody; 1:5000, Abcam, Hong Kong, China), and anti-TRPV1 antibody (anti-rat TRPV1 antibody; 1:200, Alomone, Isreal). Blots were washed and incubated with horseradish peroxidase-conjugated secondary antibody (1:8000, Boster, Wuhan, China) for 2 h at room temperature. All antibodies were diluted in blocking buffer. Immunoblots were detected with an enhanced chemiluminescence kit (Millipore, USA) and visualized on GeneSnap image acquisition software (Syngene, UK). The density of the bands was quantified using densitometric analysis on Quantity One software (Bio-Rad, USA). The intensity of mGluR5 or TRPV1 bands was normalized to their β-actin intensity.
Quantitative real-time reverse-transcription polymerase chain reactions
Total RNA was isolated from the frozen lumbar spinal cord enlargement using Trizol reagent (Life Technologies, CA, USA). RNA concentration was measured with the NanoDrop ND-1000 Spectrometer (NanoDrop Technologies, Wilmington, DE). Two-step reverse transcription-polymerase chain reaction (RT-PCR) was performed using reagents and kits from Takara (China). Starting with 2.5 µg of total RNA, PCR amplification was performed using a SYBR PrimeScript Quantitative Real-Time RT-PCR Kit (Takara, China) according to the manufacturer’s instruction. The specific forward and reverse primers are shown in Table 1.
Forward and reverse primers of mGluR5, TRPV1, and β-actin.

mGluR5: metabotropic glutamate receptor 5; TRPV1: transient receptor potential vanilloid 1.
The selected primers amplify all known splice variants of mGluR5. PCR amplification was performed on the CFX96 Real-Time PCR Detection System (Bio-Rad, USA). Relative changes in mGluR5 and TRPV1 messenger RNA levels were calculated using the ΔΔCt method, and the reference gene β-actin was used as a normalizer. Reactions without any template were included as negative controls.
Immunochistochemistry
Rats were perfused transcardially with 4% paraformaldehyde at 4°C for the immunohistochemistry experiments. Tissue removed from perfused animals was then post-fixed for 5 h in 4% paraformaldehyde and then cryoprotected overnight in 30% sucrose in phosphate-buffered saline (PBS) at 4°C. Tissue was cryosectioned (8 µm), mounted on microscope slides, incubated with normal goat serum (Boster, Wuhan, China) in PBS for 2 h to block non-specific staining, and finally incubated with primary antibodies (rabbit polyclonal anti-c-Fos antibody, Santa Cruz, USA) at 4°C overnight. The sections were then washed in PBS and incubated with FITC-conjugated goat anti-rabbit antibody (Beyotime, Shanghai, China) for 30 min at room temperature. In the control experiments, tissues were processed using the same staining protocol, but without applying the primary antibodies. For co-localization of mGluR5 and TRPV1 in the spinal cord, fluorescent probes (Anti-Rat-TRPV1-ATTO-488, 1:50; Anti-mGluR5-ATTO-594, 1:50; Alomone, Isreal) were applied simultaneously to the sections. Coverslips were mounted using Antifade Mounting Medium (Beyotime, Shanghai, China) and viewed using a fluorescence microscope (Eclipse Ni-E, Nikon, Shanghai, China). Images were analyzed using the Image J 1.50i (National Institute of Health, USA). On the images, a line was drawn surrounding the area of spinal lamina III-V, and an observer blinded to the groups counted the number of Fos-positive neurons within this region. The total number of nuclei in the same area was calculated, and the proportion of Fos-positive neurons to the number of nuclei of the same region is presented in the results.
Statistical analysis
Data are expressed as mean ± SD. PCR and Western blot data after UCD were analyzed by one-way analysis of variance for overall differences among groups, with Bonferroni’s multiple comparisons test for pairwise comparisons. The semi-quantified data of spinal c-Fos expression in each group were compared using two-tailed paired student t test. Statistical analysis was performed using SPSS Statistics Version 22.0 (IBM Corporation, Armonk, NY). P < 0.05 was considered statistically significant.
