Abstract
Ion channels are very important in the peripheral sensitization in neuropathic pain. Our present study aims to investigate the possible contribution of CaV3.2 T-type calcium channels in damaged dorsal root ganglion neurons in neuropathic pain. We established a neuropathic pain model of rats with spared nerve injury. In these model rats, it was easy to distinguish damaged dorsal root ganglion neurons (of tibial nerve and common peroneal nerve) from intact dorsal root ganglion neurons (of sural nerves). Our results showed that CaV3.2 protein expression increased in medium-sized neurons from the damaged dorsal root ganglions but not in the intact ones. With whole cell patch clamp recording technique, it was found that after-depolarizing amplitudes of the damaged medium-sized dorsal root ganglion neurons increased significantly at membrane potentials of −85 mV and −95 mV. These results indicate a functional up-regulation of CaV3.2 T-type calcium channels in the damaged medium-sized neurons after spared nerve injury. Behaviorally, blockade of CaV3.2 with antisense oligodeoxynucleotides could significantly reverse mechanical allodynia. These results suggest that CaV3.2 T-type calcium channels in damaged medium-sized dorsal root ganglion neurons might contribute to neuropathic pain after peripheral nerve injury.
Introduction
Neuropathic pain results from lesions of the peripheral nervous system, its symptoms include spontaneous pain, mechanical allodynia, and hyperalgesia.1–3 Different kinds of ion channels, including voltage- as well as ligand-gated ion ones,3–5 are essential for pain transmission.3,6,7
T-type calcium channels are low voltage-activated calcium channels. In contrast with high voltage-activated calcium channels, T-type calcium channels are activated at more negative membrane potentials, with faster kinetics of macroscopic voltage-dependent inactivation, slower kinetics of deactivation, and smaller single-channel conductance.8–10 Since this characteristic, T-type calcium channels are considered to be a basis for after-depolarizing potentials (ADP) in a sub-population of dorsal root ganglion (DRG) neurons, 11 and thus to control the neuronal excitability.
T-type calcium channels include three subtypes, namely, CaV3.1, CaV3.2, and CaV3.3.12,13 Bourinet et al. 12 showed that CaV3.2, but not CaV3.1 or CaV3.3, antisense treatment resulted in antinociceptive, antihyperalgesic, and antiallodynic effects, suggesting that CaV3.2 plays an important function in sensory transduction in pain. 13
Peripheral sensitization is a major player in neuropathic pain, which originates from both damaged and intact nerve fibers and from damaged and intact DRG.14–17 In the present study, we focused on the role of CaV3.2 T-type calcium channels in damaged DRG neurons completely separated from intact ones in neuropathic pain model of rats with spared nerve injury (SNI). We demonstrated that CaV3.2 T-type calcium channels in damaged medium-sized DRG neurons might contribute to the development of neuropathic pain in SNI rats.
Experimental procedures
Animals
Male Sprague-Dawley rats weighing 200 to 250 g (eight to nine weeks old) were used. They were provided by Department of Experimental Animal Sciences, Peking University Health Science Center. Animals had free access to food and water and were raised under natural diurnal cycles. All experiment protocols were approved by the Animal Use and Care Committee of Peking University Health Science Center, which followed the Guidelines of Animal Use and Protection adopted from the National Institutes of Health, USA. Every possible measure was taken to minimize discomfort to the animals.
Establishment of SNI model of rats with neuropathic pain
SNI operation was performed as previously described.18,19 After anesthesia, the left tibial and common peroneal nerves were tightly ligated and sectioned distal to the ligation, leaving the sural nerve intact. Muscle and skin were sutured in two layers. Sham operation control was similar in exposure of the sciatic nerve and its branches but without any further lesion. All operation procedures were in the sterile operation.
Fifty percent paw withdrawal threshold test for mechanical allodynia in SNI rats
Mechanical allodynia of left hind paw was tested before and at different time points after SNI surgery. The 50% paw withdrawal threshold in response to a series of von Frey filaments was examined by the up and down method as described previously.19,20 Eight filaments with approximately equal logarithmic incremental (0.224) bending forces were chosen (0.41, 0.70, 1.20, 2.00, 3.63, 5.50, 8.50, and 15.10 g). Only those rats with 50% paw withdrawal thresholds less than 4 g were selected and used in the subsequent morphological, pharmacological, and electrophysiological studies.