Results
Effect of UCD on spinal mGluR5 expression
The mGluR5 antibody detected a Western blot band at 137 kDa, while the β-actin antibody detected a Western blot band at 45 kDa (Figure 1(A)). The integrated optical density (IOD) ratio of each band was determined, and fold increase in IOD is presented in the figures. UCD significantly increased spinal mGluR5 mRNA and protein expression in a stimulus intensity-dependent manner (Figure 1(B) and (C)). The immunohistochemistry results also demonstrated that the change in spinal mGluR5 levels was primarily found in the deep dorsal horn (lamina III∼V) region of the spinal cord. While lamina I and II expressed dense mGluR5, its expression did not change significantly between the stimulated and sham groups (Figure 1(D)). In addition, UCD seemed to elicite a more drastic increase of TRPV1 expression in rat dorsal root ganglion neurons comepared with control group.( Supplemental figure 11).

Western blot of mGluR5 shows UCD stimulus intensity-dependent response. (A) Western blot showing expression of spinal mGluR5. Loading control with β-actin is shown in lower portion. The numbers on the bottom of the Western blot represent the distension forces. (B) Histogram showing fold change in density of mGluR5 protein level normalized to β-actin in response to UCD stimulation (n=9). Data are presented as mean ± SD. (C) Histogram showing fold change in mGluR5 mRNA levels as quantified by real-time RT-PCR analysis in response to UCD stimulation (n=9). Spinal mGluR5 mRNA expression increased as the distension force increased in the rat cervix. Data are presented as fold change from the control (distention strength of 0 g) as mean ± SD. ***P < 0.001 when compared with sham group, ###P < 0.001 when compared with 25 g group, △△△P < 0.001 when compared with 50 g group. (D) mGluR5 immunoactive neurons in the spinal cord of sham- and UCD- (75 g) treated group. mGluR5 increased in the UCD group compared to sham (a, d). Lamina I, II of the dorsal horn expressed more mGluR5 positive neurons, and this region did not show significant change between sham and UCD group (b, e), Lamina III-V (deep dorsal horn region as framed out by the rectangular) demonstrated an increase in mGluR5 with UCD stimulation (c, f).
Effect of UCD on spinal TRPV1 expression
The TRPV1 antibody detected a Western blot band at 97 kDa, while the β-actin antibody detected a Western blot band at 45 kDa (Figure 2(A)). The IOD ratio of each band was determined, and fold increase in IOD is presented in the figures. UCD significantly increased spinal TRPV1 mRNA and protein expression in a stimulus intensity-dependent manner (Figure 2(B) and (C)). The immunohistochemistry results also showed that the change in spinal TRPV1 levels was primarily found in the deep dorsal horn (lamina III-V) region of the spinal cord. Lamina I and II expressed dense TRPV1, but its expression did not change significantly between the stimulated and sham groups (Figure 2(D)). In addition, UCD seemed to elicite a more drastic increase of TRPV1 expression in rat dorsal root ganglion neurons comepared with control group.( Supplemental figure 11).