Intrathecal administration of oligodeoxynucleotide antisense-CaV3.2
Antisense oligonucleotide and mismatched oligonucleotide to CaV3.2 with sequence as previously reported 12 were synthesized in AuGCT (Beijing, China). Sequence for the antisense-CaV3.2 (AS) was CCACCTTCTTACGCCAGCGG and the mismatched-CaV3.2 (MS) was TACTGTACTTGCGAGGCCAC. Oligodeoxynucleotide (ODNs) were dissolved in sterile saline. To visualize the ODN uptake, the 5’-end of the AS-CaV3.2 was coupled to a fluorescein isothiocyanate group.
ODN administration was carried out through intrathecal catheterization into rats. After implantation surgery, the rats were allowed to recover for three to five days before further experiments. ODNs (12.5 μg/rat) or saline was administrated in a volume of 10 μl one day before SNI surgery, and repetitively twice a day for consecutive eight days. Mechanical allodynia was measured before ODN administrations and then on every other day from day 3 to day 21 at the same time-point in the morning. Treatments were randomized and all behavioral experiments were performed blindly.
Retrograde labeling and immunohistochemistry staining of DRG neurons
According to the method described in previous report, 21 DRG neurons from the injured tibial nerve or common peroneal nerve were labeled retrogradely with Fluorogold (FG, Fluorochrome, LLC, USA). After transaction of either the tibial or common peroneal nerve, 2% FG (1–2 μl) was injected slowly with a microsyringe into the proximal stump, and then the injection site was clamped with microforceps for 1 s to ensure maximal labeling. 3, 7, and 14 days later, the rat was deeply anaesthetized and lumbar 4 (L4) and lumbar 5 (L5) DRGs in the injected site were harvested for immunohistochemical or electrophysiological studies.
As described in our previous study, 22 the rat was perfused with normal saline followed by 4% paraformaldehyde in 0.1 M phosphorate buffer. Bilateral L4 and L5 DRGs were removed, post-fixed in 4% paraformaldehyde for 4 h, and then dehydrated in 30% sugar solution. Several days later, these tissues were cut in 10-μm thick serial longitudinal sections and mounted on gelatin/chrome alum-coated glass slides. After blocking in normal goat serum, these sections were incubated with primary antibodies over two nights at 4°C, followed by incubation with secondary antibodies overnight at 4°C. The following are the primary antibodies and their dilution ratios used in the present experiment: rabbit anti-CaV3.2 polyclonal antibody (1:200; Alomone Labs, Israel), mouse anti-neurofilament-200 (NF-200) monoclonal antibody (1:1000; Sigma-Aldrich, USA), fluorescein isothiocyanate-conjugated IB4 lectin (10 μg/ml; Sigma-Aldrich), or mouse anti-calcitonin gene-related peptide monoclonal antibody (1:1000; Sigma-Aldrich). Except IB4-treated DRG sections, all other sections were treated by a mixture of Alexa Fluor 488 goat anti-rabbit IgG (H + L) and Alexa Fluor 568 goat anti-mouse IgG (H + L) (1:500; Invitrogen, Life TechnologiesTM, USA). The stained section was captured with a fluorescence microscope (Leica, Germany), and the Image-Pro Plus software (Media Cybernetics, USA) was used for quantification of cell area.
To measure cell area, neurons with apparent nuclear region were graphically highlighted. Total number of neurons and CaV3.2 positive ones from three sections of each DRG were counted. The percentage of positive neurons to total neurons and the percentage of FG positive neurons to CaV3.2 positive neurons were calculated and statistically analyzed.
Cross-sectional area distribution in positive neurons was calculated in 3, 7, and 14 days after SNI. DRG neurons were classified as small (<700 μm2), medium-sized (700–1200 μm2), and large (>1200 μm2) one according to the measured cross-sectional area.16,23
Western blot detection of CaV3.2 proteins
Total protein was extracted as previously described. 22 Briefly, whole L4 and L5 DRGs were disrupted in lysis buffer (HEPES (pH 7.0) 4 mM, sucrose 320 mM, and EDTA (pH 8.0) 5 mM with protease inhibitor). The samples were sonicated on ice and then centrifuged at 10,000 g for 15 min at 4°C to isolate the supernatant containing total protein samples. For membrane protein extraction, 24 six L4 and L5 DRGs were collected as one sample. The sample was homogenized in the lysis buffer mentioned above and centrifuged at 2000 g for 10 min. The supernatant was centrifuged again at 2 × 105 g for 60 min. This supernatant was the cytosolic protein sample. The pellet was solubilized in the lysis buffer as the membrane protein sample.