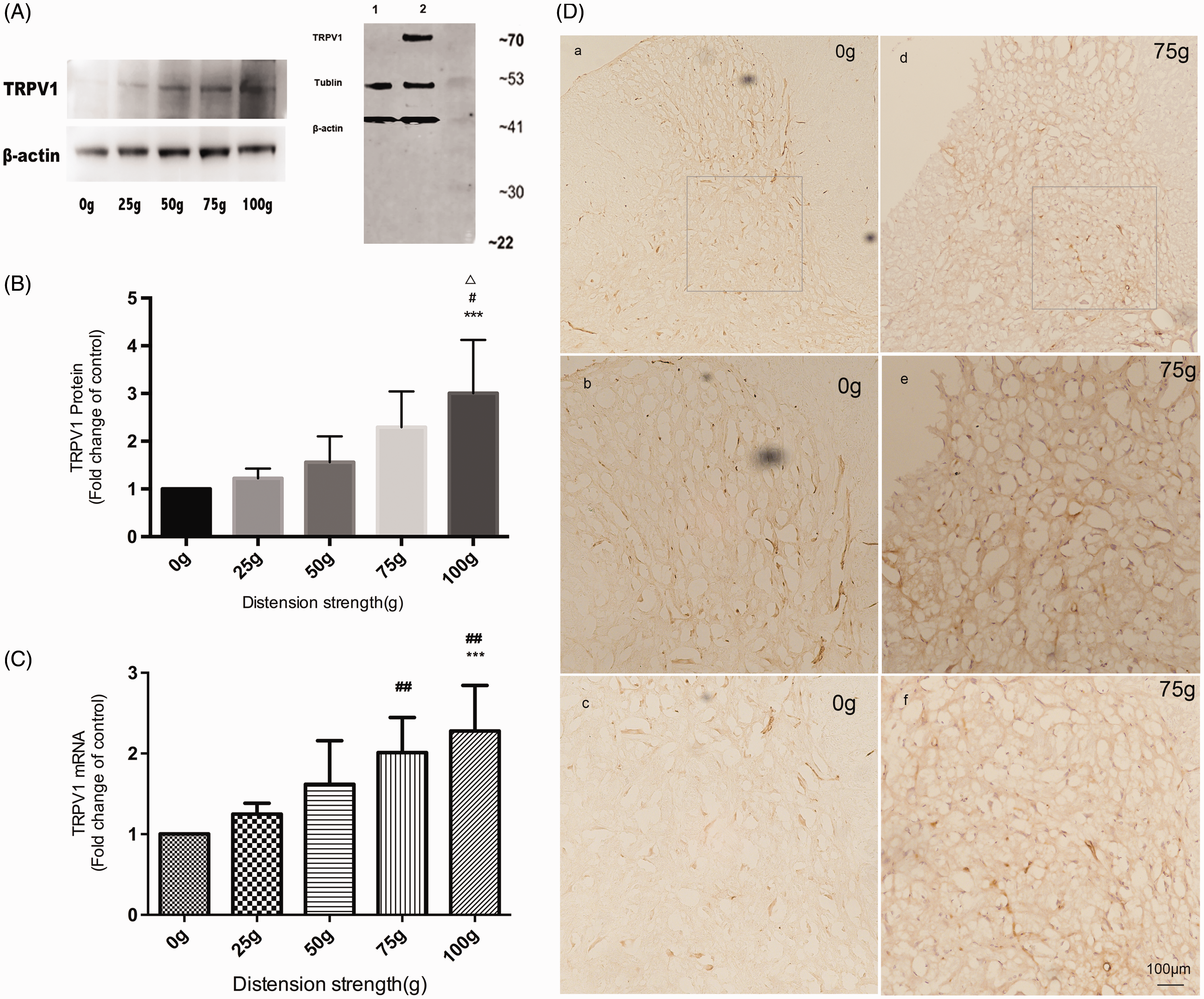
Western blot of TRPV1 shows UCD stimulus intensity-dependent response. (A) Western blot showing expression of spinal TRPV1. Loading control with β-actin is shown in lower portion. The numbers on the bottom of the Western blot represent the distension forces. Western blot analysis with anti-TRPV1 antibody (1:100, Alomone Labs, #ACC-030). 1. Results of rat DRG lysate; 2. Results of rat ventral spinal cord. (B) Histogram showing fold change in density of TRPV1 protein level normalized to β- actin in response to UCD stimulation (n=9). Data are presented as mean ± SD. (C) Histogram showing fold change in TRPV1 mRNA levels as quantified by real-time RT-PCR analysis in response to UCD stimulation (n=9). Spinal TRPV1 mRNA expression increased as the distension force increased in the rat cervix. Data are presented as fold change from the control (distention strength of 0 g) as mean ± SD. ***P < 0.001 when compared with sham group, ###P < 0.001 when compared with 25 g group, △△△P < 0.001 when compared with 50 g group. (D) TRPV1 immunoactive neurons in the spinal cord of sham- and UCD- (75 g) treated group. TRPV1 increased in the UCD group compared with sham (a, d).Lamina I, II of the dorsal horn generally expressed more TRPV1 positive neurons, and this region did not show significant change between sham and UCD group (b, e), Lamina III∼V (deep dorsal horn deep dorsal horn region as framed out by the rectangular) show clearly TRPV1 increase with UCD stimulation (c, f).