The protein samples were separated on SDS–PAGE gels and transferred to PVDF membranes. After blocking with 5% non-fat dried milk in TBST (20 mM Tris-HCl, 150 mM NaCl, 0.05% Tween 20, pH 7.6), the PVDF membrane was incubated with rabbit anti-CaV3.2 (1:1000), anti-β-actin (1:5000), or anti-α1 Na+-K+-ATPase (1:8000) antibodies in 5% non-fat dried milk in TBST overnight at 4°C. After washing with TBST, the membrane was incubated with goat anti-rabbit antibody or goat anti-mouse antibody (HRP labeled) diluted with 5% non-fat dried milk in TBST and detected with ECL reagents (Amersham Biosciences, Arlington Heights, IL). Blots were scanned with Spot Advanced and Adobe Photoshop 10.0 (Adobe, Inc.), and band densities were compared with TotalLAB software.
Acute dissociation of DRG neurons for patch clamp recording
Neurons were acutely isolated from ipsilateral L4 and L5 DRG of rats using methods as previously described. 24 Briefly, freshly dissected ganglia were subjected to digestion at 37°C with collagenase (3 mg/ml, type IA, Sigma-Aldrich) for 47 min, followed by trypsin (2 mg/ml, Type II-S, Sigma-Aldrich) for 12 min. The enzymatic digestion was stopped by washing the cells with DMEM containing 10% fetal bovine serum, and the remaining pieces of ganglia were gently triturated by using a fire-polished glass Pasteur pipette. The suspended solution was placed onto poly-D-lysine (0.1 mg/ml, Sigma-Aldrich)-pretreated glass coverslips within sterile 24-well tissue culture plates and kept in an incubator at 5% CO2, 37°C for 4 to 5 h before patch-clamp recording.
Electrophysiological patch clamp recording of DRG neurons
Whole-cell current-clamp recordings were performed at room temperature using an EPC-10 amplifier and Patchmaster software (HEKA, Germany). Patch pipettes were pulled from borosilicate glass capillaries with a tip resistance of 4 to 5 MΩ when filled with internal solution containing (in mM) KCl 130, HEPES 40, MgCl2 5, EGTA 1, Mg-ATP 2, and Na-GTP 0.1 adjusted to pH 7.3 with KOH. The external solution contained (in mM) NaCl 140, KCl 4, CaCl2 2, MgCl2 2, HEPES 10, and glucose 10, adjusted to pH 7.4 with NaOH.
Action potentials (APs) were measured with pipette and membrane capacitance cancellation, filtered at 2 kHz and digitized at 10 kHz. Series resistance was compensated at 80% to 90%. The membrane capacitance (Cm) was read from the amplifier by software Patchmaster for determining the size of the cell.
For current-clamp recordings, the cell was held at 0 pA, and the firing threshold of the DRG neuron was first measured by a series of 20-ms depolarizing current injection in 50-pA steps from 0 pA to elicit the first AP. For after-depolarizing (ADP) recording, 5-ms long, 800 pA depolarizing pulses were used to elicit APs with an advanced 400-ms long hyperpolarizing current injection holding the membrane potentials at −85 mV or −95 mV. A hyperpolarizing current injection in 200 ms, −200 pA was used to measure membrane input resistance, which was assessed from the value of the evoked membrane potential divided by the injected hyperpolarizing current (−200 pA). The following values were also measured: resting membrane potential (RMP), the depolarized current threshold for eliciting the first AP, threshold potential (TP), rising rate of TP ((RMP − TP) ÷ duration from RMP to TP), amplitude of AP, overshot of AP, duration of AP, and amplitude of ADP at −60 mV, −85 mV, and −95 mV.
Data analysis and statistics
Statistical analyses were performed with GraphPad Prism 5 for Windows (GraphPad Software, Inc., USA). All data were expressed as mean ± SEM. Two-tailed unpaired Student’s t test was used for the comparison of the mean values between the two groups. One-way analysis of variance followed by Tukey’s post-test or two-way analysis of variance followed by Bonferroni post hoc test was used for multiple comparison. Differences with p < 0.05 were considered statistically significant.
Results
Expression of CaV3.2 in all types of DRG neurons in naïve rats
In naïve rats, expression pattern of CaV3.2 T-type calcium channel proteins in DRG neurons was observed with immunofluorescent staining. CaV3.2-immunoreactivity (-ir) was detected in the cytoplasm of small, medium-sized, and large DRG neurons, with co-localization of isolectin B4 (a marker for non-peptidergic small DRG neurons), NF-200 (a marker for the medium-sized to large DRG neurons), and calcinonin gene-related peptide (a marker for peptidergic small DRG neurons) (Figure 1).

Immunofluorescent double labeling of CaV3.2 protein in three kinds of DRG neurons. (a) to (c) The expression of CaV3.2. (d) to (f) The expression of IB4, NF-200, and CGRP. (g) to (i) Co-localization of CaV3.2 and neuronal markers. CaV3.2 was expressed in all three kinds of DRG neurons, showing good co-localization with IB4, NF 200, and CGRP. Scale bar: 75 μm.
Expression of CaV3.2 proteins increased in the membrane of damaged medium-sized DRG neurons in SNI rats
In order to quantify the change of CaV3.2 in neuropathic pain rats after SNI, we performed Western blotting analysis. In samples from whole DRGs, there was no obvious change of CaV3.2 expression in total (Figure 2(a) and (b)), membrane, or cytosolic (Figure 2(c) and (d)) proteins.

Total protein expression CaV3.2 in DRG after SNI with Western blotting analysis. (a) and (b) Total expression in L4 (a) or L5 (b) DRG at 3, 7, and 14 days after SNI. No significant changes of total immunoreactivity in L4 or L5 DRG after SNI were observed. Upper: Representative Western blotting bands of CaV3.2 and β-actin. Lower: Statistical analysis of band density ratios of CaV3.2 to β-actin. (c) Immunoreactivity of membrane CaV3.2 in L4/L5 DRGs 14 days after SNI. No significant change was observed. Upper: Representative Western blotting bands of CaV3.2 and α1 Na+,K+-ATPase. Lower: Statistical analysis of band density ratios of CaV3.2 to α1 Na+,K+-ATPase. n = 5 rats in each group. (d) Immunoreactivity of cytosolic CaV3.2 in L4/L5 DRGs 14 days after SNI. No significant change was observed. Upper: Representative Western blotting bands of CaV3.2 and β-actin. Lower: Statistical analysis of band density ratios of CaV3.2 to β-actin. n = 5 rats in each group. The molecular weight of CaV3.2 was ∼250 kDa.
This phenomenon could result from its different expression pattern in the intact and the damaged neurons (Figure 3(a) and (b)), and from the low percentage of neurons with CaV3.2 expression on the neuronal membrane (Figure 3(c) and (d)).
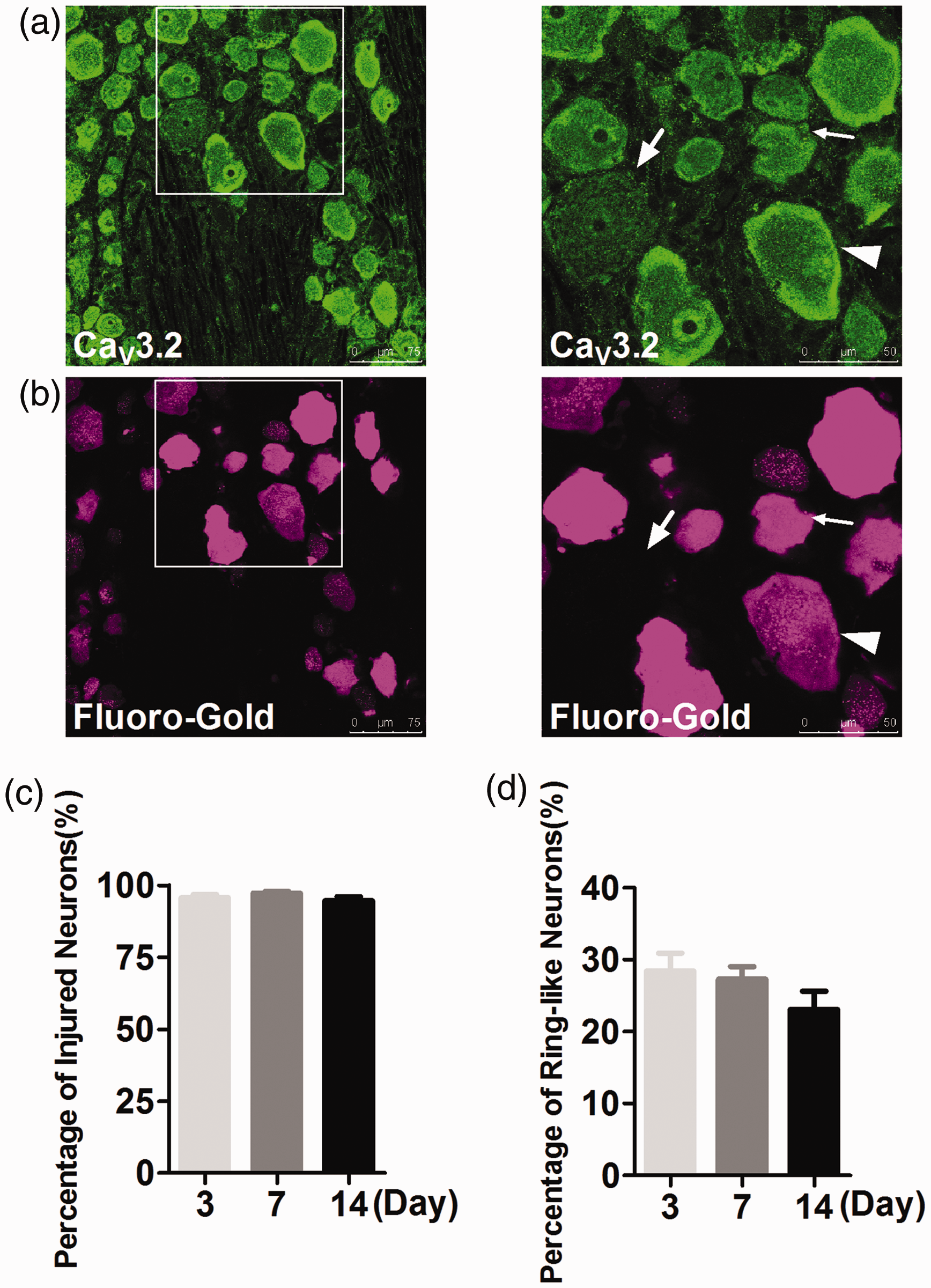