Effect of MPEP on TRPV1 and SB-366791 on mGluR5 in rats undergoing UCD
Spinal TRPV1 mRNA and protein expression by Western blot was significantly reduced in rats that had intrathecal administration of MPEP prior to UCD (Figure 3(a) to (c)). Intrathecal administration of MPEP did not have a significant effect on protein or mRNA expression of mGluR5 (Figure 3(d) to (f)). Spinal mGluR5 protein expression and its mRNA level were similar in rats with intrathecal administration of SB-366791, control, and vehicle (Figure 4). There was no significant difference in the expression of TRPV1 between those rats that had intrathecal injection of DMSO or no drug (Figure 3).

Change of TRPV1 expression in the setting of MPEP administration. Spinal TRPV1 protein (a) and (b) and mRNA (c) expression in response to UCD with MPEP pretreatment (n=4). Fold change for the density of TRPV1 level was normalized to β-actin. Data are presented as mean ± SD. *P < 0.05 compared with control or DMSO-treated group. TRPV1: transient receptor potential vanilloid 1; mGluR5: metabotropic glutamate receptor 5; DMSO: dimethyl sulfoxide; MPEP: 2-methyl-6-(phenylethynyl)-pyridine.
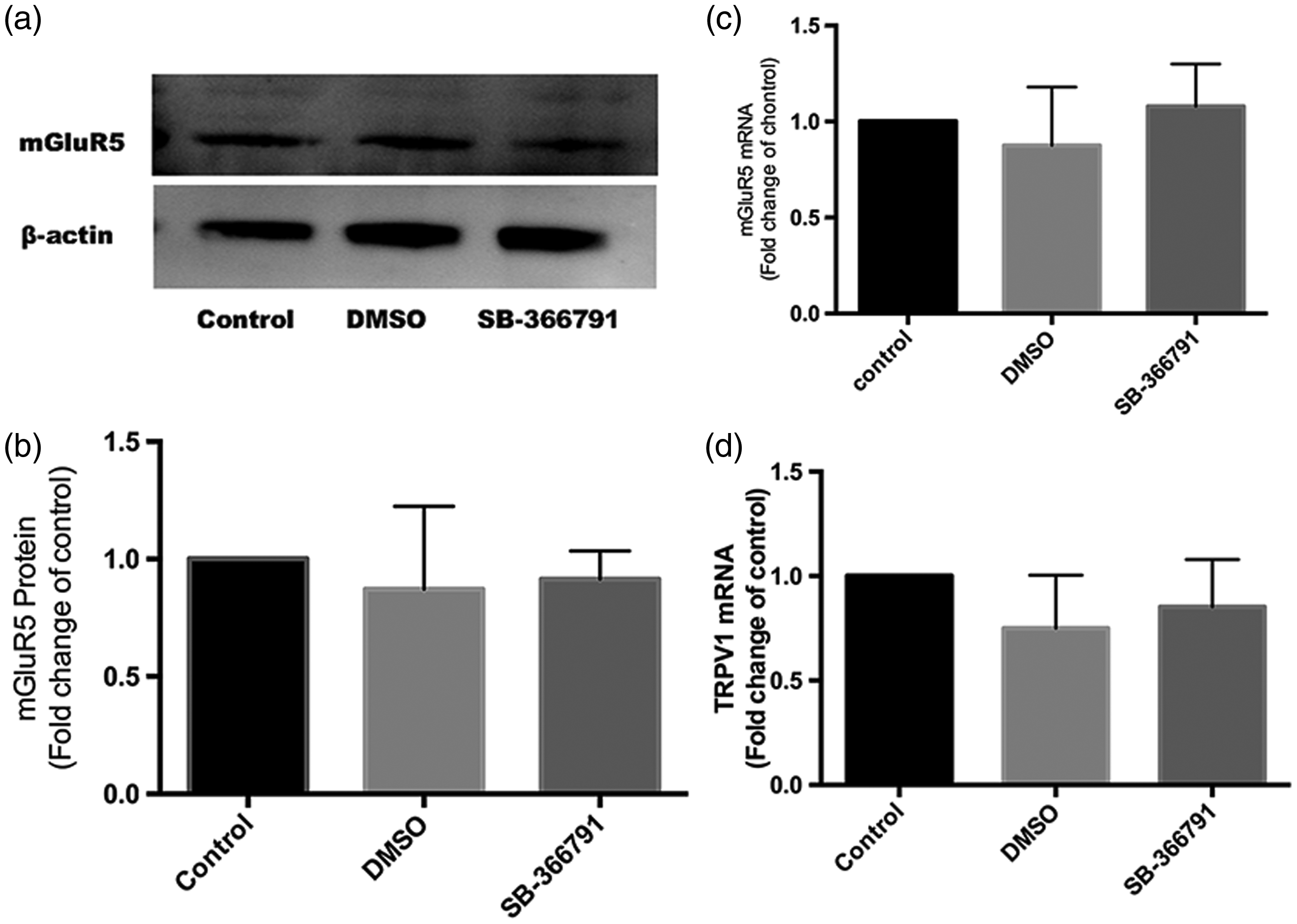
Change of mGluR5 expression in the setting of SB-366791 administration. (a) and (b) mGluR5 protein level did not change significantly between the control, DMSO, and inhibitor groups (n=3). Fold change for the density of mGluR5 level was normalized to β-actin. Data are presented as mean ± SD. (c) and (d) mGluR5 mRNA expression did not change significantly between the control, DMSO, and inhibitor groups.
Localization of mGluR5 and TRPV1 receptors in rat spinal cord
In rats subjected to a distension force of 75 g, cells expressing both mGluR5 and TRPV1 located densely in the deep dorsal horn region (lamina III-V) (Figure 5). In addition, mGluR5 and TRPV1 were expressed in naïve rat spinal cord, with the most dense expression in lamina I-II of the dorsal horn. Neurons co-expressing mGluR5 and TRPV1 in lamina I-II of the spinal cord in both sham- and UCD-treated rats were also detected (Supplemental Figure 9).