CaV3.2 expression pattern in damaged DRG neurons after SNI with immunofluorescent staining. (a) (Left): CaV3.2 expression in DRG neurons 14 days after SNI. Scale bar: 75 μm. (Right): CaV3.2 expression with higher magnification. Scale bar: 50 μm. (b) (Left): Fluoro-Gold (FG) labeling of DRG neurons in the same field with (a). (Right): Fluor-Gold labeling with higher magnification. CaV3.2 expressed on the membrane of damaged (FG positive) medium-sized neurons at 14 days after SNI. Membrane expression of CaV3.2 was observed mainly in damaged medium DRG neurons (arrowheads), (c) Statistical analysis of percentage of damaged DRG neurons in CaV3.2 membrane-positive neurons. Almost all CaV3.2 membrane-positive neurons were damaged ones in SNI rats. (d) Statistical analysis of percentage of CaV3.2 membrane-positive neurons (ring-like neurons) in DRGs. About 20% to 30% DRG neurons were CaV3.2 membrane-positive after SNI.
In SNI rats, FG was used to retrogradely mask damaged DRG neurons, and it was found that CaV3.2 was expressed in totally different patterns after SNI. CaV3.2 was uniformly distributed in the cytoplasm of sham-operation DRG neurons (data not shown), while in SNI groups, CaV3.2 turned to be in the membrane of neurons (Figure 3(a) and (b)). CaV3.2 was expressed in about 25% of membrane of DRG neurons of SNI rats (Figure 3(d)), most of which were damaged neurons (Figure 3(c)).
In order to further confirm the change of CaV3.2 expression pattern in damaged DRG neurons after SNI, the ratio of expression of CaV3.2 in DRG neurons after SNI was quantified according to cell area (Figure 4(a)), with cell areas ranging from 0 to over 4000 μm2. It was found that CaV3.2 distributed mainly in the membrane of medium-sized neurons. As shown in Figure 4(b), the ratio of expression of CaV3.2 in the medium-sized DRG neurons at 7 and 14 days after SNI increased significantly compared with 3 days (24.0 ± 1.8% and 29.1 ± 3.5% vs. 18.7 ± 0.7%, p < 0.05). Compared with that at 7 days, the ratio of expression of CaV3.2 in the medium-sized DRG neurons at 14 days after SNI increased, but with no statistical differences.

Distribution of CaV3.2 membrane-positive neurons in DRG neurons after SNI. (a) Each column represents the ratio of the number of membrane-positive neurons to total number of neurons at 3, 7, and 14 days after SNI. (b) Each column represents the ratio of the number of small, medium-sized, and large neurons to total number of neurons at 3, 7, and 14 days after SNI. Note that the ratio of the number of the medium-sized neurons to total number of neurons at 7 and 14 days after SNI increased significantly. *p < 0.05, compared with three days after SNI.
It was found that most CaV3.2-positive neurons were damaged neurons (Figure 3(c)), and the expression of CaV3.2 increased in the medium-sized DRG neurons after 14 days of SNI (Figure 4(b)). These results indicate that expression of CaV3.2 increased in the membrane of damaged medium-sized DRG neurons after SNI.
Functional up-regulation of T-type calcium channels in damaged medium-sized DRG neurons after SNI
T-type calcium channels play a crucial role in neuronal excitability because of their control over ADP. 11 As the predominant subtype in DRG neurons, CaV3.2 T-type calcium channels mainly distribute in small and the medium-sized neurons and represent electrophysiological properties.25,26 To further test the function of CaV3.2 expressed in the damaged DRG neurons of SNI rats, we then examined ADP amplitudes at resting state (−60 mV) and at two hyperpolarization conditions (−85 mV and −95 mV). Based on cell size, these neurons were then categorized into small (Cm < 30 pF), medium-sized (30 pF < Cm < 50 pF), and large (Cm > 50 pF) ones.
As shown in Figure 5, in the damaged small or large DRG neurons in SNI group, ADP amplitudes did not show obvious changes either at resting or at hyperpolarization states compared with those in sham-operation and naïve groups (Figure 5(a) to (d), left and right columns). In the damaged medium-sized DRG neurons, ADP amplitudes showed significant increase at −85 mV (17.2 ± 1.4 mV vs. 13.5 ± 0.9 mV and 11.2 ± 0.5 mV, p < 0.05) (Figure 5(a) and (c), middle column). Similar results were observed at −95 mV (Figure 5(a) and (d), middle column). These results indicate a functional up-regulation of CaV3.2 T-type calcium channels in the damaged medium-sized neurons after SNI. Original data were shown in Supplementary Table 1.

Increase of after-depolarizing potential (ADP) amplitudes of damaged DRG neurons in SNI rats. (a) A representative of action potentials (APs) evoked by 5-ms, 800-pA depolarizing current pulses, recorded from small to large DRG neurons at −95 mV. Note the ADPs. The red traces represent the discharges from damaged DRG neurons. (b) to (d) ADP amplitudes at −60 mV (b), −85 mV (c), and −95 mV (d) in naïve, sham-operation, and damaged small to large neurons (middle to right: the medium-sized and large). Compared with that in naïve and sham-operation rats, ADP amplitudes increased significantly in damaged medium-sized neurons at −85 mV (c) or at −95 mV (d). *p < 0.05, one-way ANOVA followed by Tukey’s post-test.
Silencing of CaV3.2 with antisense–reduced mechanical allodynia after SNI
To test the role of CaV3.2 in the development of neuropathic pain, we further studied the effects of local CaV3.2 AS knock-down in the DRGs on mechanical allodynia. CaV3.2 antisense by intrathecal injection led to preferential uptake in the corresponding DRGs (data not shown).
As shown in Figure 6(a), mechanical allodynia remained because the mechanical thresholds were still low in normal saline group (2.8 ± 0.8 g) and in MS group (2.7 ± 0.8 g), indicating the hyperalgesic state seven days after SNI. Interestingly, in CaV3.2-AS group, the mechanical allodynia was largely reduced as shown by a significant increase in mechanical thresholds (to maximal 8.9 ± 1.5 g), and this effect lasted for 10 days. This anti-nociceptive efficacy was displayed more clearly when the mechanical thresholds were analyzed with area under the curves of time course (Figure 6(b)).

Antinociceptive effects of CaV3.2 antisense oligonucleotides (ODNs) in SNI rats. (a) Time course of the effect of i.t. injection of CaV3.2 antisense on mechanical thresholds in SNI rat. SNI rats were treated with either AS-CaV3.2 ODN, mismatched ODN, or saline vehicle. Mechanical thresholds were measured before (one day before SNI, day −1) and after SNI (day 3), and then at every other day (till day 21) after injection. The AS injection produced a significant antinociceptive effects lasting for 10 days. *p < 0.05, ***p < 0.001, two-way ANOVA followed by Bonferroni post-test. #p < 0.05, unpaired t test. (b) Statistical analysis of the area under the time-course curves of variations in mechanical thresholds. The AS injection produced significant analgesic effects as compared with normal saline and mismatched injection. ***p < 0.05, one-way ANOVA followed by Tukey’s post-test.