Co-localization of mGluR5 and TRPV1 in the deep dorsal horn region of the spinal cord. The upper four images (magnified 10×) show neurons expressing both mGluR5 and TRPV1 in the L1 segment of rats subjected to the 75 g distention force (n=4). The lower four images show the same spinal region magnified 20×, with arrows showing neurons most densely expressing the two receptors.
Validation of UCD model by measurement of spinal c-Fos expression and EMG
Deep dorsal horn c-Fos expression in response to UCD was observed on immunofluorescence (Figure 6(a)). c-Fos positive cells were quantificated against their nuclei conterstains (Supplemental figure 12), the expression quantification demonstrated a stimulus intensity-dependent increase of c-Fos positive neurons, and the changes concentrated mainly around lamina III-V of the spinal cord (Figure 6(a) and (b)). Increasing UCD forces also resulted in increased EMG signal intensity (Figure 6(c) and (d)). In rats treated with MPEP or SB-366791, deep dorsal horn (lamina III-V) c-Fos expression on immunofluorescence as well as the EMG signal was significantly reduced compared with the DMSO-treated group (Figures 7 and 8). Negative controls without incubation of the first antibodies produced no signal (Supplemental Figure 10).

Immunohistochemistry of Fos-positive neurons and EMG intensity in response to UCD. (a) c-Fos immunoreactive neurons are seen concentrated in the III-V laminae of the spinal cord (T12-L2). Individual microphotographs represent spinal cord sections of 8 µm. The scale bar represents 100 µm. (b) Histogram showing quantification of c-Fos expression in lamina III-V of the spinal cord (percentage of all cells) of different UCD groups (n=4). For each animal and each spinal level, the total number of c-Fos positive neurons in the deep dorsal horn region (lamina III-V) was counted in 3 to 5 non-serial 8-µm sections and averaged. (c) and (d) Increase in EMG signal during the UCD process. Images show the change in EMG intensity during the 20-s stimulation period with every distension cycle among groups (n=4). The graph shows changes in accumulated EMG signal (AUC) during the 1-h distension phase. Data are presented as mean ± SD, ***P < 0.001 when compared with sham group, ###P < 0.001 when compared with 25 g group, △△△P < 0.001 when compared with 50 g group.