Discussion
In the present study, we found that expression in the cell membrane and the function of CaV3.2 T-type calcium channels increased in damaged medium-sized DRG neurons in neuropathic pain rats after SNI. We also found that inhibition by antisense could significantly relieve mechanical allodynia at 14 days after SNI. These results indicate that CaV3.2 T-type calcium channels in the damaged medium-sized DRG neurons might play an important role in the development of neuropathic pain after SNI.
T-type calcium currents were first discovered in 1989. This channel participates in the generation of membrane potential depolarization that often lead to the firing of a short series or bursts of AP. 11 T-type calcium channels include three subtypes, namely, CaV3.1, CaV3.2, and CaV3.3.12,13 In the present study, we found that CaV3.2 was expressed in all DRG neurons in naïve rats (Figure 1), which is different from the results of previous studies that T-type currents are expressed in the majority of small and the medium-sized DRG neurons.27–29 In fact, Rose et al. 26 also reported that a smaller proportion of DRG cells were co-labeled with CaV3.2 and NF-200. In addition, Yue et al. 30 found that SNI significantly increased current density of T-type Ca2+ channels in the medium-sized and large DRG neurons, supporting that CaV3.2 T-type calcium channels were expressed in all types of DRG neurons.
In DRG neurons, from naïve or sham-operation rats, CaV3.2 distributed mainly in the cytoplasm, while in SNI rats, CaV3.2 turned to the membrane of the damaged neurons (Figure 3(a) and (b)). It is well known that ion channels in the membrane are functional compared to those in the cytoplasm in a neuron, so CaV3.2 re-distribution into the membrane after SNI has important significance.
As shown in Figure 4, the ratio of CaV3.2 expression in the damaged medium-sized DRG neurons after SNI increased significantly. There are other papers that show damaged medium-sized DRG neurons play a role in neuropathic pain following nerve injury,17,31,32 but there exist contradictory reports. For example, Andre et al. 31 reported that nerve injury induced a decrease of T-type current in medium-sized DRG neurons after chronic constrictive injury in mice, and McCallum et al. 33 reported the loss of T-type current in the medium-sized DRG neurons in rats with chronic constrictive injury. These authors considered that the decrease of T-type currents in medium-sized neurons might have important roles in neuropathic pain. But Hilaire et al. 32 reported that intracellular calcium entry through voltage-gated calcium channels plays an important role in the increase of excitability of mouse DRG axotomized neurons. Thus, in different experimental conditions or experimental models or in different types of neurons, nerve injury might have different effects on the expression of T-type calcium channels in the medium-sized DRG neurons. In view of importance of the medium-sized DRG neurons in pain,17,31–36 the membrane CaV3.2 in medium-sized DRG neurons could take important roles in neuropathic pain.
Intracellular calcium is an important intracellular messenger,37,38 and intracellular calcium entry through voltage-gated calcium channels plays an important role in increasing the excitability of mouse DRG axotomized neurons. 32 Peripheral sensitization is a major player in neuropathic pain, which originates from both damaged and intact nerve fibers and from damaged and intact dorsal root ganglia (DRG),14–16 so the membrane CaV3.2 in medium-sized damaged DRG neurons could participate in ectopic discharges by increasing intracellular calcium and then participate in neuropathic pain.
Molecular mechanisms underlying CaV3.2 channel trafficking to the membrane of the damaged neurons after SNI is a further interesting question. One possibility is inflammatory mediators (such as interleukin-6) released from activated macrophages and satellite glial cells combined to their receptors and thus upregulated the membrane CaV3.2 in medium-sized damaged DRG neurons.39,40 Another possibility may be neuropoietic cytokines such as ciliary neurotrophic factor and leukemia inhibitory factor which regulate trafficking of T-type Ca2+ channels in damaged DRG neurons through JAK and ERK signaling pathways. 41
Although we did not observe obvious increase of neuronal excitability of damaged DRG neurons from SNI rats with patch clamping recording methods (see discussion below), we observed an significant increase of ADP amplitudes in the damaged medium-sized DRG neurons at −85 mV and at −95 mV (Figure 5), indicating a functional up-regulation of CaV3.2 T-type calcium channels in the damaged medium-sized neurons after SNI. White et al. 11 found that the transient low-threshold Ca2+ current triggered burst firings through ADP in an adult mammalian neurons. These results suggest that once the excitation of a DRG neuron is initiated, the excitability will be strengthened by the increased ADP amplitude under the functional up-regulation of membrane CaV3.2 T-type calcium channels.
In SNI-induced neuropathic pain rats, we measured the neuronal intrinsic membrane properties by whole-cell patch clamp recording. Unexpectedly, the excitability of damaged DRG neurons showed no obvious changes (Supplementary Figure 1, original data were shown in Supplementary Table 2). These results suggest that CaV3.2 T-type calcium channels were not correlated with neuronal excitability of DRG neurons. In fact, the neuronal excitability of DRG neurons following axotomy is more likely dependent on comprehensive changes in several kinds of ion channels like Na+, K+, HCN, and TRPV1.3,22,36
In the present study, we found that CaV3.2 T-type calcium channels increased significantly in damaged medium-sized neurons (Figure 3) with significant trafficking to neuronal cell membrane (Figure 3). To examine the possible effects of CaV3.2 in SNI-induced neuropathic pain, we further examined the effects of CaV3.2 AS in the DRGs on mechanical allodynia. Intrathecal injection of CaV3.2 antisense ODNs could reverse mechanical allodynia (Figure 6). Additionally, we found that there were no obvious changes of CaV3.2 expression in total, membrane, or cytosolic proteins in DRGs after SNI (Figure 2) and no obvious changes of CaV3.2 expression in total proteins in DRGs after the application of antisense (data not shown), but CaV3.2 expression increased in sural nerve after SNI and CaV3.2 decreased with the application of antisense in sural nerve after SNI but not the MS-Cav3.2 (data not shown). These data indicated successful AS-mediated knock-down of Cav3.2 T-type calcium channels after SNI. All of these results suggest that CaV3.2 protein in the damaged DRG neurons might make important contributions to neuropathic pain.
In our study, we focused on the role of CaV3.2 T-type calcium channels in neuropathic pain. In fact, CaV3.2 T-type calcium channels also have an important role in inflammation pain. For example, Zhang et al. 42 found that CaV3.2 is involved in IGF-1R-mediated pain hypersensitivity in CFA-induced inflammatory pain behaviors. All of the researches about the role of CaV3.2 T-type calcium channels in neuropathic pain and inflammation pain provided the possibility of CaV3.2 T-type calcium channels as a target of development of new analgesics.
In conclusion, CaV3.2 T-type calcium channels in the damaged medium-sized DRG neurons might play an important role in neuropathic pain of SNI rats.
Supplemental Material
Supplementary Table 1 -Supplemental material for Increased expression of CaV3.2 T-type calcium channels in damaged DRG neurons contributes to neuropathic pain in rats with spared nerve injury
Supplemental material, Supplementary Table 1 for Increased expression of CaV3.2 T-type calcium channels in damaged DRG neurons contributes to neuropathic pain in rats with spared nerve injury by Xue-Jing Kang, Ye-Nan Chi, Wen Chen, Feng-Yu Liu, Shuang Cui, Fei-Fei Liao, Jie Cai and You Wan in Molecular Pain
Supplemental Material
Supplementary Table 2 -Supplemental material for Increased expression of CaV3.2 T-type calcium channels in damaged DRG neurons contributes to neuropathic pain in rats with spared nerve injury
Supplemental material, Supplementary Table 2 for Increased expression of CaV3.2 T-type calcium channels in damaged DRG neurons contributes to neuropathic pain in rats with spared nerve injury by Xue-Jing Kang, Ye-Nan Chi, Wen Chen, Feng-Yu Liu, Shuang Cui, Fei-Fei Liao, Jie Cai and You Wan in Molecular Pain
Supplemental Material
Figure -Supplemental material for Increased expression of CaV3.2 T-type calcium channels in damaged DRG neurons contributes to neuropathic pain in rats with spared nerve injury
Supplemental material, Figure for Increased expression of CaV3.2 T-type calcium channels in damaged DRG neurons contributes to neuropathic pain in rats with spared nerve injury by Xue-Jing Kang, Ye-Nan Chi, Wen Chen, Feng-Yu Liu, Shuang Cui, Fei-Fei Liao, Jie Cai and You Wan in Molecular Pain
Footnotes
Author Contributions
XJK and YW raised the hypothesis. XJK and YNC conducted the experiments. WC and FYL helped design the study. SC, FFL, and JC provided technical advices. XJK, YNC, and YW analyzed the data. XJK and YW drafted the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from National Natural Science Foundation of China (81571067, 91732107, 81521063, 30600173) and the “111” Project” of Ministry of Education of China (B07001).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