Immunohistochemistry of spinal c-Fos expression with administration of inhibitors. (A) Individual images show c-Fos immunoactivity in the same spinal segment (L1) based on administration of inhibitors. (B) and (C) Histogram showing the quantification of c-Fos expression (percentage of all cells) in lamina III-V of the spinal cord among different groups (n=4). The control group represents rats subjected to catheter placement and UCD (75 g) without administration of vehicle or drug. Data are presented as mean ± SD. ***P < 0.001 compared with control or DMSO-treated group.

EMG signal in the settings of inhibitor administration. (A) Decrease of EMG signal with administration of inhibitors. (B) Bar graph showing digitalized EMG intensities (AUC) among groups. The control group represents rats subjected to catheter placement and UCD (75 g) without administration of vehicle or drug. Data are presented as mean ± SD. *P < 0.05 compared with control group.
Discussion
In this study, we found that UCD in rats results in increased spinal mGluR5 and TRPV1 mRNA and protein expression in a stimulus intensity-dependent manner. We also found that intrathecal administration of MPEP 30 min prior to UCD results in decreased spinal TRPV1 mRNA and protein expression, while intrathecal administration of SB-366791 30 min prior to UCD did not affect spinal mGluR5 mRNA and protein expression. Finally, we found that cells expressing changes in both mGluR5 and TRPV1 located most densely to the deep dorsal horn region.
The finding that mGluR5 and TRPV1 expression was increased with UCD is not surprising, as similar results have been found in gastrointestinal and urinary models of visceral pain. mGluR5 has been implicated in playing a role in the transmission of visceral pain in a colorectal distension rat model. 7 TRPV1 has also been shown to play an important role in the transmission of stretch-evoked nociceptive signals in the bladder. 8 Our study further confirms that mGluR5 and TRPV1 play a role in the transmission of nociceptive signals in many viscera, including the uterine cervix.
Our finding that the effect of UCD exerted on neuronal transmission occurs within such a rapid time frame is consistent with other models of acute visceral nociception (e.g., 70 min in colorectal distension and 1 h in bladder distension models7,8). Furthermore, spinal protein kinase A levels increase in a stimulus-dependent manner after intra-colonic noxious stimulation within a timeframe of 60 min, 19 and the mGluR1/5 agonist DHPG enhances TRPV1 phosphorylation in a protein kinase C-dependent manner within a timeframe of 45 min in dissociated rat trigeminal ganglion neurons. 20 Given that protein kinase A and protein kinase C are downstream messengers of mGluR5 and potential TRPV1 modulators, these results further support our findings that the rapid effect of acute visceral nociception in our UCD model on mGluR5 and TRPV1 expression are valid.
Decreased levels of TRPV1 were found in MPEP-treated rats, but no change in mGluR5 expression was demonstrated in SB-366791-treated rats, suggesting that mGluR5 may be an upstream regulator of TRPV1 expression in uterine cervical nociception. This finding is supported by prior studies that have suggested mGluR5 activation results in phosphorylation-induced sensitization of TRPV1, thereby enhancing thermal and mechanical hyperalgesia.11,20 Similarly, other studies have found that activation of spinal mGluR5 elicits Ca2+ influx via the non-selective cation channel TRPV1.21,22
Our findings offer a more extensive understanding of the interplay between mGluR5 and TRPV1. First, the finding that mGluR5 and TRPV1 co-localized in the deep dorsal horn in rats subjected to UCD suggests that TRPV1 and mGluR5 may be coupled in the sensory neurons of the spinal cord. Second, the finding that TRPV1 mRNA expression was significantly decreased by MPEP suggests that mGluR5 participates pre-translationally. Such a theory is supported by an electrophysiological study performed by Hu et al., 23 which demonstrated that activation of mGluR5 enhanced TRPV1 expression via protein kinase A and cyclooxygenase pathways. Finally, our results suggest that mechanical hypersensitivity during UCD may be modulated by mGluR5 through regulation of TRPV1. This idea is supported by Chung et al. 20 who demonstrated that mGluR5 activation results in enhanced mechanical hyperalgesia through TRPV1 phosphorylation in trigeminal sensory neurons.
Our study has several important limitations. First, while MPEP has mGluR5 inhibitor properties, it also expresses NMDA receptor antagonist properties. 24 Given that activation of NMDA receptors sensitizes TRPV1 channels, 25 we cannot definitively determine if MPEP resulted in a decrease in TRPV1 through decreasing mGluR5 or NMDA expression. Second, MPEP retains analgesic properties in mGluR5 knockout mice, 3 thus questioning whether our results may have been influenced by its actions on the NMDA receptor. Finally, locomotor side effects have been associated with the use of MPEP, which may limit clinical utility. 2
Despite these limitations, one of the most important strengths of our study is that we had a well-functioning UCD model for investigating visceral pain originating from the uterine cervix. We modified the UCD rat model developed by Tong et al. 14 by using a lighter depth of anesthesia that prevented purposeful escape but still allowed for detection of rectus abdominis visceromotor responses. The validation experiments demonstrated that UCD resulted in a stimulus intensity-dependent increase in EMG and spinal c-Fos, and a decrease in EMG and spinal c-Fos in the setting of antagonist administration, indicating that our UCD model successfully elicited visceral pain at the uterine cervix.
In summary, our study demonstrates that UCD stimulates spinal mGluR5 and TRPV1 expression, MPEP decreases TRPV1 expression, but SB-366791 does not change mGluR5 expression. These results suggest that mGluR5 and TRPV1 may play an important role in the modulation of nociceptive signals during uterine contractions, and that mGluR5 may be an upstream regulator of TRPV1 expression. Further studies are needed to better understand the role these and other receptors and channels play in the transmission of visceral nociception during labor.
Supplemental Material
Supplemental Material1 - Supplemental material for Involvement of mGluR5 and TRPV1 in visceral nociception in a rat model of uterine cervical distension
Supplemental material, Supplemental Material1 for Involvement of mGluR5 and TRPV1 in visceral nociception in a rat model of uterine cervical distension by Wenxin Zhang, Dan Drzymalski, Lihong Sun, Qi Xu, Cuicui Jiao, Luyang Wang, Shufang Xie, Xiaowei Qian, Hui Wu, Fei Xiao, Feng Fu, Ying Feng and Xinzhong Chen in Molecular Pain
Supplemental Material
Supplemental Material2 - Supplemental material for Involvement of mGluR5 and TRPV1 in visceral nociception in a rat model of uterine cervical distension
Supplemental material, Supplemental Material2 for Involvement of mGluR5 and TRPV1 in visceral nociception in a rat model of uterine cervical distension by Wenxin Zhang, Dan Drzymalski, Lihong Sun, Qi Xu, Cuicui Jiao, Luyang Wang, Shufang Xie, Xiaowei Qian, Hui Wu, Fei Xiao, Feng Fu, Ying Feng and Xinzhong Chen in Molecular Pain
Supplemental Material
Supplemental Material3 - Supplemental material for Involvement of mGluR5 and TRPV1 in visceral nociception in a rat model of uterine cervical distension
Supplemental material, Supplemental Material3 for Involvement of mGluR5 and TRPV1 in visceral nociception in a rat model of uterine cervical distension by Wenxin Zhang, Dan Drzymalski, Lihong Sun, Qi Xu, Cuicui Jiao, Luyang Wang, Shufang Xie, Xiaowei Qian, Hui Wu, Fei Xiao, Feng Fu, Ying Feng and Xinzhong Chen in Molecular Pain
Footnotes
Acknowledgments
The authors thank Professor Shucai Ling (Zhejiang University School of Medicine, Zhejiang, China) for his technological guidance in the experiments performed as part of this study.
Author Contributions
XC designed the experiments in this study and drafted the manuscript. WZ, LS, QX, and HW performed the experiments and data analysis. DD performed revisions and contributed to the intellectual development of the manuscript. CJ, LW, SX, FX, FF, YF, and XQ contributed to the design of the study and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by funding from the National Natural Science Foundation of China (NSFC, No. 81271237, No. 81471126, and No. 81501702) and the Zhejiang Provincial Natural Foundation of China (Grant No. LY15H150007).
Supplemental Material
Supplemental material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
